Abstract
The study aims to evaluate the prognosis and risk factors of sepsis-associated thrombocytopenia (SAT) among patients with coagulopathy, and to provide evidence of the relationship between adverse outcomes and potential risks. Patients with sepsis-associated coagulopathy were included in the study from January 2014 to December 2022. The primary outcome was sepsis-associated thrombocytopenia (platelet count less than 100 *109/L), which was evaluated by logistic regression models adjusted for demographic characteristics and comorbidities. Among patients in the SAT group, 54% developed severe SAT, while 16% of these patients recovered from thrombocytopenia. The in-hospital mortality rate was significantly higher in the SAT group compared to the non-SAT group (31% in SAT group vs 23.9% in non-SAT group, p = 0.029). Even after adjusting for age, gender, Charlson comorbidity, white blood cell, and Sequential Organ Failure Assessment score, the differences in mortality rate persisted (Odds Ratio 0.72, [95% Confidence Interval 0.52–0.92]). Correlation analyses revealed that prothrombin time (r = 0.08, p = 0.50), international normalized ratio (r = 0.08, p = 0.42), prothrombin activity (r = −0.06, p > 0.999), D-dimer (r = −0.02, p > 0.999), and inflammatory parameters such as C-reactive protein (r = −0.11, p = 0.37) were not significantly correlated with platelet counts. According to subgroup analyses, patients with lung infection complicated by SAT had slightly higher mortality (OR 0.66, [95% CI, 0.46 to 0.94]). Sepsis-associated coagulopathy indicates a subset of critical ill patients, with those experiencing thrombocytopenia at greater risk for in-hospital death compared to those without it.
Keywords
Introduction
Sepsis-associated thrombocytopenia (SAT) is a common complication of sepsis patients, with an incidence rate ranging from 30% to 70%.1,2 There are varous studies suggested that thrombocytopenia in critically ill patients developed a poor prognosis.3,4 The prognosis of sepsis patients with concomitant thrombocytopenia is worse than for those without it. 1 The negative impacts on prognosis include increased bleeding, transfusion requirements, application of life-support, longer length of hospital stay, and higher mortality rate.
However, though the importance of SAT has been proved in previous studies, the risk factors are still controversial. Low platelet count is a typical change observed while the relationship between sepsis and thrombocytopenia has not yet been clarified. According to some studies, enhanced platelet consumption, reduced platelet production probably attributes to systemic inflammation response with severe infections, leading to SAT.5,6 Besides of the inflammation factors, male sex, and concommitant liver dysfunction is related to the development of thrombocytopenia during intensive care unit (ICU) stay.5,7
Bacterial endotoxins, especially in Gram-negative infections, are known to strongly activate both the coagulation and fibrinolytic systems. 8 Previous research has highlighted the respiratory tract as the most common site of infection that leads to sepsis and septic disseminated intravascular coagulopathy (DIC).9‐12 The distinct inflammatory reactions triggered by severe pneumonia compared to bacteremia and their respective contributions to coagulopathy during sepsis remain unexplored. Whether the different infection sites influence SAT remains to be proved.
The current study aimed to elucidate the prognosis and risk factors of SAT, provide evidence to the relationship between adverse outcomes and the potential risks.
Methods
This study was appoved by the the the Ethics Committee of Guangdong Provincial People's Hospital (NO. S2024-700-02). Data were gathered from ICU patients diagnosed with sepsis or suspected sepsis in Guangdong Provincial Peoples’ Hospital and Yantai Affiliated Hospital of Binzhou Medical University from January 2014 to December 2022. The data included patient demographics, ICU admission specifics, hospital stay duration, laboratory tests, blood transfusions, vasopressor use, and Sequential Organ Failure Assessment (SOFA) scores to assess severity.
The inclusion criteria were: 1) confirmed or suspected infection along with organ dysfunction (SOFA score ≥ 2); 2) evidence of coagulation dysfunction. Exclusion criteria included age <18 years, pregnancy, diagnosis of any active malignant disease or liver cirrhosis, receipt of chemotherapy within 3 months, and insufficient coagulation test data. Sepsis-associated thrombocytopenia was defined as a platelet count less than 100 *109/L within the initial 72 h (h) of ICU admission. Severe SAT was further specified as a platelet count below 50 *109/L, determined by the lowest count recorded within the initial 72 h post-ICU admission. Disseminated intravascular coagulation (DIC) diagnosis followed the International Society for Thrombosis and Hemostasis (ISTH) criteria, with a score of ≥ 5 points confirming DIC presence.
Descriptive statistics were utilized to summarize continuous variables, presented as means with standard deviations (SD), while categorical variables were depicted as frequencies and percentages. Quantitative data were evaluated using Student's t-test. Chi-squared analysis was employed to calculate p-values for all categorical data, and Fisher's Exact Test was used for relative risks. Pearson correlation was conducted to explore the relationship between in-hospital data and SAT. Furthermore, a logistics regression model was utilized to identify risk factors for mortality and SAT. All statistical analyses were performed using R 4.2.2 (The R Foundation for Statistical Computing, Vienna, Austria). A two-sided p-value of less than 0.05 was deemed statistically significant.
Results
The study's workflow is illustrated in Figure 1 and the baseline characteristics are presented in Table 1. The mean age (p = 0.196), male gender (p = 0.730), ICU stay (p = 0.425), and SOFA score (p = 0.143) were comparable between the SAT and non-SAT groups. Patients in SAT group tended to have higher costs (p = 0.507) and shorter in-hospital stays (p = 0.050) compared to those in the non-SAT group. Furthermore, individuals with blood group O were less likely to experience SAT than those with other blood types (p < 0.001). The SAT group were slightly older (58 [IQR, 43–69] years vs 60 [IQR, 45–70] years, p = 0.196) and had a higher prevalence of comorbidities (Charlson comorbidity index 1–5, 69.78% vs 53.51%, p < 0.001), compared to the non-SAT group.
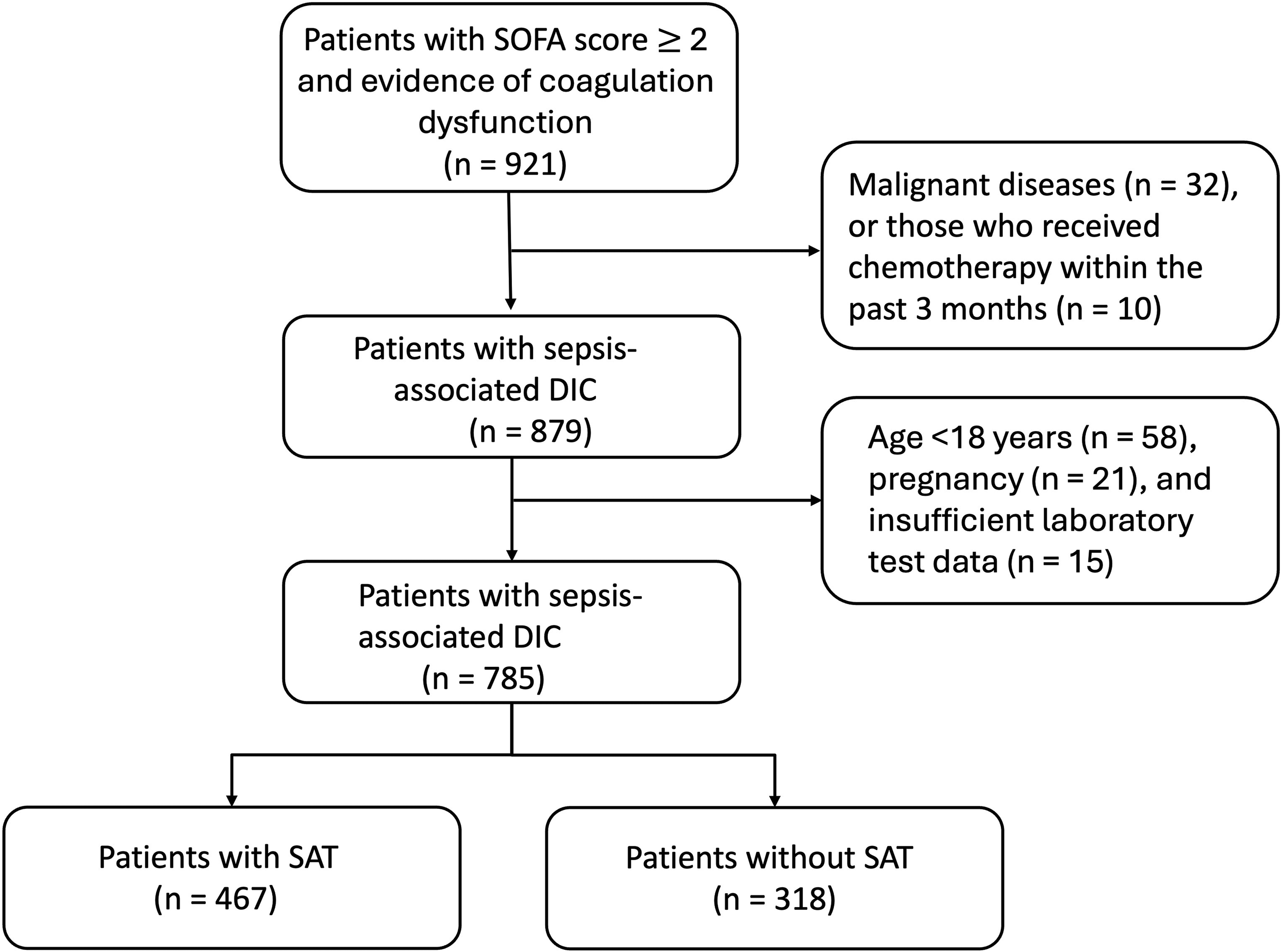
Flow-chart of the study.
Patient Demographics and Baseline Characteristics.
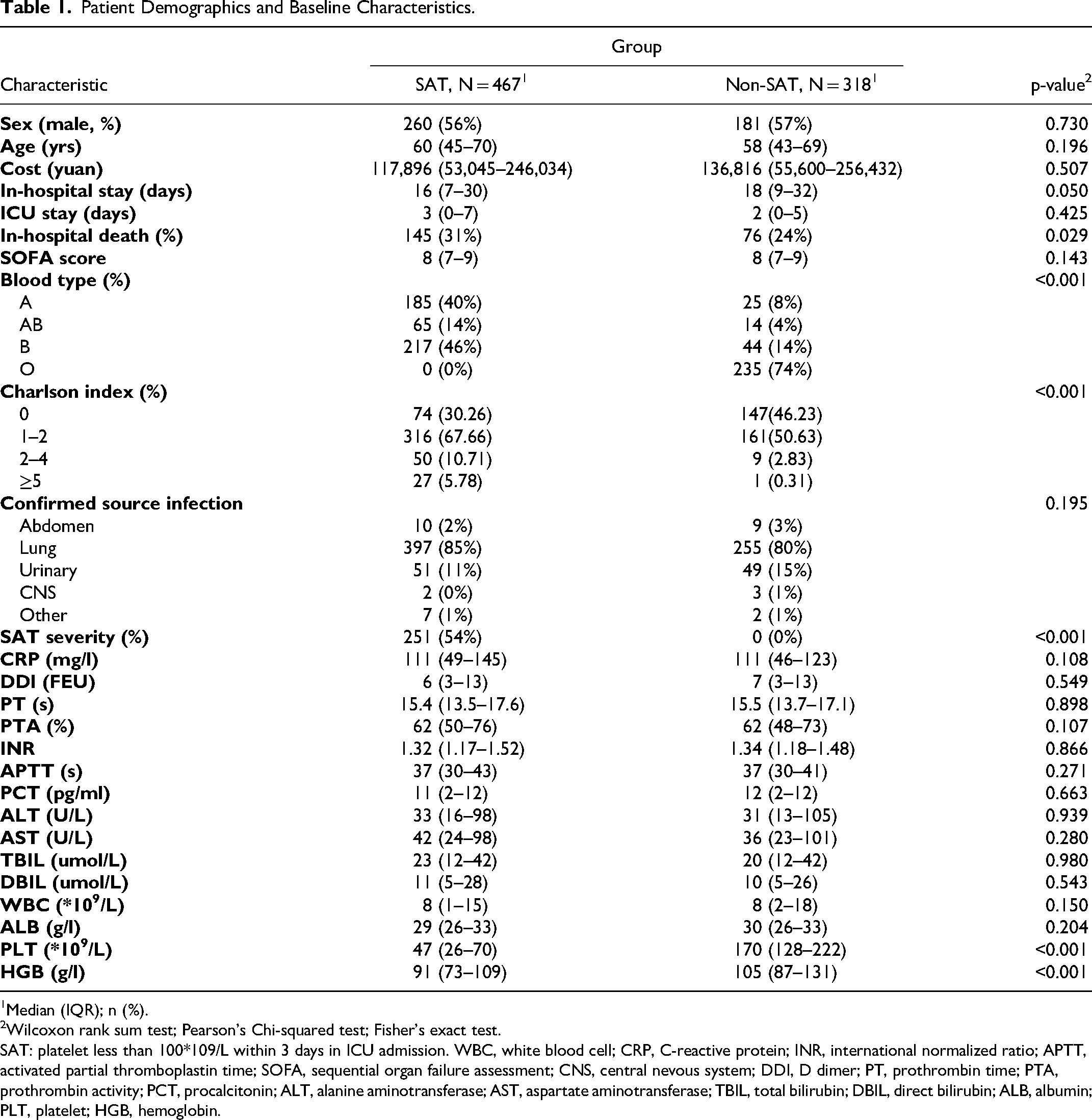
Median (IQR); n (%).
Wilcoxon rank sum test; Pearson's Chi-squared test; Fisher's exact test.
SAT: platelet less than 100*109/L within 3 days in ICU admission. WBC, white blood cell; CRP, C-reactive protein; INR, international normalized ratio; APTT, activated partial thromboplastin time; SOFA, sequential organ failure assessment; CNS, central nevous system; DDI, D dimer; PT, prothrombin time; PTA, prothrombin activity; PCT, procalcitonin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; TBIL, total bilirubin; DBIL, direct bilirubin; ALB, albumin; PLT, platelet; HGB, hemoglobin.
Among patients in the SAT group, 54% developed severe SAT, while 16% recovered from thrombocytopenia. The in-hospital mortality rate was significantly higher in the SAT group than in the non-SAT group (31% vs 23.9%, p = 0.029, Table 2). Even after adjusted for age, gender, Charlson comorbidity, WBC, and SOFA score, the differences in mortality rate persisted (OR 0.72, [95% CI 0.52–0.92], Figure 2). However, there were no significant differences between the SAT and non-SAT groups in coagulation parameters, including prothrombin time (PT), prothrombin activity (PTA), activated partial thromboplastin time (APTT), D-dimer, and international normalized ratio (INR). Similarly, the white blood cell (WBC) count, procalcitonin (PCT), C-rective protein (CRP), alanine transaminase (ALT) and albumin did not differ significantly between the two groups.
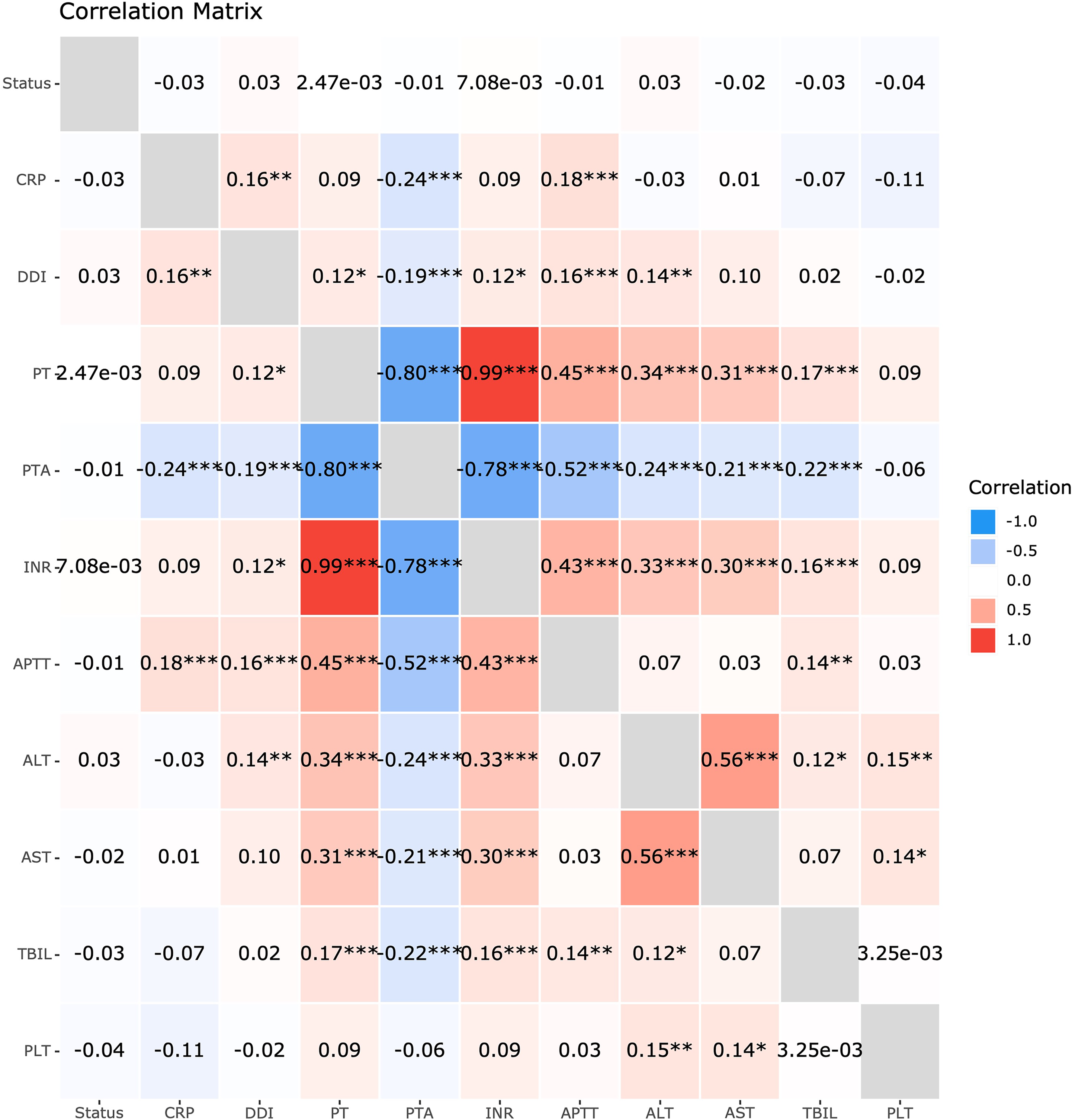
Correlation among the lab parameters and platelet counts. CRP, C-reactive protein; INR, international normalized ratio; APTT, activated partial thromboplastin time; DDI, D dimer; PT, prothrombin time; PTA, prothrombin activity; ALT, alanine aminotransferase; AST, aspartate aminotransferase; TBIL, total bilirubin; DBIL, direct bilirubin; PLT, platelet.
Logistic Regression for in-Hospital Mortality in SAT Group.
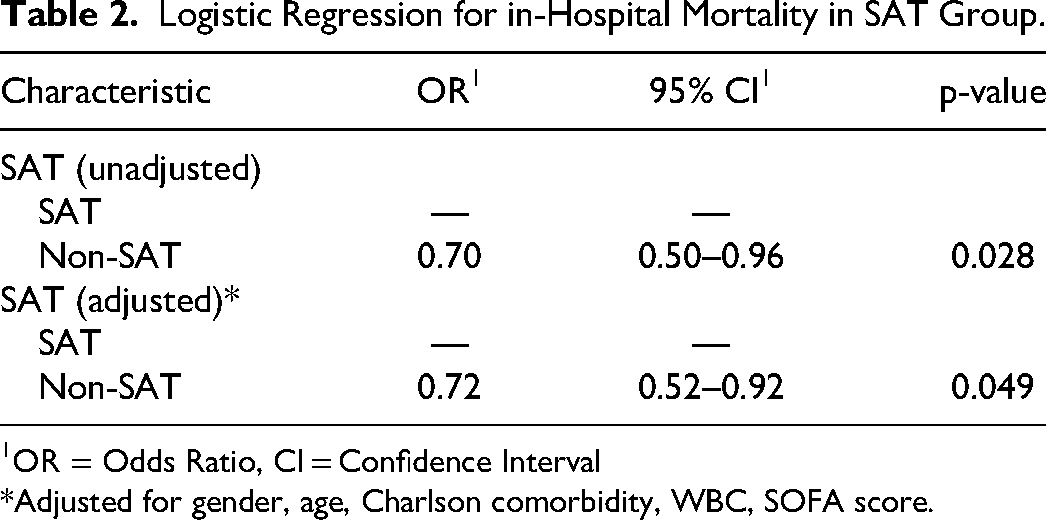
OR = Odds Ratio, CI = Confidence Interval
*Adjusted for gender, age, Charlson comorbidity, WBC, SOFA score.
To identify robust predictors of SAT, we conducted univariate and multivariate logistic regression analyses. Male sex, age, SOFA score, CRP, and INR were not significantly related to SAT in either univariable or multivariable logistic regression. A Charlson comorbidity index of 2–4 (OR 1.15 [95% CI, 1.00 to 1.45]) and greater than 5 (OR 1.32 [95% CI, 1.05 to 2.08]) were significantly related to SAT in multivariable logistic regression. Infection in the central nevous system (CNS) (OR 1.59 [95% CI, 1.16 to 2.19] increased the risk of SAT onset compared to lung infection, although there are not significant differences among different infection sites in logistic regression (Table 3).
Univariate and Multivariate Analysis of Influencing Factors for Sepsis-Associated Thrombocytopenia (SAT).
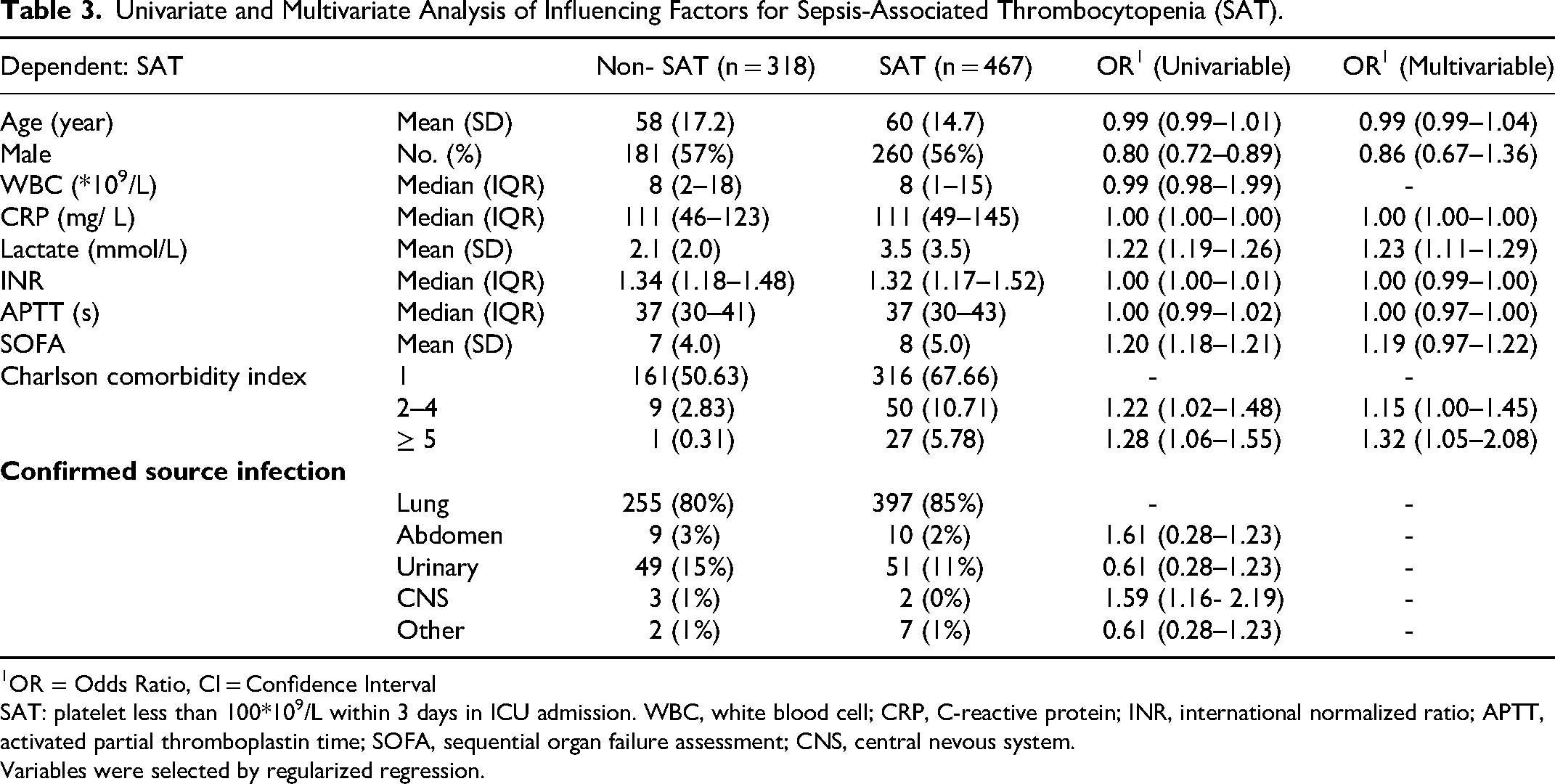
OR = Odds Ratio, CI = Confidence Interval
SAT: platelet less than 100*109/L within 3 days in ICU admission. WBC, white blood cell; CRP, C-reactive protein; INR, international normalized ratio; APTT, activated partial thromboplastin time; SOFA, sequential organ failure assessment; CNS, central nevous system.
Variables were selected by regularized regression.
Correlation analysis revealed that PT (r = 0.08, p = 0.50), INR (r = 0.08, p = 0.42), PTA (r = −0.06, p > 0.999), D-dimer (r = −0.02, p > 0.999), and inflammatory parameters such as CRP (r = −0.11, p = 0.37) were not significantly correlated with platelet counts (Figure 2). Alanine aminotransferase (r = 0.15, p = 0.001) and aspartate aminotransferase (r = 0.14, p = 0.036) were strongly correlated with platelet counts and the severity of thrombocytopenia. However, APTT (p > 0.999), PTA (p > 0.999), D-dimer (p > 0.999), and TBIL (p > 0.999) level show no significant correlation with in-hospital mortality.
Subgroup analyses were performed according to several baseline characteristics. The effect of thrombocytopenia on in-hospital mortality varied according to baseling age subgroup. For patients aged 60 or greater, the odds ratio was 0.54 (95% CI, 0.34 to 0.84). Among patients with a SOFA score of 10 or greater, the odds ratio for in-hospital mortality was 0.67 (95% CI, 0.43 to 1.01), whereas among patients with a SOFA score less than 10 the odds ratio was 0.74 (95% CI, 0.44 to 1.22). Although there was no significant difference in SAT effect among the confirmed source infection subgroups, patients with lung infection complicated by SAT had higher mortality (HR 0.66, [95% CI, 0.46 to 0.94]). There was no evidence of heterogeneity of SAT effects in the other subgroups (Figure 3).
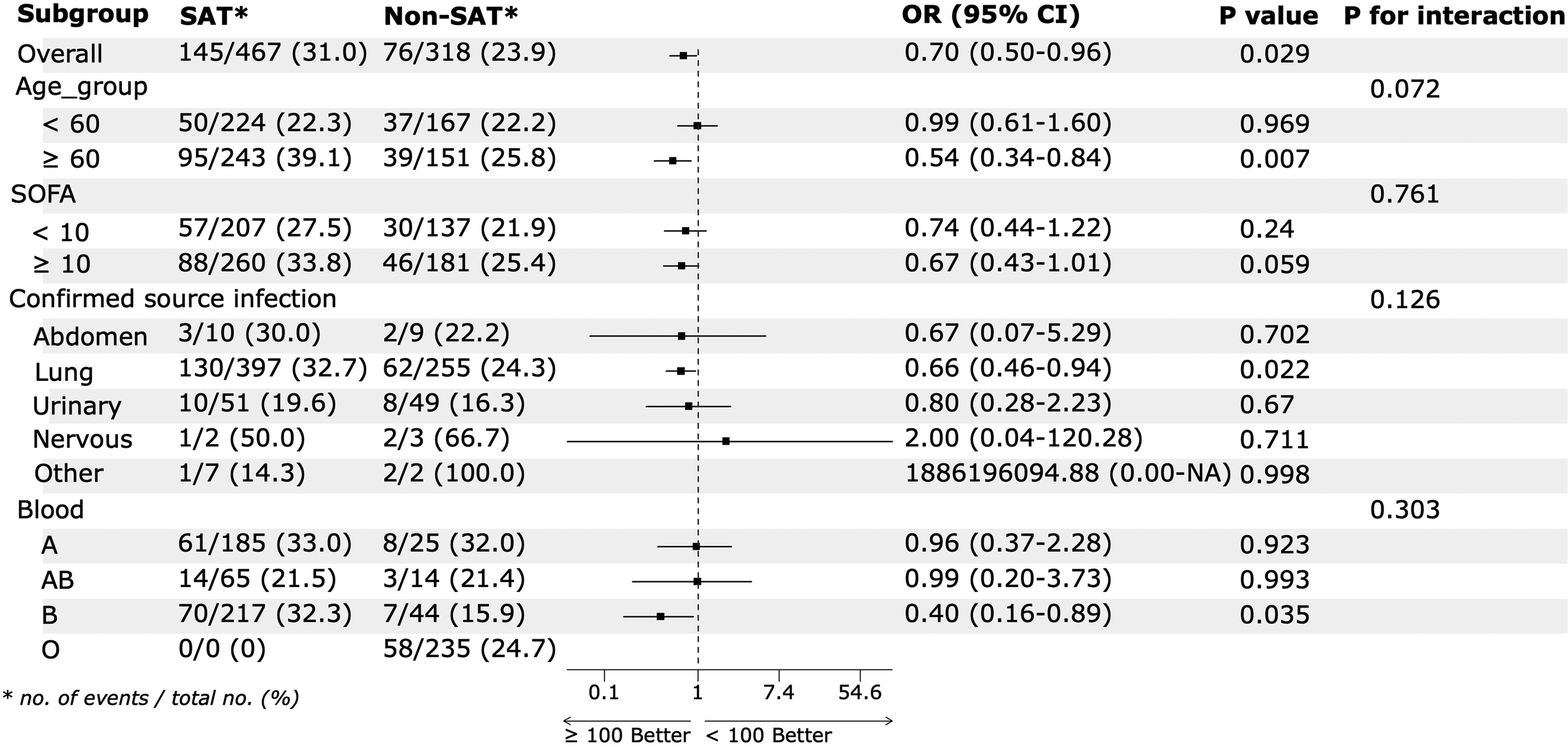
Subgroup analyses for the in-hospital mortality among patients with sepsis-associated thrombocytopenia. SOFA, sequential organ failure assessment.
Discussion
In the current study, we found that the prevalence of SAT among patients with sepsis-associated coagulation dysfunction was 59.5%, with severe SAT occurring in 54% of these cases. Our study revealed that thrombocytopenia was not significantly related to certain coagulation parameters, such as PT and INR. Although the confirmed source infection subgroup did not alter the prognosis, patients with pneumonia in the SAT group had significant higher in-hospital mortality rate.
Thrombocytopenia is a common event in critically ill patients with sepsis, with reported incidence ranging from 20% to 50%. 13 Thrombocytopenia and inflammation are closely intertwined, with a bi-directional positive feedback relationship.13,14 Numerous studies have shown that thrombocytopenia in sepsis patients is linked to poor prognosis, including prolonged hospital stays, lower survival rates, shock, bleeding, and acute kidney injury.15‐18 Thrombocytopenia at ICU admission is common, and the nadir platelet count likely occurs at day 3, according to previous studies.19‐21 In our study, we identified that the lower platelet counts were associated with higher mortality rates among patients with sepsis-induced DIC. The mortality rate in the severe SAT group was significantly higher than that in non-SAT group. Based on these results, we carefully suggest that thrombocytopenia or severe thrombocytopenia leads to poorer prognosis comparing to patients with coagulation dysfunction, as indicated by abnormalities of coagulation parameters.
A comprehensive view of the coagulation progress involves the contribution of platelets, fibrinogen function, clot firmness, and the status of clot lysis. 22 Coagulopathy and thrombocytopenia are critical hemostatic disorders that frequently co-occur, complicating the clinical management of affected patients. Some studies have indicated the intricate relationship between coagulopathy and thrombocytopenia by examining the pathophysiology, diagnostic criteria, and therapeutic approaches.23‐26 According to previous research, thrombocytopenia is a criteria of ISTH overt DIC, while platelet activation results in systemic thrombosis. 27 Thus, changes in platelet count can influence the scoring of DIC, which encompasses deficiencies in coagulation factors and excessive fibrinolysis. The common etiologies between coagulopathy and thrombocytopenia make distinguishing or differentiating diagnoses more difficult, as they complicate the interpretation of coagulation tests and mask underlying disorders.28,29 In the sepsis cohort, coagulation and endothelial cell activation are triggered by the systemic inflammatory response, likely involving platelets. 30 The current study included cases diagnosed with sepsis-induced DIC and reported different prognosis for SAT and non-SAT among these DIC patients. The overall mortality rate was 28.2% (221/785), comparable with previous studies on sepsis-associated DIC.6,31 Efforts were made to identify patients at greater risk of coagulation dysfunction, including hypofibrinogenemia, bleeding, and thrombogenesis. In this cohort, no patient experienced major bleeding or thrombosis events.
Concomitant with pneumonia in this cohort indicated a higher risk of in-hospital mortality. Pneumonia is a common infection known to be associated with platelet activation, particularly via pneumococcus. 32 Previous study have shown that community-acquired pneumonia is associated with an increased thrombosis, with serum levels of endotoxins playing a role in enhanced thrombin generation. 33 Additionally, severe thrombocytopenia has been reported to impaired host defense during pnenumonia-derived sepsis. 30 The evidence from previous studies suggests a potential association between pneumonia and sepsis associated coagulopathy. T Tagami et al demonstrated that there might be little association between the use of recombinant human soluble thrombomodulin and mortality in severe pneumonia patients with sepsis-associated DIC. Our study is consistent with previous research, showing that pneumonia complicating with SAT had a higher mortality rate. However, within the sepsis-associated DIC cohort, the infection site was not a significant risk factor associated with SAT.
Limitation
There are several limitations in the current study. It is subject to the common limitations of a retrospective study, such as potential selection bias. The cohort included patients with sepsis-associated DIC, who have a relatively higher mortality rate. Critical ill patient might not have been able to undergo all the necessary coagulation tests or bacterial cultures for potential infection sites due to the rapid progression of their clinical condition. Furthermore, the cohort focused on the poor prognosis of thrombocytopenia in DIC patients without accounting for the blood transfusion or anti-coagulation drugs.
Conclusions
Sepsis-associated DIC indicated a subset of critical ill patients, with those experiencing thrombocytopenia at greater risk for in-hospital death compared to those without it. Although the confirmed source infection site was not the risk factor for prognoses, patients with pneumonia in the SAT group had significant higher mortality rate.
Footnotes
Data Sharing Section
The datasets used or analysed during the current study are available from the corresponding author on reasonable request.
Competing Interests
None.
IRB
This retrospective study was approved by the Ethics Committee of Guangdong Provincial People's Hospital (NO.S2024-700-02).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China initiated supporting funds for the project (No. 8230020134) and Science and Technology Planning Project of Guangdong Province (2020B1111170011).
