Abstract
Patients undergoing laparoscopic-guided radiofrequency ablation (LRFA) for the treatment of a renal mass are commonly prescribed antithrombotic agents for the management of comorbid medical diseases. We retrospectively evaluated the safety of LRFA in this group. From October 2005 to June 2010, 109 patients underwent LRFA. Antithrombotic therapy was prescribed to 52 of these patients. Agents were managed the week of surgery per current practice guidelines from the American College of Chest Physicians. Intraoperatively, patients prescribed at least one antithrombotic agent lost a median of 10 mL of blood, while patients not on an antithrombotic agent also lost 10 mL of blood (P = .828). Both groups had a similar rate of procedure-related complications (intraoperative, P = 1.00; postoperative, P = .673). No patient required a blood transfusion or experienced a postoperative thromboembolic event. In conclusion, when current practice guidelines are followed, LRFA is safe among patients prescribed antithrombotic agents.
Introduction
Guidelines from the American Urologic Society (AUA) promote partial nephrectomy as the standard of care for the management of small renal masses. 1 For elderly patients and those with a significant comorbid disease burden, AUA guidelines alternatively suggest treatment with an ablative technology such as radiofrequency ablation. Laparoscopic-guided radiofrequency ablation (LRFA) involves identification of a renal tumor with either direct visualization or laparoscopic ultrasound. Once the tumor is located, a radiofrequency probe is inserted and an electric current is passed causing the tumor to heat and necrose. 2 This nephron-sparing approach has been shown to be safe with acceptable oncologic outcomes.3,4
Current practice places patients on antiplatelet (AP) therapy for a variety of indications including primary and secondary prevention of thromboembolic events. 5 Further, many patients receive vitamin K antagonists (VKAs), given a history of atrial fibrillation, presence of mechanical heart valve, or venous thromboembolism. For this reason, many of the patients undergoing LRFA are often prescribed one or more AP and/or VKA. Data are lacking, however, on the safety of LRFA in patients receiving these agents. We evaluated the safety of LRFA in patients taking antithrombotic agents and discuss our results along with current practice guidelines.
Patients and Methods
Data Collection
Following Institutional Review Board approval, we retrospectively reviewed the medical records of patients who underwent LRFA for the management of a small renal mass. Records were reviewed for demographic information, past medical history, medications, pre- and postoperative serum creatinine values, American Society of Anesthesiology score, estimated blood loss, procedure-related complications, tumor pathology, length of hospital stay, and perioperative thromboembolic events (ie, myocardial infarction, deep venous thrombosis, transient ischemic attack, and stroke). Estimated glomerular filtration rate was calculated with the modification of diet in renal disease (MDRD) formula. 6
Statistical Analysis
Data was analyzed in PASW Statistics 18.0 (IBM, Somers, New York). Following data collection, patients were dichotomized into 2 groups: those not taking an antithrombotic agent (group 1) and those taking at least 1 agent (group 2). Continuous variables were compared between groups with the nonparametric test of medians and proportions were compared with chi-square analysis. Fisher’s exact test was used for proportions of small size. A P value of ≤.05 was considered statistically significant.
Laparoscopic-Guided Radiofrequency Ablation Procedure
Previously we have described our multipass approach for LRFA. 3 In brief, LRFA was performed by a conventional transperitoneal laparoscopic approach. Tumors were localized with the aid of laparoscopic ultrasound. Prior to ablation, multiple biopsy samples were obtained with an 18-gauge spring-loaded core biopsy needle. One of two different laparoscopic probes was used at the preference of the surgeon: the 14-gauge Star-Burst XL (Angiodynamics, Queensbury, New York) or the Cool-Tip (Valleylab, Boulder, Colorado) probe. For tumors larger than 3.5 cm, a Cool-Tip switch box with three 3-cm probes was routinely used with multiple deployments as required to achieve the temperature goal throughout the tumor. Thermal monitoring was used in cases of larger masses to evaluate ablation at the tumor periphery.
Perioperative Management of Antithrombotic Agents
Current guidelines from the American College of Chest Physicians (ACCP) on the perioperative management of patients receiving antithrombotic therapy recommend the cessation of AP agents 7 to 10 days prior to surgery. 7 These guidelines also suggest stopping warfarin 4 to 5 days prior to surgery without bridging for patients at a low risk of thromboembolism and with bridging for those at moderate-to-high risk of an event. Our practice of managing antithrombotic agents was based on these guidelines. Following adequate hemostasis at the time of surgery, all oral agents were restarted at 24 hours after ablation.
Results
Between October 2005 and June 2010, 109 patients underwent LRFA for the management of a small renal mass. Groups 1 (no antithrombotic agent) and 2 (at least 1 antithrombotic agent) were comprised of 55 and 52 patients, respectively. Table 1 compares the 2 groups. Patients were well matched in terms of age (P = 291), sex (P = .551), race (P = .364), body mass index (P = .689), pre- (P = .479) and postoperative (P = .312) renal function, American Society of Anesthesiology score (P = .301), tumor side (P = .442), location (P = .973), extension (P = .326), and pathology (P = .212). Patients in group 2 had larger tumors (P = .044) and a higher frequency of comorbid diseases including hypertension (P < .0001), coronary artery disease (P < .0001), and history of stroke or transient ischemic attack (P = .005). No patient in group 1 had a history of atrial fibrillation, while 12 (23.1%) patients in group 2 had this diagnosis (P < .0001), all of whom were on warfarin.
Study Cohort Characteristics
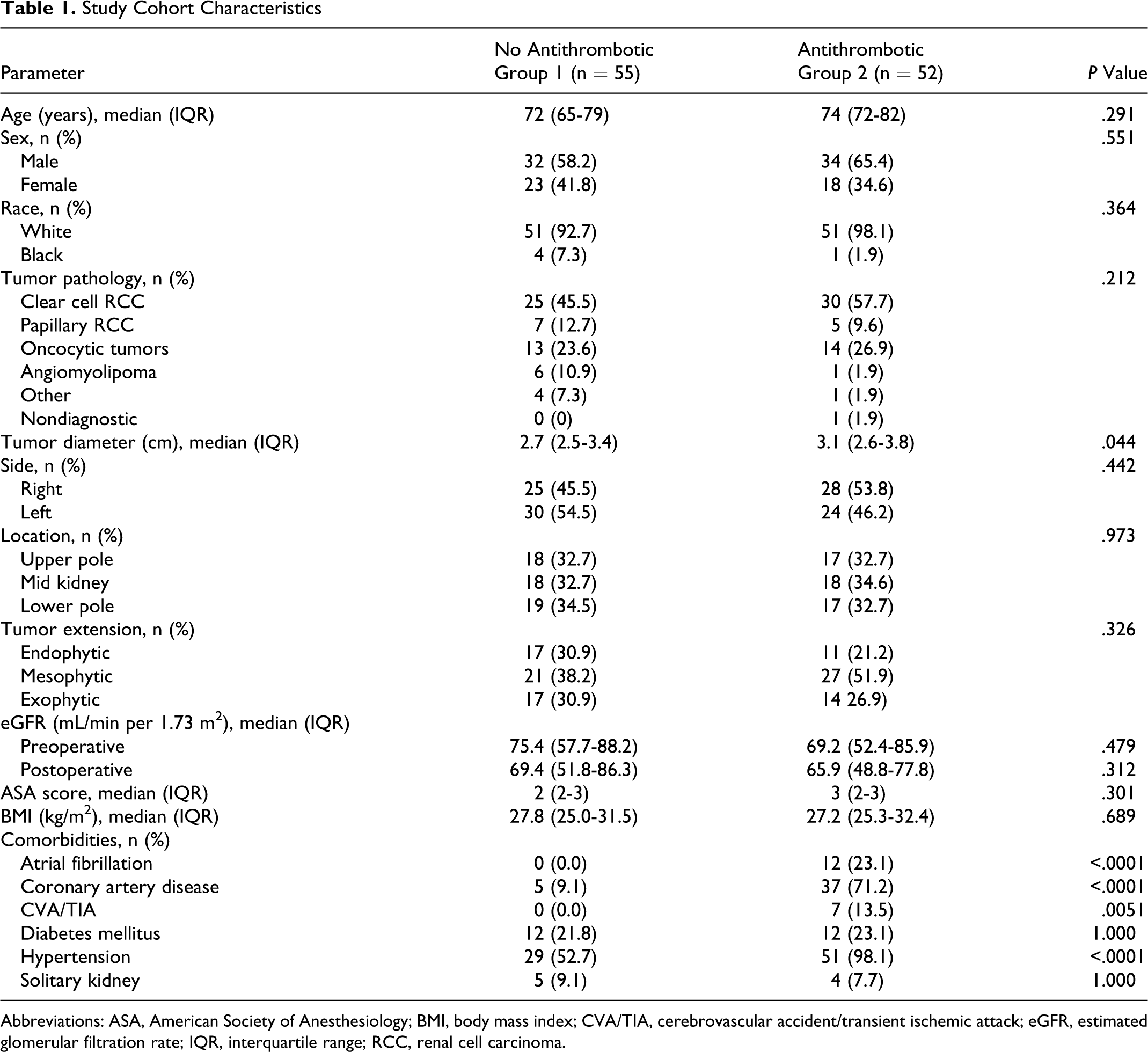
Abbreviations: ASA, American Society of Anesthesiology; BMI, body mass index; CVA/TIA, cerebrovascular accident/transient ischemic attack; eGFR, estimated glomerular filtration rate; IQR, interquartile range; RCC, renal cell carcinoma.
Patients in group 2 were on at least 1 antithrombotic agent. In total, 29 (55.8%), 3 (5.8%), and 8 (15.4%) patients were on aspirin, clopidogrel, and warfarin in isolation, respectively (Table 2). In addition, 8 (15.4%) patients were on both aspirin and clopidogrel, while 4 (7.7%) patients were on warfarin in combination with an AP. Table 2 details the indications for taking these agents.
Antithrombotic Agents and Indication
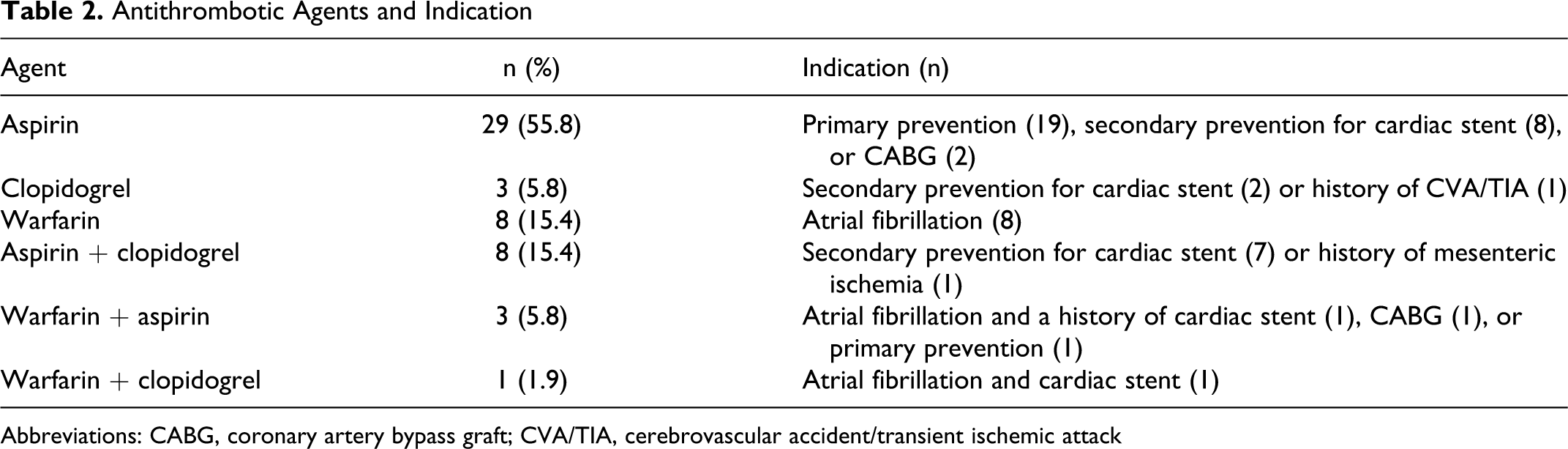
Abbreviations: CABG, coronary artery bypass graft; CVA/TIA, cerebrovascular accident/transient ischemic attack
Estimated blood loss at the time of surgery was similar between groups. Patients in group 1 lost a median of 10 mL (interquartile range [IQR] 10-25) of blood while patients in group 2 lost 10 mL (IQR 10-23) of blood (P = .828; Table 3). The length of hospital stay (P = .703) and rate of procedure-related complications were similar between groups (intraoperative, P = 1.00; postoperative, P = .673) with no patient in either group requiring a blood transfusion. In addition, no patient experienced a thromboembolic event in the immediate postoperative period.
Comparison of Blood Loss and Complications
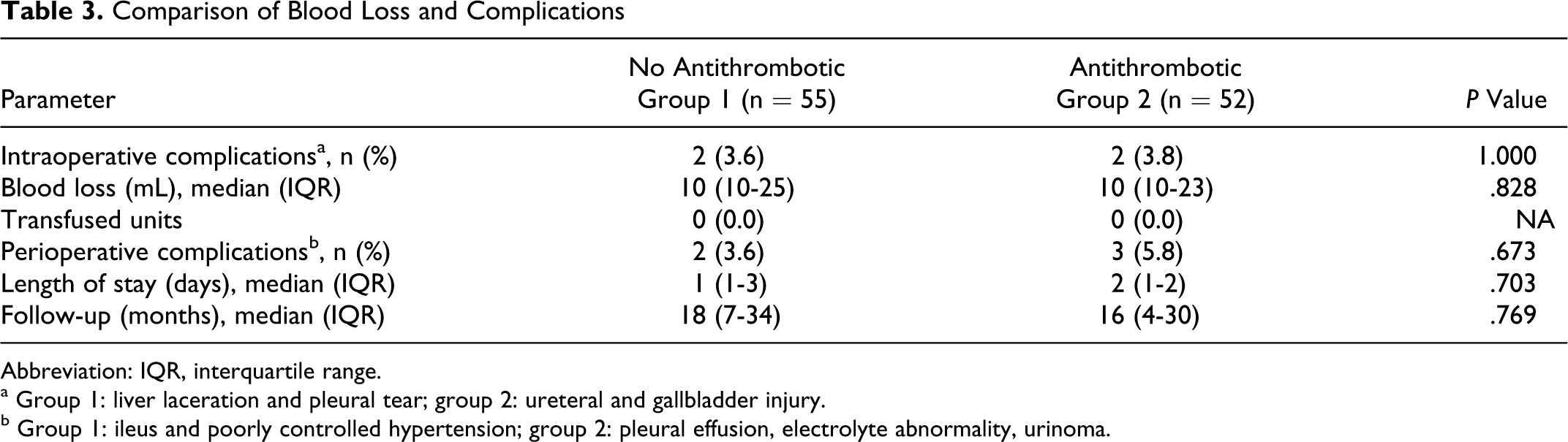
Abbreviation: IQR, interquartile range.
a Group 1: liver laceration and pleural tear; group 2: ureteral and gallbladder injury.
b Group 1: ileus and poorly controlled hypertension; group 2: pleural effusion, electrolyte abnormality, urinoma.
Discussion
Currently no specific guidelines exist on the administration of antithrombotic agents in the perioperative period among patients undergoing urologic surgery. The extent to which the continuation of AP or VKA drugs confer an increased risk of bleeding in the perioperative period is not clear. 8 While there is data demonstrating an increased risk of hemorrhage when these agents are continued at the time of surgery,9–11 other reports have shown it may be safe to continue these agents during certain procedures such as prostate biopsy and ureteroscopy for nephrolithiasis.12,13 To date, however, many urologic surgeons continue to stop antithrombotic agents prior to procedures due to the perceived risk of bleeding. This is particularly true when operating on highly vascularized organs such as the prostate and kidney.
In the presented work, we examined the bleeding risk of patients prescribed antithrombotic agents undergoing LRFA. We found no difference in blood loss between those who received AP and/or VKA treatment and those who did not. Further, there were no blood transfusions or required intervention for bleeding in either arm of the study. These findings suggest that LRFA can be safely performed in patients receiving AP if it is stopped within 7 days or VKA if stopped within 5 days prior to this procedure, with reinstitution of these medical therapies within 24 hours postprocedure. Although the study was not powered to detect adverse cardiac or thromboembolic events, none were observed in the perioperative period.
There have been several suggested algorithms for the perioperative management of AP therapy in the urologic patient. 14 These algorithms, however, are not based on randomized trials but rather on cautious expert opinion of mostly observational studies. Further, these suggested guides are mostly founded on extrapolations of nonurologic surgical series.
A modified algorithm published by Eberli et al 15 recommended stoppage of single-agent AP therapy 5 days prior to surgery in cases of primary prevention and continued treatment of a single agent for all cases of secondary prevention for all nonemergent urologic surgeries. While this may be reasonable for some surgeries, LRFA involves placing a large gauge needle (typically 14 gauge) into a well-vascularized organ with a potential for significant bleeding. Therefore, our initial experience was performed after AP and VKA therapy was held. Further, because small renal masses have a low risk of progression or metastasis in the short term, 16 we were comfortable in delaying treatment until the cardiac risk was minimized as in cases of patients after cardiac stenting.
Alternatives to LRFA for the treatment of small renal masses include laparoscopic or open partial nephrectomy, cryoablation, and observation. 17 However, each of these approaches to treatment are not without their own drawbacks. Observation may be a safe alternative in selected cases but may be associated with significant patient anxiety and increased number of imaging studies. In addition, data for surveillance are still sparse and not yet mature. 18 In contrast, partial nephrectomy is associated with an approximate 5% risk of hemorrhage and a 2.7% incidence of blood transfusion, 19 while cryoablation is associated with a 4.6% risk of hemorrhage. 20 Radiofrequency ablation, however, confers a minimal risk of hemorrhage and thus is a desirable treatment for patients receiving AP and VKA therapy. Unlike cryoablation, which results in freezing of the tumor and potential kidney “cracking,” radiofrequency ablation is ideally suited for patients taking AP and/or VKA therapy because the heat generated causes coagulative necrosis of the tissue, thus limiting the risk of bleeding. In our study, a median blood loss of 10 mL (IQR 10-23) was observed in patients on AP or VKA therapy, with no patients requiring a blood transfusion.
As an initial study, we have shown that LRFA when performed on patients taking AP or VKA confers no increased risk of hemorrhage when current guidelines are followed. However, there exists a subset of patients who are deemed to have intermediate to high cardiac risk and hence should ideally not have their AP or VKA therapy stopped. This group includes those patients who are less than 6 or 12 months out from the placement of either a bare-metal or a drug-eluting stent. Another group of concern are those patients with a history of persistent atrial fibrillation with a high CHADS2 score. 21 In the former group, discontinuation of AP agents confers >50% risk of a major cardiac event with a 20% mortality. 22 While observation and delay of surgery can move some out of high-risk status, it is unclear just how long surgery can be delayed before influencing oncologic outcomes. For this reason, the next step would be to perform LRFA in carefully selected patients who cannot have their AP or VKA stopped.
Limitations of the presented work include its retrospective design and small sample size. Although retrospective, patients were well matched for nearly all variables (Table 1). Of note, patients receiving antithrombotic agents had a larger median tumor size (2.7 cm vs 3.1 cm, P = .044). Because tumor size correlates with the extent of tumor ablation and therefore risk of hemorrhage, we would have expected patients in this group to be at a higher risk of bleeding without consideration of medications. Despite this added risk, patients receiving an antithrombotic agent experienced no additional blood loss. With regard to sample size, while small, we did have adequate power to evaluate differences in intraoperative blood loss. This study was not powered to detect differences in perioperative thromboembolic events. However, no patient experienced an adverse event related to stopping antithrombotic therapy in the immediate postoperative period. While inconclusive, this finding speaks to the safety of current guidelines.
In conclusion, LRFA has a minimal risk of hemorrhage among patients taking antithrombotic agents when these medications are stopped prior to surgery. Antithrombotic therapies can be safely reinstituted in the immediate postoperative period. Future work aims to evaluate the safety of LRFA without the discontinuation of antithrombotic agents at the time of surgery. This approach may be best suited for those at moderate to high risk of thromboembolic events in the perioperative period.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
