Abstract
Background
This study aims to investigate the potential association between the incidence of portal vein tumor thrombosis (PVTT) and a panel of inflammatory indices—namely the systemic immune-inflammatory index (SII), platelet-to-lymphocyte ratio (PLR), and neutrophil-to-lymphocyte ratio (NLR)—in patients with HBV-related hepatocellular carcinoma (HCC).
Methods
Covariates were identified through univariate and multivariate logistic regression analyses alongside the variance inflation factor (VIF). The associations of inflammatory biomarkers were evaluated using subgroup analyses, multivariate logistic regression, and restricted cubic spline (RCS) models. A nomogram was developed based on the selected variables, and calibration curves were plotted to assess the predictive accuracy of the model. Receiver operating characteristic (ROC) curves were generated, and the area under the curve (AUC) was calculated to evaluate the diagnostic performance of the model.
Results
In this study of 504 HBV-related HCC patients, PVTT was identified in 22.02% of cases. Multivariate analysis confirmed SII and PLR as independent risk factors for PVTT, with the third quartile of SII and PLR associated with 109.7% and 122.6% increased risk, respectively. A nonlinear dose-response relationship for SII was identified with an inflection point at 467.5. A nomogram incorporating SII and five clinical variables demonstrated good predictive accuracy (AUC = 0.760) and high specificity (84%), supporting its utility for clinical risk stratification.
Conclusions
The SII was identified as an independent risk factor for PVTT in patients with HBV-related HCC.
Keywords
Background
Hepatocellular carcinoma (HCC) ranks among the most frequently diagnosed cancers, with over 865,269 new cases (the sixth highest incidence of all cancers) and 757,948 fatalities (the third highest of all cancers) recorded in 2022.1,2 Primary liver cancer mostly comprises HCC (75%-85% of cases) and intrahepatic cholangiocarcinoma (10%-15% of cases), with chronic hepatitis B virus (HBV) or hepatitis C virus (HCV) infection responsible for 21% to 55% of HCC cases globally.3,4 China bears the greatest burden of HCC globally, with hepatitis B virus (HBV) accounting for over 190,000 instances of HCC in the country. 5 A significant proportion of patients with HCC exhibit vascular invasion during disease exacerbation, recurrence, or progression. 6 These vascular invasions, particularly portal vein invasions, lead to severe stages of the disease for which therapeutic treatments are no longer available. 7 Research indicates that the prevalence of portal vein tumor thrombosis (PVTT) is greater in patients with HBV-associated HCC at 31%, compared to 22% in those with HCV-associated HCC and 29% in patients with non-HBV, non-HCV-associated HCC. 8
PVTT, a significant intrahepatic vascular infiltration in HCC patients, poses a substantial challenge in the management of HCC, affecting around 44% to 62% of individuals with the condition. 9 Increasing research elucidates numerous molecular processes, encompassing genetic and epigenetic regulation, cancer stem cells, the immunosuppressive microenvironment, and hypoxia, among others. 10 Because of their affordability, convenience of use, and simplicity, inflammatory indices based on blood cell counts have garnered a lot of attention since their inception. There is a substantial association between the prognosis of gallbladder cancer, intrahepatic cholangiocarcinoma, and HCC and the systemic immune inflammatory index (SII).11,12 In addition, the SII is a validated assessment of HCC recurrence after liver transplantation and hepatic resection. Platelet-to-lymphocyte ratio (PLR) has been shown to be strongly associated with the prognosis of HCC patients after radiofrequency ablation and liver transplantation.13,14 Neutrophil-to-lymphocyte ratio (NLR) has been previously demonstrated to be an independent predictor of HCC. 15 An elevated NLR in HCC patients has been linked to an undesirable prognosis in individuals with primary HCC, according to another study.16,17 It is noteworthy that preoperative NLR has been identified as a predictive factor in patients with PVTT and HCC following hepatectomy. Derived from complete blood count profiles, these composite biomarkers may reflect integrated immunological responses through multiple biological pathways. Their simultaneous quantification provides a comprehensive perspective on systemic inflammatory status, surpassing conventional single-parameter assessments.
Therefore, this study aimed to develop and internally validate a predictive nomogram that integrates systemic inflammatory biomarkers (SII, PLR, and NLR) with key clinical variables to estimate the individualized risk of PVTT in patients with HBV- HCC. This model may facilitate early risk stratification and guide clinical decision-making for the prevention and management of PVTT in this high-risk population.
Methods
Study Population
Initially, 2200 patients diagnosed with primary HCC at Tianjin Third Central Hospital between 2018 and 2023 were included in this study, and in this cohort, 1608 individuals diagnosed with primary HCC for the first time during their hospital stay were considered for further analyses, and it was ensured that patients had not previously received systemic treatments for HCC (eg, transarterial chemoembolization (TACE), targeted therapies, immune checkpoint inhibitors, or chemotherapies); all patients had been screened for PVTT by enhanced CT/MRI during their hospital stay, and there was no missing personal history information such as smoking history and alcohol consumption. Strict exclusion standards were used to guarantee the precision and dependability of the findings. Patients with a combination of other malignancies (including cholangiocellular carcinoma, lung cancer and breast cancer) were excluded. In addition, people with other causes of HCC (including HCV infection, alcoholic liver disease, metabolism-associated steatohepatopathy, and autoimmune liver disease), confirmed portal vein thrombosis, and hematological abnormalities (leukemia, myelodysplastic syndromes, and thrombocythemia) were excluded from the analysis. To reduce bias, white blood cell (WBC) count, neutrophil (NEU) count, lymphocyte (LYM) count, platelet (PLT) count, albumin (ALB) level, hepatitis B surface antigen (HBsAg), hepatitis B surface antibody (HBsAb), hepatitis B e antigen (HBeAg), hepatitis B e antibody (HBeAb), hepatitis B core antibody (HBcAb), alpha-fetoprotein (AFP), carcinoembryonic antigen (CEA), glycoprotein 19-9 (CA19-9), aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP) and plasminogen time (PT) data were excluded. The final analytical cohort comprised 504 eligible participants, with detailed recruitment flow depicted in Figure 1.
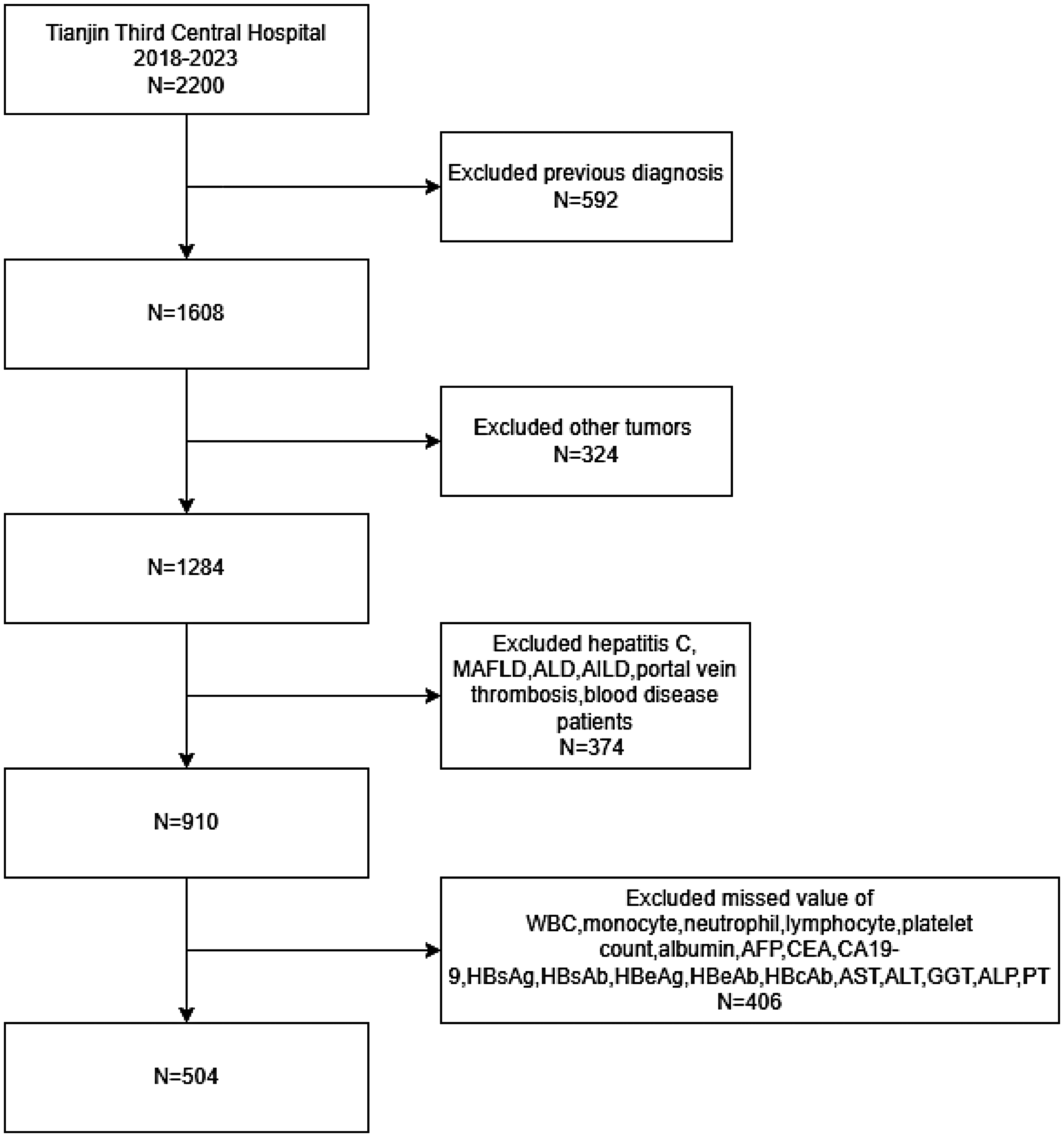
Flow chart of participants selection. MAFLD metabolic dysfunction-associated fatty liver disease, ALD alcoholic liver disease, AILD autoimmune liver disease.
Data Collection and Laboratory Methods
Clinical data, examination results, lifestyle factors, medical history, and medication regimens from the time of initial presentation through hospital admission were included in the comprehensive study dataset. Admission records encompassed demographic characteristics (age, sex) and anthropometric measurements (blood pressure, height, weight, body mass index).
Smoking status was defined as lifetime consumption of at least 100 cigarettes. Alcohol consumption was classified as a history exceeding 5 years. Diabetes mellitus was defined by self-reported diagnosis, use of oral hypoglycemic agents, or insulin therapy. Hypertension was diagnosed based on systolic blood pressure >140 mm Hg, diastolic blood pressure >90 mm Hg, self-reported history, or current use of antihypertensive medication. Cerebral ischemic events were defined according to standardized criteria: persistent focal neurological deficits (>24 h), neuroimaging-confirmed infarction (CT or MRI), or documented history of cerebral infarction/transient ischemic attack.
Fasting venous blood samples (8-10 h) were collected for standardized laboratory analysis. Measurements were performed using an automated analyzer according to the manufacturer's protocol. Complete blood count analysis determined white blood cell, neutrophil, lymphocyte, and platelet counts. The following indices were calculated:
- SII = platelet count ×neutrophil count / lymphocyte count - NLR = neutrophil count / lymphocyte count - PLR = platelet count / lymphocyte count
The Diagnosis of Hepatitis B-Related HCC and PVTT
The study enrolled individuals who were positive for HBsAg for over six months or had detectable HBV DNA, while excluding those with other viral hepatitis (eg, HCV, HDV), alcoholic liver disease, metabolic dysfunction-associated steatotic liver disease (MASLD), autoimmune liver disease, and other etiologies of HCC. Those among them with a confirmed diagnosis of HCC via liver biopsy or imaging studies were classified as having hepatitis B-associated HCC. The diagnosis of HCC with PVTT was established when the HCC diagnosis was unequivocal and there was demonstrable evidence of PVTT, characterized by distinct space-occupying lesions in the portal vein across all phases of CT/MRI, exhibiting partial enhancement in the arterial phase and filling deficiencies in the portal vein phase.
Statistical Analyses
To ensure model stability and the reliability of parameter estimates, we first performed multicollinearity analysis, excluding variables with a variance inflation factor (VIF) ≥ 10. Subsequently, univariate logistic regression was conducted. Variables yielding a P-value <.05 were further included in the multivariate logistic regression analysis. Variables with a P-value ≤.05 in this analysis were selected as covariates for subsequent steps. Three distinct models were constructed: Model 1with no adjustment, Model 2 adjusting for specific covariates (Alcohol Use, Child Pugh), and Model 3 adjusting for variables selected by multivariate logistic (Alcohol Use, Child Pugh, AFP, GGT, FIB). Stratified analysis and interaction effect analysis were subsequently performed across all covariates, with the results visualized using forest plots. We then employed restricted cubic spline (RCS) regression and threshold effect analysis to investigate potential nonlinear relationships and threshold effects between SII, PLR, NLR, and PVTT. Furthermore, a nomogram was constructed using the screened variables to visually depict the relative importance of each factor in predicting PVTT occurrence. The calibration curve was plotted to assess the predictive accuracy of this model. Finally, receiver operating characteristic (ROC) curves were generated, and the area under the curve (AUC) was calculated to evaluate the diagnostic performance of the models. Decision curve analysis was performed to evaluate the clinical utility of the nomogram by quantifying its net benefit across a range of threshold probabilities. All analyses were performed using R statistical software (version 4.4.1). All statistical tests were two-sided, and a P-value <.05 was considered statistically significant.
Results
Characteristics of the Study Population
The analytical cohort comprised 504 patients with hepatitis B-related hepatocellular carcinoma (Table 1), demonstrating significant gender disparity (male: 80.36%; female: 19.64%) and mean age (58.97 ± 9.89 years). PVTT was radiologically confirmed in 22.02% of cases (n = 111), establishing this subgroup as the primary focus of subsequent analyses. There were significant differences between the PVTT and non-PVTT groups (P < .05) in the liver function indices (total bilirubin, direct bilirubin, alkaline phosphatase, gamma-glutamyl transpeptidase, glutamyltransferase, and glutamic oxalyltransferase) and individual components of systemic inflammatory biomarkers, such as the platelet, neutrophil, and lymphocyte counts. Significant differences in systemic inflammatory biomarkers, ie SII, PLR, NLR, were observed between the PVTT and non-PVTT groups (P < .05). These differential patterns remained consistent across both continuous and quartile-stratified analyses. Moreover, significant dose-response relationships emerged for NLR and PLR (Figure 2). Compared to Q1 reference, progressively elevated PVTT risks were observed across quartiles (Q2-Q4), demonstrating monotonic increasing trends.

Inflammatory burden (by biomarker quartiles) and risk of PVTT in hepatocellular carcinoma patients.
Characteristics of PVTT Versus Non-PVTT among Patients.

Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; TBIL, total bilirubin; ALP, alkaline Phosphatase; GGT, gamma-glutamyl transpeptidase; FIB, fibrinogen; AFP, alpha-fetoprotein; SII, systemic immune-inflammation index; PLR, platelet-to-lymphocyte ratio; NLR, neutrophil-to-lymphocyte ratio.
Covariate Selection
To simplify the model structure and ensure its stability, we first used the VIF to assess multicollinearity among variables, identifying and excluding those with a VIF value > 10 (Figure 3). Subsequently, we performed univariate logistic regression analysis, and variables with a P-value <.05 were further incorporated into the multivariate logistic analysis (Table 2). Ultimately, we identified alcohol consumption, AFP, Child-Pugh grade, GGT, and FIB as covariates for subsequent analysis.

Multicollinearity analysis of variables associated with inflammatory markers, quantified by variance inflation factors (VIFs). Abbreviations: WBC white blood cells, N neutrophils, L lymphocytes, PLT platelets, APTT activated partial thromboplastin time, FIB fibrinogen, TT thrombin time, PTA prothrombin activity, ALT alanine aminotransferase, AST aspartate aminotransferase, GGT gamma-glutamyl transpeptidase, TBIL total bilirubin, DBIL direct bilirubin, TBA total bile acids, PT prothrombin time, BUN serum urea nitrogen, Cr serum creatinine, PALB prealbumin, ALB albumin, CHE cholinesterase, AFP alpha-fetoprotein, CEA carcinoembryonic antigen, CA19-9 carbohydrate antigen 19-9, HBsAg hepatitis B surface antigen, HBsAb hepatitis B surface antibody, HBeAg hepatitis B e antigen, HBeAb hepatitis B e antibody, HBcAb hepatitis B core antibody.
Univariate and Multivariate Logistic Regression Analysis.
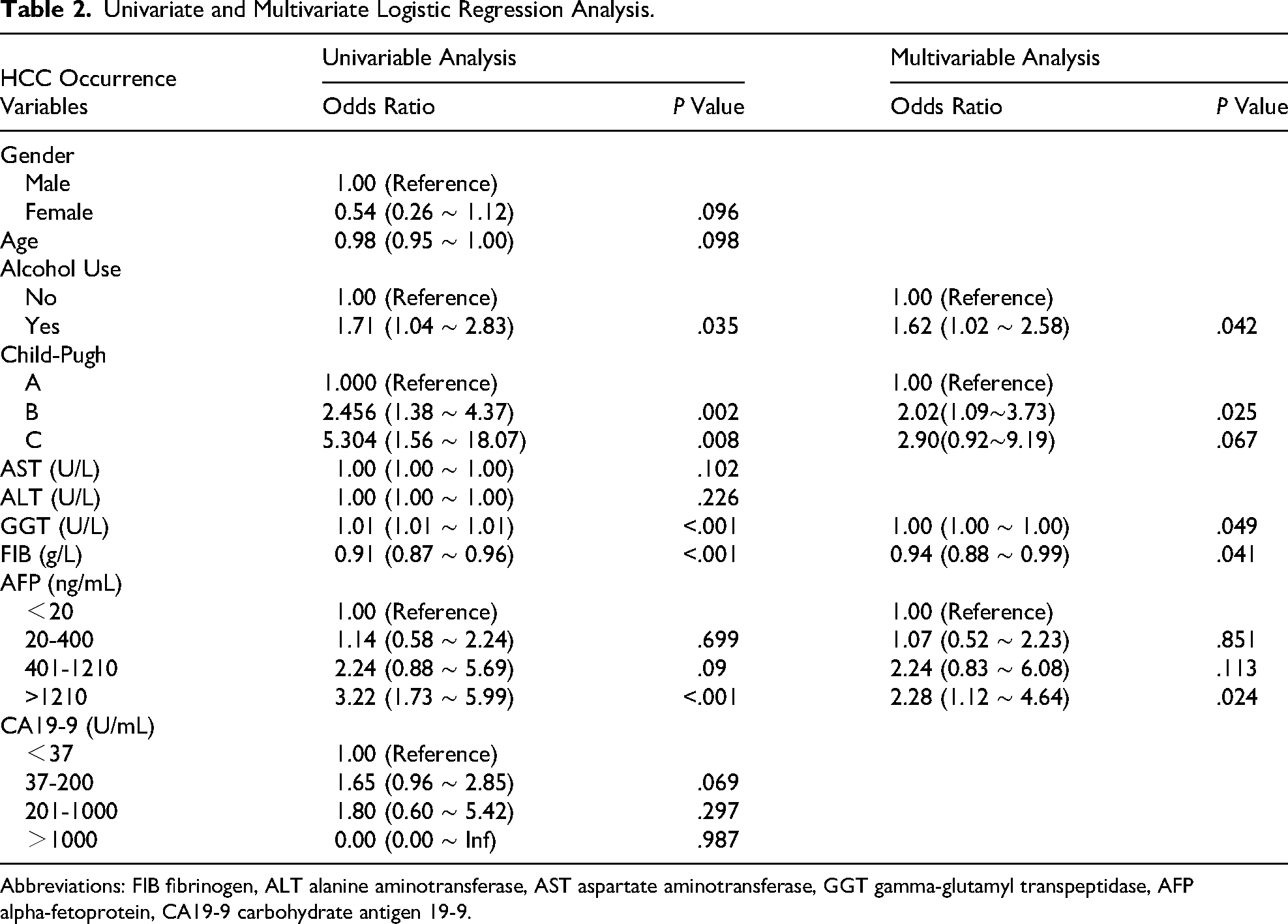
Abbreviations: FIB fibrinogen, ALT alanine aminotransferase, AST aspartate aminotransferase, GGT gamma-glutamyl transpeptidase, AFP alpha-fetoprotein, CA19-9 carbohydrate antigen 19-9.
Association Between Systemic Inflammatory Biomarkers and PVTT Risk
We subsequently performed multivariate logistic regression analysis (Table 3). In the unadjusted model, higher levels of SII, PLR, and NLR (quartiles Q3 and Q4) all demonstrated an increased trend in PVTT risk. In Model 3, adjusted for all covariates, the third quartile (Q3) of both SII and PLR remained significantly associated with PVTT. Compared to the lowest quartile (Q1), patients in the Q3 of SII group exhibited a 109.7% increased risk of PVTT (OR = 2.097; 95% CI = 1.076-4.194, P = .032), while those in the Q3 of PLR group showed a 122.6% increased risk (OR = 2.226; 95% CI = 1.103-4.648, P = .028).
Association Between Systemic Inflammatory Biomarkers and PVTT Risk.

Subgroup and Interaction Analyses
To further examine the association between systemic inflammatory biomarkers and the risk of PVTT, subgroup and interaction analyses were conducted. The results revealed no statistically significant heterogeneity in the association of SII and NLR with increased PVTT risk across subgroups (Figure 4A and B). However, PLR exhibited a stronger association with elevated PVTT risk specifically in participants within the third quartile (Q3) of GGT levels (Figure 4C).

Inflammatory biomarkers and PVTT risk: subgroup analyses by demographic factors. (A) Subgroup Analysis Forest Plot of SII. (B) Subgroup Analysis Forest Plot of NLR. (C) Subgroup Analysis Forest Plot of PLR.
Dose-Response Relationship Between Inflammatory Biomarkers and PVTT Susceptibility
We used the RCS regression model to perform a further investigation of the non-linear connection between continuous systemic inflammatory biomarkers and the risk of PVTT in order to enhance the accuracy of our findings. In the unadjusted model 1 (Figure 5A), both SII and NLR demonstrated significant nonlinear associations with PVTT risk (P-overall <.05, P-non-linear <.05), while PLR exhibited a significant linear association with PVTT (P-overall <.05). After full adjustment for covariates (Figure 5B), SII maintained a significant nonlinear relationship with PVTT risk (P-overall = .018, P-non-linear = .009). Further threshold effect analysis revealed a critical inflection point at SII = 467.541 (Table 4). When SII ≤ 467.541, each unit increase in SII was associ0ated with a 0.3% increased PVTT risk (OR = 1.003; 95% CI = 1.000-1.007, P = .040). When SII > 467.541, no significant association was observed between SII and PVTT risk (OR = 1.000; 95% CI = 1.000-1.000, P = .639).

(A) The restricted cubic splines (RCS) curve shows the association between inflammatory biomarkers and PVTT in all study participants. We did not conduct variable adjustments. (B) Nonlinear relationship between inflammatory biomarkers and PVTT: in the RCS regression, adjustments were made for Alcohol Use, Child Pugh, AFP, GGT, and FIB.
Threshold Analysis Result (Adjusted for Alcohol use, Child Pugh, AFP, GGT, FIB.).

Construction and Evaluation of the Nomogram Model
The nomogram, constructed by integrating five variables selected through univariate and multivariate logistic regression analyses along with the Systemic Immune-Inflammation Index (SII), enables individualized PVTT risk prediction by assigning point values to each predictor, summing these into a total score, and projecting this score onto a risk probability axis (Figure 6A). Internal validation with 1000 bootstrap resamples demonstrated excellent calibration (mean absolute error = 0.018; Figure 6B) and good discriminative power (AUC = 0.760; Figure 7). Based on the maximized Youden index, the optimal risk probability threshold for classifying PVTT was determined to be 0.287, meaning patients with a predicted probability > 0.287 were classified as high-risk for PVTT. At this threshold, the nomogram achieved 59% sensitivity and 84% specificity, confirming its strong rule-out capability for clinical exclusion of non-PVTT cases. To assess the clinical utility of the nomogram, we performed decision curve analysis (Figure 8). The results demonstrated that the nomogram provided a positive net benefit across a wide range of clinically reasonable threshold probabilities (from 10% to 58%), indicating that using the nomogram to guide clinical decisions would add more benefit than either the “treat-all” or “treat-none” strategies.
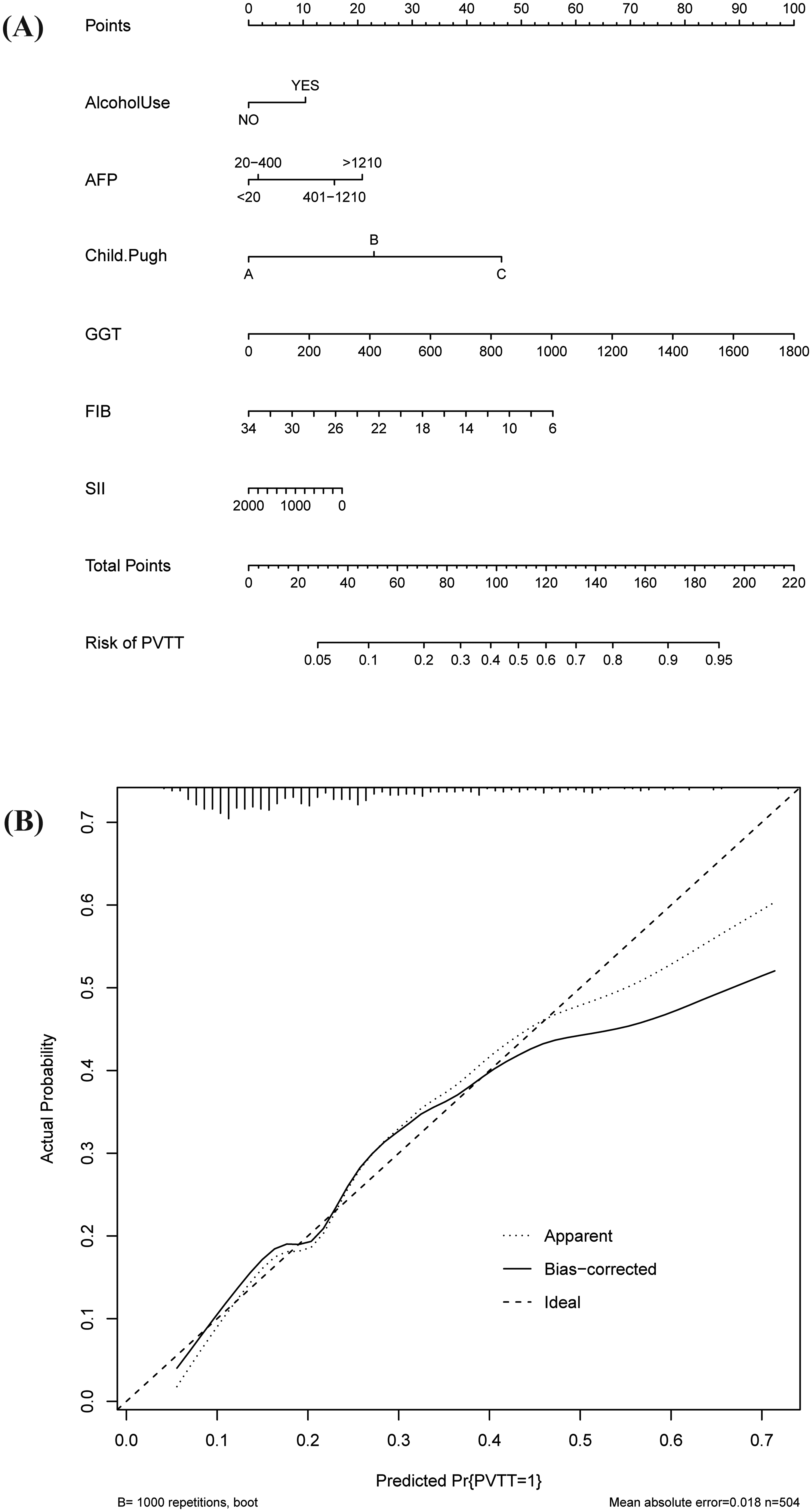
(A) A nomogram was used to estimate the risk of PVTT related to SII. (B) Internal validation with 1000 bootstrap resamples.

The receiver operating characteristic (ROC) curves of all variable models.

Decision curve analysis for the nomogram predicting PVTT in patients with HBV-related HCC.
Discussion
Multivariable logistic regression analysis revealed that alcohol consumption, AFP > 1210 ng/mL, Child-Pugh grade A, GGT level, and FIB level were independent risk factors for PVTT.
The study results demonstrated a significant positive correlation between SII and an increased risk of PVTT development in patients with HBV-related HCC. First, in the logistic regression analysis, a significant positive association between SII and the risk of PVTT was consistently observed, both before and after adjusting for covariates. Furthermore, the RCS curve and corresponding threshold effect analysis revealed a non-linear relationship between SII and the risk of PVTT, with an inflection point identified at 467.541. When SII was below this threshold, the risk of PVTT increased with rising SII levels. This finding, consistent with the logistic regression analysis, solidifies the association between elevated SII and PVTT risk in HBV-HCC patients. The findings of this study indicate that lower SII levels are associated with a reduced risk of PVTT, suggesting that modulating systemic inflammation may represent a potential preventive avenue. Although this observational study cannot establish causality, it provides strategy-oriented insights for future research and clinical practice. The identified inflection point (approximately 468) could serve as a practical clinical monitoring threshold. Dynamic assessment of SII may help identify patients with escalating inflammatory risk, thereby prompting intensified imaging surveillance for early detection of vascular invasion. Furthermore, given that SII reflects a pro-inflammatory and immune-imbalanced state, investigating safe long-term anti-inflammatory agents or actively managing concurrent inflammatory drivers—such as hepatitis B viremia or metabolic syndrome—to lower SII levels warrants further exploration. Additionally, for patients undergoing curative-intent treatment, exploring preoperative or adjuvant immunomodulatory strategies aimed at reducing SII may help diminish the risk of microvascular invasion and subsequent PVTT development. These strategies remain hypothetical and require validation in prospective interventional studies. Nonetheless, this work positions SII not only as a prognostic indicator but also as a potential monitoring tool and therapeutic target for preventing this severe complication in HBV-related HCC.
It is unclear exactly which biological processes connect inflammatory indices to PVTT. Nevertheless, a number of theories might be put out in light of our findings, the pathophysiology of PVTT, and the part inflammation plays in metabolic diseases. Inflammatory markers may contribute to portal invasion by activating hepatic stellate cells (HSCs) or exacerbating oxidative stress, leading to elevated GGT, which in turn releases metalloproteinases (eg MMP-9) that may promote extracellular matrix (ECM) degradation and thus tumor cell invasion through the vascular basement membrane.18–20 Secondly, elevated inflammatory factors can cause hepatocyte damage and mitochondrial dysfunction, which promotes the release of GGT, which in turn promotes the generation of reactive oxygen species (ROS) through the catabolism of extracellular glutathione, destroying the integrity of the vascular endothelium or directly contributing to the development of PVTT.21–23 The elevation of SII suggests that the organism is in a state of chronic inflammation, which accelerates the progression of hepatic fibrosis and promotes the elevation of portal venous pressure, leading to the development of portal invasion. Elevated portal venous pressure leads to altered vascular shear forces, contributing to endothelial cell injury or platelet activation, portal microthrombosis, leading to tumor cell adhesion to the thrombus surface, and accelerating PVTT proliferation.24,25
This study demonstrates a significant association between systemic inflammatory biomarkers, particularly the SII, and the presence of PVTT in patients with HBV-related HCC. The consistency of our findings across multiple regression models and sensitivity analyses strengthens the reliability of the observed relationships. Nonetheless, several important limitations must be acknowledged to contextualize the results. First, the single-center, cross-sectional design inherently limits causal inference. While we identified significant associations, we cannot definitively establish whether elevated inflammatory markers precede and contribute to PVTT development or are merely a consequence of more advanced tumor biology. Second, the relatively small sample size, especially with only 111 PVTT-positive cases, may constrain the statistical power for detailed subgroup analyses and increase the risk of type II errors. Third, although we performed extensive adjustments for available clinical covariates, residual confounding from unmeasured or unrecorded factors (eg, detailed portal hypertension metrics, specific anticancer treatments prior to enrollment, or genetic background) remains possible. Fourth, a critical methodological limitation concerns the development and validation of our nomogram. The model was constructed and evaluated on the same dataset without an internal validation cohort or an external cohort from a different institution. This absence of robust validation limits the assessment of its potential for overfitting and its generalizable clinical applicability. Fifth, although we developed a nomogram for predicting PVTT in HBV-related HCC, we were unable to directly compare its performance with existing predictive models. This is because, to the best of our knowledge, there is currently no universally accepted or widely validated clinical prediction model specifically designed for preoperative PVTT risk assessment in this patient population. The existing tools vary substantially in terms of target populations, predictor sets, outcome definition, and timing of prediction. Given this heterogeneity, a direct head-to-head comparison using our current dataset may not yield clinically meaningful or generalizable insights. Finally, the generalizability of our findings may be limited by the ethnic homogeneity (predominantly Han Chinese) of our cohort, as genetic and regional environmental factors could influence inflammatory pathways.
To address these limitations and advance this field, future multi-center, prospective longitudinal or cohort studies with larger, more diverse populations are essential. Such studies are needed to verify the temporal relationship between inflammation and PVTT, refine risk prediction, and validate our proposed nomogram in independent patient cohorts. Furthermore, translational research integrating mechanistic investigations is warranted to elucidate the precise pathophysiological links between systemic inflammation and portal vein thrombogenesis in HCC.
Conclusions
The SII was identified as an independent risk factor for PVTT in patients with HBV-related HCC. In our study, approximately 22% of the patients experienced PVTT events. We successfully developed a nomogram model to predict distinct risk trajectories associated with PVTT development in this patient population, which may facilitate earlier imaging examinations to confirm the presence of PVTT and prompt timely intervention for high-risk individuals.
Supplemental Material
sj-pdf-1-cat-10.1177_10760296261433671 - Supplemental material for The Systemic Immune-Inflammation Index Predicts Portal Vein Tumor Thrombosis and Informs a Clinical Nomogram in HBV-Related Hepatocellular Carcinoma
Supplemental material, sj-pdf-1-cat-10.1177_10760296261433671 for The Systemic Immune-Inflammation Index Predicts Portal Vein Tumor Thrombosis and Informs a Clinical Nomogram in HBV-Related Hepatocellular Carcinoma by Runci Jiang, Shuqin Cheng, Lu Jiang, Yuexi Yu, Menghua Lin, Yiping Zhang, Jingyu Shen, Guliziba Abudumijiti, Boyuan Xiao, Haoyang Pang and Fengmei Wang in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Patient Consent Statement
This study followed the Declaration of Helsinki and was approved by the Medical Ethical Committee of the Tianjin Third Center Hospital (approval number: IRB2022-036-02). The informed consent was waived because this is a retrospective study conducted by a review of medical records only.
Author Contributions
Conceptualization: JRC and CSQ; methodology: JRC and JL; software: XBY and CSQ; validation: ZYP and YYX; formal analysis: JRC, CSQ, LMH, and JL; data curation: JRC and PHY; writing: all authors; writing—review and editing: JRC, CSQ, YYX, and SJY; visualization: JRC, GLZB and CSQ; supervision: WFM.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Incubation Fund of Tianjin First Central Hospital, Tianjin Natural Science Foundation Project, Tianjin Municipal Education Commission Scientific Research Program Project, Tianjin Key Medical Construction Project, (grant number 2025FYMS07, 25JCZDJC00960, 2025ZXZD017, TWJ2022XK027).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data included in this study are available upon request by contacting with the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
