Abstract
Background
Immune thrombocytopenic purpura (ITP) is an autoimmune hematologic disorder characterized by thrombocytopenia and clinical manifestations such as petechiae, ecchymosis, and bleeding. Recently, simple and accessible inflammatory indices including neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and hemoglobin-to-platelet ratio (HPR) have been proposed as potential biomarkers for disease assessment. The aim of this study was to evaluate the diagnostic and clinical value of NLR, PLR, and HPR in patients with ITP in Southeast Iran.
Methods
This retrospective cross-sectional study included 114 ITP patients and 115 control. Hematologic parameters and inflammatory indices (NLR, PLR, HPR) were assessed before and after treatment. Diagnostic accuracy was evaluated using receiver operating characteristic (ROC) curves.
Results
Significant differences were observed between patients and controls across all parameters. HPR was notably higher in ITP patients. Bleeding symptoms were associated with elevated NLR and HPR (P < .001). After treatment, patients with petechiae showed decreased NLR (P = .016). ROC analysis revealed high sensitivity and specificity for NLR, PLR, and HPR.
Conclusion
PLR and HPR may serve as supportive tools for the diagnosis and management of ITP when combined with clinical and hematologic features. NLR should be interpreted cautiously and used alongside other markers. Further large-scale studies are warranted to validate these findings.
Keywords
Introduction
In ITP, autoantibodies primarily target platelet surface glycoproteins specifically GPIIb/IIIa and GPIb/IX resulting in accelerated platelet destruction by the reticuloendothelial system. These autoantibodies also bind to megakaryocytes in the bone marrow, impairing platelet production. The involvement of T-cell-mediated mechanisms in antibody production highlights the complex interplay between humoral and cellular immunity in disease pathophysiology.3,4
Diagnostic evaluation of ITP typically includes complete blood counts, screening for autoimmune markers, and virological assays. Bone marrow aspiration is reserved for select scenarios such as advanced age, organomegaly, or suspicion of hematologic malignancy. Immune dysregulation in ITP is further evidenced by elevated circulating inflammatory cytokines including IL-10, IL-17, IL-18, interferon-alpha (IFN-α), and tumor necrosis factor-alpha (TNF-α) which contribute to disease activity and progression. Despite their diagnostic and therapeutic relevance, routine cytokine profiling is often limited by cost and logistical constraints, particularly in resource-limited settings.5–7
Chronic inflammation underlies the pathogenesis of many autoimmune diseases, including ITP. Immune cell activation and recruitment comprising neutrophils, lymphocytes, monocytes, and platelets amplify inflammatory cascades that exacerbate thrombocytopenia. Platelets, beyond their hemostatic role, actively participate in inflammatory processes by releasing pro-inflammatory mediators and interacting with leukocytes. Recent studies have linked extensive platelet destruction in ITP with secondary inflammatory complications, such as pulmonary inflammation.3,8
Neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR) have emerged as accessible, cost-effective biomarkers for systemic inflammation. These indices have demonstrated prognostic utility in various clinical conditions, including infectious diseases, ischemic stroke, and myocardial infarction, and have been correlated with adverse outcomes in chronic illnesses such as type 2 diabetes mellitus, hypertension, and cardiovascular disease.9,10
Neutrophils and lymphocytes constitute the frontline immune response to pathogenic insults and are integral to both acute and chronic inflammatory states. The NLR, easily derived from routine hematological tests, serves as a surrogate marker of immune balance and inflammation severity. Elevated NLR values have been consistently associated with autoimmune disease activity and may provide critical prognostic information.11,12
Similarly, the PLR reflects systemic inflammatory status and is often utilized in conjunction with NLR to enhance diagnostic accuracy and severity stratification across diverse pathologies, including malignancies and inflammatory disorders.12,13
The hemoglobin-to-platelet ratio (HPR) is an emerging biomarker reflecting the interplay between inflammation, oxygen delivery, vascular function, and immune status. In pathologies characterized by tissue hypoxia and fibrosis, reduced hemoglobin coupled with thrombocytosis can exacerbate hypoxic injury by impairing microvascular blood flow and promoting inflammatory responses. 14
Given the pivotal role of hematological inflammatory parameters in the immunopathogenesis of autoimmune diseases like ITP, the present study aims to evaluate the diagnostic utility of NLR, PLR, and HPR readily available and cost-effective biomarkers to better understand their clinical significance in the diagnosis and management of ITP patients.
Materials & Methods
Population Sample
This retrospective cross-sectional study reviewed the medical records of all patients diagnosed with immune thrombocytopenic purpura (ITP) admitted to Ali ebn Abitaleb Hospital, Zahedan Province, from January 2019 to December 2023. The control group comprised healthy individuals who presented to the same hospital for routine health check-ups during the same period. Controls were selected based on normal platelet counts adjusted for age and sex and absence of clinical manifestations consistent with ITP, including platelet counts <100 × 109/L, bleeding episodes, thrombosis, or any chronic or inflammatory diseases. Data were extracted from hospital archives using standardized data collection checklists.
Inclusion criteria for the ITP group were platelet counts below 100 × 109/L, presence of bleeding symptoms, non-smoking status, and no history of hookah or narcotic use. Additionally, patients with no other immunological diseases or alternative etiologies for thrombocytopenia were included.
Exclusion criteria comprised patients with incomplete clinical records or a documented history of systemic lupus erythematosus, rheumatoid arthritis, Behçet's disease, ankylosing spondylitis, thrombotic thrombocytopenic purpura (TTP), or HELLP syndrome. These criteria were established based on recent and validated literature to minimize selection bias.
Extracted data included demographic variables (age, sex), clinical parameters (length of hospitalization, symptoms, history of drug use, splenectomy status, season of disease onset), and laboratory values (complete blood counts including platelets, white blood cells, red blood cells, hematocrit, hemoglobin). Additionally, inflammatory indices neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and hemoglobin-to-platelet ratio (HPR) were calculated for pre-treatment (a) and post-treatment (b) time points.
Statistical analyses were performed using SPSS version 22. The normality of continuous data was assessed using the Kolmogorov-Smirnov test with Lilliefors correction. Parametric tests (Student's t-test) were applied for normally distributed data, while non-parametric tests (Mann-Whitney U test) were used for non-normally distributed variables.
Ethical Approval
The study protocol was approved by the local ethics committee of Zahedan University of Medical Sciences (Ethics code: IR.ZAUMS.REC.1402.024). The ethical approval is accessible at https://ethics.research.ac.ir/PortalProposalList.php?code = ir.zaums.rec.1402.024. Written informed consent was obtained from all participants prior to inclusion in the study.
Statistical Analysis
The data analysis was performed using SPSS software version 26. Analyses were performed based on two main groups: case and control. The case group was further subdivided into two subgroups: “before treatment (at the time of diagnosis)” and “after treatment.” Categorical variables (nominal or ordinal scale) were analyzed as the frequency with percentages, and to describe the continuous variables, mean ± standard deviation (mean ± SD), Median and interquartile range (IQR) was reported. We assessed the normality of data using the Kolmogorov–Smirnov test. Associations between different indices with continuous scales were assessed through Pearson or Spearman correlation tests. The nonparametric Wilcoxon–Mann–Whitney and Kruskal–Wallis H tests were utilized to compare variables between the control group and ITP patients. All tests were two-sided. In all the analyses, the P value of <.05 was considered statistically significant. Receiver operating characteristic (ROC) curve analysis was conducted to evaluate the diagnostic performance sensitivity and specificity of NLR, PLR, and HPR as inflammatory markers. The area under the curve (AUC) and optimal cut-off values were determined using GraphPad Prism version 8.4. Optimal thresholds were selected based on the maximal Youden index, balancing sensitivity and specificity.
Diagnostic performance was categorized according to AUC values as follows:
Class I (Excellent): > 90% Class II (Good): 80%-90% Class III (Acceptable): 70%-80% Class IV (Poor): 60%-70% Class V (Fail): ≤ 60%
Statistical significance was set at a P-value < .05.
Results
A total of 229 subjects were included in this study, divided almost equally into two groups based on the inclusion criteria: ITP patients (49.7%) and controls (50.3%). Within the ITP group, 68 patients (59.6%) were female and 46 (40.4%) were male. The control group included 68 females (59.1%) and 47 males (40.9%) (Table 1). The majority of participants in both groups were under 10 years of age, accounting for 60.5% of the ITP group and 46.1% of controls.
The Demographic Information of Participants and Hematological Parameters of ITP Patients and Control Group.

Mann-Whitney U test analysis (Table 1) revealed statistically significant differences (P < .05) between the ITP and control groups in almost all hematological parameters, including white blood cell count (WBCa), red blood cell count (RBCa), hemoglobin (Hba), hematocrit (HCTa), platelet count (PLTa), neutrophils (Neua), lymphocytes (LYMa), hemoglobin-to-platelet ratio (Hb/PLTa), neutrophil-to-lymphocyte ratio (NLRa), platelet-to-lymphocyte ratio (PLRa), and absolute lymphocyte count (ALCa). The only parameter not showing significant difference was ANCa.
Clinical Symptom Frequency in ITP Patients
Among ITP patients, the most common clinical symptoms observed were ecchymosis (68.4%), petechiae (56.1%), bruising (56.1%), bleeding (55.3%), and fever (22.8%). Ecchymosis, petechiae, and bruising were notably more prevalent. No significant association was found between clinical symptoms and length of hospitalization (P > .05).
Association Between Laboratory Parameters and Clinical Manifestations
Pre-treatment hematological parameters (PLTa, PLRa, NLRa, and Hb/PLTa ratio) were analyzed in relation to clinical symptoms among ITP patients. Notably, the mean Hb/PLTa ratio was significantly higher in patients presenting with bruising (11.98 vs 9.09; P = .041) and bleeding (12.13 vs 9.01; P = .05) (Table 2).
The Association Between Laboratory Parameters and Clinical Findings (Before-Treatment).

Abbreviation: IQR: interquartile range (Q1, Q3).*
Patients with bleeding also had a significantly elevated mean NLRa (2.53 vs 1.35; P < .001), whereas those with petechiae had a lower mean NLRa (1.70 vs 2.40; P = .016).
Conversely, PLRa was significantly reduced in patients exhibiting petechiae (11.88 vs 26.95; P = .005) and bruising (11.04 vs 28.43; P = .01). Platelet counts PLT were also significantly lower in patients with bruising (24,951.61 vs 65 600; P = .047) and bleeding (31 450 vs 55 783.33; P = .024) (Table 2).
Overall, PLRa showed significant differences across various clinical symptoms (P = .011), whereas Hb/PLTa ratio (P = .146), NLRa (P = .182), and PLTa (P = .061) did not. Post-hoc analysis indicated that PLRa was lowest in patients with the combined presentation of petechiae, purpura, and bruising (P < .05) (Table 3). No significant association was found between hospitalization length and multiple clinical findings (P = .204).
The Results of Posthoc Analysis Between various Multiple Clinical Symptoms and PLR Mean (Before-Treatment).

*Mean (absence of SD is due to the presence of one data).
Association Between Treatment Strategy and Outcomes
Intravenous immunoglobulin (IVIG) was the most frequently administered therapy (73.7%), followed by prednisolone (37.7%), cortisone (33.3%), tranexamic acid (15.8%), and rituximab (1.8%). No significant association was observed between treatment type and length of hospitalization (P > .05). Analysis of combined treatments also showed no significant impact on hospitalization duration (P = .505).
Association Between Treatment Strategy and Laboratory Parameters
Patients treated with IVIG exhibited significantly lower post-treatment NLR values (NLRb) compared to those not receiving IVIG (2.11 vs 4.42; P < .001). In contrast, NLRb was significantly higher in patients receiving cortisone (3.56 vs 2.14; P < .001) (Table 4).
The Association Between Treatment Strategy and Laboratory Findings.

*a is before-treatment and b is after-treatment.
Additionally, platelet counts (PLTb) were significantly lower post-treatment in patients administered prednisolone (83 333.33 vs 100 725.81; P = .042) (Table 4).
According to the results of the Kruskal-Walli's test, the mean of NLRb and ANCb variables in the categories of multiple drugs received had a significant difference (P-value .004, P-value .008, respectively) (Table 5). Therefore, in Table 6, the mean difference between NLRb and ANCb for categories of drugs received in combination is reported, the significance level of which is less than 0.05.
Statistics of Laboratory Findings by Multiple Drug (After-Treatment).

*Mean (absence of SD is due to the presence of one data).
The Results of Post-hoc Analysis Between various Treatment Strategies and NLRb Means (After-Treatment).

*Mean (absence of SD is due to the presence of one data).
Association Between Splenectomy, Hospitalization Length, and Laboratory Parameters
Splenectomy (one of the treatment options) was performed in 3.5% of patients. No significant association was found between splenectomy and length of hospitalization (P = .2), nor between splenectomy and laboratory parameters including Hb/PLTb, NLRb, PLRb, and PLTb (all P > .05).
Association Between Seasons, Hospitalization Length, and Laboratory Parameters
Seasonal variation was evaluated for its effect on hospitalization length and laboratory indices. The shortest and longest hospitalization durations were observed in winter (3.64 ± 3.08 days) and spring (6.65 ± 6.80 days), respectively; however, this difference was not statistically significant (P = .185). No significant seasonal associations were identified for Hb/PLTb, NLRb, PLRb, or PLTb (P > .05).
Diagnostic Accuracy of Laboratory Parameters
Receiver operating characteristic (ROC) analysis assessed the diagnostic utility of Hb/PLTa ratio, NLRa, and PLRa for ITP detection compared to controls. The Hb/PLTa ratio demonstrated excellent diagnostic performance with an optimal cut-off >0.8950, yielding 92.59% sensitivity (95% CI: 86.06-96.20) and 99.13% specificity (95% CI: 95.24-99.96), with an area under the curve (AUC) of 0.9534 (P < .001) (Table 7, Figure 1).

The AUC of Hb/PLT (Before-Treatment) Ratio in ITP Diagnosis.
Diagnostic Accuracy of Hematological Inflammatory Parameters in ITP Diagnosis (Before-Treatment).
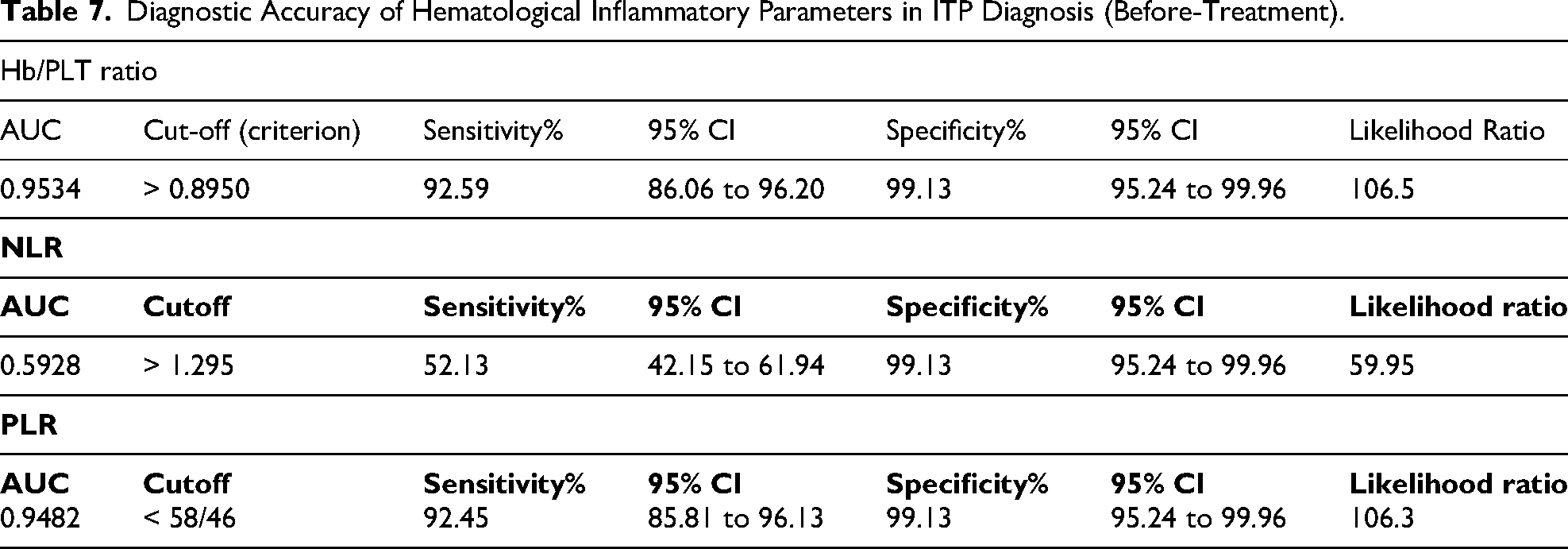
NLRa, with a cut-off >1.295, showed lower sensitivity at 52.13% (95% CI: 42.15-61.94%) but maintained high specificity at 99.13% (95% CI: 95.24-99.96%) (AUC = 0.5928, P = .0210) (Table 7, Figure 2).
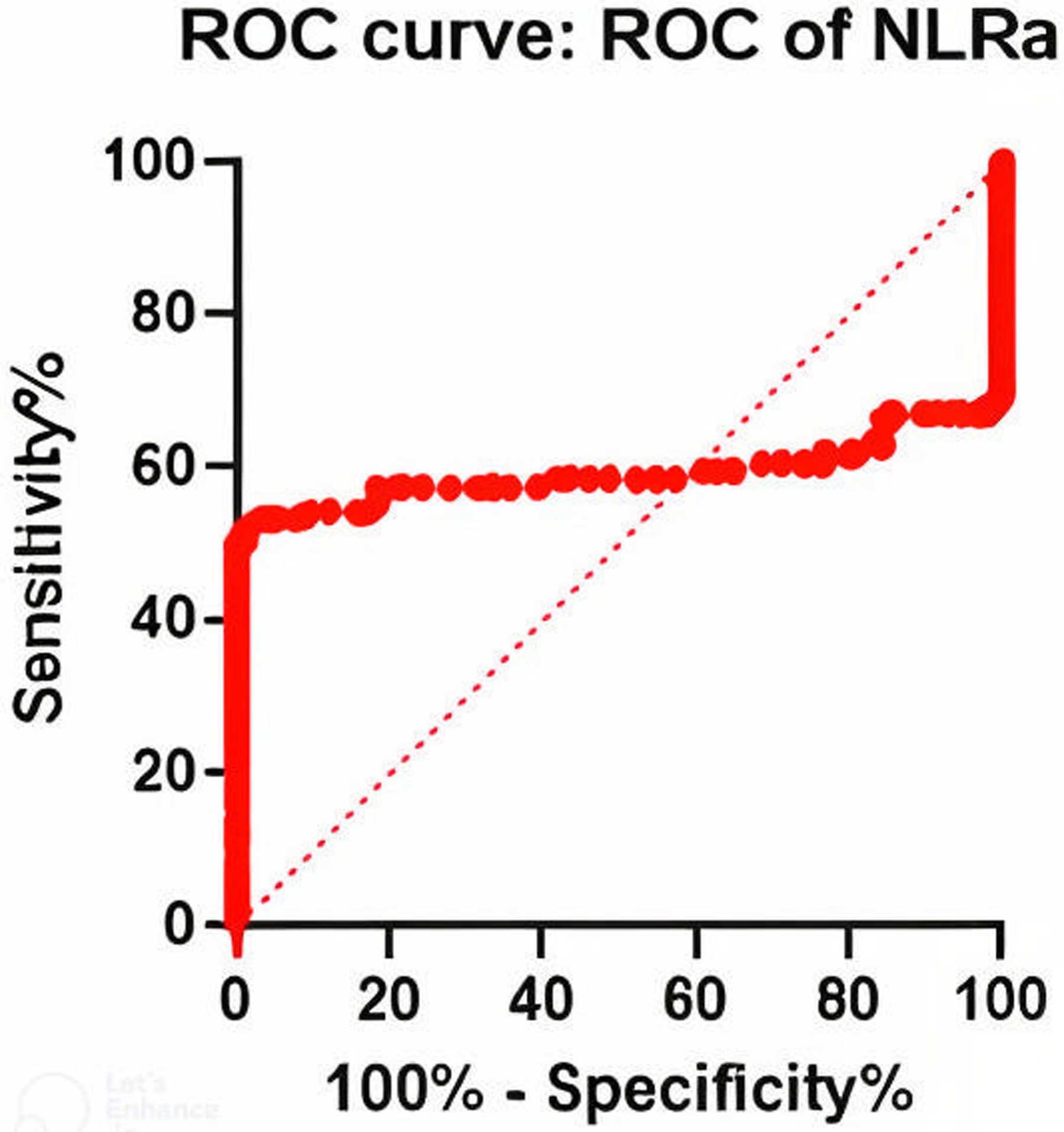
The AUC of NLR (Before-Treatment) in ITP Diagnosis.
PLRa, with a cut-off <58.46, also exhibited excellent diagnostic accuracy, with sensitivity of 92.45% (95% CI: 85.81-96.13%) and specificity of 99.13% (95% CI: 95.24-99.96%), and an AUC of 0.9482 (P < .001) (Table 7, Figure 3).

The AUC of PLR (Before-Treatment) in ITP Diagnosis.
Overall, the Hb/PLTa ratio and PLRa demonstrated excellent diagnostic utility (AUC > 0.90), while NLRa showed moderate performance (AUC ∼0.59). Nonetheless, all three indices serve as valuable, accessible biomarkers to aid in the diagnosis of ITP.
Discussion
ITP is a heterogeneous autoimmune disorder that occurs more frequently in children and older adults. The disease has a complex pathophysiology involving dysregulation of both humoral and cellular immune responses. Thrombocytopenia in ITP is primarily caused by the production of autoantibodies directed against platelet surface glycoproteins, particularly GPIb/IX and GPIIb/IIIa, which are also expressed on megakaryocytes. Once sensitized with these autoantibodies, platelets are targeted and cleared by the reticuloendothelial system, leading to a reduction in circulating platelet levels. Although the precise triggers for autoantibody production remain unclear, recent research suggests that T-cell-mediated immune responses contribute significantly to their generation. In addition, various inflammatory processes and cytokines especially interleukin-18 (IL-18) have been implicated in the initiation and amplification of this autoimmune activity.
In recent years, the diagnostic significance of inflammatory cell indices in various inflammatory and systemic diseases has been increasingly recognized. Indices such as the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and hemoglobin-to-platelet ratio (HPR) have emerged as accessible and cost-effective markers of systemic inflammation. These parameters reflect underlying immune activity and are now widely used in clinical settings. For example, a study by Xiankun Wang et al demonstrated that elevated NLR levels in patients with severe fever with thrombocytopenia syndrome (SFTS) are associated with an increased risk of mortality and reduced overall survival, highlighting NLR's prognostic value in disease management. 15 Similarly, research by Ceyhun Aksakal et al found that elevated PLR levels in patients with epistaxis suggest a role for inflammation in its pathogenesis. 9 Additionally, Guanghao Qin et al reported that both PLR and NLR can aid in the diagnosis of retinal artery and vein occlusion, reinforcing their diagnostic utility in vascular inflammatory conditions. 16 Furthermore, a study by Fauzia Imtiaz et al identified NLR as a reliable index for assessing systemic inflammation, noting its elevation in patients with chronic conditions such as hypertension and diabetes. 17 Regarding the inflammatory parameter HPR, a decrease in platelet count in patients with ITP purpura directly increases the ratio of this parameter. Usually, in the early stages, hemoglobin levels usually remain within the normal range, chronic or severe bleeding, especially mucosal and cutaneous bleeding such as petechiae, purpura, and ecchymoses, can cause a decrease in hemoglobin levels and, as a result, affect the value of the HPR ratio. Recent studies suggest that HPR, as a composite index, reflects a complex balance between platelet destruction, bleeding severity, and the patient's systemic inflammatory status. As a low-cost and available biomarker, this parameter can play an effective role in assessing disease severity and predicting treatment response in ITP patients. A significant increase in HPR in patients with bleeding symptoms is a sign of effective platelet reduction and indicates the need for more careful monitoring of these patients.18,19
The diagnostic value of inflammatory indices such as HPR, NLR, and PLR has been established in a wide range of clinical conditions, including autoimmune rheumatic diseases (ARDs), hepatitis C virus infection, COVID-19, pre-eclampsia, colorectal cancer, myocardial infarction, pneumonia, and various malignancies. Although data specifically addressing the correlation of these indices with clinical outcomes in patients with ITP remain limited, their role as surrogate markers of immune system activity is gaining attention. Given their accessibility, low cost, and ease of measurement, these parameters offer practical advantages over more complex and expensive methods such as cytokine profiling. As such, monitoring changes in NLR, PLR, and HPR may serve as a valuable adjunct in the diagnosis, prognosis, and follow-up of ITP patients.
The present study demonstrated significant differences in average cell counts and other hematological parameters between ITP patients and controls. Specifically, white blood cells WBC and neutrophil counts were lower in ITP patients, whereas lymphocyte count and HPR were elevated compared to the control group. These findings contrast with those of Mohammad Hossein Ahmadi et al, who reported increased WBC and neutrophil counts in ITP patients. However, consistent with our results, their study also found elevated HPR levels in patients relative to healthy individuals. 18 Furthermore, our findings differ from those of Ibrahim Ahmed et al, who suggested that lower lymphocyte counts may serve as a predictive marker for ITP. 20 Similarly, the study by Michael D. Deel et al indicated that lymphopenia is a strong predictor of ITP. Nevertheless, in agreement with our data, they also observed reduced WBC counts among affected individuals. 21
Furthermore, in contrast to our findings, the study by R. Zahorec et al on inflammatory diseases reported elevated neutrophil counts and reduced lymphocyte counts, which they attributed to activation of the innate immune response under inflammatory conditions. Similarly, other studies have indicated that in ITP patients, increased neutrophil levels and decreased lymphocyte levels may correlate with a poorer prognosis. 22
In our study, laboratory parameters such as hemoglobin, red blood cell RBC count, and hematocrit (HCT) were significantly lower in ITP patients compared to the control group. These results are consistent with those of Mohammad Hossein Ahmadi et al, who also observed reduced RBC, HCT, and hemoglobin levels in ITP patients relative to healthy individuals. 18
Following clinical examination of the ITP group, our study found that the most common clinical symptoms were ecchymosis, petechiae, and bruising. This finding aligns with the study by Jyotiranjan Champatiray et al, which identified petechiae as a key diagnostic feature, with additional presentations including gastrointestinal bleeding, hematuria, and menorrhagia. 1 Similarly, the study by Mohammad Hossein Ahmadi et al reported that petechiae and ecchymosis were more prevalent than other clinical manifestations in ITP patients. 18
In our study, no significant relationship was found between clinical symptoms and the length of hospitalization. However, this contrasts with the findings of Mohammad Hossein Ahmadi et al, who reported that fever in ITP patients may significantly correlate with extended hospital stays. 18 Supporting this, Ruopeng An et al demonstrated a significant association between clinical symptoms such as sepsis and bleeding and longer hospitalization durations. 23 Additionally, Neel S. Bhatt et al observed that bleeding was associated with prolonged hospital stays in ITP patients. 24
Our study demonstrated that the inflammatory parameters NLR and HPR increased significantly in ITP patients presenting with bleeding. In patients with bruising, HPR levels were elevated, whereas PLR decreased. Conversely, patients with petechiae exhibited decreased levels of both NLR and PLR. These findings are consistent with the study by Mohammad Hossein Ahmadi et al, which reported reduced platelet counts in ITP patients exhibiting petechial manifestations, followed by a corresponding decline in PLR. 18 Due to the scarcity of studies examining other clinical manifestations and their associations with inflammatory markers, further comparisons were not possible.
Furthermore, the current study found no significant relationship between treatment strategies and the length of hospitalization. This observation highlights a gap in the existing literature, as few studies have directly explored this relationship. Supporting this, Lawrence Rice et al acknowledged that the impact of pharmacologic interventions on hospitalization duration in ITP patients remains under investigation. 25
Among therapeutic agents, glucocorticoids such as Cortone remain the first-line treatment for ITP patients. Our study found that patients receiving IVIg exhibited lower baseline NLRb, whereas those treated with corticosteroids showed higher NLRb values. Additionally, the mean baseline PLTb was significantly higher in patients administered prednisolone. These findings align with the study by Jyotiranjan Champatiray and colleagues, which also reported a decrease in platelet counts following steroid therapy. 1
Patients with ITP can be treated with medications or surgery, such as splenectomy. Splenectomy is an invasive procedure used to manage ITP, which can effectively increase platelet counts and reduce bleeding. However, it carries risks including infections with encapsulated bacteria and thrombosis. Splenectomy typically results in increased platelet and leukocyte counts, leading to a decrease in HPR and an increase in NLR. Due to the potential for long-term complications, splenectomy is generally reserved for select cases.
In the present study, no significant association was found between splenectomy and the length of hospitalization. Similarly, Antoine Finianos and colleagues reported no significant change in hospital stay duration following splenectomy. 26 Conversely, Soames Boyle et al found that splenectomy was associated with increased mortality and longer hospital stays. 27 Additional studies by Ruopeng An, Michael D. Tarantino, and Mark D. Danese also reported longer hospitalization periods in splenectomy patients.23,28,29
Regarding inflammatory parameters, our study observed no significant changes in HPR, NLR, or PLR after splenectomy. In contrast, Arife Simsek et al demonstrated that platelet counts increased while NLR and PLR decreased following splenectomy. 30 Similarly, Mohammad Hossein Ahmadi et al found that splenectomy patients had significantly elevated platelet counts and reduced HPR levels. 18 Shruti Chaturvedi et al also reported that splenectomy is associated with increased platelets and decreased HPR. 31
According to the present study, the shortest and longest hospitalization durations for ITP patients occurred in winter and spring, respectively. This finding aligns with the study by Guillaume Moulis et al, which reported that the majority of hospitalizations (39%) occurred in spring. 32 Seasonal variations in hospitalization may be influenced by the increased incidence of viral and respiratory infections during spring and winter. However, no significant differences were observed in the incidence of ITP or the laboratory parameters across different seasons. It is important to note that only a limited number of studies have explored the relationship between seasonal variation and ITP clinical or laboratory outcomes.
In our study, analysis of the inflammatory indices NLR, PLR, and HPR revealed that average NLR and HPR levels were elevated in ITP patients compared to controls, while PLR levels were decreased. Similarly, Mohammad Hossein Ahmadi et al reported increased NLR and HPR levels and a rise in PLR in ITP patients relative to healthy subjects. 18 Contrarily, Zhen-yu Song et al found higher PLR levels in ITP patients compared to controls. 33 Additionally, Aisha Arshad et al demonstrated that an increase in NLR and decrease in PLR could serve as predictors of prognosis, treatment response, and disease severity in ITP patients. 34 Consistent with our findings, Inas Abdelmoaty Mohamed and colleagues showed that a decrease in PLR may indicate ITP or disease recurrence. 5
In the design of this study, considering the potential influence of confounding factors on hematological inflammatory parameters such as the HPR, NLR, and PLR, efforts were made to minimize the effect of unwanted variables through meticulous screening of clinical histories, laboratory examinations, and comprehensive review of patient records. Accordingly, patients with a history of other hematological disorders, anemia due to unrelated causes (iron deficiency, vitamin B12 or folate deficiency), active infections, chronic inflammatory diseases, and coexisting metabolic or nutritional conditions were identified and excluded from the study when applicable. Additionally, detailed information regarding the use of influencing medications (such as corticosteroids, immunosuppressants) and relevant surgical interventions (such as splenectomy) was collected and considered in the data analysis.
Moreover, differences in age, sex, disease severity, and duration of ITP among the patient and control groups were controlled as much as possible to ensure greater homogeneity between groups. Through these measures, the findings predominantly reflect changes related to the autoimmune pathophysiology of ITP and its associated inflammatory responses, with the impact of confounding factors considerably reduced.
Nonetheless, given the cross-sectional nature of the study and inherent limitations in documenting all underlying asymptomatic conditions and disorders, the possibility of residual confounding effects cannot be entirely excluded and should be taken into account when interpreting the overall results. Considering the controls implemented, the results of this study are reliable and valid, and their value in elucidating the role of hematological inflammatory parameters in the diagnosis and monitoring of ITP patients is highlighted. It is recommended that future studies with larger populations and even more rigorous control of confounding variables be conducted to enhance the inferential power of the findings.
Conclusion
This study demonstrated that NLR and HPR were elevated, whereas PLR was decreased in patients with ITP. ROC curve analysis indicated that PLR and HPR possess high diagnostic value for identifying ITP patients, while the discriminative ability of NLR was relatively limited. Accordingly, inflammatory parameters—particularly PLR and HPR—when interpreted in conjunction with clinical manifestations such as petechiae, ecchymosis, and bruising, as well as hematologic indices including WBC, RBC, neutrophil and lymphocyte counts, hemoglobin, and hematocrit levels, may serve as valuable adjunctive tools in the diagnosis and management of ITP. Nevertheless, NLR should be interpreted with caution and used alongside other markers to improve diagnostic accuracy. Further investigations with larger study populations and extended follow-up periods are warranted to confirm and refine these findings.
Limitations and Strengths
One of the main limitations of this study is its retrospective design, which may introduce biases in data collection and evaluation. Additionally, selecting patients and controls from a single medical center increases the possibility of selection bias, potentially limiting the generalizability of the findings to broader populations. Moreover, the lack of longitudinal data and long-term follow-up on disease progression and treatment response hinders a more comprehensive understanding of the clinical course and prognosis of ITP patients.
However, the strengths of this study include the use of readily accessible, cost-effective, and easily measurable inflammatory indices such as NLR, PLR, and HPR, which can serve as valuable supportive tools in the diagnosis and management of ITP when combined with clinical manifestations and hematologic parameters. The study also benefits from careful control of confounding factors through meticulous screening and exclusion of patients with coexisting diseases or conditions, enhancing the validity and reliability of the results.
Therefore, future studies with prospective designs, larger sample sizes, and extended follow-up periods are warranted to validate and generalize these findings.
Footnotes
Acknowledgments
We would like to thank all of our colleagues who have helped us with this study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Zahedan University of Medical Sciences, (grant number 10865).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
