Abstract
Introduction
Even after percutaneous coronary intervention (PCI), some patients with coronary heart disease (CHD) still have cardiac and cerebrovascular adverse events. Patients with CHD and diabetes mellitus (DM) often have worse clinical outcomes. 1 It is generally believed that patients with CHD and DM not only have a higher risk of thrombotic events, but also an increased risk of bleeding events, 2 the mechanisms of which may be related to the abnormal glucose metabolism and inflammatory response, including endothelial dysfunction, abnormal prethrombotic coagulation, enhanced oxidative stress, and platelet dysfunction.3,4 It is important to apply reasonable and effective antithrombotic strategies in patients with CHD complicated with DM.5,6
Bivalirudin is a direct thrombin inhibitor, which is used in anticoagulant therapy during PCI, and compared with unfractioned heparin (UFH), it has the advantages of specificity, reversibility, short half-life, and stronger dose response on thrombin inhibition. 7 Previously, EUROMAX (The European Ambulance Acute Coronary Syndrome Angiography) trial 8 showed that compared with UFH, bivalirudin reduced the risk of bleeding events in patients undergoing PCI, but increased the risk of stent thrombosis. Nevertheless, BRIGHT (Bivalirudin in Acute Myocardial Infarction vs Heparin and GPI Plus Heparin Trial) 9 showed that continuous postoperative infusion of bivalirudin reduced the risk of bleeding without increasing the risk of stent thrombosis and major adverse cardiovascular events (MACE). In patients with DM, Horizons-AMI (Harmonizing Outcomes with Revascularization and Stents in Acute Myocardial Infarction) Trial of DM subgroup analysis 10 suggested that DM was not an independent risk factor for bivalirudin in acute myocardial infarction patients undergoing primary PCI. However, whether DM is an independent risk factor for thrombotic and bleeding events in elective PCI patients anticoagulated with bivalirudin has not been reported before.
Therefore, it is urgent to study whether DM affects the occurrence of thrombotic and bleeding events after elective PCI in patients anticoagulated with bivalirudin. This study intends to investigate the impact of DM on one-year thrombotic and bleeding events in patients undergoing elective PCI and anticoagulated with bivalirudin, so as to provide reference for clinical decision-making.
Methods
Study Design and Population
This study was a prospective and multicenter cohort study. Consecutive patients who underwent elective PCI and were anticoagulated with bivalirudin were enrolled from January 2017 to August 2018 in Fuwai Hospital, Northern Theatre General Hospital, and Xinxiang Central Hospital. The inclusion criteria were as follows: patients were aged from 18–85 years old, underwent elective PCI, and were anticoagulated with bivalirudin in the operation. The exclusion criteria were as follows: acute coronary syndrome (ACS) patients who underwent primary PCI, or patients who took oral anticoagulants with warfarin or non-vitamin K antagonists. Finally, 1152 patients were included in the current analysis. The Declaration of Helsinki protocols were followed. The research protocol was approved by the ethics committee of Fuwai Hospital (Ethics number: 2018-1107). All participants signed informed consents and agreed to follow-up.
Definition and Group Division of DM
DM was defined according to the guidelines on Prevention and Treatment of Diabetes in China. The diagnostic criteria for diabetes were diabetic symptoms with random venous blood glucose levels ≥ 11.1 mmol/L, fasting venous blood glucose levels ≥ 7.0 mmol/L, oral glucose tolerance test 2-h blood glucose levels ≥ 11.1 mmol/L, or use of antidiabetic drugs. According to the diagnosis of DM, patients were divided into DM group (n = 472) and non-DM group (n = 680).
Study Procedures
Patients were routinely given aspirin and P2Y12 receptor inhibitor (clopidogrel or ticagrelor) preoperatively, while patients who had not previously taken P2Y12 receptor inhibitor were given a loading dose of clopidogrel 300mg or ticagrelor 180 mg one day preoperatively. After PCI, patients were prescribed aspirin 100 mg once daily and clopidogrel 75 mg once daily (or ticagrelor 90 mg twice daily) for at least 1 year. Bivalirudin (Shenzhen Salubris Pharmaceuticals Co., Ltd) was given 0.75 mg/kg through intravenous injection during PCI, followed by 1.75 mg kg−1 h−1 intravenous infusion, and maintained until 3-4 h after PCI. The dose was adjusted according to activated clotting time. Clinical baseline information, procedural characteristics, and outcome data were collected in detail by independent research personnel.
Endpoints and Follow-Up
Primary endpoints included thrombotic endpoint and bleeding endpoint. The thrombotic endpoint was Major Adverse Cardiac and Cerebrovascular Events (MACCE), a composite endpoint consisting of all-cause death, myocardial infarction, stent thrombosis, ischemic revascularization, and stroke. Myocardial infarction was defined according to the third global general definition of myocardial infarction. 11 Stent thrombosis was classified as positive, probable or impossible according to the standard definition of Academic Research Consortium. 12 Stroke was diagnosed by a neurologist. The bleeding endpoint was according to the Bleeding Academic Research Consortium (BARC) 2, 3 or 5 bleeding. 13 Patients were followed-up at one year after discharge by hospital visit or telephone, and the occurrence of MACCE and bleeding events were recorded in detail. All events were confirmed and recorded by two cardiologists.
Statistical Analysis
Mean with standard deviation was used to describe normally distributed continuous variables, and Student’s t-test was used to compare the differences. M (Q1, Q3) was used to describe abnormally distributed continuous variables, and Mann-Whitney U test was used to compare the differences. Frequency and percentage were used to describe categorical variables, and Pearson chi-square test and Fisher's exact probability method were used to compare the differences. Kaplan-Meier curve analysis was used to estimate the cumulative survival rates, and log-rank test was used for statistical test. Univariate and multivariate Cox regression analyses were used to explore the influencing factors of MACCE and bleeding events. The Cox proportional hazard model was used to estimate hazard ratios (HRs) and their corresponding confidence intervals (CIs) for the MACCE and BARC 2, 3 or 5 bleeding events. In multivariate Cox regression analysis, MACCE and bleeding events were taken as dependent variables, and DM, gender, age, body mass index (BMI) and factors with statistically significant differences in univariate Cox regression analysis were included as independent variables. All statistical tests were two-sided with a significant level of 0.05. Statistical analyses were performed with SPSS 25.0 software (IBM Corp., Armonk, New York, USA).
Results
Patients’ Characteristics
A total of 1152 patients undergoing elective PCI and anticoagulated with bivalirudin were included in the study. Among them, 472 patients (41.0%) were in the DM group and 680 patients (59.0%) were in the non-DM group. The age of the enrolled patients was (68.6 ± 10.1) years old, including 781 males (67.8%). Among these patients, 178 (15.5%) had ST-elevation myocardial infarction, 115 (10.0%) had non-ST-elevation myocardial infarction, and 859 (74.6%) had angina pectoris. There were 982 patients (85.2%) taking clopidogrel, and 170 patients (14.8%) taking ticagrelor. Compared with the non-DM group, the DM group had more females, higher BMI, more hyperlipidemia and hypertension, less smoking history, more myocardial infarction and PCI history, and lower low-density lipoprotein cholesterol (all P < .05) (Table 1).
Baseline Characteristics of Study Population.

Abbreviations: DM, diabetes mellitus; BMI, body mass index; COPD, chronic obstructive pulmonary disease; CHD, coronary heart disease; CVD, cerebrovascular disease; PAD, peripheral artery disease; MI, myocardial infarction; PCI, percutaneous coronary intervention; CABG, coronary artery bypass grafting; LVEF, left ventricular ejection fraction; WBC, white blood count; HB, hemoglobin; PLT, platelet; LDL-C, low density lipoprotein cholesterol; hs-CRP, high sensitive – C reactive protein; IABP, intra-aortic balloon pump.
Clinical Outcomes and Kaplan-Meier Curve Analysis
All patients completed one-year follow-up, and there were 89 (7.7%) MACCE, and 21 (1.8%) BARC 2, 3 or 5 bleeding events occurred. MACCE (8.1% vs 7.5%, P = .731) and its components (including all-cause death, myocardial infarction, ischemic revascularization, stent thrombosis, and stroke), and BARC 2, 3 or 5 bleeding (1.5% vs 2.1%, P = .473) had no statistically significant difference between DM-group and non-DM group (all P > .05) (Table 2). Kaplan-Meier curve analysis showed there was no significant difference in the cumulative incidence of MACCE and BARC 2, 3 or 5 bleeding (log-rank test, all P > .05) (Figure 1).

Kaplan-Meier curve analysis of MACCE (A) and BARC 2, 3 or 5 bleeding (B).
Clinical Outcomes Between DM and Non-DM Group.

Abbreviations: MACCE, major adverse cardiac and cerebrovascular events; BARC, bleeding academic research consortium.
Univariate Cox Regression Analysis
Univariate Cox regression analysis showed DM had no effect on the occurrence of one-year MACCE in patients anticoagulated with bivalirudin during elective PCI (HR: 1.081, 95%CI: 0.711-1.646, P = .715), while peripheral artery disease (PAD) history (HR: 2.238, 95%CI: 1.319-3.796, P = .003) and white blood cell counts (HR: 1.110, 95%CI: 1.004-1.227, P = .042) were associated with MACCE; DM had no effect on the occurrence of one-year BARC 2, 3 or 5 bleeding (HR: 0.718, 95%CI: 0.290-1.779, P = .474), while age (HR: 1.070, 95%CI: 1.011-1.131, P = .018), PAD history (HR: 2.913, 95%CI: 1.067-7.952, P = .037), and hemoglobin level (HR: 0.970, 95%CI: 0.948-0.993, P = .010) were associated with BARC 2, 3 or 5 bleeding. (Table 3)
Univariate Cox Regression Analysis of MACCE and BARC 2, 3 or 5 Bleeding.
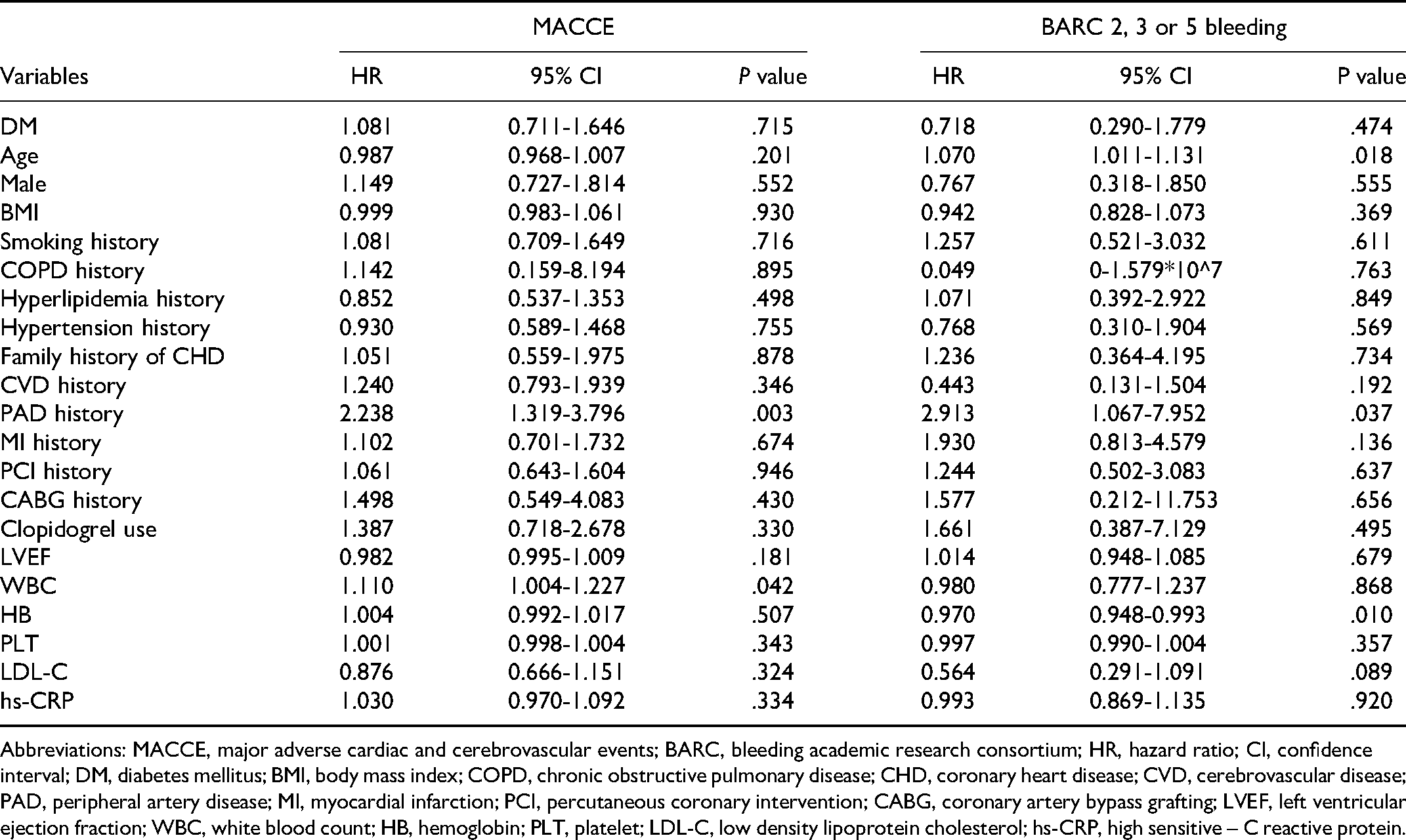
Abbreviations: MACCE, major adverse cardiac and cerebrovascular events; BARC, bleeding academic research consortium; HR, hazard ratio; CI, confidence interval; DM, diabetes mellitus; BMI, body mass index; COPD, chronic obstructive pulmonary disease; CHD, coronary heart disease; CVD, cerebrovascular disease; PAD, peripheral artery disease; MI, myocardial infarction; PCI, percutaneous coronary intervention; CABG, coronary artery bypass grafting; LVEF, left ventricular ejection fraction; WBC, white blood count; HB, hemoglobin; PLT, platelet; LDL-C, low density lipoprotein cholesterol; hs-CRP, high sensitive – C reactive protein.
Multivariate Cox Regression Analysis
After adjusting for age, gender, BMI, and factors with statistically significant differences in univariate Cox regression analysis, multivariate Cox regression analysis showed that DM was not an independent risk factor for one-year MACCE in patients anticoagulated with bivalirudin during elective PCI (HR: 1.029, 95%CI: 0.674-1.573, P = .893), while PAD history (HR: 2.200, 95%CI: 1.290-3.751, P = .004) was an independent risk factor for MACCE; DM was not an independent risk factor for BARC 2, 3 or 5 bleeding (HR: 0.732, 95%CI: 0.293-1.831, P = .505), while PAD history (HR: 3.029, 95%CI: 1.102-8.332, P = .032) and low hemoglobin level (HR: 0.972, 95%CI: 0.947-0.998, P = .036) were independent risk factors for BARC 2, 3 or 5 bleeding. (Table 4)
Multivariate Cox Regression Analysis of MACCE and BARC 2, 3 or 5 Bleeding.
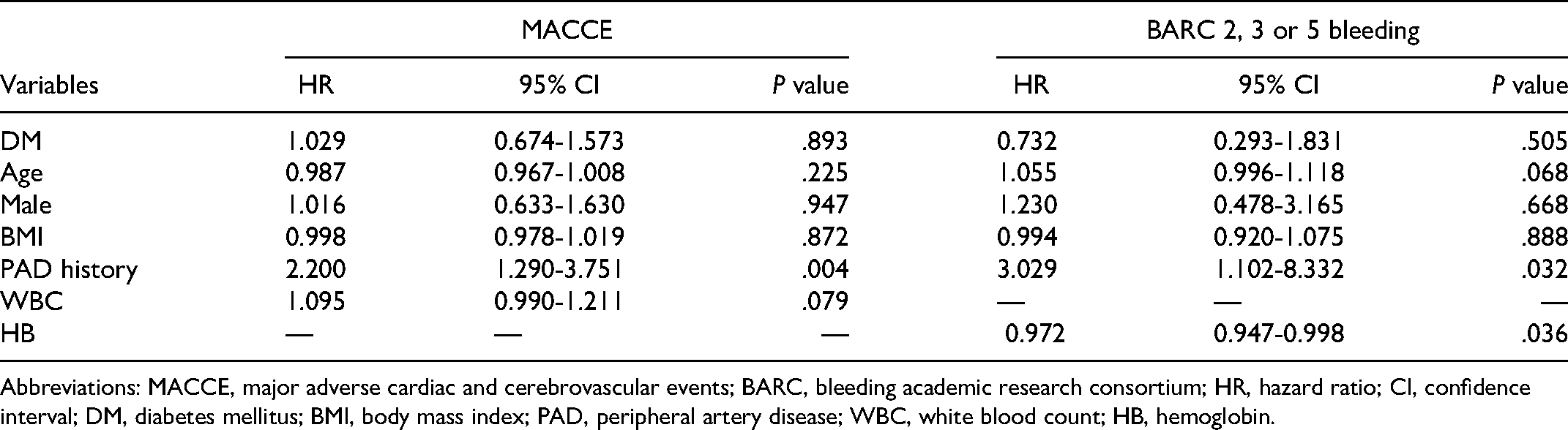
Abbreviations: MACCE, major adverse cardiac and cerebrovascular events; BARC, bleeding academic research consortium; HR, hazard ratio; CI, confidence interval; DM, diabetes mellitus; BMI, body mass index; PAD, peripheral artery disease; WBC, white blood count; HB, hemoglobin.
Discussion
This study is the first to investigate whether DM affects the occurrence of one-year MACCE and bleeding events in patients undergoing elective PCI and anticoagulated with bivalirudin. The results showed as follows: DM did not affect the occurrence of one-year MACCE in patients anticoagulated with bivalirudin during elective PCI, while PAD history was an independent risk factor for MACCE; DM did not affect the occurrence of one-year BARC 2, 3 or 5 bleeding in patients anticoagulated with bivalirudin during elective PCI, while PAD history and low hemoglobin level were independent risk factors for BARC 2, 3 or 5 bleeding.
In this study, DM did not affect the occurrence of one-year MACCE in patients anticoagulated with bivalirudin during elective PCI. At present, the conclusions about whether DM can affect the thrombotic events in patients undergoing PCI are not uniform. For patients anticoagulated with heparin during PCI, EPICOR Asia Study (long-term follow-up of Antithrombotic Management Patterns in Acute Coronary Syndrome Patients in Asia Study) 14 indicated that, DM significantly increased the risk of two-year thrombotic events after PCI in ACS patients. However, Wang HH et al 15 did not find that DM was an independent predictor of two-year MACE after PCI in a cohort study of 10 724 patients undergoing PCI, including patients with ACS and chronic coronary syndrome. In a five-year long-term follow-up study, Zibaeenezhad et al 16 also concluded that DM could not be an independent predictor of MACE after PCI. The above results are inconsistent, which may be related to whether the ACS population are included, and additionally, the patients’ year of inclusion. It should be noted that heparin was used for anticoagulation in all the studies above. We reported for the first time that, in patients anticoagulated with bivalirudin during elective PCI, DM was not a risk factor for one-year MACCE. However, our study enrolled patients form January 2017 to August 2018, which is not relatively new. In recent years, the new drug therapies and drug-eluting stents are developing rapidly in CHD area. New hypoglycemic drugs, such as sodium-glucose cotransporter protein-2 inhibitors and glucagon-like peptide-1 receptor agonists, were reported to lower death, non-fatal myocardial infarction and other cardiovascular outcomes in DM patients. 17 As a result, further investigation could be carried out to test their effect in DM patients undergoing elective PCI and anticoagulated with bivalirudin.
In addition, our study also found that PAD history was an independent risk factor for one-year MACCE in patients anticoagulated with bivalirudin during elective PCI. In patients with stable CHD, Cheng et al 18 suggested that PAD was an independent risk factor for cardiovascular events after PCI. Franzone et al 19 showed that among patients with stable CHD and ACS who received PCI, patients with PAD had poor prognosis. Our findings are consistent with the above studies, suggesting that PAD history is an independent risk factor for thrombotic events after PCI regardless of using bivalirudin or heparin. The current guidelines also recommend focusing on revascularization strategies for CHD patients with PAD, so as to reduce the occurrence of adverse clinical outcomes in such patients. 5
Our study also indicated that DM did not affect the occurrence of one-year BARC 2, 3 or 5 bleeding in patients anticoagulated with bivalirudin during elective PCI. At present, the conclusions about whether DM can affect the bleeding events in patients undergoing PCI are also controversial. Recently, Luca et al 20 showed that DM did not increase the risk of one-year ischemic events after PCI in elderly patients anticoagulated with heparin, but significantly increased the risk of one-year bleeding events after PCI. Similarly, Jarrah et al's study 2 on PCI population showed that DM significantly increased the risk of bleeding after PCI. However, Guan S et al's study 14 on the ACS population and Pembele et al's study 21 both showed that diabetes was not an independent influencing factor of bleeding events after PCI. Notably, heparin was used in all of the studies above, while bivalirudin was used in our study. Our study showed that DM was not a risk factor for increased bleeding events in patients anticoagulated with bivalirudin during elective PCI. The reason for this result may be related to the attention paid to patients with high bleeding risk in recent years, so that clinicians have taken effective management of relevant patients. More patients with high bleeding risk have used proton pump inhibitors, and the majority of patients choose the radial artery approach in PCI procedure. Evidence supported that the above measures could reduce the risk of bleeding after PCI,22,23 and the application of bivalirudin in DM patients also reduced the risk of bleeding events compared with heparin. 24
Additionally, our study also found that PAD history and low hemoglobin level were independent risk factors for one-year BARC 2, 3 or 5 bleeding in patients anticoagulated with bivalirudin during elective PCI. Previous studies using heparin for anticoagulation also supported the independent correlation between PVD and hemoglobin and bleeding events after PCI. PAD history is an important component of the CREDO-Kyoto bleeding prediction model, 25 and it is also an important indicator of the CRUSADE bleeding score. 26 Low baseline hemoglobin level is an important risk factor in PARIS score 27 and PRECISE-DAPT score. 28 Kalra et al 29 suggested that low baseline hemoglobin level was associated with an increased risk of bleeding after PCI for stable CHD patients. Faggioni et al 30 demonstrated that low baseline hemoglobin level was an independent risk factor for bleeding after PCI in ACS patients. These conclusions are consistent with the results of our study, in which patients were anticoagulated with bivalirudin. The current guidelines also recommend focusing on CHD patients with anemia to reduce the incidence of bleeding outcomes in these patients. 6
In summary, the impact of diabetes on patients anticoagulated with heparin during PCI remains controversial. We conducted an exploratory study in the real world of patients undergoing elective PCI and anticoagulated with bivalirudin, and the results showed that DM was not a risk factor for one-year MACCE and bleeding events, which indicates that the use of bivalirudin is not affected by DM. In addition, our study suggests that in elective PCI patients anticoagulated with bivalirudin, more attention should be paid to PAD history and hemoglobin level to identify patients with potential high risk of thrombotic and bleeding events, and individualized treatment may reduce the occurrence of adverse clinical outcomes.
Limitations
There are some limitations in our study. First, this study is an observational study, and randomized controlled trial with a larger sample size could be carried out in the future to evaluate the impact of DM on clinical outcomes in patients anticoagulated with bivalirudin during elective PCI. Second, this study enrolled patients form January 2017 to August 2018, which is not relatively new and did not evaluate the effect of new drug therapy on diabetes. Third, the lack of duration of DM may be a limitation of this study.
Conclusions
In patients undergoing elective PCI and anticoagulated with bivalirudin, DM was not an independent risk factor for one-year thrombotic and bleeding events. More attention should be paid to PAD history and hemoglobin level to identify patients with potential high risk of thrombotic and bleeding events.
Footnotes
Acknowledgements
The authors are grateful to the individuals who participated in this study, and grateful to all the funding supported us: the CS Optimizing Antithrombotic Research Fund (CRFH20180082), Young and middle-aged talents in the XPCC Science and Technology Project (2020CB012), CAMS Innovation Fund for Medical Sciences (CIFMS) (2020-I2M-C&T-B-052) and the National Key Research and Development Program of China (No. 2016YFC1301300 and No. 2016YFC1301301).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the National Key Research and Development Program of China, Young and middle-aged talents in the XPCC Science and Technology Project, the CS Optimizing Antithrombotic Research Fund, CAMS Innovation Fund for Medical Sciences (CIFMS), (grant number No. 2016YFC1301300 and No. 2016YFC1301301, 2020CB012, CRFH20180082, 2020-I2M-C&T-B-052).
Ethics Approval and Consent to Participate
Ethical approval to report this study was obtained from the local ethics committee of the Fuwai hospital‘s Research Ethics Committee (Ethics number: 2018-1107). Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
