Abstract
Objective
To analyze 180-day survival and associated factors in patients with chronic kidney disease (CKD) who initiated anticoagulation therapy during hospitalization.
Methods
A survival analysis study was conducted using Kaplan-Meier curves on a retrospective cohort of CKD patients on anticoagulation. Cox regression identified factors associated with survival differences.
Results
The study included 224 patients (50.90% male, 71.80% > 65 years, 30.80% with atrial fibrillation (AF), and 50% with venous thromboembolism (VTE)). Mortality was 40.63%, with higher survival in the AF group versus VTE (p = 0.0107), CKD stage 3a versus stage 4 (p = 0.0009), and stage 5 versus stage 4 (p = 0.0277). Mortality risk was higher in patients with subsidized healthcare (HR 1.47; 95% CI 1.17–3.12; p = 0.010), cancer (HR 1.77; 95% CI 1.15-2.71; p = 0.008), and VTE (HR 1.87; 95% CI 1.22–2.86; p = 0.004), Arterial hypertension (HR 0.44; 95% CI 0.29-0.68; p = 0.001), coronary artery disease (HR 0.42; 95% CI 0.20-0.48; p = 0.022), and antiplatelet monotherapy (HR 0.56; 95% CI 0.32-0.96; p = 0.037) did not influence survival.
Conclusions
CKD patients on anticoagulation therapy have high early mortality. Cancer, subsidized healthcare affiliation, and venous thromboembolism were independent risk factors associated with mortality, whereas hypertension and single antiplatelet therapy were not. This suggests that intensified follow-up and individualized management strategies may be particularly beneficial in these high-risk groups. Larger, prospective studies are needed to optimize anticoagulation management in this high-risk population.
Keywords
Introduction
Chronic kidney disease (CKD) is a major global public health problem with increasing prevalence and a high burden of morbidity and mortality. According to the Global Burden of Disease (GBD) study, the global incidence of CKD was 18.98 million in 2019, more than double the incidence of CKD in 1990 (7.8 million). 1 Currently, approximately 10% of the world's population is affected by CKD to some degree. The number of disability-adjusted life years (DALYs) lost due to CKD exceeds 41 million, and the global mortality rate from CKD is approaching 3%. It is estimated that CKD will become the fifth leading cause of death worldwide by 2040, representing one of the most significant increases among leading causes of death.1,2
CKD-specific pathophysiological mechanisms contribute to the development of cardiovascular complications and venous thromboembolic disease due to increased proinflammatory and procoagulant mediators.3–5 The incidence of major cardiovascular events in patients with CKD is 7 cases per 100 person-years compared to 4 cases per 100 person-years in patients without CKD (p < 0.0001). 6 CKD patients also have high hemorrhagic risk. 7 Furthermore, a progressive relationship between decreasing estimated glomerular filtration rate (eGFR) and mortality has been demonstrated.3,6 As renal clearance decreases, there is a progressive increase in the risk of concurrent thromboembolic and hemorrhagic events.3,8
Anticoagulant therapy is an important intervention in the prevention of thromboembolic events in atrial fibrillation (AF) and in the treatment of venous thromboembolic disease (VTE), particularly in the prevention of recurrence, but there is limited evidence to support its use in patients with CKD, particularly in advanced stages.9–11 Much of the evidence available for anticoagulated patients with CKD has been extrapolated from the AF indication. As a result, treatment patterns and outcomes in other indications remain unknown. The results of randomized controlled clinical trials suggest that warfarin and direct oral anticoagulants (DOACs) are effective in patients with mild to moderate chronic kidney disease (CKD); however, available data on the use of these anticoagulants in advanced stages of CKD and in patients on renal replacement therapy remains limited and controversial.11,12 Patients with an estimated glomerular filtration rate (eGFR) of less than 25 mL/min were excluded from studies of apixaban, 13 and patients with an eGFR of less than 30 mL/min were excluded from clinical trials of rivaroxaban and dabigatran.14,15
Although survival analyses have been performed in patients with chronic kidney disease, these studies have not focused on the specific population receiving anticoagulation therapy. As a result, the outcomes of anticoagulation therapy, particularly in relation to the degree of renal impairment and the indication for anticoagulation, remain poorly understood, as do the potential risk factors associated with mortality in this population. The aim of this study is to perform a survival analysis and describe the factors associated with it in patients with chronic kidney disease (CKD) who have initiated anticoagulation therapy.
Methods
A survival analysis study was conducted in a retrospective cohort of patients with a history of chronic kidney disease, older than 18 years, who initiated anticoagulation therapy during hospitalization and were treated at Hospital Universitario San Ignacio in Bogotá, Colombia. Patients were identified through the Hospital Universitario San Ignacio Institutional Anticoagulation Registry (RAC-HUSI) between 2019 and 2021. The institutional registry prospectively includes all adults (≥18 years) in whom therapeutic-dose anticoagulation is initiated during the index hospitalization for indications diagnosed or confirmed during that admission. We included all patients who met the KDIGO definition of chronic kidney disease 16 and who started anticoagulation in-hospital for any of the following indications: atrial fibrillation; atrial flutter; mechanical or biological valve prosthesis; acute or chronic pulmonary embolism; lower or upper extremity deep vein thrombosis; splanchnic thrombosis; cerebral venous thrombosis; left ventricular thrombus, were included. Patients referred to other institutions were excluded from the study. The study was approved by the institutional ethics committee (FM-CIE- 256/2021).
The REDCap electronic data capture tools, hosted at the Hospital Universitario San Ignacio, were used to collect and manage the information. 17 Patients are identified daily through a pharmacy database of dispensed anticoagulants. Trained personnel systematically collected data, including clinical and demographic variables, during hospital care. All patients were asked to provide informed consent for telephone follow-up after hospital discharge at 30, 90, or 180 days. Those who consented were assessed at 30, 90, and 180 days after discharge. Variables related to thrombotic and hemorrhagic complications and vital status were collected. Regular audits of the data collection process were performed to identify areas for improvement and to ensure data quality. When missing information was identified, it was supplemented by a retrospective review of institutional electronic medical records. Patients who could not be contacted for follow-up were censored to the last known date of complication information. To determine whether patients lost to follow-up were still alive, a search was performed in the single database of members of the General Social Security Health System (BDUA-SGSSS). The information recorded in this website is reported by government agencies as part of the mandatory reporting to the Colombian Social Security Health System.
Stages of chronic kidney disease were defined according to the kidney disease: Improving Global Outcomes (KDIGO) classification, 16 eGFR was calculated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) 2021 equation, which has been shown to be more accurate than other existing formulas. 18 Within the group of patients with stage 5 CKD, a distinction was made between those on and those not on renal replacement therapy, such as hemodialysis or peritoneal dialysis.
Hemorrhagic complications were classified according to the definition of major or minor bleeding of the International Society of Thrombosis and Hemostasis. 19 Major bleeding was defined as that associated with hemodynamic compromise, located at an anatomically critical site, requiring transfusion (≥2 units of red blood cell concentrate), or a decrease in hemoglobin ≥2 g/dL. The remainder were defined as minor bleeding.
For the purposes of this study, the period from initiation of anticoagulation therapy to the occurrence of a bleeding or thrombotic event was defined as the time during which the patient remained free of complications. Survival was defined as the time from initiation of anticoagulation to the date of death. A death was classified as anticoagulation-attributable when the primary cause of death recorded in the electronic health record explicitly indicated a thrombotic or hemorrhagic event temporally related to the anticoagulation period. In cases with multiple potential contributors (eg, cancer, infection, VTE), attribution required documentation of a thrombotic/hemorrhagic cause; otherwise, deaths were coded non-attributable.
SARS-CoV-2 infection status was not systematically captured for all patients due to evolving testing practices during the study period, with reduced routine testing in later months. Because missingness was substantial and likely non-random, COVID-19 status was not included as a covariate in multivariable models to avoid exposure misclassification and biased estimates.
One of the variables under investigation is the affiliation regime to the Colombian healthcare system, which is divided into two categories: contributive and subsidized. The contributive regime is a system in which individuals with the capacity to pay contribute to the system through their work income and have access to health services by paying a monthly contribution. The subsidized regime is designed for low-income individuals who are unable to afford the contribution to the health system. Consequently, the state subsidizes and provides services free of charge. Both regimes are designed to ensure that the Colombian population has access to health services.
Categorical variables were expressed as absolute values and percentages. Continuous variables were analyzed using the mean and standard deviation or the median and interquartile range, depending on data distribution. The Shapiro-Wilk test was used to assess the assumption of normality. Statistical significance between groups was determined based on the nature of the variables using Student's t-test, Chi-square test, or Mann-Whitney U test.
The evaluation of survival functions according to disease stage were evaluated using Kaplan-Meier curves and the log-rank test for subgroup comparisons. The Nelson-Aalen cumulative hazard function was used to analyze hemorrhagic complications, with right-censoring applied. Finally, a multivariate Cox regression analysis was conducted to determine which variables were associated with survival outcomes. We first performed univariate Cox models for each variable. The multivariable model initially included variables that were significant in univariate analyses and others considered clinically relevant based on prior studies. The final model was selected using a backward stepwise approach. Variables were kept if they remained significant or clearly improved the model. A p-value < 0.05 was considered statistically significant. The proportional hazards assumption was assessed using the Schoenfeld residuals test, confirming the results of the graphical analysis of log(-log(S(t))) versus t or log(t). All analyses were performed using Stata 14 (StataCorp LP, College Station, TX).”
Results
A total of 224 patients were included in the study; 46 (20%) were in stage 4 and 45 (20.5%) in stage 5 of chronic kidney disease. Among stage 5 patients, 29 were receiving renal replacement therapy (RRT), with 27 on hemodialysis and 2 on peritoneal dialysis. In the overall population, 161 patients (71.8%) were older than 65 years, with a high prevalence of comorbidities, including arterial hypertension (71.85%), diabetes mellitus (27.23%), cancer (28.12%), and heart failure (25%).
Table 1 provides details information on the demographics and clinical characteristics of the general population and two subgroups stratified by eGFR ≥30 ml/min and <30 ml/min. In the subgroup analysis, eGFR ≥30 ml/min were older and had a higher frequency of DOAC use, in contrast to the lower eGFR group where warfarin was more prevalent. Additionally, this group had a higher estimated bleeding risk as assessed by the HASBLED score, with a value of ≥3. Regarding anticoagulant class by insurance scheme, subsidized patients were mostly treated with non-warfarin regimens. Among warfarin-treated patients (n = 142), 24 (16.9%) belonged to the subsidized scheme. In this subsidized-warfarin subgroup, no major bleeding was recorded and two clinically relevant non-major bleeding events occurred at 30 and 60 days.
Demographic and Clinical Characteristics of Participants.
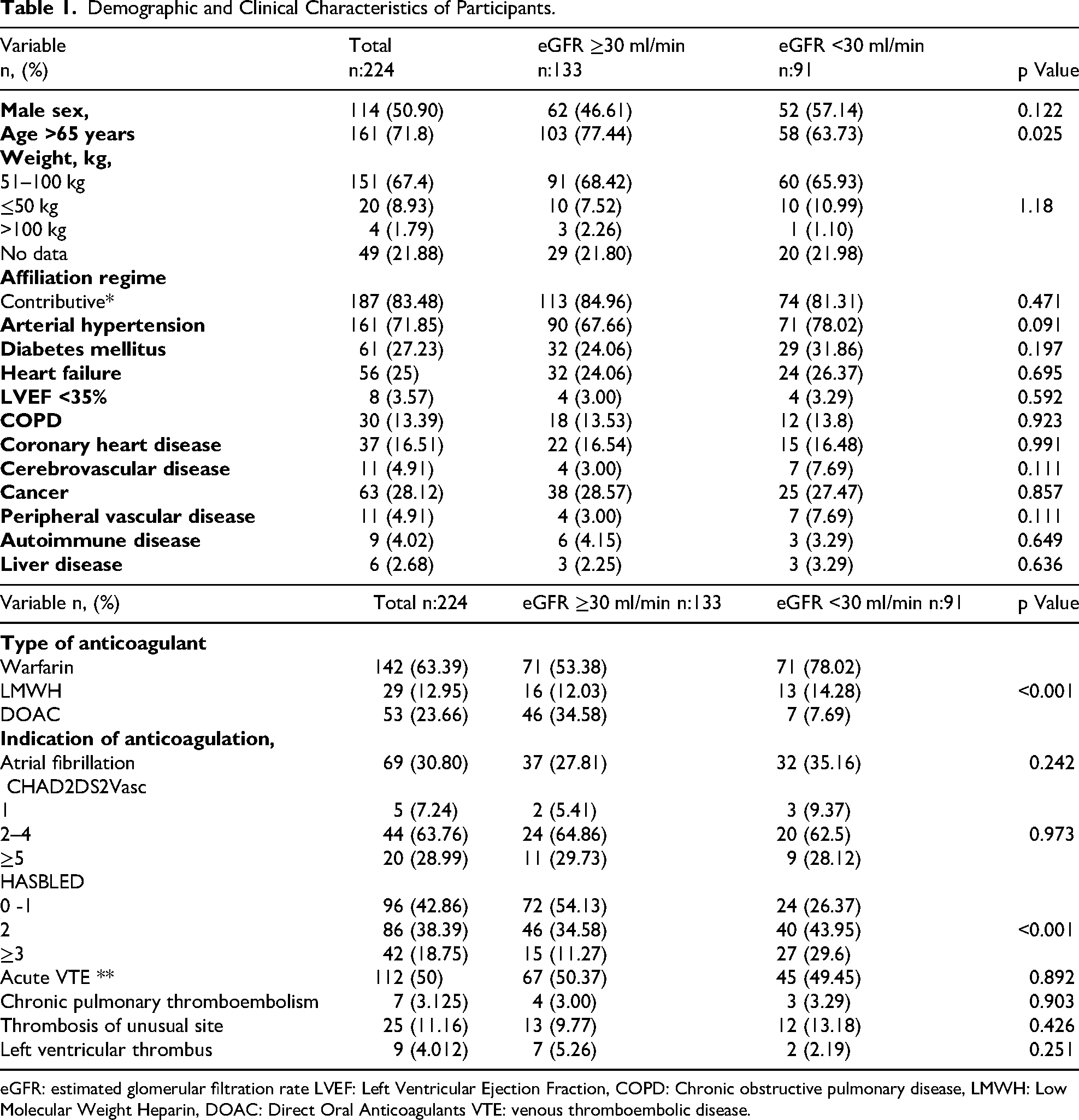
eGFR: estimated glomerular filtration rate LVEF: Left Ventricular Ejection Fraction, COPD: Chronic obstructive pulmonary disease, LMWH: Low Molecular Weight Heparin, DOAC: Direct Oral Anticoagulants VTE: venous thromboembolic disease.
Among patients receiving antiplatelet therapy (n = 65), aspirin (acetylsalicylic acid) monotherapy was most frequent (n = 60). Dual antiplatelet therapy, aspirin plus a P2Y12 inhibitor, was used in 5 patients.
Table 2 provides a detailed description of the complications and mortality events presented in the study. During the initial 30-day follow-up period, 30 cases of hemorrhagic complications were observed, 13 of which were classified as major bleeding. Among major bleeding complications in patients with stage 5 CKD, gastrointestinal bleeding was the most common, occurring twice as often as intracranial bleeding and more than twice as often as in patients with lower stages of CKD. In addition, thrombotic complications were more common in patients with stage 5 CKD. Within stage 5 CKD (n = 45), 29 patients (64.4%) received renal replacement therapy (27 hemodialysis, 2 peritoneal dialysis). In the hemodialysis subgroup, three in-hospital bleeding events occurred (11.1%); one fulfilled the ISTH criteria for major bleeding and the death was attributed to this event. During ambulatory follow-up, two additional clinically relevant non-major bleeding episodes were recorded, one at 30 days and one at 90 days; no bleeding events were observed at 180 days, and no thrombotic complications were documented over follow-up. In the peritoneal dialysis subgroup (n = 2), no bleeding events were observed at any time point and one thrombotic event occurred at 180 days, a deep vein thrombosis. The remaining 16 stage 5 patients were not on renal replacement therapy. Per center protocol, no intradialytic heparin was administered during hemodyalisis sessions. Notably, while patients with stage 5 CKD had a higher incidence of general hemorrhagic complications, patients with stage 3a CKD had a higher incidence of major bleeding events compared to other stages.
Complications and Mortality in Patients with CKD and Anticoagulation Therapy According to CKD Stage.
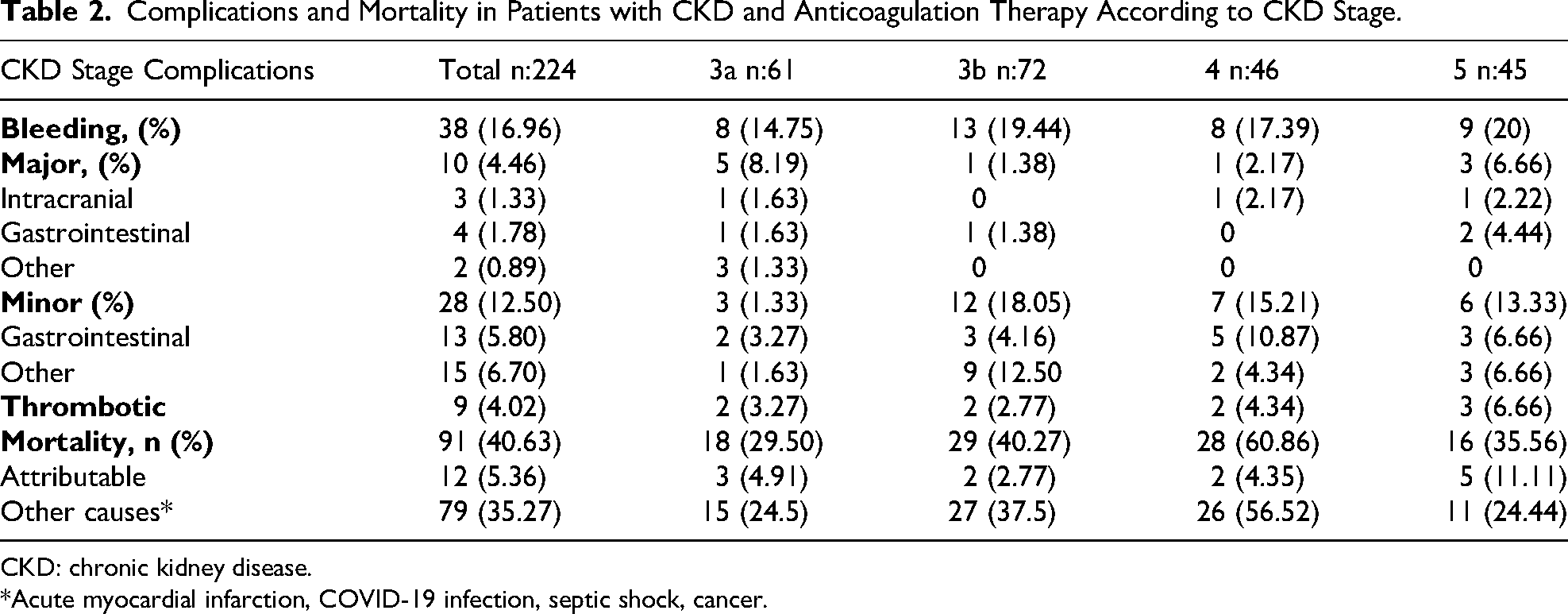
CKD: chronic kidney disease.
*Acute myocardial infarction, COVID-19 infection, septic shock, cancer.
Among the 91 recorded deaths, 79 (86.8%) were attributed to causes not directly related to anticoagulation therapy. Notably, 17 (18.6%) were attributed to COVID-19 infection. According to our prespecified definitions, these deaths were non-attributable to anticoagulation, as the cause was neither thrombotic nor hemorrhagic. Other non-anticoagulation-related causes included septic shock (29.7%), cancer (15.4%), acute myocardial infarction (14.3%) and others causes. However, the number of deaths attributable to anticoagulation was more than twice as high in stage 5 CKD compared to other stages.
Kaplan-Meier analysis of the survival analysis for overall study population is shown in Figure 1. It suggests that mortality is higher in the first 30 days following anticoagulation initiation, with subsequent stabilization of function up to 180 days, at which point the survival rate exceeds 50%.
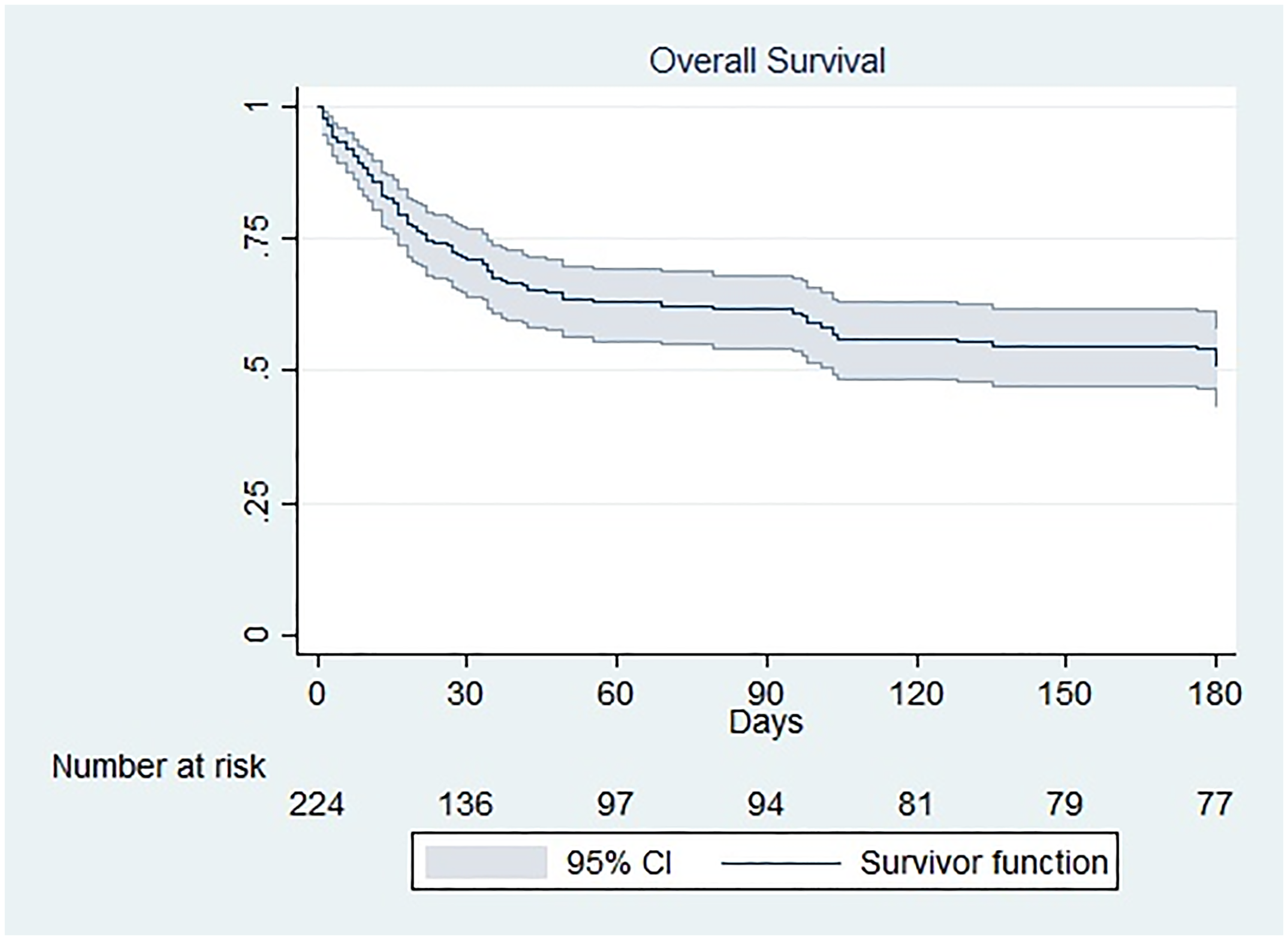
Overall Survival of Patients with Anticoagulation Therapy and a History of Chronic Kidney Disease.
When survival functions by eGFR were compared, using the log-rank test, higher survival was observed in patients with stage 3a compared to stage 4 (log-rank test p = 0.0009) and in patients with stage 5 compared to stage 4 (log-rank test p = 0.0277) (Figure 2).
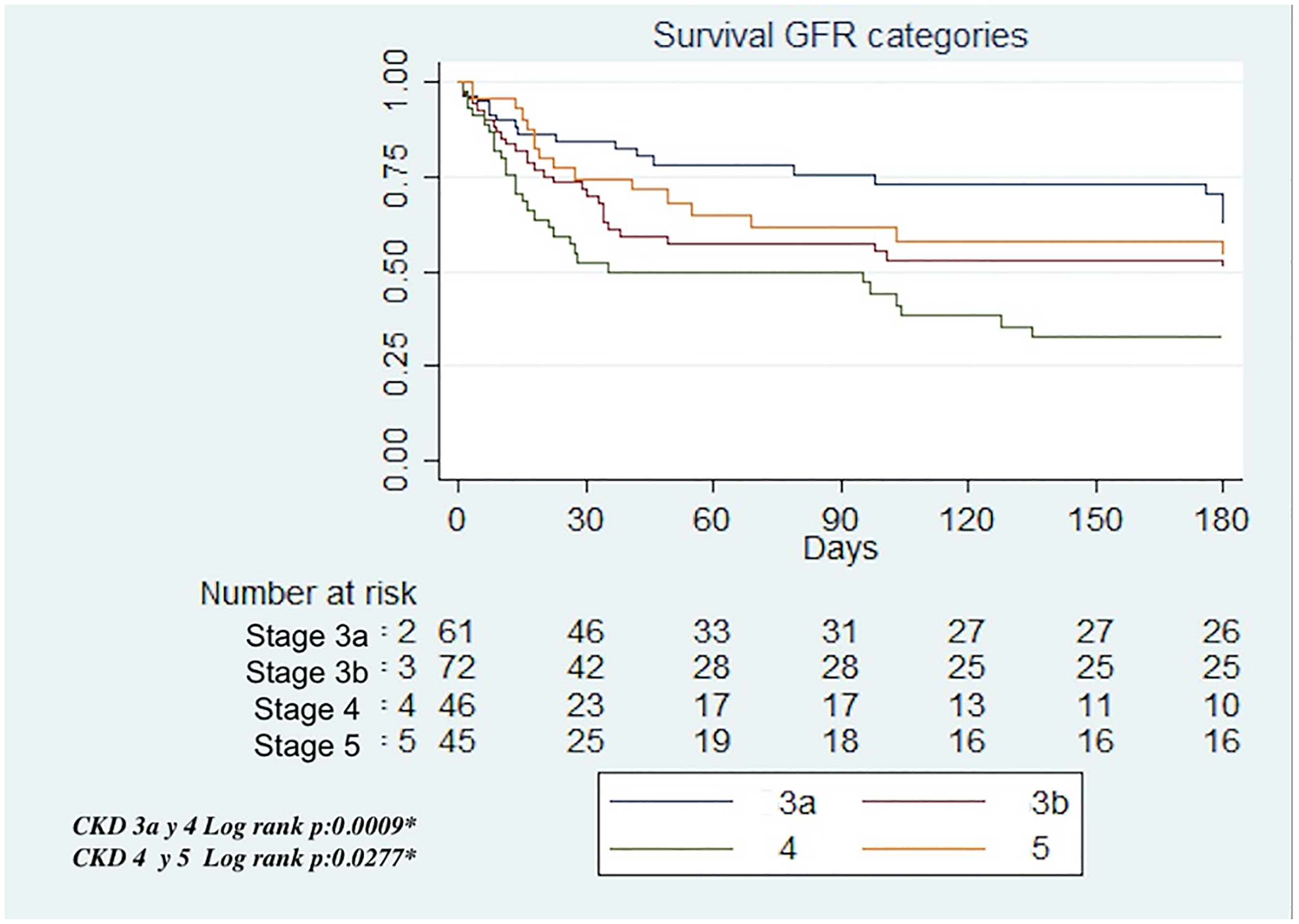
Survival in Patients with Anticoagulation Therapy, According to eGFR Stage.
Kaplan-Meier analysis of survival according to indication for anticoagulant therapy is shown in Figure 3, which shows a higher survival rate in the AF group compared to the VTE group (log-rank test p = 0.0107).

Survival of Patients with Chronic Kidney Disease According to Indication of Anticoagulation Therapy.
The cumulative incidence function of bleeding risk is illustrated in Figure 4. A significant increase in the cumulative risk of bleeding during the first 30 days after anticoagulation initiation. Subsequently, there is a decrease in the slope by day 90, which correlates with a stabilization of the cumulative bleeding risk.
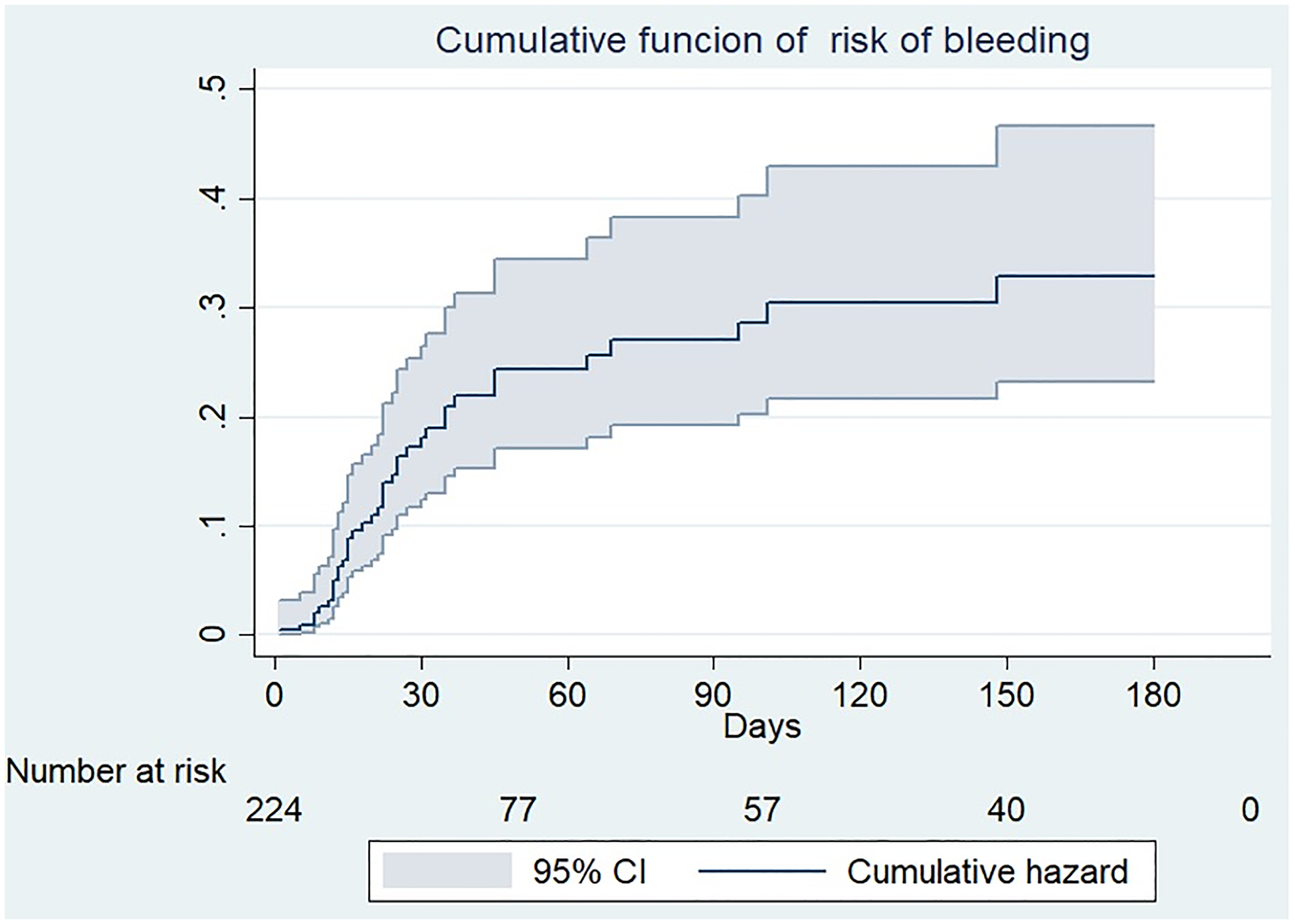
Function of Cumulative Risk of Bleeding Complications.
Finally, univariate analysis of the Cox proportional hazards model (Table 3) revealed a significant association between a higher mortality risk and the following variables: being in a subsidized health care system (HR 1.47; 95% CI 1.17–3.12; p = 0.010), cancer (HR 1.77; 95% CI 1.15–2.71; p = 0.008), and venous thromboembolic disease (HR 1.87; 95% CI 1.22–2.86; p = 0.004). In contrast, no effect on survival was observed for arterial hypertension (HR 0.44; 95% CI 0.29–0.68; p = 0.001), coronary heart disease (HR 0.42; 95% CI 0.20–0.48; p = 0.022), and antiplatelet monotherapy, (HR 0.56; 95% CI 0.32–0.96; p = 0.037).
Univariate and Multivariate Analysis of Factors Associated with Mortality in Patients with CKD and Anticoagulation Therapy.

eGFR: estimated glomerular filtration rate, AC: anticoagulation, VTE: venous thromboembolic disease * Alcohol consumption defined as AUDIT >7.
On multivariate analysis, the final model retained a higher risk of mortality associated with affiliation to a subsidized regime (HR 1.89; 95% CI 1.17–3.06; p = 0.009) and with cancer (HR 1.68; 95% CI 1.10–2.58; p = 0.017), but not with comorbid hypertension (HR 0.47; 95% CI 0.31–0.72; p = <0.001). Single antiplatelet therapy, venous thromboembolic disease and coronary heart diseas were included in the initial model but were not retained because their association with mortality was not significant after adjustment and it did not improve model performance.
The proportional hazards assumption was satisfied according to the Schoenfeld residuals test (p = 0.9177).
Discussion
This study we performed a 180-day survival analysis in anticoagulated patients with chronic kidney disease (CKD) and an eGFR <60 mL/min. The results indicate a higher mortality rate within the first 30 days after anticoagulation initiation, a period also marked by an increased incidence of hemorrhagic complications. However, only 12 (13.19%) of deaths were directly attributed to anticoagulation therapy, suggesting that other factors contributed significantly to early mortality. Mortality varied by CKD stage and anticoagulation indication, with AF associated with better survival compared to VTE.
These findings are consistent with previous studies highlighting the high bleeding risk in CKD patients. Specifically, patients with eGFR = 15 to <30 ml/min per 1.73 m(2) had adjusted relative risks of hemorrhage of 1.9 (95% confidence interval [95% CI], 1.5 to 2.4) especially at initiation of treatment, underlining the importance of close monitoring early in treatment.20–22 The higher rate of hemorrhagic events in the first 30 days suggests that early bleeding risk remains a major concern, particularly in patients with advanced CKD. However, similar to our study, overall mortality attributable to anticoagulation remained relatively low. Although mortality is higher in patients with CKD than in those without CKD with a mortality rate 1.3 to 3.6 times higher, 23 there is evidence that in CKD patients with AF, those taking anticoagulants had a lower rate of all-cause mortality (HR 0.82 IC 95% 0.74 to 0.91) than those not taking anticoagulants, despite higher rates of stroke and bleeding. 24
Mortality varied according to CKD stage and anticoagulation indication. Notably, patients with CKD stage 3a had better survival compared to stage 4, and stage 5 patients had better survival compared to stage 4. This differs from previous reports, which typically show progressive worsening of survival with advancing CKD stage. For example, Rein et al showed that the continuous variable decline in kidney function significantly predicted death (HR 1.32, 95% CI 1.03 to 1.70, p = 0.032), while another study showed that after a median of 5.7 years, patients with severe progression of CKD had a significantly increased risk for mortality (HR 1.54; 95% CI 1.30 to 1.82) compared with those with mild progression.25,26 Several factors may explain our findings. First, selection and survivor bias are likely: stage 5 patients offered anticoagulation were carefully chosen, and many were already receiving dialysis with close monitoring. In contrast, stage 4 patients had generally not entered a structured dialysis program. As a result, care intensity differs, dialysis patients undergo frequent laboratory testing and medication review, which may allow earlier identification of bleeding or dosing problems. Differences in case mix may also contribute, although our study was not powered for stage-specific models. Subgroup sizes were modest, so random variation cannot be excluded. Consistent with these considerations, in the adjusted Cox model the dichotomy eGFR <30 versus ≥30 mL/min/1.73 m² was not independently associated with mortality, suggesting that factors beyond kidney function level alone likely drive the observed difference.
Additionally, in our study, patients anticoagulated for venous thromboembolism (VTE) had worse survival compared to those with atrial fibrillation (AF), which is consistent with previous studies. Goto et al demonstrated that patients with moderate to severe CKD had a higher risk of all-cause mortality (adjusted hazard ratio [aHR], 1.44; 95% CI, 1.21–1.73) and major bleeding (aHR, 1.40; 95% CI, 1.03–1.90) than patients with mild to no CKD. 27 This may be due to the inflammatory and prothrombotic burden associated with VTE, particularly in CKD patients, as well as differences in the underlying disease severity and treatment response.27–29 Although the study was conducted during the COVID-19 pandemic, which may have influenced overall mortality and thrombotic event rates, COVID-19 status could not be reliably adjusted for because of incomplete and heterogeneous testing, which may have introduced residual confounding. In this cohort, COVID-19 was not the leading cause of death. Prior studies indicate that COVID-19 independently increases thrombotic risk, with event rates exceeding 20% among hospitalized patients, and that infection further elevates the risk of venous thromboembolism and mortality in CKD, with additional risk in patients with cancer.30,31 These considerations underscore the need for prospective studies with standardized ascertainment of SARS-CoV-2 infection to allow covariate-level adjustment and to disentangle COVID-19–related mortality from outcomes driven by CKD and anticoagulation.
Being enrolled in the subsidized healthcare scheme remained an independent predictor of mortality after adjustment for measured covariates, suggesting that the excess risk is unlikely to be explained by the clinical factors included in our model. This system serves individuals who cannot contribute financially to health insurance. In the Colombian context, prior studies document structural barriers that disproportionately affect subsidized affiliates: longer waiting times, more administrative steps for authorizations and referrals, less preventive care, delays in diagnostic testing, and gaps in continuity of care.32,33 These health-system determinants plausibly contribute to delayed diagnosis, suboptimal follow-up, and more complications, thereby worsening survival even after accounting for baseline clinical differences. While unmeasured confounding cannot be completely excluded, our findings and the national evidence support targeted strategies to reduce access delays, strengthen early post-discharge anticoagulation follow-up, and improve coordination between dialysis units and anticoagulation services for medication review and complication surveillance in the subsidized scheme.
Cancer was another factor that was significantly associated with mortality, which aligns with previous studies indicating that CKD patients, particularly in advanced stages, have high cancer-related mortality rates and other adverse outcomes.34–37 Additionally, patients with cancer-associated thrombosis have higher rates of adverse outcomes. 38 The annualized death rate for VTE was 448 per 100,000 patients, representing a remarkable 47-fold increase (95% CI, 6-89, p = 0.03) compared to the general population. 37 This is attributed to cancer-induced hypercoagulability, tumor-related endothelial dysfunction, systemic inflammation, and treatment-related toxicity (chemotherapy, radiation therapy). These mechanisms collectively increase both thrombotic and bleeding risks, complicating anticoagulation management in this population. 38
The identification of hypertension as a protective factor for survival may be explained by the well-documented inverse relationship between blood pressure and mortality in CKD patients.39,40 This phenomenon, often referred to as the reverse epidemiology in CKD, 41 suggests that normal or low blood pressure is associated with worse outcomes, possibly due to reduced perfusion and inadequate cardiovascular adaptation. Additionally, in patients on renal replacement therapy, hypertension may reflect better volume status and hemodynamic stability, contributing to improved survival. 42
These findings have important clinical implications for anticoagulation management in CKD. Much of the current evidence on anticoagulation in CKD excludes patients with advanced stages and indications other than AF.11,43 The higher mortality observed in individuals with VTE highlights the need for a careful risk-benefit assessment of anticoagulation in this subgroup. Furthermore, the identification of cancer and subsidized healthcare affiliation as independent risk factors for mortality underscores the importance of personalized treatment strategies and closer follow-up in high-risk populations. In this context, our study provides a substantial sample size that includes different stages of CKD and multiple indications for anticoagulation. Data collection also included assessments of whether mortality was directly related to anticoagulation therapy, providing further insight into treatment outcomes in this complex patient population.
An important advantage of our study is that, unlike other studies, it specifically analyzed the population of patients with CKD and the data were collected in the context of a medical registry, which increases accuracy and fidelity. The study provides valuable insights into a population with limited available evidence, contributing to an understudied area. To enhance data reliability, we employed a systematic patient identification process using the institutional anticoagulation registry and conducted regular audits. The inclusion of patients in a real-world clinical setting provides valuable insights into the effectiveness and safety of anticoagulation in routine practice. The implementation of a systematic follow-up at 30, 90, and 180 days allows for a more precise assessment of patient outcomes and the detection of relevant clinical events. Additionally, the study not only examined the relationship between renal function and survival but also explored the impact of other comorbidities and clinical variables, offering a more comprehensive understanding of the factors associated with mortality in this population. Given the limited evidence on anticoagulation in patients with chronic kidney disease, this study provides relevant data that may serve as a foundation for future research.
The main limitation of our study is its retrospective nature and single institution setting; on the other hand, some of the observed differences were not statistically significant, probably because, for example, there was a significant amount of missing data on patient weight. Weight had the highest degree of missingness (21.8%), largely because this variable is not mandatory in the electronic record for general inpatient admissions and, in some cases, could not be measured due to clinical status. Other variables had minimal missing data. Although we performed targeted chart review, residual missingness may have reduced power and obscured potential associations, given that previous studies have shown that extreme body weight can significantly influence survival outcomes in anticoagulated patients. In addition, conducting the study during the COVID-19 pandemic adds complexity to data interpretation, as the pandemic may have affected mortality rates and resource availability.
In conclusion, this study provides important insights into the survival of anticoagulated patients with CKD at different stages, including advanced disease and indications beyond atrial fibrillation. The higher mortality observed within the first 30 days of anticoagulation underscores the need for close early monitoring, yet only a small fraction of these deaths was directly linked to anticoagulation. The identification of venous thromboembolism, subsidized healthcare affiliation, and cancer as independent risk factors for mortality suggests that intensified follow-up and individualized management strategies may be particularly beneficial in these high-risk groups. The study addresses a significant gap in the literature, highlighting the need for larger, prospective, and multicenter research to optimize anticoagulation practices in CKD populations.
Footnotes
Ethical Considerations and Consent to Participate
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Ethics Committee (FM-CIE- 256/2021), all patients were asked to provide informed consent for telephone follow-up after hospital discharge.
Consent for Publication
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
