Abstract
Data describing physicians’ and patients’ perspectives towards immune thrombocytopenia (ITP) management and impact of disease in Iran are limited. This ITP World Impact Survey was conducted between October 2019 and October 2020. Of the 114 patients included in the survey, 17 were aged ≤18 years. Forty-seven physicians, including 22 pediatric hematologists, participated in the survey. Fatigue and anxiety around stable platelet counts were frequent patient-reported symptoms at diagnosis and at survey completion. According to physicians, “watch-and-wait” was the preferred treatment option for mean (standard deviation) proportion of 50.1 (24.1) and 48.6 (21.8) of their adult and pediatric patients, respectively, following first diagnosis. Per adult and pediatric hematologists, the most prescribed treatments for newly diagnosed patients based on available answers were steroids (100%, n = 20/20; 89%, n = 16/18), respectively. Forty percent of adult (n = 10/25) and 38% of pediatric hematologists (n = 8/21) reported that ITP reduced patients’ quality of life. Energy levels (46%, n = 52/112) and ability to concentrate on everyday activities (42%, n = 47/113) were the most affected aspects of patients' lives. This I-WISh study in Iran underlined the negative impact of ITP on patients.
Keywords
Introduction
Primary immune thrombocytopenia (ITP) is an autoimmune disorder traditionally defined by a platelet count <100 × 103/µL affecting both the pediatric and the adult population. 1 Patients with ITP may have no symptoms or present with minimal bruising; however, some patients experience serious bleeding caused by an impaired blood-clotting mechanism. 2 Primary ITP differs from secondary ITP, with the latter occurring in association with other disorders. 3
Management approaches vary based on disease duration, platelet count, patient and physician preferences, impact on quality-of-life (QoL) and accessibility to care. 4 ITP may have a sudden onset and follow an extended course in adult patients, whereas in children, onset occurs following a viral infection or immunization, with the majority of patients recovering spontaneously within few months; 2 thus, ITP management in this patient group in the absence of or with mild bleeding is often observation (watch-and-wait), regardless of platelet count. 5 In asymptomatic patients with a platelet count ≥30 × 103/µL, management with watch-and-wait is often adopted, whereas corticosteroids and/or intravenous immune globulin (IVIG), or Rho(D) immune globulin (anti-RhD) in steroid-resistant cases are widely used as first-line treatment in other patients. 5 In refractory or non-responsive ITP, second-line therapies include splenectomy or use of monoclonal antibodies against the CD20 antigen (anti-CD20) and thrombopoietin receptor agonists (TPO-RAs). 5 In Iran, physicians follow current guidelines from the American Society of Hematology 5 when treating patients with ITP and adapt therapy based on their judgment, patients’ needs, treatment access and affordability.
Despite recent advancements in the management of ITP, the impact of the disease and its treatments on the patient overall physical and emotional functioning is multifaceted and often underappreciated. Previous work has demonstrated that ITP impairs patients’ health-related QoL (HRQoL) in many respects and patients confront problems such as fatigue, effect of the disease on occupation, lifestyle activities, and family life.6, 7 The global ITP World Impact Survey (I-WISh) study was a large, international, cross-sectional survey of 1507 patients with ITP and 472 physicians aimed to evaluate patient and physician perceptions regarding ITP symptoms, impact on QoL and disease management across 13 countries. 8 The study revealed a detrimental impact of ITP on patients' HRQoL both directly for patients and from a physician's perspective, with patients reporting that ITP affected their energy levels, their capacity to exercise, and limited their ability to perform daily tasks. 9 From the patient's perspective, fatigue, along with anxiety around platelet counts, were two of the top symptoms patients would most like to be resolved 8 ; however, it remains unclear how to effectively approach these in clinical practice, underlining an important unmet medical need. In addition, while results from the global I-WISh survey indicated that both patients and physicians have a good understanding of the overall burden of ITP, 8 more can be done to refine patient-physician communication on treatment goals, preferences, and concerns to improve the overall patient experience with ITP management and limit disease burden.
Data describing physician and patient perspectives towards ITP management and impact of disease on patients' HRQoL in Iran remain limited. To understand which aspects of ITP are perceived as having the greatest impact on patients in Iran and explore potential differences between the adult and the pediatric population of patients, we conducted a local I-WISh survey. Here we present findings from the study and discuss differences and similarities with the results of the global I-WISh survey.
Methods
Study Design, Participants, and Survey Questionnaire
This was a local, exploratory, cross-sectional, I-WISh study conducted in Iran between October 2019 and October 2020, after completion of the Global I-WISh study, in the form of a research contract between the Clinical Trial Center of Tehran University of Medical Sciences and Novartis Pharmaceuticals, Iran. Both pediatric and adult patients with ITP and pediatric, and adult hematologists with experience in treating patients with ITP participated in the survey. To participate, hematologists had to have >3 patients with ITP as an active caseload and had to be responsible for their treatment decisions. Patients aged >6 years with a diagnosis of ITP were recruited. Caretakers had to complete the survey for patients aged <18 years. Patients with ITP who were hospitalized were excluded. Eligible patients were initially identified by participating physicians, but participation in the survey was voluntary. No patient access groups were involved in patient selection. Survey questionnaires used in the Global I-WISh study were translated into local language with the guidance of Novartis consultants and sent in an online format to the study participants after verbal and multimedia training. The patients’ questionnaire comprised six sections and collected information on demographic characteristics (seven questions), symptoms of ITP (four questions), HRQoL associated with ITP (12 questions), treatment received (17 questions), impact of ITP on job and economic status (15 questions), and patient-physician relationship (seven questions). The physicians' questionnaire also comprised six sections and collected information on demographic characteristics (two questions), diagnosis of ITP (seven questions), symptoms of ITP (five questions), HRQoL associated with ITP (11 questions), treatment patterns (13 questions), patient-physician relationship (four questions). In addition, the survey was contextualized to pediatric patients by removing the questions that were not applicable to this patient population. The ITP Life Quality Index (ILQI) questionnaire (10 questions) was used to identify the degree of impact that ITP had on patients' HRQoL, emotional health, social, personal, and work life (in the last month). The response options were “never”, “sometimes”, “more than half the time”, and “all of the time”. Patients who missed to answer any of these 10 questions were excluded from the ILQI analysis. The ILQI was initially developed by clinical experts in the field of ITP and the format of the items and response options was based on the Dermatology Quality of Life Index (DLQI).10, 11
Data Collection and Statistical Analysis
Data were collected between February 2020 and May 2020. Online surveys took approximately 40 min for respondents to complete. Fully deidentified respondent information was collated and aggregated by local fieldwork partners such that surveys were unlinked and anonymized. Data were analyzed using the IBM SPSS Statistics program, version 26 and STATA software, version 14. Analyses were presented by adult versus pediatric hematologists and by adult (>18 years) versus pediatric (≤18 years) patients. The aim of the I-WISh survey was to collect information from physicians and patients to elicit understanding of the disease impact of ITP in terms of symptoms, emotional and economic burden, impact on daily living and work productivity, and patient/physician perceptions on disease treatment and management. Therefore, as the study did not aim to confirm or reject pre-defined hypotheses, all statistical analyses were purely descriptive. For numeric variables, the respondent mean, median and range were reported. For categorical variables, the percentages and corresponding absolute counts with denominators equalling to the total number of answered items excluding missing values were reported.
Results
Baseline Characteristics
A total of 114 patients, including 17 patients aged ≤18 years, and 47 physicians, including 22 pediatric oncologists, participated to this local I-WISh survey. Patient characteristics are summarized in Table 1. Overall mean (SD) age was 38.2 (17.9) years, 68% were female, and the median (range) duration of ITP was 5.1 (0.8 − 21.6) years. Most adult and pediatric physicians were private hospital-based and qualified during the 1981 − 1993 period. Each physician reported a mean caseload at the time of survey completion of 36 patients for adult hematologists, and 45 patients for pediatric hematologists (Table 2). The majority of patients (65%; n = 74/113) reported their current health as “high”.
Patient Demographics and Social Characteristics.
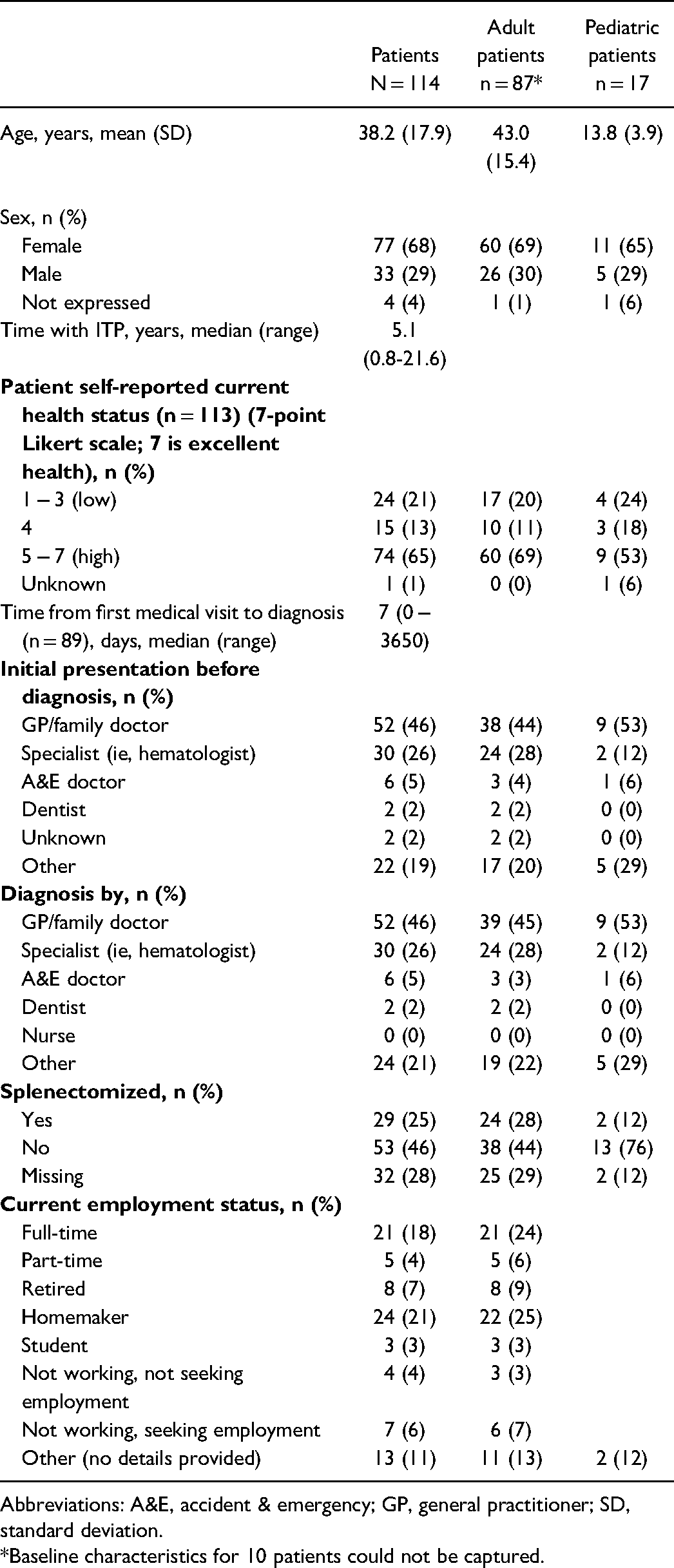
Abbreviations: A&E, accident & emergency; GP, general practitioner; SD, standard deviation.
*Baseline characteristics for 10 patients could not be captured.
Physician Professional Characteristics.

Abbreviations: APS, antiphospholipid syndrome; CLL, chronic lymphocytic leukemia; CVD, cardiovascular disease; DITP, drug-induced immune thrombocytopenia; ES, Evans syndrome; HIV, human immunodeficiency virus; ITP, immune thrombocytopenia; SLE, systemic lupus erythematosus.
*Mean proportion of patients with either primary or secondary ITP as estimated by physicians.
Diagnosis
Patient initial presentation was mostly to a general practitioner/family doctor (46%; n = 52/114) or a specialist (26%, n = 30/114) and the median (range) number of healthcare professionals seen before ITP diagnosis was 2 (1 − 10) (Table 1). Seventy-seven percent of physicians (n = 37/47) estimated that it would take multiple weeks to formally diagnose a patient with ITP following their initial symptom presentation to a doctor, and ∼80% of them (n = 37/46; adult hematologists [n = 20/25], pediatric hematologists [n = 17/21]) believed that misdiagnosis occurred in up to 25% of patients, with the most common misdiagnoses being aplastic anemia/ myelodysplastic syndromes (adult physicians, 65%; pediatric physicians, 74%), drug induced thrombocytopenia (adult physicians, 61%; pediatric physicians, 42%), leukemia (adult physicians, 26%; pediatric physicians, 63%), and hereditary thrombocytopenia (adult physicians 26%, pediatric physicians, 58%). Thirty-two percent of patients felt there was a long delay in confirming ITP diagnosis and believed that the most common reasons were “waiting time for additional tests to confirm diagnosis” (42%), “misdiagnosis” (33%), and “delay in referral to a specialist” (17%). Following diagnosis, 63% of patients reached for support to physicians, 51% of patients to family and friends, 32% of patients to patient support groups, 10% of patients to a nurse, and 8% of patients felt they had no support. Sixty percent of patients liked to have had more support at the time of their diagnosis from physicians (58%), patient support group (18%), family/friends (17%), nurse (6%), other patients (5%), and other type of support (18%).
ITP Symptoms and Severity
Patient Perception
Bruising of unknown origin, fatigue, petechiae, and anxiety around maintaining stable platelet counts were among the most frequent patient-reported signs and symptoms at diagnosis and at survey completion, as summarized in Figure 1. Anxiety around unstable platelet counts was reported as most severe (scored ≥5 on a Likert scale 1 − 7; 7 = the worst imaginable) at diagnosis (adults [77%, n = 23/30]; pediatric [67%, n = 4/6]) and at survey completion (adults [52%, n = 11/21]; pediatric [67%, n = 4/6]). Fatigue was reported as a severe sign/symptom in 56% (n = 22/39) of adult and 83% (n = 5/6) pediatric patients at diagnosis, and in 45% (n = 17/38) of adult and 50% (n = 3/6) pediatric patients at survey completion. In addition, heavy menstrual bleeding (adult patients [75%, n = 15/20]; pediatric patients [100%, n = 2/2], at diagnosis), extra bleeding during or immediately after surgery (adult patients at diagnosis [83%, n = 5/6] and pediatric patients at survey completion [100%, n = 4/4]), and depression (adult patients at diagnosis [57%, n = 12/21]; pediatric patients at diagnosis [75%, n = 3/4]; adult patients at survey completion [52%, n = 11/21]; pediatric patients at survey completion [100%, n = 1/1]) were also reported. The top signs and symptoms that patients mostly wanted resolved included fatigue (overall 63% [n = 39/62]; adult patients, 67% [n = 34/51]; pediatric patients, 45% [n = 5/11]), bruising (overall 37% [n = 23/62], adult patients 35% [n = 18/51]; pediatric patients 45% [n = 5/11]), petechiae (overall 23% [n = 14/62]; adult patients, 18% [n = 9/51]; pediatric patients, 45% [n = 5/11]), increased number of moderate to severe headaches (overall 26% [n = 16/62]; adult patients, 27% [n = 14/51]; pediatric patients, 18% [n = 2/11]) and heavy menstrual bleeding (overall 18% [n = 11/62]; adult patients, 16% [n = 8/51]; pediatric patients 27% [n = 3/11]). Only a minority of patients reported no symptoms at diagnosis (4%) and at survey completion (8%).

Patient perception at (a) diagnosis and (b) at time of survey completion. (c) Physician perception on overall of occurrence and severity of ITP signs, and symptoms at time of the survey.
Physician Perception
The most common signs and symptoms adult and pediatric hematologists reported hearing about from their patients at any time (overall) were similar to those reported at diagnosis (overall: petechiae [96%, 95%], purpura [92%, 95%], epistaxis [88%, 90%], bleeding gums [68%, 81%] and menorrhagia [76%, 48%]; diagnosis: petechiae [100%, 95%], purpura [92%, 95%], epistaxis [88%, 95%], bleeding gums [72%, 71%], and menorrhagia [80%, 48%] for adult and pediatric hematologists, respectively). Forty percent of adult hematologists (n = 10/25) and 38% of pediatric hematologists (n = 8/21) reported that ITP symptoms reduced a patient HRQoL. Adult and pediatric hematologists, respectively, believed several signs and symptoms had a major negative impact on patient HRQoL (scored ≥5 on a Likert scale 1 − 7; 7 = a great deal), including thrombosis (68%, 68%), hematuria, melena or rectal bleeding (67%, 57%), profuse bleeding during surgery (67%, 65%), and menorrhagia (48%, 67%), and anxiety surrounding unstable platelet count (52%, 52%). Approximately a third of physicians reported that fatigue substantially reduced patient HRQoL (score ≥5 on a Likert scale 1 − 7; 7 = a great deal). The symptoms that adult and pediatric hematologists believed had a major negative impact on patient HRQoL are summarized in Figure 1C. Adult (n = 25) and pediatric hematologists (n = 21) reported that a mean proportion (SD) of 26.0 (25.5) and 18.9 (21.5) of their patients experienced fatigue, respectively, and was significant in 7 − 8% of them (score ≥5 on a Likert scale 1 − 7; 7 = completely fatigued), (Figure 2A). Forty-four percent of adult hematologists (n = 11/25) and 40% (n = 8/20) of pediatric hematologists believed that platelet counts <10 × 109/L were associated with increased fatigue severity (Figure 2B).

(a) Physician perception of their patients' severity of fatigue.* (b) Physician perceiving highest likelihood of fatigue by platelet groups.† *Physician-reported perceived severity of fatigue on a 7-point Likert scale where 1 = low level and 7 = completely. †Physicians reported likelihood of fatigue scored ≥ 5 on a 7-point Likert scale where 1 = not at all and 7 = very likely.
Treatment and Management
Adult and pediatric hematologists, respectively, indicated that during the first 6 months following diagnosis, watch-and-wait rather than pharmacological intervention was the preferred option for 50% and 49% of their patients. Adult and pediatric hematologists also indicated that watch-and-wait was the preferred option for 45% of previously treated adult patients and 48% of previously treated pediatric patients with ITP beyond 12 months of diagnosis or recurrent ITP. As per adult hematologists and pediatric hematologists, factors (>50%) influencing watch-and-wait instead of recommending drug treatment following their initial diagnosis were platelet levels not low enough (90% [n = 19/21]; 89% [n = 16/18]), patient being asymptomatic (81% [n = 17/21]; 78% [n = 14/18]), and absence of severe bleeding symptoms (24% [n = 5/21]; 67% [n = 12/18]). Over one-third of the patients (37%, n = 34/91 [adult patients: 38%, n = 26/68; pediatric patients: 13%, n = 2/15]) reported they had been on watch-and-wait management at some time.
Splenectomy
When physicians were asked what proportion of patients would undergo splenectomy, adult hematologists and pediatric hematologists estimated that 14% and 2% of patients, respectively, with newly diagnosed ITP, and 51% and 23% of patients, respectively, with persistent or chronic or recurrent ITP would undergo a splenectomy. At the time of the survey, 39% of adult patients (n = 24/62) and 13% of pediatric patients (n = 2/15) had been splenectomized.
Pharmacological Treatments
Per adult and pediatric hematologists, the most commonly prescribed treatments for newly diagnosed patients (prior to first relapse) were corticosteroids or steroids, such as prednisolone, methylprednisolone, or dexamethasone (100%, n = 20/20; 89%, n = 16/18), IVIG (90%, 18/20; 89%, n = 16/18), and anti-fibrinolytics (75%, n = 15/20; 56%, n = 10/18), respectively. For persistent, chronic or recurrent ITP, the most common treatments adult and pediatric hematologists would prescribe were anti-CD20 (100%, n = 20/20; 83%, n = 15/18), other immunosuppressants (90%, n = 18/20; 94%, n = 17/18), cyclosporine (90%, n = 18/20; 83%, n = 15/18), androgens such as danazol (85%, n = 17/20; 67%, n = 12/18) and TPO-RAs (80%, n = 16/20; 89%, n = 16/18). Adult hematologists would most commonly prescribe TPO-RAs at first relapse (57%) rather than at second-relapse (43%), whereas pediatric hematologists would most commonly prescribe TPO-RAs at second relapse (56%) rather than at first relapse (6%). A summary of treatments prescribed by physicians following relapse are shown in Figure 3. The most common reasons for adult and pediatric hematologists changing therapy were treatment-related side effects (95%, n = 20/21; 83%; n = 15/18), lack of efficacy (81%, n = 17/21; 89%, n = 16/18), cost/coverage (71%, n = 15/21; 67%, n = 12/18) and disease progression (67%, n = 14/21; 56%, n = 10/18), respectively. Other reasons included, change in blood counts (43%, n = 9/21; 28%, n = 5/18), patient preference (33%, n = 7/21; 17%, n = 3/18), change of symptoms (19%, n = 4/21; 33%, n = 6/18) and others (5%, n = 1/21; 6%, n = 1/18), for adult and pediatric hematologists, respectively. Among the attributes considered for treatment decision making, adult hematologists (n = 20 − 21) and pediatric hematologists (n = 17 − 18) both identified, respectively, keeping side effects to a minimum (mean score [SD]: 82.20 [25.10], 89.94 [20.96]), reduction of bleeding risk (86.33 [20.93], 85.50 [21.94]), and the ability to offer sustained remission or cure of ITP (82.95 [18.41], 80.24 [25.38]) as attributes with highest importance (on a scale of 1 to 100; 100 = of the highest importance). Patients (n = 49 − 62) identified the ability to offer sustained remission or cure of ITP (96.27 [16.55]), keeping side effects minimum (95.05 [17.78]), reduction of bleeding risk (92.05 [21.16]), avoiding immunosuppression (91.04 [19.20]), and slower recovery from relapse but potentially longer term (89.71 [23.21] as attributes with highest importance (on a scale of 1 to 100; 100 = of the highest importance).

Treatment prescribed by (a) adult physicians and (b) pediatric physicians following relapse of ITP.
Stability of Platelet Counts
Stability of platelet counts over the previous 2 − 6 months and over one year was reported in 9% (n = 4/43; adult patients, 11% [n = 4/36]) and 28% (n = 12/43, adult patients 25% [n = 9/36]; pediatric patients, 43% [n = 3/7]) of patients, respectively.
Impact of ITP on Functional and Emotional Well-Being
In the ILQI questionnaire, which explored the impact of ITP on aspects of patients' daily lives within the last month, 39% (n = 28/71) of patients answered “sometimes” to “all the time” to all 10 questions. The aspects most affected (“more than half the time” or “all the time”) were energy levels (46%, n = 52/112 [adult patients: 48%, n = 41/86; pediatric patients: 44%, n = 7/16]), ability to concentrate on everyday activities (42%, n = 47/113 [adult patients: 40%, n = 35/87; pediatric patients: 50%, n = 8/16]) and capacity to exercise (41%, n = 46/113 [adult patients: 43%, n = 37/87; pediatric patients: 38%, n = 6/16]) (Figure 4A–B). Forty-four percent of patients (n = 50/113) felt that ITP negatively impacted their psychological and emotional well-being (score ≥5 on a Likert scale 1 − 7; 7 = a great deal). Patients were concerned that their condition would worsen (61%, n = 69/113 [adult patients: 61%, n = 53/87; pediatric patients: 75%, n = 12/16]), about unexplained fluctuations in platelet levels (62%, n = 70/113 [adult patients: 60%, n = 52/87; pediatric patients: 75%, n = 12/16]), of having stable and safe platelet levels (77%, n = 87/113 [adult patients: 76%, n = 66/87; pediatric patients: 88%, n = 14/16]), of feeling stressed about ITP (50%, n = 56/113 [adult patients: 46%, n = 40/87; pediatric patients: 69%, n = 11/16]), and of feeling anxious/nervous about their platelet counts (40%, n = 45/113 [adult patients: 36%, n = 31/87; pediatric patients: 63%, n = 10/16]). Twenty-three percent of patients (n = 26/113) were worried about dying (adult patients: 24%, n = 21/87; pediatric patients: 25%, n = 4/16), 28% of patients (n = 32/113) indicated substantial concern about physical appearance (ie, bruising, rashes, and lumps [adult patients: 18%, n = 16/87; pediatric patients: 69%, n = 11/16]), and 13% of patients (n = 15/113 [adult patients: 7%, n = 6/87; pediatric patients: 44%, n = 7/16]) reported wearing long-sleeved clothing to hide evidence of bleeding (score ≥5 on a Likert scale 1 − 7; 7 = a great deal) (Figure 4C–D). Fifty-two percent of adult hematologists (n = 13/25) and 70% of pediatric hematologists (n = 14/20) felt that patients' anxiety about their platelet levels had a substantial negative impact on their emotional well-being (score 6 or 7 on a Likert scale 1 − 7; 7 = a great deal). Adult and pediatric hematologists also felt that their patients were concerned about stress (52%, n = 13/25; 53%, n = 10/19), long-term rare disease (32%, n = 8/25; 60%, n = 12/20), fear around disease (32%, n = 8/25; 50%, n = 10/20), feeling of helplessness (28%, n = 7/25; 45%, n = 9/20), physical appearance (24%, n = 6/25; 30%, n = 6/20), and dying (24%, n = 6/25; 25%, n = 5/20), respectively (score ≥5 on a Likert scale 1 − 7; 7 = a great deal).

Impact of ITP on daily living (ILQI) in (a) adult patients and (b) pediatric patients. Perceived factors driving a psychological/emotional impact in ITP in (c) adult patients and (d) in pediatric patients.
Impact of ITP on Work and Other Activities
At the time of the survey, 26 patients reported working full- or part-time. Of these, 20% (n = 5/24) felt ITP negatively affected on their productivity at work (score ≥5 on a scale of 1 − 7; 7 = completely prevented from working) (Figure 5A). Twenty-two percent of respondents (n = 11/51) reported that they had reduced their working hours because of their ITP, 12% (n = 5/43) had declined a promotion, 11% (n = 5/46) had considered terminating their employment, and 4% (n = 2/47) were forced to stop working because of their ITP (Figure 5B). One of four pediatric patients and 30% (n = 23/77) of adult patients reported that their ITP had a negative impact on their ability to undertake daily activities outside of work/school (described as work around the house ie, food preparation, housework, gardening, childcare, oral hygiene [score ≥5 on a scale of 1 − 10; 10 = completely prevented productivity]) (Figure 5C). Thirty-five percent (n = 7/20) of adult hematologists and 20% (n = 5/25) of pediatric hematologists believed ITP had a negative impact on their patients' ability to undertake daily activities and another 28% of adult hematologists (n = 7/25) and 60% of pediatric hematologists (n = 12/20), also felt that ITP had a negative effect on the overall level of physical activity that their patients were able to undertake (a score ≥5 on a 7-point Likert scale; 7 = a great deal). In addition, adult and pediatric hematologists, respectively, felt that ITP had a negative effect on out-of-pocket expenses (18%, n = 4/22; 63%, n = 12/19), patients’ relationship with spouse/partner (33%, n = 8/24; 38%, n = 6/16), patients’ family or social life (20%, n = 5/25; 35%, n = 7/20), and the relationship with the patient caregiver (12%, n = 3/25; 30%, n = 6/20). Adult hematologists believed that ITP also had a negative impact on patients’ sexual activities (32%, n = 8/25) and sexual desire (8%, n = 4/22), as well as ITP affected patients’ travel plans with concerns about taking medication abroad (40%, n = 10/25), the burden to consult about their platelet levels before travelling (34%, n = 12/35), and the need for travel insurance (44%, n = 11/25) (a score ≥5 on a 7-point Likert scale; 7 = a great deal).

Effect of ITP on (a) productivity at work; (b) on employment status; (c) regular activities.
Satisfaction and Perception of Treatment and Other Aspects of Care
When looking at patients’ satisfaction with current treatment among patients being treated with anti-CD20s, TPO-RAs, IVIG or corticosteroids, the few patients being treated with anti-CD20s reported the greatest overall satisfaction regarding control of their ITP (100%, n = 2/2), followed by TPO-RAs (66%, n = 2/3), IVIG (40%, n = 2/5) and corticosteroids (32%, n = 8/25; score ≥5 on a Likert scale 1 − 7; 7 = strongly agree). Physicians were also asked about their satisfaction with the current pharmacological interventions being used for treating their patients. Sixty-seven percent (n = 14/21) of the adult hematologists and 39% (n = 7/18) of the pediatric hematologists were highly satisfied with the current pharmacological interventions available for the management of ITP (score ≥5 on a Likert scale 1 − 7; 7 = completely satisfied). The most common reasons (>30% in either of the groups) for adult hematologists (n = 21) and pediatric hematologists (n = 18) being satisfied with current treatment options include the ability to improve QoL for patients (43%, n = 9/21; 61%, n = 11/18), wide selection of treatments (38%, n = 8/21; 17%, n = 3/18), and ability to improve disease symptoms (33%, n = 7/21; 67%, n = 12/18), respectively. When asked about reasons for not being satisfied with current treatment options, adult hematologists and pediatric hematologists mentioned, respectively, unacceptable side effects (48%, n = 10/21, 22%, n = 4/18), limited treatment options (38%, n = 18/21, 56%, n = 10/18), not able to provide deep/stable remission for patients (38%, n = 8/21, 56%, n = 10/18), not able to reach the treatment goals set (33%, n = 7/21; 28%, n = 5/18), and patients become refractory to treatment (33%, n = 7/21, 50%, n = 9/18). Thinking about the three most important treatment goals beyond a cure for ITP, patients reported healthy blood counts (85%, n = 82/97 [adult patients: 83%, n = 60/72; pediatric patients: 88%, n = 14/16]), increasing energy levels (61%, n = 59/97 [adult patients: 63%, n = 45/72; pediatric patients: 50%, n = 8/16]), and improvement in overall symptoms (60%, n = 58/97 [adult patients: 61%, n = 44/72; pediatric patients: 63%, n = 10/16]) (Figure 6A−B), whereas adult hematologists and pediatric hematologists reported, respectively, reduction of spontaneous bleeding (71%, n = 16/23, 78%, n = 15/19), healthy blood counts (57%, n = 13/23; 39%, n = 7/19), better quality of life (38%, n = 9/23; 72%, n = 14/19), and symptom improvement (38%, n = 9/23; 56%, n = 11/19) (Figure 6C−D). The majority of patients (78%, n = 88/113) reported they were highly satisfied with the management and treatment of their ITP condition, that the physician responsible for their care understood and supported their treatment goals (76%, n = 86/113), and that their physician considered their needs when planning their treatment goals (76%, n = 85/112) (score ≥5 on a Likert scale 1 − 7; 7 = completely satisfied). Adult and pediatric hematologists felt that patients were highly satisfied with the treatment goals that had been set (82%, n = 18/22; 58%, n = 11/19), with the ease and convenience of prescribed medication (82%, n = 18/22; 50%, n = 9/18), with the treatment decisions that have been made (86%, n = 19/22; 47%, n = 9/19), with the management of their condition (91%, n = 20/22; 63%, n = 12/19), and with the level of communication about their disease and treatment (91%, n = 20/22; 68% n = 13/19), respectively (set score ≥5 on a Likert scale 1 − 7; 7 = completely satisfied). Patients’ and physicians’ level of agreement regarding patient-physician relationship is shown in Figure 7.

Top three treatment goals identified by (a) adult patients and (b) pediatric patients and (c) adult physicians and (d) pediatric physicians.
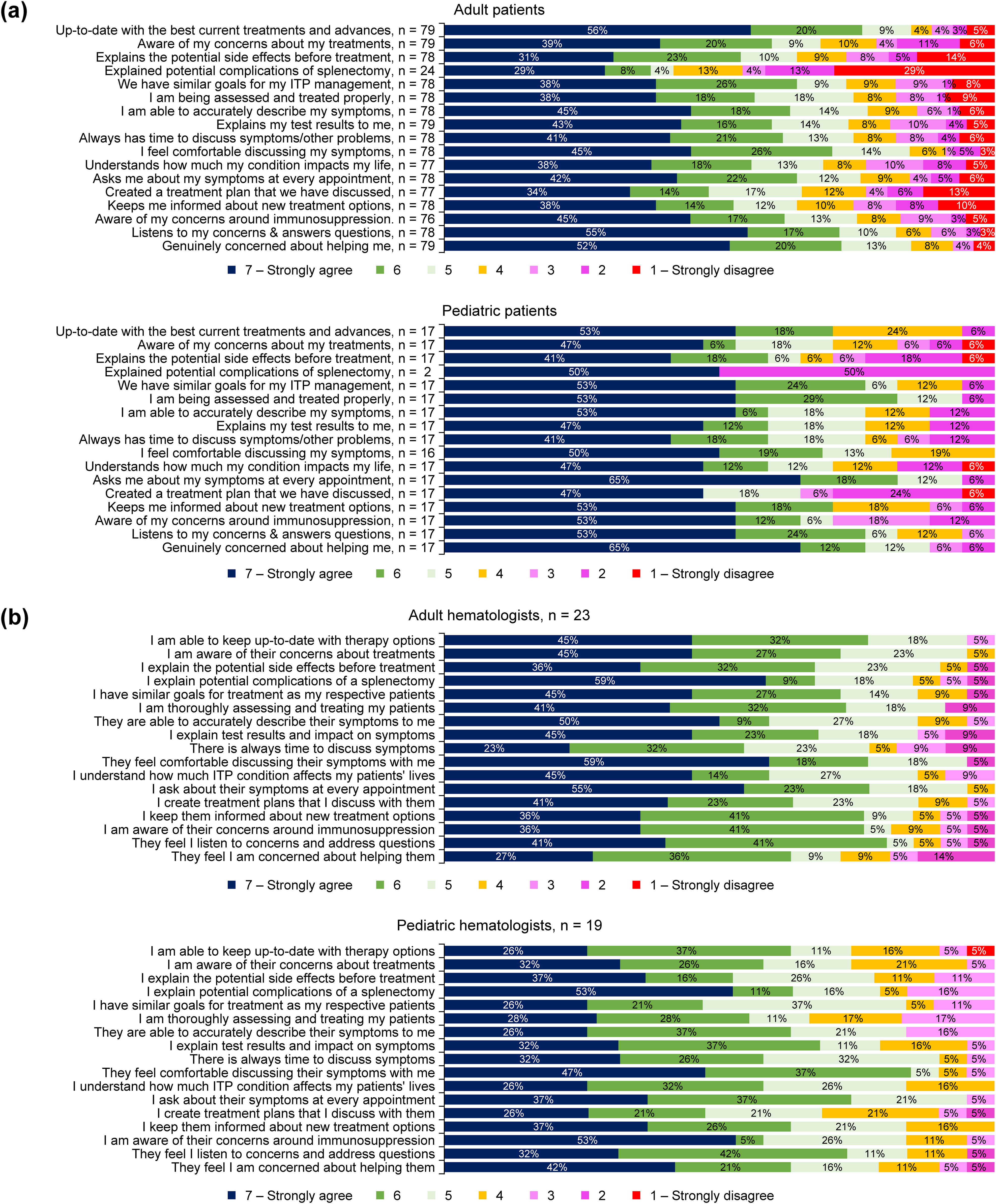
(a) Patients’ level of agreement regarding patient-physician relationship; (b) Physicians’ level of agreement regarding patient-physician relationship.
Discussion
The global I-WISh online survey recently uncovered the extent of the adverse real-world impact of ITP on patients' QoL, and highlighted the need for physicians to adapt disease management to the individual.8, 9 This local I-WISh survey further expanded on the global findings and contextualized them to Iranian clinical practices. This survey is also the first to investigate differences and similarities between the pediatric and adult populations of patients with ITP.
When looking at time to diagnosis from initial presentation, most patients expressed a positive experience with the Iranian healthcare system whereas some patients who experienced delays in diagnosis felt that these were caused by a delay in consulting a specialist. Therefore, it can be inferred that direct accessibility to medical specialists in Iran can lead to early diagnosis and improved patient satisfaction. Despite this positive finding, some patients experienced misdiagnosis, or their ITP diagnosis was delayed due to the need for additional tests, which may be reflective of the lack of a definitive laboratory test to diagnose this disease. As with the findings from the global I-WISh survey, petechiae and bruising were two of the most frequently reported patient symptoms at diagnosis but were reduced by half at survey completion except in the pediatric population. This may reflect the watch-and-wait management approach that is often adopted in this patient group, in line with current international guidelines. 5 Instead, fatigue and anxiety about stable platelet counts did not substantially decrease over time in either subgroup. Reasons for these outcomes have not been explored in the current analysis and require further research; although, fatigue is a common symptom among patients with ITP, and appears to be independently associated with platelets counts, making its management particularly challenging. 12 Fatigue was one of the top symptoms patients mostly wanted resolved, even though it was not one of the most common signs and symptoms adult and pediatric hematologists reported hearing about from their patients. This may indicate that even if patients felt fatigue as the most burdensome symptom, they were not proactive towards it. The rate of splenectomy in this patient population was relatively high, and most likely reflected the lack of availability of TPO-RAs and/or the high costs associated with these therapies, which prevent their wide adoption in Iranian clinical practice. However, more adult than pediatric patients had been splenectomized, in line with current guidelines recommending delaying splenectomy because of the potential for spontaneous remission in the first year particularly among the pediatric population, but also because of the complications associated with this intervention. 5 For both patients within six months and beyond 12 months of diagnosis, nearly half of physicians indicated a preference for a watch-and-wait approach with no difference between the adult and pediatric populations, primarily driven by patients’ high platelet counts or absence of symptoms. If pharmacological treatments were needed, patients newly diagnosed or at first relapse were most likely treated with corticosteroids whereas patients with persistent/chronic disease or after subsequent relapse had access to several treatment options, including anti-CD20, other immunosuppressants, TPO-RAs, and cyclosporine. The considerable variability in ITP management after first relapse may be reflective of a lack of availability/affordability of certain treatment options in Iran, such as anti-CD20 and TPO-RAs; however, this cohort of patients with ITP in Iran appeared to receive treatments in line with the findings from the global I-WISh study. 8 When looking at differences between the adult and pediatric populations, TPO-RAs were most commonly prescribed to adult rather than pediatric patients at first relapse, and to pediatric rather than adult patients at second and third relapse. In addition, platelet transfusions were mostly used in adult rather than pediatric patients, most likely prior to splenectomy.
Patients often cited reduced energy levels, difficulty to concentrate on everyday tasks and reduced ability to exercise as symptoms that negatively affected their QoL. The majority of pediatric patients believed that ITP impacted their studies. More pediatric than adult hematologists believed ITP had a negative impact on the patient's ability to exercise, which may be due to concerns around the risk of serious bleeding and intracranial hemorrhage during exercise and the notion that children and adolescents tend to be more active than adults. Pediatric patients also felt ITP had a negative impact on practising a hobby. However, given the limited number of respondents, caution is warranted when interpreting these findings. A meaningful proportion of patients also felt that ITP negatively impacted their psychological and emotional well-being, with patients mostly concerned that their condition would worsen and platelet levels would unexpectedly go down. Of note, more pediatric than adult patients hid the signs of bleeding with clothing, possibly to avoid social isolation and stigmatization. However, most patients were satisfied with the management and treatment of their ITP condition, despite only few reported stable platelet counts, suggesting that patients felt their symptoms were overall well-managed and that the physician responsible for their care understood and supported their treatment goals. It is worth noting that adult and pediatric patients seemed to align on goals of therapy. Among the reasons that negatively impacted patients’ satisfaction with treatment, lack of effectiveness of current therapies and treatment side effects were often mentioned, underling the need for future therapies to provide effective treatment while minimizing adverse events.
Limitations of the study included the risk of selection biases, which is inherent to non-interventional studies. The robustness of data might be impacted due to the low sample size, particularly in the pediatric subgroup, and by potential biases such as differences in patients’ motivations to participate to the survey. Some data were presented based on the available responses per item and were not based on the overall population. Thus, interpretation of these results warrants caution. The survey had fixed questions with multiple choice answers, with no opportunity to further comment, which may limit interpretation of the answers. Recall bias is another limitation because patients were asked to describe past experiences to estimate their QoL at the time of diagnosis and, therefore, caution is advised when interpreting these retrospective data. Interpretability of patients’ and physicians’ satisfaction with specific therapies is limited by the low number of responses. All participating physicians had experience in ITP, and likely have more awareness of both the burden of ITP on patients' lives and the benefits and limitations of available treatment options than most hematologists and hemato-oncologists in general practice have.
In conclusion, this local I-WISh survey underlined the extent to which ITP negatively impacted patients' life in Iran, in line with the global findings.8, 9 Energy levels, ability to concentrate on everyday activities, and ability to exercise were the most affected aspects of patients' daily lives. With the possible exception of ITP treatment, management and perception of ITP impact on patients’ lives appeared to align between adult and pediatric patients and between adult and pediatric hematologists. Fatigue was perceived as one of the top symptom patients wanted resolved, even though it was not commonly reported to hematologists. However, most patients felt satisfied with the care they received, suggesting that most symptoms (except for fatigue) were generally well managed and that physicians had experience in managing patients with ITP and had a good understanding of the benefits and the limitations of the currently available treatments in Iran. We hope that the present study may assist physicians in optimizing the clinical and emotional management of patients with ITP in Iran.
Footnotes
Acknowledgements
The authors would like to thank all patients and physicians for participating in the survey. The authors would also like to thank Sabrina Giavara, PhD, of Novartis UK Ltd, and Pranitha Manchanapalli, Pharm D, Novartis Healthcare Private Limited, for providing medical writing support in accordance with Good Publication Practice (GPP3) guidelines (![]() ).
).
Authors’ Contributions
PE, FA, AH, HH, NM, AA, NS, and MF provided substantial contribution to the conception, the design, and the planning of the study. AAA, MB, HH, and MF contributed to the acquisition and analysis of the data. All authors contributed to the interpretation of the data, critically reviewed, and revised the manuscript for important intellectual content and approved the final version of the article.
Availability of Data and Materials
Novartis is committed to sharing with qualified external researchers, access to patient-level data and supporting clinical documents from eligible studies. These requests are reviewed and approved by an independent review panel on the basis of scientific merit. All data provided is anonymized to respect the privacy of patients who have participated in the trial in line with applicable laws and regulations. This trial data availability is in accordance with the criteria and process described on ![]() .
.
Declaration of Conflicting Interests
PE received an honorarium from the Clinical Trial Center of Tehran University of Medical Sciences. FA acted as an invited speaker to Cobel Darou and Novartis. HH and AAA were contracted by Novartis Pharmaceuticals, Iran to carry out this project, HH in quality of CEO of the trial CRO and AAA as the manager of the Data Management unit. AA and NS are employees of Novartis Pharma, Iran. HA, MB, AH, NM, and MF had no conflict of interest to declare.
Ethics Approval and Consent to Participate
Study materials and protocol were approved by the research ethics committees of Iran University of Medical Sciences (IR.IUMS.FMD.REC.1400.229) and Shahid Beheshti University of Medical Sciences (IR.SBMU.RICH.REC.1398.013), Iran. Patients and physicians were given an overview of the study and the ethical approval details; those who wished to participate had to provide consent before undertaking the survey.
Funding
This work was supported by Novartis Pharmaceuticals, Iran.
