Abstract
Background
Venous thromboembolism (VTE) is a potentially life-threatening complication in patients undergoing total hip arthroplasty (THA) and total knee arthroplasty (TKA). Multiple antithrombotic agents are routinely used to prevent VTE after THA and TKA. However, it remains unclear which anticoagulant is optimal in real-world clinical practice. This study aimed to assess the effectiveness and safety of aspirin and other antithrombotic agents for VTE prophylaxis following THA and TKA.
Methods
Several electronic databases, including PubMed, Embase, and the Cochrane Library, were systematically searched from inception to January 13th, 2025. The search identified real-world studies assessing the efficacy or safety of aspirin versus other anticoagulants for VTE prophylaxis after THA or TKA. The pooled rate of thromboembolic and bleeding events was estimated using a random-effects model. Two reviewers independently screened the literature and extracted data. Subgroup analyses were conducted based on study type, dose regimen and geographic region.
Results
A total of 41 studies were identified for inclusion. The overall incidence of VTE after THA and TKA with low-molecular-weight heparin (LMWH) was 1.20% (95% CI: 0.90%-1.60%). The incidence of VTE for aspirin, warfarin and direct oral anticoagulants (DOACs) was 0.80%, 0.9% and 0.90%, respectively. The pooled incidence of major bleeding for aspirin (1.90%, 95% CI: 0.00%-4.60%) was statistically significantly different from that in patients receiving LMWH (3.50%, 95% CI: 0.00%-7.80%) and DOACs (3.10%, 95% CI: 1.20%- 5.00%). When analyzing regional subgroups among several antithrombotic agents, VTE rate was significantly higher in Asia (LMWH: 21.80%, aspirin: 2.40%, DOACs: 9.40%) compared to other regions. In terms of major bleeding, the higher rate associated with DOACs (5.00%, 95% CI: 0.90%-9.20%) was found in North America compared to other regions, especially among patients treated with rivaroxaban (4.10%, 95% CI: 1.20%-7.00%).
Conclusion
To date, this study provides the most comprehensive real-world evidence regarding antithrombotic agents for VTE prophylaxis after THA and TKA. Aspirin appears to be as effective as other antithrombotic agents for VTE prophylaxis following THA and TKA. Furthermore, its use is associated with a significantly lower risk of bleeding. Nevertheless, these findings should be interpreted carefully.
Keywords
Introduction
Venous thromboembolism (VTE), including deep vein thrombosis (DVT) and pulmonary embolism (PE), is a potentially life-threatening complication following total hip arthroplasty (THA) and total knee arthroplasty (TKA). 1 Although the incidence of VTE events in patients undergoing THA or TKA is estimated to be low, rate of VTE in those without prophylactic measures are high, leading to increased mortality and substantial medical costs, thus necessitating rigorous chemoprophylaxis.2,3 Currently, both anticoagulants and antiplatelet agents are used for VTE prophylaxis after THA and TKA, including aspirin, factor Xa-inhibitors, low-molecular-weight heparin (LMWH), direct oral anticoagulants (DOACs), and warfarin. Nevertheless, there remains no consensus on the optimal chemoprophylactic strategy for preventing VTE.
An optimal thromboprophylactic strategy should effectively prevent thrombosis, while minimizing the risk of bleeding complications. In current clinical practice, LMWH is widely used over other antithrombotic agents for thrombosis prophylaxis. However, its use is limited by drawbacks such as injection site reactions, heparin- induced thrombocytopenia, and bleeding risk. Besides LMWH, DOACs are also employed for pharmacological thromboprophylaxis in patients undergoing THA or TKA. 4 DOACs are reported to offer greater efficacy in VTE prevention, particularly among individuals at high risk.5,6 Aspirin, a widely used antiplatelet agent, has demonstrated comparable efficacy to other anticoagulants such as warfarin and DOACs in VTE prophylaxis. Additionally, due to its favorable safety profile, lack of requirement for routine monitoring, and cost-effectiveness, its clinical acceptability has expanded significantly.7–11 Based on accumulated evidence from previous studies, the American Academy of Orthopedic Surgeons (AAOS) has recommended aspirin as an alternative thromboprophylaxis agent.12,13 However, the bleeding risk in patients receiving aspirin after THA and TKA remains incompletely characterized.
In recent years, an increasing number of studies have evaluated the efficacy and safety of different anticoagulants for VTE prevention following THA or TKA.14–17 However, conflicting evidence exists regarding the comparative effectiveness and safety of aspirin versus other anticoagulants in real-world setting.18–20 A growing body of evidence suggests that, compared to non-aspirin anticoagulants, aspirin is effective in VTE prophylaxis and is associated with a lower risk of wound complications and infection.11,21,22 Conversely, some studies indicate that DOACs may be superior to aspirin without an increased risk of bleeding. 23 Furthermore, the complication profiles of these agents in patients undergoing THA and TKA are not yet fully elucidated. 24 Large-scale, real-world studies are therefore needed to better characterize the incidence of VTE and adverse outcomes across different antithrombotic regimens.
Given the current scarcity and inconsistency of evidence, this study aims to compare the real-world effectiveness and safety of various antithrombotic agents in preventing VTE among patients undergoing THA or TKA. The findings will provide comprehensive real-world evidence to inform optimal thromboprophylactic strategies in this patient population.
Methods
This study was conducted following the Systematic Reviews and Meta-analyses (PRISMA) guidelines, 25 and this study was pre-registered in the International Prospective Register of Systematic Reviews.
Search Strategy
A comprehensive literature search was performed independently by two reviewers, with language restriction of English. Electronic databases including PubMed, Embase, and the Cochrane Library were systematically searched from inception until January 13th, 2025. The detailed search strategy is provided in eTableS1. After removal of duplicate records, the eligibility of each identified study was independently reviewed by two investigators. To minimize the risk of missing relevant studies, reference lists of all included articles were manually screened. Any discrepancies were resolved through consultation with a third reviewer.
Selection of Studies
The inclusion criteria were as follows: (1) Real-world data from studies with case-control, observational, prospective or retrospective design; (2) Studies involving patients receiving antithrombotic therapy after THA or TKA; (3) Antithrombotic agents including aspirin, factor Xa-inhibitors, LMWH, DOACs, or warfarin, etc; (4) Studies reporting outcomes related to VTE, major bleeding, or thromboembolic events following THA or TKA. VTE was defined as deep vein thrombosis (DVT) or pulmonary embolism (PE) confirmed by clinical evaluation or diagnostic testing. Major bleeding was defined as intracranial hemorrhage, gastrointestinal bleed, or any bleeding requiring a return to theater. Low-dose aspirin was defined as ≤162 mg per day, and high-dose aspirin was defined as > 162 mg per day.
Exclusion criteria: (1) Incomplete outcome data; (2) Studies evaluating bleeding or thromboembolic events without anticoagulant or antiplatelet therapy; (3) Review articles, conference abstract, case report, editorials,study protocol.
Literature Screening and Data Extraction
Two reviewers independently conducted literature screening and extracted basic characteristics from the eligible studies. During literature screening, irrelevant literature was excluded based on title and abstract. Subsequently, full texts of potentially relevant articles were further determined in final analysis. The extracted data mainly included the following: study title, first author's name, publication year, region or country, patients characteristics, sample size, study design, type of antithrombotic agents, primary outcome (VTE, DVT, or PE), and secondary outcomes (major bleeding, or other complications). All data extraction was performed independently by two reviewers and compared at the end to minimize selection bias. In case of disagreements, a third reviewer reviewed the relevant records and resolved discrepancies through consensus.
Quality Assessment
Two investigators independently assessed the risk of bias in each included study using a modified version of Newcastle-Ottawa Scale (NOS), which evaluates the following domains: representativeness of sample population, sample size, participation rate, outcome assessment, and analytical methods to control for bias. 26 The overall score ranges from 0 to 9, with a cumulative score > 7 indicating high quality and a score < 4 indicating low quality (eTableS2). Any disagreement was resolved through consultation with a senior author.
Statistical Analyses
The outcomes were presented as pooled rate with corresponding 95% confidence interval (CI) using a random-effects model. 27 Heterogeneity across studies was assessed using I2 test, with I2 > 50% indicating significant heterogeneity. Subgroup analyses were conducted based on antithrombotic agents, study design, dose regimen, and geographical regions (Asia, North America, and Europe) to explore potential source of heterogeneity. Comparability between subgroups was evaluated through interaction analyses. Sensitivity analysis was conducted by sequentially excluding individual studies to determine the stability of results. When data from 10 or more studies were available, potential publication bias was evaluated using funnel plots and Egger’ s regression test. Meta-regression was performed to identify factors associated with heterogeneity. P-value < 0.05 was considered statistically significant. All statistical analyses were conducted using STATA version 13.0 (Statacorp, College Station, Texas, United States).
Results
Literature Search
The detailed literature screening process is presented in Figure 1. The initial database search identified 4474 records, including 2655 from Pubmed, 1815 from Embase, and 4 from the Cochrane library. After removal of 151 duplicates, 4323 references remained for screening based on titles and abstracts. Subsequently, 59 full-text articles were assessed for eligibility in accordance with the inclusion and exclusion criteria. Of these, 18 full-text articles were excluded, and specific reasons provided in eTable S3. The excluded studies comprised 3 review articles, 2 that did not involve THA or TKA procedures, 7 lacking outcome data, 3 did not evaluste antithrombotic drugs, and 3 randomized controlled trials. Finally, 41 studies were eligible for inclusion in synthetic analysis.

Flow diagram for the selection of eligible studies.
Study Characteristics
The characteristics of all included studies were summarized in Table 1. Thirty-three were retrospective cohort studies and eight were prospective studies. Geographically, twenty-one were performed in North America, eleven in Europe, six in Asia, 2 in Oceania and 1 in South America. Ten articles assessed the efficacy and safety of anticoagulants for VTE prophylaxis in patients undergoing TKA, eight focused on THA, and twenty-three included both TKA and THA populations. A total of 1,813,377 individuals received antithrombotic therapy for VTE prevention, with sample sizes ranging from 71 to 408,038.
Detailed Characteristics of the Included Studies. THA: Total Hip Arthroplasty; TKA: Total Knee Arthroplasty.

THA: total hip arthroplasty; TKA: total knee arthroplasty; LMWH: low-molecular-weight heparin; MBL: major bleeding; NR: not reported.
Patient Characteristics and Quality Assessment
The clinical characteristics of the patients enrolled in the studies are summarized in eTable S4. The median age of participants was 66.23 years, with male accounting for 75.11%. Common cardiovascular comorbidities included hypertension, diabetes mellitus, heart failure, and atrial fibrillation. The quality scores of included studies are summarized in eTable S5. Overall, the risk of bias was low, and all studies were of moderate to high quality, with scores ranging from 6 to 9.
Primary VTE Outcomes
The incidence of VTE in patients receiving antithrombotic therapy for VTE prophylaxis after THA or TKA was the primary effective outcome, and the results are summarized in Figure 2.

Pooled rate of VTE events in patients receiving aspirin and other anticoagulants after total hip and knee arthroplasty. No.: number of included studies; VTE: venous thromboembolism; LMWH: low-molecular-weight heparin; DOACs: direct oral anticoagulants.
Overall Analysis for VTE Prophylaxis
A total of 16 studies involving 277,438 individuals reported LMWH treatment, with a pooled incidence of VTE was 1.20% (95% CI: 0.90%-1.60%) (eFigure S1). Thirty-four studies evaluated the risk of VTE in patients receiving aspirin after THA or TKA, yielding an overall VTE incidence of 0.80% (95% CI: 0.70%-1.00%) (eFigure S2). Among patients receiving warfarin for VTE prophylaxis after THA or TKA, the combined VTE incidence was 0.90% (95% CI: 0.40%- 1.40%) (eFigure S3). Additionally, twenty studies assessed VTE risk in patients with DOACs, reporting an overall incidence of 0.90% (95% CI: 0.70%-1.20%) (eFigure S4). In the overall analysis, the pooled VTE incidence rate among patients receiving aspirin did not differ significantly from those receiving other anticoagulant agents.
Subgroup Analysis for VTE Prophylaxis
In subgroup analysis by geographic region, patients receiving LMWH therapy demonstrated the highest VTE incidence in Asia (21.80%, 95% CI: 10.50%-33.10%), followed by Oceania (6.00%, 95% CI: 0.80%-11.20%), Europe (1.10%, 95% CI: 0.50%-1.70%) and North America (0.80%, 95% CI: 0.50%-1.20%)(eFigure S5). Among patients treated with DOACs, the highest VTE rate was observed in Asia (9.40%, 95% CI: 1.30%-17.60%), followed by Oceania (3.40%, 95% CI: 1.30%-5.40%), North America (0.60%, 95% CI: 0.20%-1.00%), and Europe (0.80%, 0.50%, and 1.20%) (eFigure S6). Similarly, in aspirin therapy group, the highest rate was reported in Europe (3.00%, 95% CI: 1.70%-4.30%) and Asia (2.40%, 95% CI: 0.20%-4.60%), followed by Oceania (1.60%, 95% CI: 0.30%-2.90%) and North America (0.60%, 95% CI: 0.50% −0.80%) (eFigure S7). These differences were statistically significant. Nevertheless, no significant difference was observed in warfarin group across regions (Europe: 1.00% vs North America: 0.90%)(eFigure S3).
For subgroup analysis based on aspirin dosage regimen, there was no significant difference in VTE incidence between patients receiving high-dose aspirin (0.70%, 95% CI: 0.40%-1.00%) and those receiving low-dose aspirin(0.70%, 95% CI: 0.40%-1.00%) (eFigure S8). However, a significant difference was observed within the aspirin group between retrospective study (0.80%, 95% CI: 0.60%-0.90%) and prospective study (1.70%, 95% CI: 0.80%-2.50%) (eFigure S9). No significant differences in VTE incidence were found for LMWH therapy according to study design (retrospective: 1.30% vs prospective: 1.10%), nor for warfarin (retrospective: 1.00% vs prospective: 0.90%) and DOACs (retrospective: 0.80% vs prospective: 1.30%)(eFigure S10-11).
Among different DOACs, apixaban was associated with a higher risk of VTE (7.20%, 95% CI: 0.00%-16.10%) compared to dabigatran (0.70%, 95% CI: 0.10%-1.30%) and rivaroxaban (0.80%, 95% CI: 0.40%-1.10%), however, this difference was not statistically significant (eFigure S12).
Primary Safety Outcomes
The rate of major bleeding in patients receiving antithrombotic therapy for VTE prophylaxis after THA or TKA is the primary safety outcome, and the result is summarized in Figure 3.
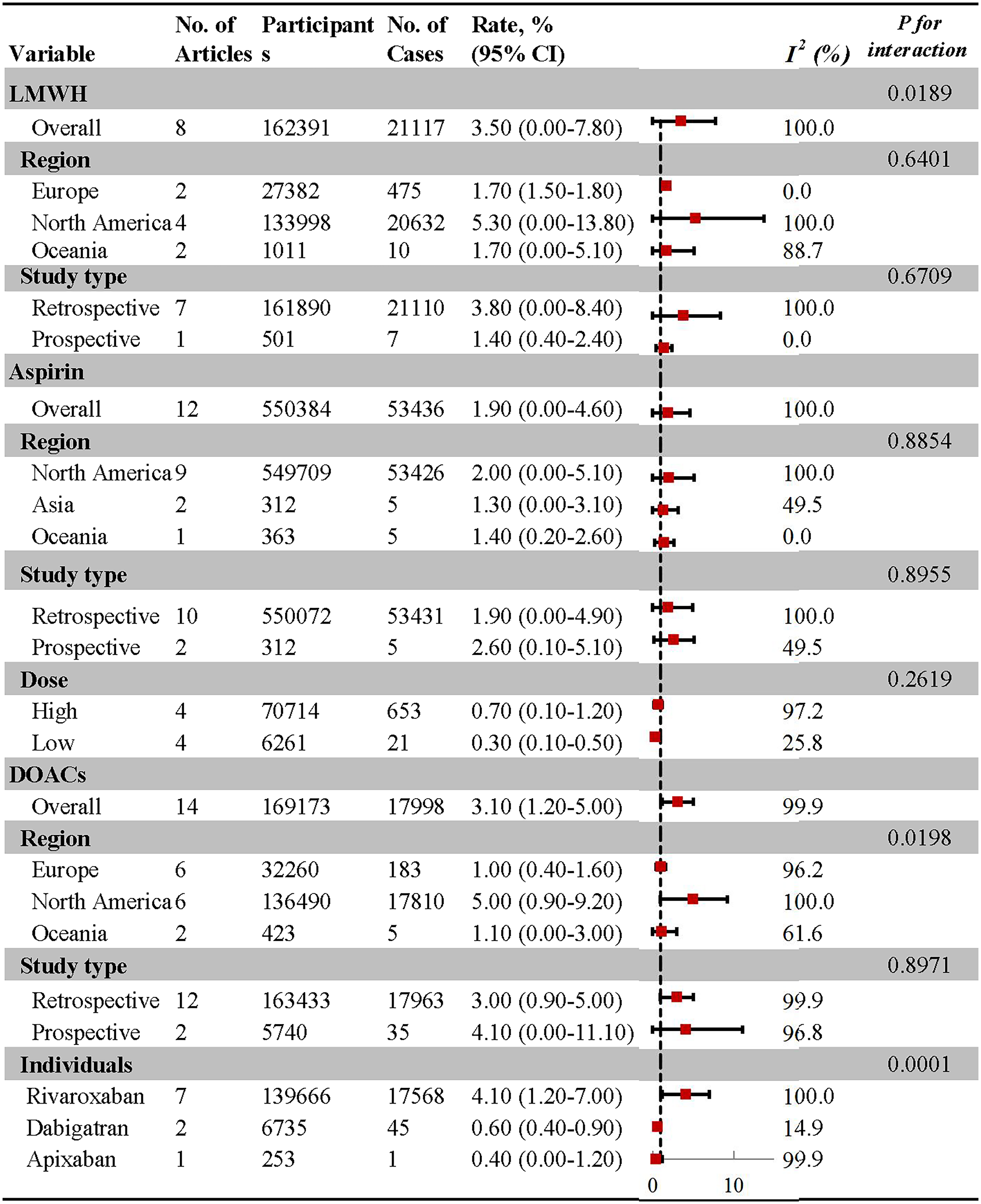
Pooled rate of major bleeding in patients receiving aspirin and other anticoagulants after total hip and knee arthroplasty. No.: number of included studies; LMWH: low-molecular-weight heparin; DOACs: direct oral anticoagulants.
Overall Analysis for Bleeding Risk
Among all anticoagulants, the lowest rate of major bleeding was observed in patients treated with aspirin (1.90%, 95% CI: 0.00%-4.60%), followed by DOACs (3.10%, 95% CI: 1.20%-5.00%) and LMWH therapy (3.50%, 95% CI: 0.00%-7.80%) (eFigure S13-15). Moreover, patients receiving aspirin therapy had a significantly reduced pooled risk of bleeding compared with those receiving LMWH and DOACs.
Subgroup Analysis for Bleeding Risk
In subgroup analysis by geographic region, pooled data indicated that rate of major bleeding associated with DOACs therapy was higher in North America (5.00%, 95% CI: 0.90%-9.20%) compared to Europe (1.00%, 95% CI: 0.40%-1.60%), and Oceania (1.10%, 95% CI: 0.00%-3.00%) (eFigure S16). Similarly, among patients receiving LMWH therapy, North American populations exhibited a higher incidence of bleeding events (5.30%, 95% CI: 0.00%–13.80%) than their counterparts in Europe (1.70%, 95% CI: 1.50%-1.80%) and Oceania (1.70%, 95% CI: 0.00%-5.10%) (eFigure S17). No significant difference was observed across regions for aspirin therapy (North America: 2.00%, Asia: 1.30%, Oceania: 1.40%)(eFigure S18).
In the combined subgroup analysis by study design, no statistically significant differences were found between retrospective (1.90%, 95% CI: 0.00%- 4.90%) and prospective studies (2.60%, 95% CI: 0.10%-5.10%), or in DOACs (retrospective: 3.00%, 95% CI: 0.90%-5.00%; prospective: 4.10%, 95% CI: 0.00%-11.10%) (eFigure S19-20). Similarly, for LMWH therapy, the difference between retrospective (3.80%, 95% CI: 0.00%-8.40%) and prospective studies (1.40%, 95% CI: 0.40%-2.40%) was not statistically significant (eFigure S21).
A subgroup analysis based on aspirin dosage regimen was also conducted. There was no significant difference in risk of bleeding between patients receiving high-dose aspirin (0.70%, 95% CI: 0.10%-1.20%) and those receiving low-dose aspirin (0.30%, 95% CI: 0.10%-0.50%) (eFigure S22). Furthermore, in the subgroup of individual DOACs, rivaroxaban significantly increased risk of major bleeding (5.00%, 95% CI: 0.90%-9.20%) compared to dabigatran (0.60%, 95% CI: 0.40%- 0.90%) and apixaban (0.40%, 95% CI: 0.00%-1.20%)(eFigure S23).
Sensitivity Analysis and Meta-Regression
Sensitivity analyses for efficacy and safety outcomes of multiple anticoagulants were performed by sequentially removing individual studies to assess influence on the pooled estimates (eTable S6-9). The results indicated that the overall incidences of VTE and major bleeding events were not materially affected by the removal of any single study, confirming the robustness of primacy findings. Meta-regression analysis was conducted to explore the potential effect of patient characteristics on risks of VTE and bleeding events (eTable S10). These analyses revealed no statistically significant correlation between variables and pooled rate.
Publication Bias
Publication bias was assessed using funnel plots and statistical tests (eFigures S24–S29). For VTE incidence, Egger's test yielded P values of 0.306 for LMWH, 0.079 for DOACs, and 0.398 for aspirin therapy. For bleeding incidence, the corresponding P values were 0.399 (LMWH), 0.270 (DOACs), and 0.780 (aspirin). Collectively, these results suggest no substantial publication bias across the anticoagulant groups, as supported by visual inspection of funnel plots as well as Begg's and Egger's tests. Due to the limited number of studies in the warfarin group (n < 8), funnel plot analysis was not conducted.
Discussion
This study evaluated the efficacy and safety of aspirin compared to other antithrombotics for VTE chemoprophylaxis following THA and TKA in real-world clinical practice. The overall incidence of VTE with aspirin was not statistically different from other antithrombotics among patients undergoing THA and TKA. Nevertheless, aspirin was associated with a significantly lower rate of major bleeding, demonstrating a favorable safety profile.
VTE represents an unexpected and potentially life-threatening complication following THA and TKA. In the absence of prophylactic measures, the incidence of postoperative VTE is notably high, anticoagulant prophylaxis has been shown to reduce VTE-related complications. 15 Despite this, current clinical practices regarding VTE chemoprophylaxis remain highly variable across institutions and regions.28,29 Considerable debate persists concerning the optimal agents among aspirin, LMWH, warfarin, and DOACs when balancing efficacy against bleeding risk. 16 Among oral medications, DOACs have demonstrated effective VTE prevention with relatively low complication rates. 17 A large meta-analysis of 13 RCTs found no significant differences in either VTE or major bleeding rates between aspirin and other thromboprophylactic agents, including LMWH and rivaroxaban. 30 Netherless, translating trial findings into routine clinical practice remains challenging, as trial populations are often younger and less comorbid than real-world patients. Recently, an increasing number of real-world studies have supported aspirin as a viable option for VTE prophylaxis after THA and TKA.10,11,31 To date, no specific systematic review has specifically assessed aspirin versus other anticoagulants in real-world setting. To our knowledge, this study synthesizes the most extensive real-world evidence comparing aspirin with other anticoagulants in terms of VTE and bleeding events, thereby addressing a critical knowledge gap.
Our findings demonstrate that postoperative aspirin use is effective for VTE prophylaxis following THA and TKA, consistent with existing literature on its preventive efficacy.32,33 Notably, we observed a higher incidence of VTE with LMWH in Asian populations compared to other regions, which may be attributed to the limited epidemiological data, widely usage rate, and reduced awareness of thrombotic disorders in Asia.34,35 In large-scale cohort studies, the VTE incidence with aspirin was comparable to that with other anticoagulants. 36 Growing evidence supports the evaluation of various aspirin regimens in VTE prophylaxis.13,37 Consistent with prior reports, 38 our study found no significant difference in VTE rates between high-dose and low-dose aspirin, both maintaining an incidence of approximately 0.7%. These findings support low-dose aspirin as a valid alternative to high-dose regimens, aligning with current guidelines recommending either dose for 14 days postoperatively until discharge. 39 The selection of the most appropriate chemoprophylactic agent and dosage should be individualized, taking into account each patient's thrombotic and bleeding risks.
Postoperative bleeding is a major concern in VTE prophylaxis. Previous studies have reported conflicting results regarding the safety of aspirin compared to other agents.18,20,40 A retrospective study found no difference in bleeding events between aspirin and DOACs. 40 While large-scale trials showed similar rate of major bleeding between aspirin and LMWH or DOAC.32,41 In contrast, two cohort studies reported a lower incidence of major bleeding with aspirin.11,42 These discrepancies may stem from the lack of standardized definitions for major bleeding across studies. However, when applying internationally accepted criteria, aspirin demonstrated superior safety with respect to bleeding risk, a finding mirrored in our study.43,44 Similarly, risk of gastrointestinal bleeding was lower with aspirin than with DOACs following THA. 45
Our study conformed a lower postoperative bleeding risk between with aspirin compared to other anticoagulants, consistent with findings from RCTs.46,47 Notably, the highest bleeding events were observed in North America, potentially due to higher baseline VTE incidence and more aggressive anticoagulant use in this region compared to Asia.48,49 Additionally, no significant difference in bleeding risk was observed between low-dose and high-dose aspirin regimens, supporting previous evidence that low-dose aspirin is equally effective and safe.13,15,37,50 Furthermore, both prospective and retrospective studies consistently report similar bleeding rates in patients receiving aspirin after THA and TKA.15,37
Beyond comparable effectiveness and superior safety, aspirin offers several advantages for VTE prophylaxis. Daily administration of aspirin is simple, safe, and does not require monitoring. 51 A survey by American Association of Hip and Knee Surgeons indicated that aspirin was thought to be the easiest to use and least likely to cause bleeding. 52 Economically, the full course of LMWH costs approximately $450, while rivaroxaban exceeds $370.53,54 Although warfarin itself is inexpensive, the need for monitoring increases its overall cost. 55 In contrary, aspirin costs about $2 per month and requires no monitoring.These cost advantages make aspirin particularly valuable in value-based care models and episode-based payment systems, where reducing healthcare expenditures without compromising outcomes is essential. Given its low cost, wide availability, and proven effectiveness, aspirin emerges as the most cost-effective thromboprophylactic option. 56 Therefore, this systematic review highlights aspirin's role as an effective, safe, and economic agent for VTE prophylaxis in patients undergoing THA and TKA.
Strengths and Limitations
The strength of this study include comprehensive data collection from multiple databases, enhancing the generalizability of findings and reflecting real-world clinical practice. However, several limitations warrant consideration. Firstly, considering the nature of included observational study design, the strength of evidence may be affected by high heterogeneity and potential bias. It must be acknowledged that clinical trials are known to require more strict eligibility criteria in comparison to the observational studies. Nevertheless, this study reflects the real-life use and consequences of VTE prophylaxis, it is therefore a valuable supplement to clinical trials. Secondly, subgroup analysis by VTE type (DVT and PE) was not feasible due to insufficient data. Nevertheless, we performed other subgroup analyses assessing the effectiveness and safety of anticoagulants in relation to VTE and major bleeding. Thirdly, the definition of major bleeding varied across included studies, contributing to inconsistent findings in the literature. To address this, our analysis applied a uniform, internationally recognized definition of major bleeding. 44 Fourthly, due to the limited number of available studies, we did not evaluate all-cause mortality in real-world settings. Additionally, understanding the status of thrombus risk assessment and prophylaxis after total hip and knee arthroplasty, is essential to reduce the thrombus disease burden. However, data in the included studies were lacking.
Finally, to improve the accuracy of the results, we only conducted article search to English language, but it may limit its potential impact on generalizability.
Despite these limitations, this study is the first systematic review to provide robust real-world evidence comparing aspirin with other anticoagulants for VTE prophylaxis after THA and TKA. we did not evaluate all-cause mortality in real-world settings.55,57,58 Considering its comparable efficacy, reduced bleeding risk, low cost, ease of use, and demonstrated non-inferiority, aspirin should be considered a preferred option for VTE prophylaxis in THA and TKA patients.
Conclusions
This systematic review provides a comprehensive assessment of the efficacy and safety of aspirin compared to other anticoagulants for thromboprophylaxis after THA and TKA in real-world clinical practice. The findings indicate that aspirin is associated with a comparable incidence of VTE relative to other anticoagulants, while demonstrating a significantly lower risk of bleeding. Considering its favorable safety profile, widespread availability, and substantial cost-effectiveness, aspirin represents a viable and promising alternative for VTE prophylaxis in patients undergoing THA and TKA in routine clinical settings. However, these conclusions should be interpreted with caution and warrant further validation through high-quality, prospective studies.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251410933 - Supplemental material for Effectiveness and Safety of Aspirin Versus Other Antithrombotics for VTE After Total Hip and Knee Arthroplasty in Real-World Setting
Supplemental material, sj-docx-1-cat-10.1177_10760296251410933 for Effectiveness and Safety of Aspirin Versus Other Antithrombotics for VTE After Total Hip and Knee Arthroplasty in Real-World Setting by Ya-Fang Zhu, MD, Hua Qian, MD, Jun-Hong Ma, MD, Nan-Nan Shen, MD, and Wei Li, PhD in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Author Contributions
Ya-Fang Zhu, Hua Qian and Wei Li designed the study. Jun-Hong Ma and Wei Li performed the search strategy. Ya-Fang Zhu and Hua Qian collected and analyzed the data. Nan-Nan Shen drafted the manuscript. Nan-Nan Shen and Wei Li proofed the final revisions. All the authors contributed to study design, critically reviewed the first draft, and approved the final manuscript. All the disagreements were resolved by Wei Li.
Funding
This study was supported by the Zhongnanshan Medical Foundation of Guangdong Province (ZNSXS-20240069), Zhejiang Province Clinical Research Program for Traditional Chinese Medicine (2026ZL0948), Shaoxing Basic public welfare special project (2025A14022), Shaoxing health science and technology plan project (2023SKY079), National Natural Science Foundation of China of Young Scientists Program (82205234), Clinical Medical Research Special Fund Project of Zhejiang Medical Association (2023ZYC-A55), Clinical Medical Research Special Fund Project of Zhejiang Medical Association (2022ZYC-Z37), Program of General Scientific Project of Zhejiang Education Department (Y202557520, Y202249053), Research Project of Grassroots health science of Zhejiang Province (2022ZD09), Zhejiang Pharmaceutical Society Hospital pharmacy special research project (2016ZYY29).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
