Abstract
Introduction
The new onset atrial fibrillation (NOAF) is a complication that should not be dismissed after an acute coronary syndrome since this association is characterized by a high ischemic risk.
Methods
We investigated the performance of C2HEST and the HARMS2-AF scores for predicting NOAF during hospitalization in patients admitted with ACS treated with percutaneous coronary intervention (PCI).
Results
Among the 763 patients eligible for the inclusion criteria, 64 patients developed NOAF during the index hospitalization, with an incidence of 8.38%. For the C2HEST score, the mean score in the NOAF group was higher than in the other group [NOAF versus No NOAF: 2.45 (± 1.79) versus 1.27 (± 1.43), p < 0.001], and ROC analysis showed good accuracy of C2HEST score in predicting NOAF with cut-off ≥3 [AUC = 0.699; 95%CI [0.665; 0.731], p < 0.001]. For the HARMS2-AF score, the mean score in the NOAF group was higher than in the other group [NOAF versus No NOAF: 9.22 (± 3.5) versus 6.72 (± 2.94), p < 0.001] and ROC analysis showed good accuracy of C2HEST score in predicting NOAF with cut-off≥7 [AUC = 0.722; 95%CI [0.689; 0.754], p < 0.001], and the comparison of the two scores found no significant difference with de long test p value at 0.326. A multivariate logistic regression analysis showed that C2HEST score >3 (OR = 1.86, p = 0.043) and HARMS2-AF >7 (OR = 3.91, p < 0.001) were independently associated with NOAF.
Conclusion
The C2HEST and HARMS2-AF are simple scores that can predict NOAF in patients with ACS treated with PCI with good accuracy.
Introduction
Acute coronary syndrome (ACS) is a major cardiovascular emergency whose prognosis primarily depends on the time required for revascularization, but above all on the complications it causes. 1 Arrhythmia is one of the major complications of the acute phase of ACS and is the leading cause of sudden death in the acute phase. 2 Arial fibrillation (AF) represents a complex association with ACS, given the high ischemic risk associated with this condition. 3 This AF can be either pre-existing or of new onset during the acute phase of ACS, with an incidence ranging from 6% to 21%, with a multifactorial mechanism involving atrial ischemia, the inflammatory response and the oxidative stress generated by myocardial ischemia. 4
Recently, two scores have been developed and validated to predict atrial fibrillation in the general population, namely the C2HEST score and the HARMS2-AF score, but to patient with ACS treated with PCI, the validity of these two scores has been poorly studied. With this aim, we investigated the validation of these two scores for predicting new onset AF (NOAF) during hospitalization in patients admitted with ACS treated with percutaneous coronary intervention (PCI).
Materials and Methods
Study Design and Aims
We performed a retrospective analysis including patients admitted to cardiac intensive care unit (CICU) for ACS treated with PCI to analyze the validation of C2HEST and HARMS2-AF scores to predict NOAF.
Population, Inclusion, and Exclusion Criteria
We included patients admitted to our ICU between 2021 and 2023 for ACS who had undergone PCI and who presented during hospitalization a NOAF. We excluded patients who had not undergone PCI, as well as those already diagnosed with AF prior to admission. All patients underwent telemetric monitoring to detect NOAF during hospitalization.
Data Extraction
Data were extracted from electronic medical records for demographic, clinical, electrical and biological parameters, as well as intra-hospital evolution. We then calculated the C2HEST and HARMS2-AF scores at admission for all patients.
Definition and Scores
An increase and/or decrease in high-sensitivity cardiac troponin above the 99th percentile, accompanied by at least one of the following: symptoms of ischemia, new ischemic changes on the ECG, pathological Q waves or signs of myocardial damage on imaging, with significant stenosis on coronary angiography (>50% for the left main and 70% for other arteries).
NOAF
A NOAF is defined as an irregular rhythm with absence of organized atrial activity in a patient with no history of AF prior to index hospitalization and during the duration of hospitalization in the CICU. This NOAF is detected by telemetric monitoring and confirmed by a 12-derivation surface ECG.
6
C2HEST
The C2HEST score is a simple clinical score for predicting atrial fibrillation in the general population, initially derived from a large Chinese cohort of 471,446 patients, with a Korean external validation cohort of 514,764 patients.
7
It included the following items: Coronary artery disease or chronic obstructive pulmonary disease [1 point each]; hypertension [1 point]; elderly [age ≥75 years, 2 points]; systolic HF [2 points]; thyroid disease [hyperthyroidism, 1 point]) with AF risk increasing significantly with higher C2HEST score.
HARMS2-AF
HARMS2-AF is a simple score for predicting AF derived from prospective data from 314,280 patients in the United Kingdom Biobank (UKB) and externally validated in 7171 patients in the Framingham Heart Study 8 : It included the following items: Hypertension [4 points]; age [Age 60–65 years, 1 point and age ≥65 years, 2 points]; BMI>30 kg/m2 [1 point]; Male sex [2 points]; Sleep apnoae [2 point], Smoking [1 point] and alcohol [7–14 standard drinks/week, 1 point and >14 standard drinks/week, 2 point], with a higher HARMS2-AF score (≥5 points) was associated with a heightened AF risk.
Statistical Analysis
Quantitative variables were described as mean and standard deviation, and analyzed between the two groups using the t-student test. Categorical variables were described as numbers and analyzed between the two groups using Pearson's chi-square test or Fisher's exact test.
Receiver characteristic curves were generated for the predictive accuracy of NOAF using the C2HEST and HARMS2-AF score. The area under the curve (AUC) of the two scores was provided and compared, using the De Long test.
Multivariable logistic regression analysis was performed to investigate factors predictive of NOAF by integrating C2HEST and HARMS2-AF according to the cut-off determined in ROC analysis with associated 95% confidence interval (CI) and two-tailed p-value indicated.
All covariates that showed a statistically significant result in the univariate analysis, with associated 95% confidence interval (CI) and two-tailed p-value indicated. The Kaplan-Meier method was used to generate the survival curves for NOAF during hospitalization according to the different subgroups of C2HEST and HARMS2-AF scores, and the Log-Rank test was used to evaluate hypotheses concerning the differences observed in the survival curves. For all tests, a p-value of less than 0.05 was considered statistically significant. Data were collected and processed using IBM SPSS 26.0 statistical software.
Ethical Approval
Given the retrospective design of this study, the local ethics committee waived the requirement for patient consent. No formal ethical approval was required for this study. Data anonymity was respected in accordance with national and international guidelines. Access to patient data was authorized and approved by CHU Mohammed VI.
Results
A) General characteristics
Among the 763 patients eligible for the inclusion criteria, 64 patients developed NOAF during the index hospitalization, with an incidence of 8.38%. Patients with NOAF were older [NOAF versus No NOAF: 67.89 years (± 9.7) versus 62.53 years (± 10.1), p < 0.001], with a higher length of hospital stay 5.8 days (± 4.4) versus 3.6 days (± 1.3), p < 0.001). However, no significant differences were observed for the different cardiovascular risk factors including hypertension (45.3% vs 38.5%, p = 0.174), diabetes (48.4% vs 37. 9%, p = 0.06), and chronic kidney disease (1.6% vs 1.3%, p = 0.214), as well as for prior coronary artery disease (9.4% vs 8.4%, p = 0.971) or prior heart failure (1.6% vs 1.7%, p = 0.897) (Table 1).
Comparison of Basic Characteristics of Patients with Acute Coronary Syndrome According to NOAF.

Abbreviations: LBBB, Left Bundle Branch Block; RBBB, Left Bundle Branch Block; EF, Ejection fraction; LV, Left ventricule; RV, Right ventricle; LM, Left main; LAD, left anterior descending; LCX, Left circumflex artery; RCA, Right coronary artery; HB, haemoglobin; GFR, Glomerular filtration rate; CRP, C-reactive protein; WBC, White blood cell; CICU, Cardiovascular intensi ve care unit; PCI: Percutaneous coronary intervention; TIMI, Thrombolysis in myocardial infarction; VA, ventricular arrhythmia CD, conduction disorder; MACE, Major adverse cardiovascular events.
Patients with NOAF consulted later than others (19 h (± 20.2) for NOAF versus 15.1 h (± 10.3) for No NOAF, p < 0.001), and had more severe heart failure symptoms (20. 3% of patients with NOAF had a New York Heart Association functional class III/ IV at admission vs 6.7% in those with No NOAF, P < .001), but with no difference in heart rate at admission (p = 0.192) or systolic blood pressure (p = 0.11). Concerning electrocardiogram findings, no difference was observed between the two groups regarding the presence or not of persistent ST segment elevation (p = 0.094), ST segment elevation in the AVR lead (p = 0.062), and the presence of left bundle branch block (p = 0.165) or right bundle branch block (p = 0.210) (Table 1).
On echocardiography, patients with NOAF had a more impaired ejection fraction than others (40.98% vs 48.27%, p < 0.001), with a higher proportion of patients with EF < 40% (48.4% vs 23.6%, p < 0. 001), with left ventricular diastolic dysfunction (31.3% vs 12%, p < 0.001), with right ventricular systolic dysfunction (15.6% vs 5.6%, p = 0.005) and left atrial enlargement (32.8% vs 8.9%, p < 0.001)
There was no significant difference in coronary angiography results or in the culprit artery. However, pre-PCI TIMI flow was more altered in the NOAF group (40.6% with pre-PCI TIMI < III) versus the other group (28.5% with pre-PCI TIMI < III) (p < 0.001) (Table 1).
In terms of biological results, patients in the NOAF group had more impaired renal function, with GFR <30 ml/min/1.73 m2 in 20.3% versus 11.3% in patients without NOAF (p = 0.033), as well as higher white blood cell counts (21.6% vs 6.6%; p < 0.001). The use of thrombolysis did not differ between the two groups, but the use of vasopressors and inotropes was higher in the NOAF group (12.5% vs 4.6%; p = 0.014) (Table 1).
In terms of in-hospital outcomes, the NOAF group had more acute heart failure (18.8% vs 5.9%; p = 0.001), more thromboembolic events (11% vs 3.1%; p < 0.001), and more cardiogenic shock (9.4% vs 3.7%; p = 0.04). However, no difference was observed in hemorrhagic events (p = 0.892), Life-threatening Ventricular arrhythmia (p = 0.186), Life-threatening conductance disorders (p = 0.057) and all-cause mortality at 30 days (p = 0. 637) (Table 1).
B) Receiver characteristic curves analysis for using the C2HEST and HARMS2-AF score to predict NOAF
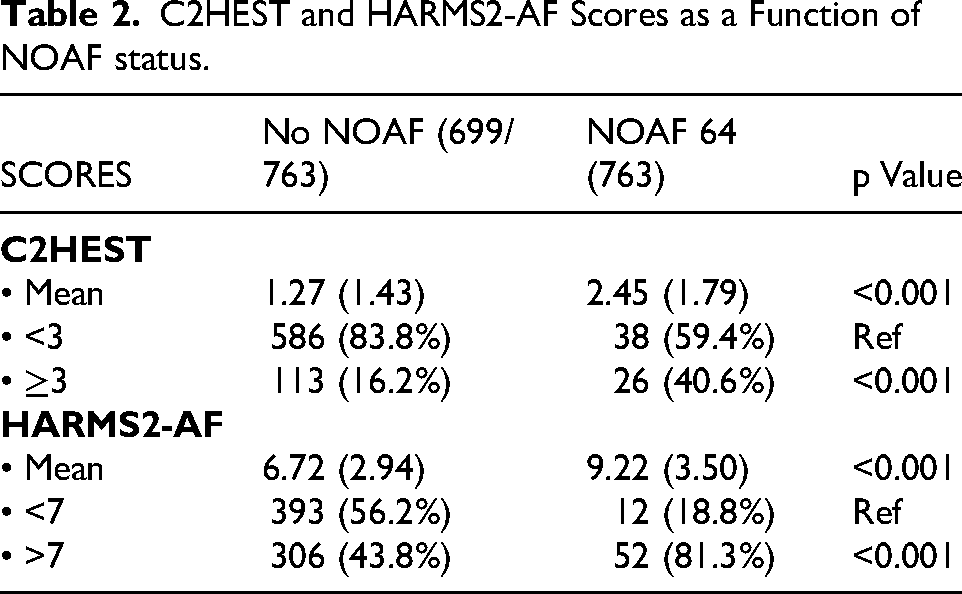
For the C2HEST score, the mean score in the NOAF group was higher than in the other group [NOAF versus No NOAF: 2.45 (± 1.79) versus 1.27 (± 1.43), p < 0.001] (Table 2). ROC analysis showed good accuracy of C2HEST score in predicting NOAF with cut-off ≥3 [AUC = 0.699; 95%CI [0.665; 0.731], p < 0.001] (Table 3) (Figure 1). The proportion of patients with C2HEST ≥3was higher in the NOAF group, with a statistically significant difference [NOAF versus No NOAF: 40.6% versus 16.2%, p < 0.001] (Table 2).

Comparison of receiver-operating characteristic (ROC) curves for predicting NOAF in-CICU according to C2HEST and HARMS2-AF score.
C2HEST and HARMS2-AF Scores as a Function of NOAF status.
Prediction of NOAF by C2HEST and HARMS2-AF Score.

•
Kaplan-Meier survival analysis of the risk of AF demonstrated a relationship between an increase in C2HEST risk score ≥3and the development of NOAF over the duration of hospitalization (Log-Rank test pValue=0.001) (Figure 2).

Kaplan-Meier survival analysis of the risk of NOAF according to C2HEST score.
For the HARMS2-AF score, the mean score in the NOAF group was higher than in the other group [NOAF versus No NOAF: 9.22 (± 3.5) versus 6.72 (± 2.94), p < 0.001] (Table 2). ROC analysis showed good accuracy of C2HEST score in predicting NOAF with cut-off≥7 [AUC = 0.722; 95%CI [0.689; 0.754], p < 0.001] (Table 3) (Figure 1). The proportion of patients with HARMS2-AF ≥7 was higher in the NOAF group, with a statistically significant difference [NOAF versus No NOAF: 81.3% versus 43.8%, p < 0.001] (Table 2).
Kaplan-Meier survival analysis of the risk of AF demonstrated a relationship between an increase in HARMS2-AF risk score ≥7 and the development of NOAF over the duration of hospitalization (Log-Rank test pValue<0.001) (Figure 3).

Kaplan-Meier survival analysis of the risk of NOAF according to HARSM2-AF.
The comparison of the C2HEST and HARMS2-AF at admission with to predict NOAF found no significant difference with de long test p value at 0.326.
C) Multivariable logistic regression
A multivariate logistic regression analysis showed that NOAF is independently associated with C2HEST score >3 (OR = 1.86, 95%CI [1.37; 4.67], p = 0.043), HARMS2-AF >7 (OR = 3.91, 95%CI [2.19; 7.47], p < 0.001). Adding to this predicting factors, RV systolic dysfunction (OR = 2.8), Atrial enlargement (OR = 3.83), Use of Vasopressor (OR = 2.91), and ejection fraction<40% (OR = 1.73) (Table 4).
Logistic Regression Analysis of Risk Factors of NOAF.

RV : Right ventricule
CKD: Chronic kidney disease
PCI: Percutaneous coronary intervention
TIMI: Thrombolysis in myocardial infarction
GFR: Glomerular filtration rate
Discussion
The main results of our study are : I) The incidence of NOAF after ACS is 8.3%, which is in accordance with the results published in the literature. II) A C2HEST score ≥3 remains a valid tool for predicting this NOAF with good accuracy [AUC = 0.699; 95%CI [0.665; 0.731], p < 0.001]. III) A HARMS2-AF score ≥7 is thus a good tool for predicting NOAF with good accuracy [AUC = 0.722; 95%CI [0.689; 0.754], p < 0.001]. IV) No significant difference between C2HEST and HARMS2-AF score to predict NOAF (De-long test =0.326).
Several mechanisms contribute to the development of NOAF during ACS including sinus node ischemia, tissue hypoxia, increased atrial pressure and neurohormonal activation.9,10 Thus. NOAF can further aggravate ventricular dysfunction by increasing oxygen demand with increased cardiac output in a failing ventricle, subsequently exposing it to complications principally ischemic stroke, ventricular arrhythmias, re-infarction and death.11,12
This NOAF can occur early or late after ACS, and with different pathophysiological mechanisms. NOAF that occurs early, within the first 24 h, is secondary to atrial ischemia with occlusion of the atrial branches, and generally has no prognostic impact. 13 However, late NOAF has a more complex mechanism with a more significant prognostic impact.13,14
In this sense, and given the morbidity and mortality of AF, several clinical scores have been studied to predict AF in the general population. Citing the CHARGE-AF score developed from a pooled analysis of 18,556 patients included in three major American studies (FHS [Framingham Heart Study], ARIC (Atherosclerosis Risk in Communities) and CHS (Cardiovascular Health Study) studies, and validated in 7672 participants included in the RS (Rotterdam Study) and AGES (Age, Gene and Environment-Reykjavik) studies. The analysis was based on 1186 incident cases of AF in the derivation cohorts and 585 in the validation cohorts. A 5-year simple predictive model including the variables age, height, weight, race, diastolic and systolic diastolic blood pressure, use of antihypertensive drugs, current smoking, diabetes, and history of heart failure and myocardial infarction showed good accuracy (AUC, 0.765; 95% CI, 0.748 to 0.781). 15
The ARICc (Atherosclerosis Risk In Communities) score derived from 14,546 patients included in the prospective ARIC (Atherosclerosis Risk In Communities) study. After 10 years of follow-up, 515 cases of AF were recorded, with age, height, race, diabetes, smoking, precordial murmur, use of antihypertensive medication, systolic blood pressure, left atrial and ventricular hypertrophy, and coronary artery disease or heart failure as clinical parameters associated with this AF with good accuracy (AUC 0.78). 16
The accuracy of the C2HEST score in predicting NOAF was assessed in a large meta-analysis of 17 studies, including a total of 11,067,496 patients, of whom 307,869 (2.78%) were diagnosed with NOAF. The pooled results showed an AUC of 0.70 (95% CI: 0.66-0.74). These results remain significant regardless of the origin of the patients or the indication, whether primary or secondary prevention. The best results were observed in patients aged <50 years, with an AUC of 0.78 (95% CI 0.76-0.79). 17
In a large meta-analysis by Salerno et al, 4 which included 151735 patients from 7 observational studies. NOAF was observed in 6597 patients (4.3%). NOAF was associated with an increase in ischemic stroke (HR 2.38, 95% CI: 1.64–3.44; P < 0.01; I2 = 50.2%9, an increase in all-cause mortality (HR 1. 36, 95% CI: 1.08–1.71; P = 0.0089; I2 = 53.25%) and finally an increased risk of AF recurrence (HR 4.68, 95% CI: 2.07–10.59; P = 0.0002; I2 = 50.2%). This means that although NOAF lasts for a short duration, its prognostic impact is important, and therefore its early detection remains of great interest. Although several data have been published to study the predictive factors of this NOAF, such as chronic kidney disease 18 or cardiogenic choc, no score has yet been developed in this sense, which will help practitioners to personalize management according to the risk of NOAF development.
For NOAF after ACS, and to our knowledge, only one study has analyzed the C2HEST score to predict NOAF: this was a prospective study of 555 patients from the Real-world observational Registry of Acute Coronary Syndrome (REALE-ACS) conducted by Flavio and al. 18 NOAF was observed in 8.1% of included patients, with a higher mean C2HEST score in the NOAF group (4.2 +/–1.7 Vs 3+/–1.5; p < 0.001) and A C2HEST score > 3 was associated with NOAF occurrence (odds ratio 4.33, 95% confidence interval 2.19–8.59, p < 0.001) and ROC curve analysis showed good accuracy of the C2HEST score (AUC 0.71, 95%CI 0.67–0.74). However, to our knowledge, no data have been published for the HARMS2-AF score in this sense.
Based on our results and those published, the question of clinical application leads us to propose two practical options. The first is to extend the duration of telemetric monitoring in patients with high scores, and the second is to indicate curative anticoagulation in these group of patients. Both options are reasonable given the high incidence of NOAF in patients with high C2HEST and HARMS2-AF scores. For this reason, our results may serve as a rationale for a clinical trial in this direction.
Limitations
As with any study, our study has its limitations, firstly its retrospective design that may underestimate transient NOAF episodes, and secondly the size of the sample. However, to be sure of generalizing our results, we need more studies evaluating these two scores, so as to be able to hierarchize the results in meta-analyses. Another limitation is that these two scores do not include other risk factors such as valvular heart disease and protective factors such as statin use.
Conclusion
NOAF is a major silent complication during the acute phase of ACS, with a major prognostic impact. These patients must be properly screened to improve their outcome, and C2HEST and HARMS2-AF are simple scores that can predict NOAF with good accuracy.
Footnotes
Acknowledgment
None.
Ethical Approval
Not required.
Consent to Publication
Not required.
Author Contributions
A.B developed the project idea, writing and revising the proofs.
W.A contributed to data collection.
S.B, Z.B, N.I, and N.E contributed to patient management
N.E supervised the project.
All authors contributed to the article and approved the submitted version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data underlying this article are available in the article.
