Abstract
Secondary failure of platelet recovery (SFPR) is a life-threatening complication that may affect up to 20% of patients after allogeneic hematopoietic stem cell transplantation (HSCT). In this study, to evaluate the efficacy of recombinant human thrombopoietin (rhTPO), we retrospectively analyzed 29 patients who received continuous rhTPO for the treatment of SFPR. Overall response and complete response were observed in 24 (82.8%) patients and 10 (34.5%) patients, at a median time of 21.5 days (range, 3-41 days) and 39.5 days (range, 7-53 days) after initiation of rhTPO treatment, respectively. Among the responders, the probability of keeping overall response and complete response at 1 year after response was 77.3% and 80.0%, respectively. In multivariate analysis, higher CD34+ cells (≥3 × 106/kg) infused during HSCT (HR: 7.22, 95% CI: 1.53-34.04, P = 0.01) and decreased ferritin after rhTPO treatment (HR: 6.16, 95% CI: 1.18-32.15, P = 0.03) were indicated to associate with complete response to rhTPO. Importantly, rhTPO was well tolerated in all patients without side effects urging withdrawal and clinical intervention. The results of this study suggest that rhTPO may be a safe and effective treatment for SFPR.
Keywords
Introduction
Secondary failure of platelet recovery (SFPR), a condition of thrombocytopenia after primary engraftment of platelet, is a critical complication after allogeneic hematopoietic stem cell transplantation (allo-HSCT) with a reported incidence varying from 12% to 20%.1-3 The most common risk factors associated with SFPR include acute GVHD (aGVHD), T-cell depletion, infections, and low number of infused stem cells.1-3 SFPR has been revealed to significantly associate with an inferior overall survival (OS) and a greater number of complications,1-3 indicating that rapid correction of severe thrombocytopenia in these patients is essential.
Thrombopoietin, a naturally occurring glycosylated peptide growth factor, is a primary regulator of platelet production. 4 It is well established that by activating the cytokine receptor c-Mpl, thrombopoietin stimulates the differentiation of hematopoietic stem cells into megakaryocyte progenitor cells and promotes megakaryocyte proliferation and maturation. 5 RhTPO is a glycosylated recombinant form of the c-Mpl ligand, containing amino acid sequence identical to endogenous thrombopoietin, 6 which is safe and well tolerated even in pregnancy. 7 Previous clinical research has demonstrated the efficacy of rhTPO for the management of thrombocytopenia caused by various reasons7-11 and promotion of platelet engraftment after HSCT.12-14 However, the effect of rhTPO in treating SFPR after allo-HSCT has not been reported in detail yet. Therefore, we carried out this retrospective study with the aim of reporting on the response rates and safety of rhTPO therapy for patients diagnosed with SFPR, as well as survival outcomes of SFPR patients.
Subjects and Methods
Patient Selection and rhTPO Treatment
SFPR was defined as a decline of platelet counts below 20 × 109/L for 7 consecutive days or requiring transfusion support to maintain a platelet count above 20 × 109/L after achieving sustained platelet counts ≥ 50 × 109/L without transfusions for 7 consecutive days after HSCT. 1 Patients who diagnosed with SFPR and received continuous rhTPO (Sunshine, Shenyang, China) were included. RhTPO was injected at 300 IU/kg/day for 42 consecutive days at most or until platelet counts ≥50 × 109/L, independent of platelet transfusion. This study was approved by our center's Institutional Review Board. All patients or their legal representative have signed the informed consent.
HSCT and GVHD Prevention
Conditioning regimen was given based on diseases: modified busulfan (Bu)/cyclophosphamide (CY)/fludarabine (FLU)/cytarabine (ARA-C) regimen for patients with acute myeloid leukemia; 15 5-day schedule of decitabine were for myelodysplastic syndrome and secondary acute myeloid leukemia as described in previous study; 16 Total body irritation/CY/Flu/Ara-C or CY/Flu/Etoposide regimen for acute lymphocytic leukemia 17 and Flu + CY ± Bu for sever aplastic anemia accordig to proliferative degree of bone marrow. 18 GVHD prophylaxis consisted of short courses of methotrexate and cyclosporine or tacrolimus, while mycophenolester and anti-thymocyte globulin were added for patients who did not receive matched sibling donor HSCT. 16
Response Evaluation and Definition
To evaluate the impact of rhTPO on megakaryocytes (MKs), morphological features of MKs were examined by two hematopathologists, who provided their opinions independently. The number of MKs was determined using a bone marrow aspirate smear in an area of 1.5 × 3 cm2. The total number of MKs on one slide was calculated under microscopy, and the number of MKs between 7 and 35 was regarded as normal. 11 The morphology of up to 25 MKs was analyzed under oil lens and classified as naïve, granular, thrombocytogenic, or naked nucleus MKs. 19 Overall response (OR) and complete response (CR) was defined as platelet ≥ 20 × 109/L and ≥50 × 109/L for 7 consecutive days without platelet transfusion support within 2 months after initiation of rhTPO, respectively. Patients who did not meet the criteria of OR were defined as "no response". OS refers to the time from diagnosis of SFPR to death of any cause. The assessment of disease risk was based on the disease risk index, 20 and patients with aplastic anemia were defined as low risk. Adverse events were graded using the Common Terminology Criteria for Adverse Events. 21
Statistical Analysis
The primary outcome was the response to rhTPO treatment, with the observation period lasting at least 3 months after rhTPO treatment. Death without reaching response was treated as competing risk for Gray test to calculate the cumulative incidence of response. The secondary outcome was OS, which was calculated by the Kaplan-Meier method. Chi-squared tests was used for comparison of categorical variables and Mann-Whitney tests for continuous variables. Counts of platelet and MKs before and after rhTPO treatment was compared using paired t-test. A Cox proportional hazards regression model was used for univariate and multivariate analysis of response to rhTPO. Variables with P<0.2 in univariate analysis were included in multivariate analysis. The history of CMV reactivation and EBV reactivation before diagnosis of SFPR were regarded as time-dependent covariates. Two-sided p-value less than 0.05 was considered to indicate statistically significant. Statistical analyses were performed using SPSS 26 (SPSS, Chicago, IL), GraphPad Prism 7 (GraphPad Software, La Jolla, CA) and R statistical software 4.0.5 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Patient Characteristics
Of 736 patients who underwent allo-HSCT in our institution from January 2017 to April 2020, 104 (14.13%) patients were diagnosed with SFPR. A total of 29 patients who were continuously treated with rhTPO for SFPR were included in this study, including 19 acute leukemia, 3 myelodysplastic syndrome and 7 aplastic anemia patients. Figure 1 shows the study flow chart. The characteristics of patients and HSCT settings were presented in Table 1. All patients received peripheral blood-derived donor stem cells with a median dose of 10.81 × 108/L mononuclear cells (range, 6.28-16.53 × 108/L) and 2.67 × 106/L CD34+ cells (range, 1.95-6.06 × 106/L). All patients achieved initial neutrophil engraftment (median, 15 days; range, 11-21) and platelet engraftment (median, 20 days; range, 11-61), with donor engraftment confirmed by full donor chimerism on bone marrow specimen. CMV immunoglobulin G antibody tested before HSCT in patients and donors were all positive.

Study design and flow diagram.
Patients' characteristics and HSCT settings.

Abbreviations: AML, acute myeloid leukemia; ALL, acute lymphoblastic leukemia; MDS, myelodysplastic syndrome; AA, aplastic anemia; ATG, antithymocyte globulin; GVHD, graft-versus-host disease; MMF, mycophenolate mofetil.
Seventeen (58.6%) patients achieved initial platelet engraftment within 28 days after HSCT, whereas 12 (41.4%) patients beyond 28 days. The median day of SFPR onset was 70 days (range, 22-358 days) post-HSCT. Nine (31.0%) patients presented concomitant neutropenia and 1 (3.4%) patient involved 3 lineage cytopenia. The median number of platelet in peripheral blood was 14 × 109/L (range, 2-18 × 109/L) before rhTPO treatment. Eleven (37.9%) patients had hypocellular bone marrow. Eight patient (27.6%) had a normal count of MKs in bone marrow before initiation of rhTPO, whereas 20 (69.0%) patients presented with a decreased MKs, and 1 (3.4%) patient did not have MKs.
Efficacy of rhTPO
Patients were treated with rhTPO immediately after diagnosis of SFPR, supported with blood transfusion of a median of 5 units of platelets (range, 0-28) and 4 units of peripheral red blood cells (range, 0-38) during rhTPO treatment. The median duration of rhTPO treatment was 18 days (range, 8-42 days). The platelet count in peripheral blood significantly increased after administration of rhTPO (Figure 2A). The platelet count (mean ± standard error) was 12.86 ± 0.73 × 109/L before rhTPO treatment, and 18.96 ± 1.48, 21.46 ± 2.32, 25.21 ± 2.43 and 34 ± 3.98 × 109/L at 7 days, 14 days, 1 month and 2 months after the initiation of rhTPO, respectively. Of note, platelet count continued to increase as the difference was statistically significant when compared 14-day versus 1-month and 1-month versus 2-month platelet count (Figure 2A). In addition, among 26 patients available for analysis of bone marrow MKs, the number of total MKs and thromocytogenic MKs was significantly higher after treatment (7.03 vs 21.58, P = 0.04; 0.31 vs 1.35, P = 0.048), while no significant increase regarding granular MKs (5.21 vs 7.58, P = 0.36, Figure 2B).
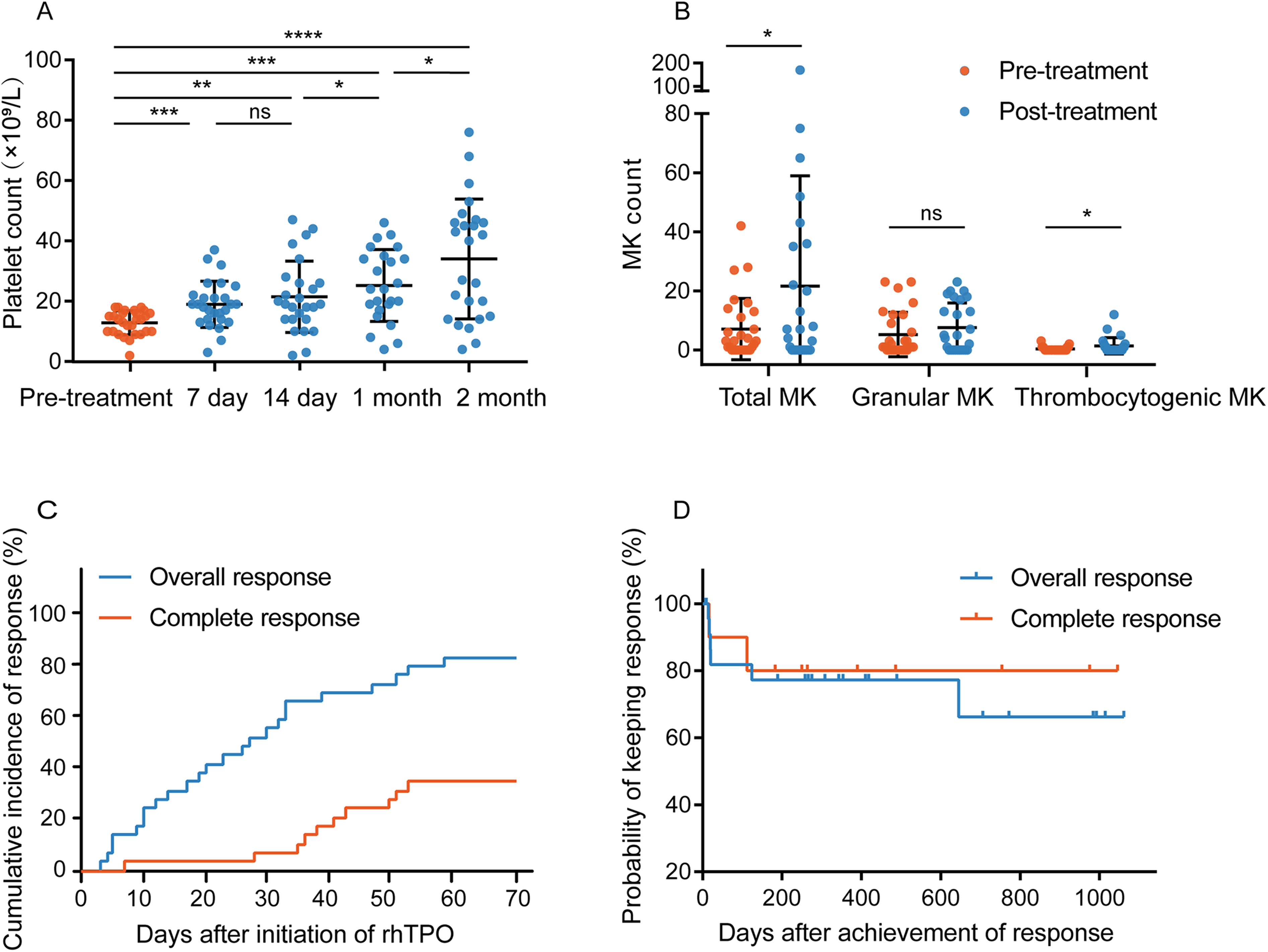
Efficacy of rhTPO. (A) The change of platelet count in peripheral blood after initiation of rhTPO treatment. (B) Megakaryocyte (MK) count in bone marrow after finish of rhTPO treatment. (C) Cumulative incidence of response to rhTPO. (D) Probability of keeping response after achievement of response. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001; NS, not significant.
In total, 24 (82.8%) patients responded to rhTPO treatment at a median time of 21.5 days (range, 3-59 days), and 10 (34.5%) patients further achieved CR at a median time of 39.5 days (range, 7-53 days) after the initiation of rhTPO treatment. The 30-day and 50-day cumulative incidence of OR was 55.2% and 72.4%, respectively, and of CR was 6.9% and 27.6%, respectively (Figure 2C). Eighteen of 24 (75.0%) overall responders remained OR and 8 of 10 (80.0%) complete responders kept CR during the follow-up period after response (median, 493 days; range, 327.5-987 days). Only 6 (25.0%) patients lost OR and 2 (20.0%) patients lost CR at a median time of 19.5 days (range, 14-645 days) and 64 days (range, 16-112 days) after response, respectively. The probability of keeping OR and CR at one-year post-response was 77.3% and 80.0%, respectively (Figure 2D).
Factors Influencing the Response of rhTPO
Table 2 shows the results of univariate and multivariate analysis for OR and CR to rhTPO treatment. With regard to OR, no variable was identified as significant independent influencing factor. For CR, multivariate analysis demonstrated that infused CD34+ cells ≥3 × 106/L during HSCT (HR, 7.22; 95% CI, 1.53-34.04; P = 0.01) and decreased ferritin after rhTPO treatment (HR, 11.28; 95% CI, 1.96-64.95; P = 0.007) were significantly associated with CR to rhTPO. Ferritin evaluation before and after rhTPO therapy was available in 21 patients. Three of 4 patients (75%) whose ferritin decreased post-rhTPO achieved CR (decreased median, 1674 ng/ml; range, 305-2939 ng/ml), while only 6 of 17 patients (35.3%) with increased ferritin achieved CR (increased median, 1287 ng/ml; range, 9-6496 ng/ml) (P = 0.27).
Univariate and multivariate analysis on response to rhTPO.

Abbreviation: HLA, human leukocyte antigen; ATG, antithymocyte globulin; MNC, mononuclear cells; MK, megakaryocyte; SFPR, secondary failure of platelet recovery; rh-TPO, recombinant human thrombopoietin. In bold are p-values <0.05.
Safety and Survival
No adverse events occurred. Importantly, no patient developed clinically meaningful abnormality of liver function and discontinued rhTPO treatment due to intolerability or adverse effects (Supplement Figure 1). 24 of 29 (82.8%) patients were alive within the follow-up period post diagnosis of SFPR (median, 438 days; range, 14-1184 days). Twenty patients were transfusion independence without rhTPO and 4 patients were transfusion dependence. Five patients died of graft-versus-host disease and infection. The probability of OS at one year after diagnosis of SFPR was 86.21% (Figure 3).
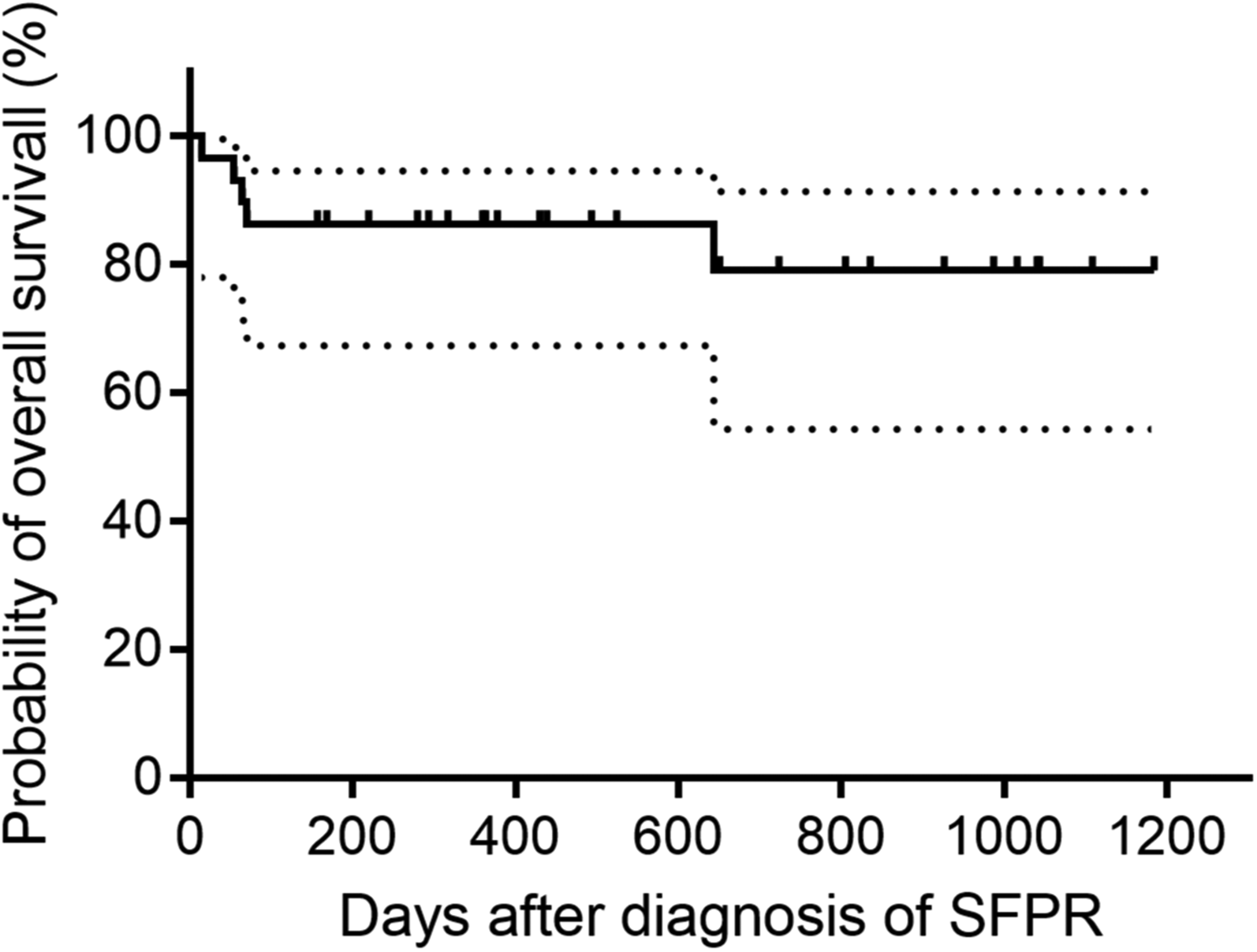
Probability of overall survival after diagnosis of SFPR.
Discussion
There is no standard therapeutic approach for SFPR at present. Administration of eltrombopag, an oral non-peptide TPO receptor agonist, was firstly reported in several case reports.22,23 Later, Fu et al. indicated that eltrombopag resulted in a rate of 52.3% patients recovering to platelet ≥50 × 109/L among 38 patients with refractory thrombocytopenia after allo-HSCT, including 15 patients with SFPR. 24 Similar response rate was achieved when using another TPO receptor agonist romiplostim, as 7 of 13 (53.8%) patients with SFPR responded. 25 However, the efficacy of rhTPO, a full-length glycosylated TPO recombinant protein, in the treatment of SFPR is still unknown. In this study, to our knowledge, we first revealed the efficacy of rhTPO on platelet recovery in patients with SFPR.
RhTPO has a variety of applications, including thrombocytopenia induced by immune disorder,7,8 sepsis, 9 and chemotherapy. 10 Randomized prospective trials showed that rhTPO administered after allo-HSCT, in the setting of graft source from umbilical cord blood or peripheral blood plus bone marrow, could significantly improve platelet engraftment and reduce platelet infusions.13,14 Furthermore, rhTPO could be used for the treatment of prolonged isolated thrombocytopenia after allo-HSCT with an overall response rate of 45.9%. 11 These studies suggest that rhTPO might be a therapeutic option for patients with SFPR after allo-HSCT.
In our cohort of SFPR, rhTPO therapy led to statistically significant increase in platelet counts in peripheral blood and megakaryocyte counts in bone marrow, which was consistent with previous studies.7,10,11,26 The OR rate was 55.2% and 72.4% at 30 days and 50 days after initiation of rhTPO, respectively. Similar OR rate was reported in a previous study using rhTPO to treat prolonged isolated thrombocytopenia, in which 45.8% patients achieved platelet ≥20 × 109/L at 28 days post rhTPO initiation. 11 In this study, we extended the duration of rhTPO treatment up to 42 days rather than reported 28 days, 11 and observed an increased OR rate at day 50 which suggested administration of rhTPO may be extended for SFPR patients for better response.
The CR rate in this study was not high as only 10 of 29 (34.5%) patients reached the criteria of platelet ≥50 × 109/L for 7 consecutive days without platelet transfusion support, while the CR rate about 50% was reported when using eltrombopag and romiplostim.24,25 However, the CR rates could not be compared directly between different research because of the small cohorts and heterogeneity. The superiority of these therapies needs to be demonstrated by further research. In addition, we found achievement of CR in SFPR patients required relatively longer treatment duration with a median of 39.5 days since rhTPO initiation, compared with a peak of platelet counts on a median day of 12 days after rhTPO in patients with cancer before chemotherapy. 26
In multivariate analysis, we found that infused CD34+ cells ≥3 × 106/kg during HSCT and decreased ferritin after rhTPO treatment were independently factors associated with CR to rhTPO. The underlying mechanisms may involve that adequate CD34+ stem cells are a prerequisite for rhTPO to promote the differentiation of hematopoietic stem cells into megakaryocyte progenitor cells and subsequent platelet production. 5 Decreased ferritin may eliminate the iron toxicity on bone marrow function which causes the accumulation of reactive oxygen species and disrupts hematopoiesis regulation. 27 Moreover, the role of CD34+ cells and ferritin on hematopoiesis was also proved by the notion that a low CD34+ cells dose and a high serum ferritin level were significant risk factors for poor graft function. 28 However, considering the association of these factors with OR was not found in our study and the number of cases was limited, the results of multivariate analysis need to be interpreted with caution.
This study has several limitations due to the retrospective nature and a small number of patients. First, it is uncertain whether the increase of platelet was result from rhTPO treatment or spontaneous recovery. Additionally, the cohort size was small and the setting of HSCT had heterogeneity, which may influence the accuracy of observed outcomes. Second, it would be better to measure the anti-rhTPO antibody prior and during rhTPO administration, despite neutralized anti-rhTPO antibody was not positive in previous results involving 33 and 81 patients, respectively.29,30 Further prospective randomized comparative studies are required to address these questions and make accurate assessments on the efficacy of rhTPO for treating SFPR. Moreover, there is an unmet need to identify the optimal regiment of rhTPO and comparison with other treatments at present.
In conclusion, this study observed significant increase of platelet counts and megakaryocytes in bone marrow after rhTPO therapy. RhTPO administered for SFPR patients led to a OR rate of 82.8% and CR rate of 34.5% without major adverse events. Multivariate analysis indicate that patients who received CD34+ cells ≥3 × 106/kg during HSCT or had a decreased ferritin after rhTPO are more likely to achieve CR to rhTPO. These preliminary results suggest rhTPO appears to be a safe and effective treatment option for SFPR after HSCT.
Supplemental Material
sj-pdf-1-cath-10.1177_10760296211068037 - Supplemental material for Efficacy of Recombinant Human Thrombopoietin for the Treatment of Secondary Failure of Platelet Recovery After Allogeneic HSCT
Supplemental material, sj-pdf-1-cath-10.1177_10760296211068037 for Efficacy of Recombinant Human Thrombopoietin for the Treatment of Secondary Failure of Platelet Recovery After Allogeneic HSCT by Yigeng Cao, Mingyang Wang, Biao Shen, Fei Zhao, Rongli Zhang, Xin Chen, Yi He, Weihua Zhai, Qiaoling Ma, Jialin Wei, Yong Huang, Donglin Yang, Aiming Pang, Sizhou Feng, Erlie Jiang and Mingzhe Han in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by Tianjin Nature Science Foundation (20JCZDJC00410), National Nature Science Foundation of China (82170217, 82070192, 81670171), Fundamental Research Funds for the central universities (3332020052), Thrombocytopenia Funding from Yeehong Business School of Shenyang Pharmaceutical University (Sansheng TCP Young Research Funding, No. 57) and Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences (2019XK320076).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
