Abstract
Background:
The association of patient and prescriber characteristics with use of warfarin versus direct-acting oral anticoagulants (DOACs) in patients with atrial fibrillation (AF) and chronic kidney disease (CKD) is not well studied.
Methods:
The 20% Centers for Medicare & Medicaid Services Parts A, B, and D claims data from 2010 to 2017 were used to identify patients with stage 3, 4, or 5 CKD and AF who received a DOAC (apixaban, dabigatran, rivaroxaban) or warfarin. Prescribers were categorized as cardiologists, primary care providers (PCPs), and others. Using logistic regression, we estimated odds ratios (ORs) for the association of baseline characteristics and prescriber specialty with first use of a DOAC, relative to warfarin.
Results:
We identified 22,739 individuals with CKD who were newly initiated on oral anticoagulation for AF. New DOAC prescriptions increased from 490 in 2011 to 3261 in 2017, and displaced warfarin over time (1849, 2011; 945, 2017). By Q4 of 2014, cardiologists prescribed DOACs as initial treatment more frequently than warfarin, but non-cardiologists did not do so until 2015. As of 2017, apixaban was the most widely prescribed anticoagulant, comprising 56% and 50% of prescriptions by cardiologists and non-cardiologists, respectively. PCPs (OR 0.54, 0.51-0.58) and other providers (OR 0.55, 0.51-0.59) were less likely than cardiologists to prescribe DOACs.
Conclusions:
DOAC prescriptions, particularly apixaban, increased over time and gradually displaced warfarin. The total number of patients with AF and CKD receiving anticoagulation increased over time. Cardiologists increased DOAC prescriptions more rapidly than non-cardiologists.
Introduction
Chronic kidney disease (CKD) is a growing health problem, affecting more than 45 million people in the United States. 1 Individuals with CKD have a high burden of cardiovascular disease, including atrial fibrillation (AF). 2,3 Both AF and CKD are independently associated with risk of ischemic stroke. 4
The importance of studying AF and its complications, such as stroke, in patients with kidney disease was recently highlighted during a Controversies Conference undertaken by a Kidney Disease: Improving Global Outcomes (KDIGO) work group. 5 In previous decades, vitamin K antagonists, such as warfarin, were the only proven stroke prevention therapy available for patients with AF. 6 Unfortunately, use of warfarin presents a major clinical challenge, with its complex drug interaction profile, narrow therapeutic window, and requirement for frequent laboratory monitoring.
More recently, direct-acting oral anticoagulants (DOACs) have emerged as an alternative, with at least similar safety and efficacy outcomes associated with their use in CKD patients compared with warfarin. 7 -11 Currently, 4 DOACs (apixaban, dabigatran, edoxaban, and rivaroxaban) are approved for use for AF in the United States, but they differ in their official indications by degree of CKD. We 12 and others 13,14 have reported how warfarin has been displaced by DOACs in patients with CKD, likely reflecting both increased availability of the drugs and growing clinician comfort and familiarity with prescribing them. There is a paucity of data regarding how patient characteristics or prescriber specialty are associated with the choice to prescribe DOACs versus warfarin in AF patients with CKD.
To better understand these prescribing patterns, we conducted a retrospective analysis to determine how oral anticoagulant prescribing for Medicare beneficiaries with AF and CKD has evolved over time. We also sought to identify the factors associated with first use of DOACs, relative to warfarin, following AF diagnosis, and the factors associated with switching from warfarin to a DOAC. We performed the study in Medicare, a near-universal entitlement for Americans aged ≥65 years encompassing >60 million beneficiaries. We hypothesized that cardiologists would be quicker to adopt use of DOACs than other providers, even in patients with CKD.
Methods and Materials
Data Sources
The 20% Centers for Medicare & Medicaid Services (CMS) Parts A, B, and D billing claims files for 2010 to 2017 were used to identify diagnoses of AF and CKD stage, subsequent anticoagulant prescriptions (warfarin, apixaban, rivaroxaban, dabigatran, and edoxaban), prescriber specialty, and other patient characteristics (described in more detail below). Medicare, a US government entitlement for nearly all older individuals (aged ≥65 years) in the US, is highly representative of the demographics of the older US population; there are no barriers to access by race/ethnicity or socioeconomic status, making study results highly generalizable. Disenrollment is rare: <2% of beneficiaries disenroll annually, meaning that patients can be followed for longitudinally for long periods when required. To minimize the possibility that patients were receiving drugs outside of the Medicare program, we studied only patients continuously enrolled with Medicare as their primary payer. Data used in this analysis are available from CMS, for a fee, to qualified individuals and institutions, subject to the terms of a Data Use Agreement.
Study Design and Cohort Construction
This study used a retrospective cohort design. Patients who used warfarin, apixaban, rivaroxaban, dabigatran, or edoxaban between January 1, 2011, and December 31, 2017, were identified. The index date was defined as the date of the first prescription during this interval. The prescriber of the index anticoagulant was categorized as 1 of 3 medical specialists: cardiologist; family medicine or internal medicine (primary care) practitioner; or other/unknown. Provider type was identified by linking the prescriber ID on the index Part D event to the prescriber characteristics file. To limit the study to new users of these drugs, we excluded patients who used any oral anticoagulant within 1 year prior to the index date (thereby constituting a washout period). Patients with AF (defined more fully below) were then identified within the previous year. Following this, we excluded patients with other putative indications for anticoagulation, such as hip or knee replacement surgery or a venous thromboembolic event (Supplemental Table S1) within the previous 60 days. We also excluded patients with a history of specific valvular heart diseases (Supplemental Table S2) or ESRD, and those without CKD stage 3, 4, or 5 in the year preceding the index date.
Identification of AF and CKD
Presence of AF was determined by ≥1 International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes 427.3X or corresponding ICD-10-CM code I48 from Medicare Part A or B claims within the year prior to the index date. As we required presence of an anticoagulant prescription and absence of a non-AF diagnosis, we used this approach to avoid excluding patients who had received an anticoagulant at the same time as (or shortly after) their first AF diagnosis. These patients could be inadvertently excluded if 2 codes separated by a time interval were required. ICD codes for CKD are provided in the Supplemental Table S3 and further detailed in the Supplemental Methods.
Outcomes
The first outcome was the type of anticoagulant prescribed on the index date (DOAC vs warfarin). For the second outcome, we considered only warfarin patients and defined time to drug switching after the index date (i.e., switching from warfarin to any DOAC during follow-up).
Demographic and Comorbidity Variables
Sociodemographic characteristics in this study included age, sex, race, enrollment status in Medicaid and Part D low-income subsidy (markers of socioeconomic disadvantage), and degree of urbanicity (urban/non-urban), defined by RUCA (rural-urban commuting area) codes. Comorbid conditions were identified over the year preceding the index date by ICD-9/10-CM diagnosis codes listed by Quan et al 15 on ≥1 inpatient or ≥2 outpatient/Part B claims separated by a minimum of 30 days. For additional covariate control, we also determined the CHA2DS2-VASc 16 and HAS-BLED 17 scores, and calculated numbers of hospitalizations, observation stays, emergency department encounters, and outpatient physician visits in the previous year.
To develop a score that reflects putative frailty, we used a variation of a previously developed claims-based algorithm to assign a proxy disability score, which we have shown is associated with adverse outcomes. 18,19 Further explanation for this approach is detailed in the Supplementary Methods and Supplemental Table S4.
Using Part D prescription claims, we also analyzed use of antiplatelet, nonsteroidal anti-inflammatory, antihypertensive, antiarrhythmic, antidiabetic, P-glycoprotein (P-gp) inhibitor, and statin medications during the 6 months before the index date. For each drug class, patients with a prescription date plus days of supply that spanned the index date were defined as baseline users.
Among baseline warfarin users, we defined covariates for CKD stage progression, ischemic stroke and systemic embolism, major bleeding, international normalized ratio (INR) testing density, and count of follow-up inpatient visits during the time after the index date and before switching to a DOAC or the end of follow-up. Ischemic stroke and systemic embolism events were defined by a hospitalization with a relevant ICD-9/10-CM diagnosis code (see Supplemental Table S5). Major bleeding events were defined using an adaptation 20 of the validated Cunningham bleeding algorithm. 21 We defined INR testing using Current Procedural Terminology (CPT) code 85610, ignoring tests occurring in the first 30 days after the warfarin index date. Patients with more than 4 INR tests within a 30-day period were defined as having high INR testing density (on the date of the fourth INR test), a presumptive marker of INR instability.
For logistic models, the outcome was the index use of an anticoagulant (warfarin or a DOAC). For Cox models, the outcome was the first DOAC to which a warfarin user switched following the index date.
Statistical Analysis
Patient baseline characteristics were examined overall and by index anticoagulant using means and standard deviations or frequencies and percentages, as appropriate. We excluded the edoxaban group due to it containing very few users. Patterns of anticoagulant prescribing were explored, by prescriber and index year and by prescriber and baseline CKD stage, by plotting frequencies and percentages.
Odds ratios (ORs) and 95% confidence intervals (CIs) for the cross-sectional association of baseline characteristics and prescriber specialty with first use of a DOAC versus warfarin were estimated using multivariable logistic regression. Wald estimates were used when calculating 95% confidence intervals. Separate models were run for DOACs overall versus warfarin (primary analysis) and for each individual DOAC versus warfarin (supplementary analyses).
Next, among the subset of patients using warfarin on the index date, we examined predictors of switching from warfarin to DOACs using Cox proportional hazards regression. For a given predictor, the Cox model estimates the relative hazard of switching for those who are exposed compared to those who are unexposed. Anticoagulant switching is measured in a time-to-event manner; each patient was followed from the date of first warfarin use until the earliest of the following: date of switching from warfarin to a DOAC, discontinuation of warfarin (i.e., a gap of more than 30 days without a refill after the end of a prescription), death, end-stage renal disease requiring renal replacement therapy/transplant, loss of Medicare Parts A, B, or D coverage, or end of the study period. Patients who are not observed to have switched during follow-up are referred to as “censored” observations. Multivariable Cox models were used to estimate hazard ratios (HRs) and 95% CIs for the association of baseline characteristics, warfarin prescriber specialty, and a set of time-varying predictors (CKD stage progression, ischemic stroke and systemic embolism, major bleeding, high INR testing density, and count of follow-up inpatient encounters) with switching from warfarin to DOAC. All analyses were conducted using SAS, version 9.4 (SAS Institute, Cary, NC, USA).
Compliance and Protection of Human Research Participants
The research protocol was approved by the institutional review board at Hennepin Healthcare. Data Use Agreements between the Hennepin Healthcare Research Institute and CMS were in place.
Results
Construction of the study cohort is shown in Figure 1. A total of 22,739 qualifying individuals with AF and CKD stages 3, 4, or 5 (not receiving dialysis) received a prescription for a studied anticoagulant between January 1, 2011, and December 31, 2017: 6738 (29.6%) apixaban, 3904 (17.2%) rivaroxaban, 1568 (6.9%) dabigatran, and 10,529 (46.3%) warfarin.

Construction of the study cohort.
Participant characteristics are summarized, by index drug, in Table 1. Mean age of the cohort was 78.8 ± 8.6 years; 11, 596 (51.0%) were women, 19,719 (86.7%) were White, and 1823 (8.0%) were Black. CKD stage 3 was present in 19,166 (84.3%) and CKD stage 5 in 213 (0.9%).
Baseline Characteristics of the Patients With CKD Prescribed an Oral Anticoagulant for Atrial Fibrillation.
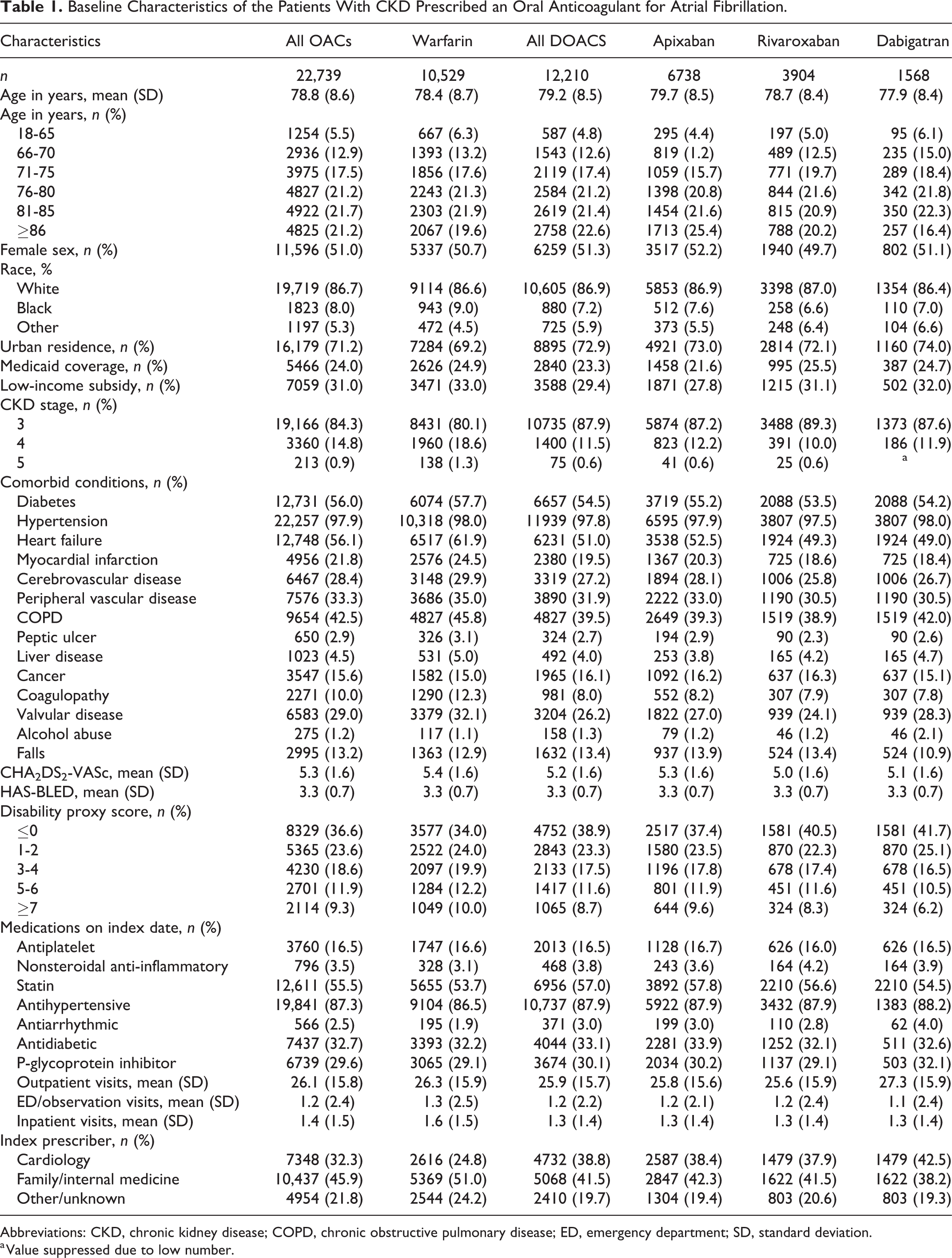
Abbreviations: CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; ED, emergency department; SD, standard deviation.
a Value suppressed due to low number.
Temporal Trends in DOAC Prescribing
Temporal trends in DOAC and warfarin prescribing for CKD patients with AF are shown, by prescriber specialty, in Figure 2. Cardiologists were the largest single prescriber specialty (7348 prescriptions, or 32.3% of the total), but, collectively, primary care providers were responsible for 10,437 (45.9%) of prescriptions.

Temporal trends in prescribing among (A) cardiologists, (B) primary care providers, (C) all other providers.
The total number of patients treated with any anticoagulant increased over time as the DOACs were introduced to the US market. Further, the number of DOAC prescriptions increased and appeared to gradually displace warfarin over time overall. However, the shift from warfarin to DOACs occurred earlier when cardiologists were the initial prescribers. For example, among cardiologists (Figure 2A), DOACs collectively were prescribed more commonly than warfarin in 2014, but not until 2015 among primary care providers and all other prescribers. Apixaban became the most widely prescribed agent among cardiologists in 2015 and among other prescribers in 2016.
Temporal Trends in Prescribing DOACs by CKD Stage and Provider Specialty
We next evaluated DOAC use, relative to warfarin, by CKD stage and prescriber specialty (Figure 3). Among patients of cardiologists, 4222 (66.5%) with CKD stage 3 and 510 (50.8%) with CKD stage 4 to 5 were prescribed a DOAC. In contrast, for patients of primary care and all other providers, DOACs were prescribed for 4409 (50.8%) (primary care providers) and 2038 (50.8%) (other providers) with CKD stage 3 and for 659 (37.5%) (primary care providers) and 306 (37.7%) (other providers) with CKD stages 4 to 5. Apixaban was prescribed most often, in CKD stage 3 for 2287 (36.0%) by cardiologists, 2462 (28.4%) by primary care providers, and 1125 (27.2%) by all other providers.

Prescribing by CKD stage among (A) cardiologists, (B) primary care providers, (C) all other providers.
Multivariable ORs for the association of baseline characteristics and prescriber specialty with initial use of any DOAC, compared with use of warfarin, in CKD patients with AF are shown in Table 2. Older age (OR 1.16, 95% CIs 1.10-1.33 for individuals aged 76-80 years and OR 1.15, 95% CIs 1.01-1.32 for individuals aged 81-85 years) and urban residence (OR 0.88, 95% CIs 0.83-0.93 for rural residence) were associated with a higher likelihood of being prescribed a DOAC versus warfarin. Increasing (worsening) CKD stage was associated with less DOAC use versus warfarin (OR 0.59, 95% CIs 0.55-0.64 for CKD stage 4 and OR 0.44, 95% CIs 0.44-0.33-0.59 for CKD stage 5). Patients with a history of cancer, alcohol abuse, falls, and use of non-steroidal anti-inflammatory drugs, antiarrhythmics, or antidiabetics were more likely to receive DOACs. Patients with comorbid conditions such as heart failure (OR 0.76, 95% CIs 0.72-0.81), history of myocardial infarction (OR 0.87, 95% CIs 0.81-0.94), or coagulopathy (OR 0.72, 95% CIs 0.66-0.79) were less likely. Notably, increasing disability proxy score (a potential marker for frailty) was associated with greater likelihood of DOAC use relative to warfarin (e.g., OR 1.27, 95% CIs 1.15-1.41 for score 5-6 and OR 1.30, 95% CIs 1.16-1.45 for score ≥7). Patients receiving an initial prescription from cardiologists, relative to non-cardiologists, were also more likely receive a DOAC than to receive warfarin (OR 0.54, 95% CIs 0.51-0.58 for family/internal medicine and OR 0.55, 95% CIs 0.51-0.59 for physicians of other/unknown type). Results were generally similar when comparing use of each individual DOAC with warfarin use (Supplemental Table S6).
Association of Baseline Characteristics and Prescriber Specialty With Use of DOACs Versus Warfarin.a

Abbreviations: CI, confidence interval; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; DOAC, direct-acting anticoagulant; ED, emergency department; LIS, low-income subsidy; INR, international normalized ratio; NSAID, non-steroidal anti-inflammatory drug; OR, odds ratio; SE, systemic embolism; TIA, transient ischemic attack.
a Odds ratios or hazard ratios for which the value is >1 represent a greater odds of initially prescribing a DOAC relative to warfarin or a greater hazard of switching to a DOAC relative to remaining on warfarin. Analogously, odds ratios or hazard ratios for which the value is <1 represent a lower odds of initially prescribing a DOAC relative to warfarin or a lesser hazard of switching to a DOAC relative to remaining on warfarin.
b Time-varying.
c More than 4 INR tests within 1 month, not counting tests within the first (initiation) month of warfarin.
The association of the CHA2DS2-VaSC stroke and HAS-BLED bleeding risk scores with anticoagulant used is shown in Supplemental Table S7. Higher CHA2DS2-VaSC score (i.e., predicting greater risk of stroke) was associated in a graded fashion with a lower likelihood of being prescribed a DOAC compared with warfarin; we did not observe a graded association between HAS-BLED score and type of drug prescribed.
Patterns of anticoagulant use during follow-up among individuals prescribed warfarin at baseline demonstrated that 3082 (29.3%) continued to use warfarin until the end of follow-up and 6677 (63.4%) ceased using any anticoagulant. Only 769 (7.3%) switched from warfarin to a DOAC: 311 (3.0%) (representing 40.4% of the switchers) changed to rivaroxaban and 285 (2.7%) (representing 37.1% of the switchers) to apixaban.
Multivariable HRs for the association of baseline characteristics, prescriber specialty, and select time-varying variables with switching from warfarin to any DOAC are also shown in Table 2. The hazard ratio of switching was higher among women (HR 1.24, 95% CIs 1.07-1.45) and lower among non-urban residents (HR 0.82, 95% CIs 0.70-0.96). CKD stage 4 was associated with a lower hazard ratio of switching (HR 0.83, 95% CIs 0.68-0.99 for CKD stage 4), there was a similar trend for CKD stage 5 (HR 0.45, 95% CIs 0.19-1.08) although power was reduced due to a lower number of patients. In contrast, ischemic stroke/systemic embolism events (HR 2.89, 95% CIs 1.63-5.10), high INR testing density (marker for INR instability) (HR 1.60, 95% CIs 1.30-1.98), and more hospitalizations during follow-up (e.g., HR 1.56, 95% CIs 1.19-2.04 for ≥2 inpatients encounters) were each associated with a greater rate of switching.
Discussion
In this retrospective cohort study, we examined temporal trends in DOAC prescriptions in a large cohort of Medicare beneficiaries with AF and CKD. We found that, over the course of just a few years, DOACs became much more commonly used than warfarin in this population. However, the total number of prescriptions for any anticoagulant roughly doubled over this relatively brief period of time. The displacement of warfarin by DOACs occurred earlier when cardiologists, rather than primary care providers, were the prescribers. Cardiologists also appeared more likely to prescribe DOACs to patients with more advanced CKD. Older age, history of cancer, and alcohol abuse were associated with greater likelihood of initial DOAC prescription, while apparent INR lability (as shown by greater INR testing density) and living in a non-urban area were associated with increased and decreased likelihood, respectively, of switching from warfarin to a DOAC. Collectively, these findings provide insights into how DOAC prescribing has evolved in Medicare beneficiaries with CKD and AF.
Use of DOACs in Medicare beneficiaries with AF and CKD increased sharply in the first quarter of 2014, soon after the US Food and Drug Administration rendered approval. Within 1 year of approval, DOACs became the most frequently prescribed initial anticoagulant class in patients with AF and CKD, at least those covered by Medicare Part D. At the beginning of our study period in 2011, dabigatran was the most common DOAC used, but its use declined substantially after rivaroxaban and, later, apixaban were introduced into clinical practice. By the last quarter of 2017, apixaban was the most commonly used drug, accounting for roughly two-thirds of all oral anticoagulant prescriptions in this population.
Given that the number of DOAC prescriptions increased roughly 2-fold over a mere 6 years, an increase unlikely to be attributable to growth in the prevalence of AF in patients with CKD, it seems possible that individuals who may not have been candidates for warfarin prior to the introduction of DOACs were considered candidates for DOACs by their prescribers over time. Previously, the decision to initiate anticoagulation in patients with CKD and AF was limited to warfarin, which is characterized by the need for frequent laboratory monitoring, a complex drug profile, and a narrow therapeutic index. That the introduction of DOACs, which have an efficacy and safety profile at least as favorable as warfarin’s, 8 -11 soon resulted in their widespread use in patients with CKD suggests that prescriber confidence rapidly increased, despite contemporaneous American Heart Association/American College of Cardiology and European Society of Cardiology guidelines suggesting that warfarin remained the anticoagulant of choice in patients with CKD and AF 22 (updated guidelines now endorse their use, albeit only to creatinine clearance of 30 mL/min 5,23 -26 ). DOACs may even be prescribed for individuals who may not have been considered suitable for warfarin using traditional criteria.
Cardiologists appeared to adopt DOACs more rapidly than non-cardiologists and to prescribe DOACs to patients at more advanced stages of CKD, even among the relatively elderly patients who characterized our study cohort. Previous reports have examined the influence of provider specialty on anticoagulation practices in patients with AF, 27,28 but there is a paucity of data in this regard for AF patients with CKD. Possibly, cardiologists rapidly gained clinical experience using DOACs in a general (non-CKD) population, and thus became more receptive to prescribing them in patients with CKD. One intriguing possibility is that primary care providers eschewed prescribing any anticoagulant to less ideal candidates, and that cardiologists were more likely to attempt DOACs in perceived higher-risk patients.
Patients more likely to receive an initial DOAC prescription were generally older and more likely to have a history of falls and a higher (worse) disability proxy score. These results suggest that prescribers may have deliberately selected DOACs over warfarin, given the trial evidence suggesting generally lower rates of hemorrhages compared with warfarin. Therefore, practitioners may perceive DOACs to be safer than warfarin, at least for CKD patients who appeared to be at especially high risk of falling. A similar decision-making process may have been operative regarding individuals with a history of alcohol abuse. Previous studies 29,30 have reported lower rates of warfarin use in patients with AF and alcohol abuse due to the perceived high risk of bleeding. DOACs may provide a safer alternative in this population in the eyes of prescribers.
We hypothesized that patients whose providers considered warfarin therapy to be unsuccessful, or even dangerous, for them would be more likely to have switched to DOACs from warfarin. Increased frequency of INR testing, a putative marker of INR lability, was, as expected, associated with a switch from warfarin to a DOAC, as was a history of ischemic stroke. In contrast, patients living in a non-urban area were less likely to be switched to a DOAC; the explanation for this is uncertain, but previous studies have suggested urban-rural differences in the care of patients with cardiac disease, leading to lower likelihood of receiving newer therapies 31 or being started on a DOAC in non-urban areas. Finally, cardiologists must be using other factors beyond traditional stroke and bleeding risk stratification, such as CHA2DS2-VaSC stroke and HAS-BLED, in deciding to switch to DOACs.
Our study has important limitations. First, we used diagnosis codes from claims instead of measured creatinine and resultant estimated glomerular filtration rate values to define CKD; this is an intrinsic limitation of using Medicare claims. Second, because our study focused only on US Medicare beneficiaries with AF and CKD, our findings may not be generalizable to other populations. Future work demonstrating external validity in a different sample of patients would be important because, in general, Medicare beneficiaries are older and have higher comorbidity burdens than patients with other types of insurance, such as employer group health plans. Third, our disability proxy score has not been validated; however, our previous work strongly suggested that it identifies factors not detectable in traditional claims-based approaches using medical comorbid conditions alone. 18,19,32 Fourth, our INR testing density metric is likely an imperfect surrogate for liability in the INR. Finally, we can make no comment about long-term use of these drugs, as our study was designed to investigate the prescribers’ “intention-to-treat”, that is, the first drug selected for initial attempts at systemic anticoagulation, and how prescribing varied over time and by prescriber type. It is possible that DOACs and warfarin differ in terms of long-term use, which is doubtless influenced by the efficacy of the respective drug classes.
Conclusions
In conclusion, in a large cohort of Medicare beneficiaries with AF and CKD stage 3, 4, and 5 not receiving dialysis, we observed that DOACs, driven primarily by apixaban, steadily displaced warfarin. The number of patients initiating oral anticoagulation over time increased substantially. Cardiologists appeared to adopt DOACs earlier, and to use them in patients with more advanced stages of CKD, than did primary care providers. Further research is needed to understand the implications of increasing use of DOACs for AF, particularly in patients with CKD.
Supplemental Material
Supplemental Material, sj-docx-1-cpt-10.1177_10742484221142220 - Prescribing Patterns of Direct-Acting Oral Anticoagulants in Patients With Atrial Fibrillation and Chronic Kidney Disease: A Retrospective Cohort Analysis
Supplemental Material, sj-docx-1-cpt-10.1177_10742484221142220 for Prescribing Patterns of Direct-Acting Oral Anticoagulants in Patients With Atrial Fibrillation and Chronic Kidney Disease: A Retrospective Cohort Analysis by Jorge L. Reyes, Charles A. Herzog, Heng Yan, Nicholas S. Roetker and James B. Wetmore in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors’ Note
This study used the Centers for Medicare & Medicaid Services (CMS) 20% sample of Parts A, B, and D billing claims files for 2010 to 2017. These data are available via the Research Data Assistance Center (ResDAC, ![]() ) to qualified US investigators, for a fee, upon completion of a Data Use Agreement between CMS and the investigators’ institution. The research protocol was approved by the Institutional Review Board at Hennepin Healthcare under approval number 18-4584. All authors reviewed and approved the manuscript for submission.
) to qualified US investigators, for a fee, upon completion of a Data Use Agreement between CMS and the investigators’ institution. The research protocol was approved by the Institutional Review Board at Hennepin Healthcare under approval number 18-4584. All authors reviewed and approved the manuscript for submission.
Acknowledgment
The authors thank Susan Everson, PhD, for manuscript editing and preparation.
Author Contributions
Research idea and study design: NSR, CAH, and JBW; data acquisition: HY and JBW; data analysis/interpretation: JLR, NSR, HY, CAH, and JBW; statistical analysis: HY and NSR; supervision or mentorship: CAH; writing of the first manuscript draft: JLR; revision of the manuscript for critical content: JLR, CAH, HY, NSR, and JBW. All authors take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CAH is a consultant for AbbVie, Amgen, AstraZeneca, Corvidia, Diamedica, FibroGen, Janssen, NxStage, Pfizer, Relypsa, Sanifit, and the University of Oxford and receives grant/research support from NIH (NIDDK and NHLBI), Amgen, Bristol-Myers (via the ARISTA program), Relypsa, and the University of British Columbia. NSR receives grant/research support from NIH and Merck. JBW receives grant/research support from NIH (NIDDK and NHLBI), Merck, Genentech, GlaxoSmithKline, Bristol-Myers Squibb (via the ARISTA program), and ACADIA.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by an investigator-initiated research award from Bristol-Myers Squibb and Pfizer, New York, New York, as part of the American Thrombosis Investigator Initiated Research Program (ARISTA-USA) (grant #CV185-725) to JBW and CAH. The funder had no role in the design of the study, the interpretation of the study findings, the drafting of the manuscript, or the decision to submit the manuscript for publication.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
