Abstract
Objective:
Real-world medical cost avoidances from a US payer perspective were estimated when new oral anticoagulants (NOACs) are used instead of warfarin for the treatment of patients with venous thromboembolism (VTE).
Methods:
Reductions in real-world event rates of recurrent VTE and MB were obtained by applying rate reductions from the NOACs versus warfarin trials to the Worcester population. Incremental annual medical costs among patients with VTE and MB from a US payer perspective were obtained from the literature or claims databases. Differences in total medical costs for patients treated with NOACs versus warfarin were then estimated. Univariate and Monte Carlo sensitivity analyses were additionally carried out.
Results:
The annual total medical cost avoidances versus warfarin were greatest for VTE patients treated with apixaban (−US$4440 per patient-year [ppy]), followed by those treated with rivaroxaban (−US$2971 ppy), edoxaban (−US$1957 ppy), and dabigatran (−US$572 ppy). The medical cost avoidances remained consistent under sensitivity analyses.
Conclusion:
Based on real-world data, when any of the evaluated NOACs are used instead of warfarin for treatment of patients with acute VTE, annual medical costs are reduced. Of the NOACs, apixaban has the greatest real-world medical cost avoidance, as its use is associated with substantial reductions in both VTE and MB event rates.
Introduction
Venous thromboembolism (VTE), including deep vein thrombosis (DVT) and pulmonary embolism (PE), is a common but underdiagnosed condition, estimated in 2011 to annually cost between US$13.5 and US$27.2 billion in the United States. 1,2 VTE is the number one cause of unexpected hospital death and estimated to contribute 300 000 deaths annually. 3,4 The American Public Health Association has called for increased awareness of VTE and better understanding of its preventability and “to take more aggressive steps” for VTE prophylaxis. 4 Current guidelines recommend initial treatment for acute DVT or PE with parenteral low-molecular-weight heparin (LMWH) or anticoagulation with rivaroxaban, and additional anticoagulation therapy (such as a vitamin K antagonist (VKA), LMWH, rivaroxaban, etc) for 3 months or longer depending on bleeding risk and whether the event was provoked, unprovoked, or associated with active cancer. 5
The clinical trials, Apixaban after the initial management of pulmonary embolism and deep vein thrombosis with first-line therapy (AMPLIFY), 6 Oral direct factor Xa inhibitor rivaroxaban in patients with acute symptomatic deep-vein thrombosis or pulmonary embolism (EINSTEIN-DVT, EINSTEIN-PE), 7 –9 Edoxaban versus warfarin for the treatment of symptomatic venous thromboembolism (Hokusai-VTE trial), 10 Dabigatran versus warfarin in the treatment of acute venous thromboembolism (RE-COVER and RE-COVER II) 11,12 demonstrated that each of the new oral anticoagulants (NOACs), apixaban, rivaroxaban, edoxaban, and dabigatran, are noninferior to warfarin for treatment of acute symptomatic VTE without increasing the risk of major bleeding (MB) events. In these trials, the rates of recurrent VTE and MB were <3% and <2%, respectively. 6 –12 The Worcester VTE study was conducted on residents from metropolitan Worcester, Massachusetts, in the years 1999, 2001, and 2003. 13 The study had a mean follow-up period of 1216 days of 1567 persons with first-time VTE and reported cumulative rates of ∼16% for recurrent VTE and 12% for MB. 13 Additionally, this study found that burden of VTE did not appear to be decreasing between 1999 and 2003 and noted that the incidence rate of recurrent VTE was similar to that reported by Prandoni et al conducted over 10 years prior to the Worcester study. 13,14
It is important to examine the efficacy and safety of new VTE prophylaxis agents in the context of the real-world setting as their introduction may impact VTE recurrence and MB rates at a more considerable level than other less efficacious interventions. All 4 of the NOACs, dabigatran, rivaroxaban, apixaban, and edoxaban are approved by the Food and Drug Adminitration (FDA) for the indication of acute VTE treatment. We have previously published the medical costs avoided when NOACs are used instead of warfarin based on rates of clinical events reported in clinical trials. 15 However, according to the Worcester study, the rates of recurrent VTE and MB in the real-world setting are different from that in the clinical trial setting. 13 Therefore, in this study, we estimated the real-world medical cost avoidances from a US payer perspective when NOACs are used instead of warfarin for the treatment of patients with VTE.
Methods
Estimation of Clinical Event Rates
The clinical events evaluated included MB and recurrent VTE, which includes VTE recurrence and VTE-related death. The real-world event rates of recurrent VTE and MB associated with standard therapy were obtained from the published data of the Worcester VTE study. 13 Standard therapy for acute VTE among the majority of the study population in the Worcester VTE study was warfarin with or without LMWH. 13 For this analysis, we referred to all of the warfarin-based therapies in the Worcester study as “warfarin treated” for simplicity and clarity. The event rates of recurrent VTE and MB among patients with VTE treated with apixaban, rivaroxaban, edoxaban, and dabigatran were estimated from the published data of the corresponding clinical trials as the percentage of patients with each of the clinical events during the trial periods. 6 –12 The analysis in this article is based on previously conducted studies and does not involve any new studies of humans performed by any of the authors.
Estimation of Medical Cost Differences
Incremental annual costs, defined as the incremental costs to a US health payer of a VTE patient experiencing a clinical event during 1 year following the initial event, were obtained from the literature or health care claims databases. In this analysis, the cost for recurrent VTE was obtained from Lefebvre et al. 16 The incremental annual cost of MB was derived from a retrospective analysis of MarketScan database claims of patients with a diagnosis of VTE (January 1, 2008-December 31, 2011). 17 The medical costs associated with clinical events were inflation adjusted to 2013 cost levels via the CPI Medical Care Index. 18 Based on the absolute event rates for each of the clinical end points, the differences in total medical costs associated with use of each the NOACs versus warfarin were determined. Our study focused on the medical cost reduction driven by clinical outcomes, with drug costs, and other additional monitoring related expenses not included in this analysis.
Sensitivity Analyses
Univariate (1-way) sensitivity analyses were conducted to determine the effects of varying a single clinical event rate or the corresponding incremental cost on the medical cost differences associated with each NOAC versus warfarin comparison. In the univariate sensitivity analysis, clinical event rates and cost estimates were varied between the ranges of their respective 95% confidence intervals. A new medical cost avoidance estimate from the model was generated when the lower end or upper end estimates of a single variable was used in the economic model. Such univariate sensitivity analysis is used to help evaluate the sensitivity of the overall medical cost avoidance with the variation in a single model parameter variable. Furthermore, since these variables are often interdependent, especially in real-world scenarios, Monte Carlo analyses were also conducted as multivariate sensitivity analyses. Gaussian distributions were assumed for the estimates of clinical event rates and the corresponding cost estimates. For each cycle of a Monte Carlo analysis, the values of the clinical event rates were taken randomly from a Gaussian distribution of the mean and standard deviation of the corresponding variables. Ten thousand such Monte Carlo iterations were conducted for each NOAC versus warfarin comparison. Descriptive statistics of the total medical cost differences were measured from the results of such 10 000 random Monte Carlo cycles. The 95% confidence intervals of the mean medical cost differences were evaluated as the range between the 2.5 and 97.5 percentiles of medical costs evaluated from the 10 000 random cycles of Monte Carlo simulation for each NOAC versus warfarin comparison.
Results
Differences in Absolute Clinical Event Rates
Real-world event rates of recurrent VTE and MB in the Worcester VTE study were 11.2% and 10.8%, respectively. Differences in real-world event rates of recurrent VTE among VTE patients treated with NOACs instead of warfarin were estimated at −1.80% for apixaban, −1.23% for rivaroxaban, −2.02% for edoxaban, and 1.02% for dabigatran (Table 1). Differences in real-world event rates of MB among patients with VTE treated with NOACs instead of warfarin were estimated at −7.48% for apixaban, −4.97% for rivaroxaban, −1.73% for edoxaban, and −2.57% for dabigatran (Table 1).
Estimate of Real-World Cinical Event Rates (Per Patient-Year) Among Patients With Venous Thromboembolism (VTE) Treated With New Oral Anticoagulants (NOACs) and Warfarin.
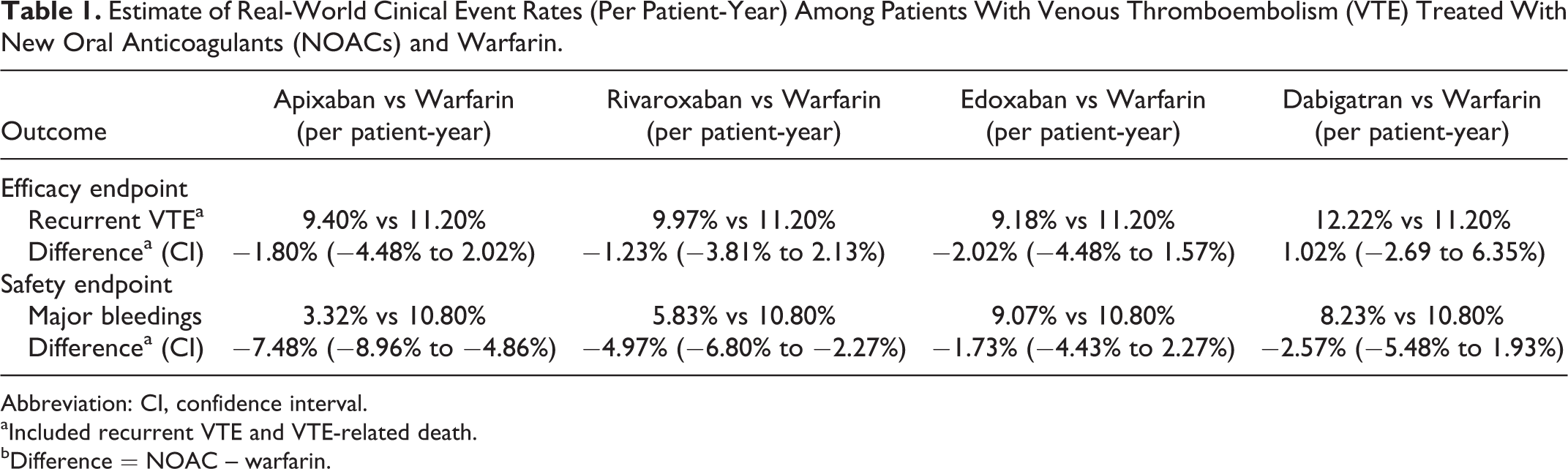
Abbreviation: CI, confidence interval.
aIncluded recurrent VTE and VTE-related death.
bDifference = NOAC – warfarin.
Medical Cost Differences
The incremental annual medical cost of a patient with a recurrent VTE event relative to a patient who did not have a VTE event in 2013 dollars was estimated at US$58 174 15 and that for a patient with an MB relative to a patient who did not have an MB at US$45 367. 16 Based on the real-world data, the annual total medical cost avoidances versus warfarin were greatest for patients with VTE treated with apixaban (−US$4440 per patient year [ppy]), followed by those treated with rivaroxaban (−US$2971 ppy), edoxaban (−US$1957 ppy), and dabigatran (−US$572 ppy; negative numbers indicate cost reduction; Table 2).
Estimates of Medical Cost Differences Among Patients With Venous Thromboembolism (VTE) Treated With NOACs Versus Warfarin Based on Clinical Trial Data and Real-World Data.
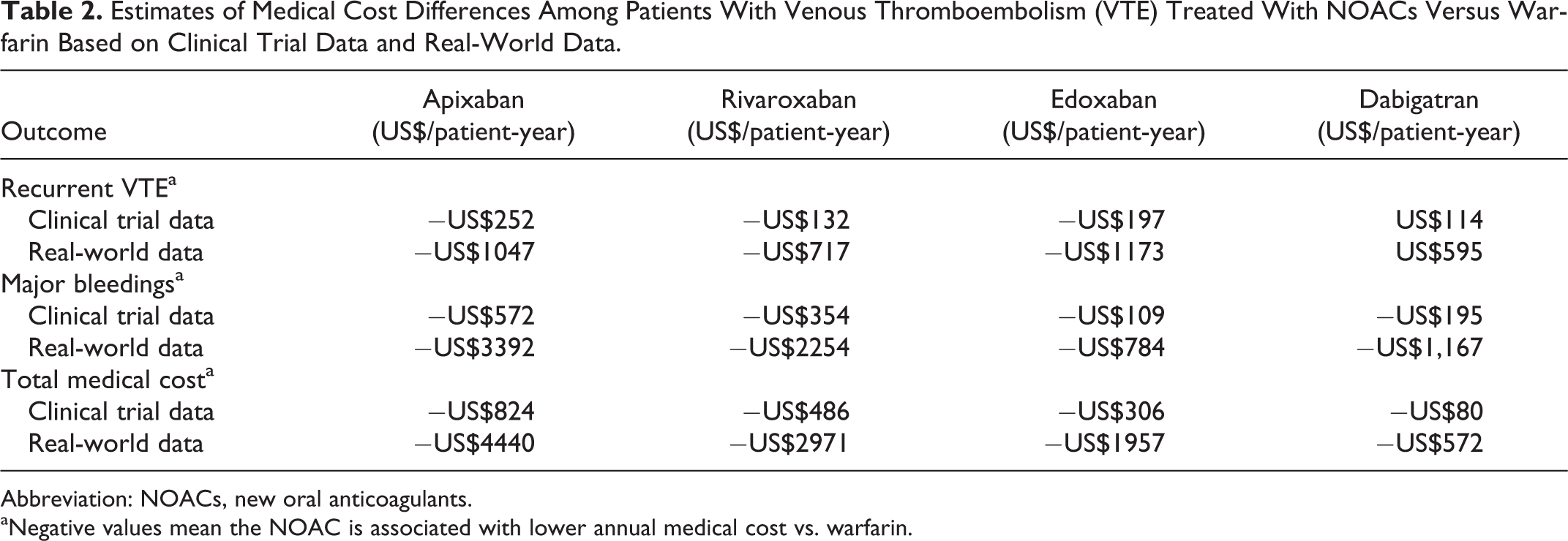
Abbreviation: NOACs, new oral anticoagulants.
aNegative values mean the NOAC is associated with lower annual medical cost vs. warfarin.
Univariate and Multivariate Sensitivity Analyses
Univariate sensitivity analysis was carried out to assess the impact of each single model parameter on the total medical cost differences estimated from the model. Figure 1 indicates how such variations in clinical event rates and event costs influenced the estimated cost differences of NOACs versus warfarin. For apixaban, rivaroxaban, edoxaban, and dabigatran, variations in event rates of VTE and MB had the greatest impact on the medical cost differences between NOACs and warfarin. Variations in VTE event rate caused the medical costs to vary from −US$5999 to −US$2220 for apixaban, from −US$4469 to −US$1016 for rivaroxaban, from −US$3390 to US$128 for edoxaban, and from −US$2731 to US$2527 for dabigatran. Variations in MB event rate caused the medical costs to vary from −US$5114 to −US$3252 for apixaban, from −US$3803 to −US$1746 for rivaroxaban, from −US$3182 to −US$144 for edoxaban, and from −US$1892 to US$1469 for dabigatran. In the case of edoxaban, variation in VTE event rates caused the medical cost difference versus warfarin to no longer be a cost reduction (cost difference >US$0). In the case of dabigatran, variations in both VTE and MB event rates caused the medical cost difference versus warfarin to no longer be a cost reduction (cost difference >US$0). Variations in costs of MB and VTE had smaller influences on the medical cost differences associated with NOACs versus warfarin.
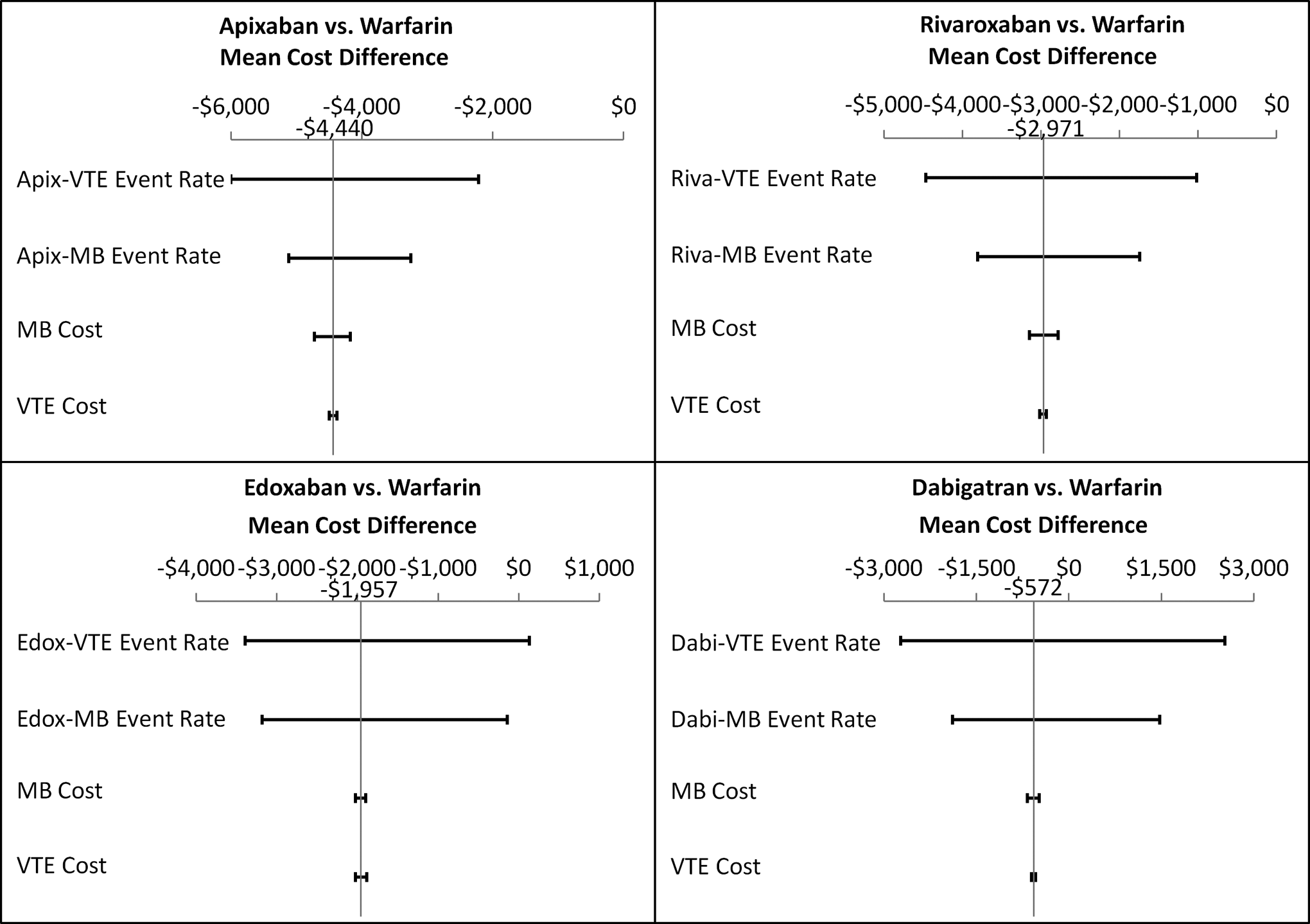
Univariate sensitivity analyses of medical cost differences of new oral anticoagulants (NOACs) versus warfarin.
Monte Carlo multivariate analyses, in which each variable of the univariate analyses were allowed to vary simultaneously for 10 000 random cycles, were used to further test the consistency of the estimates of medical cost differences associated with use of NOACs relative to warfarin (Figure 2). The mean (95% confidence interval [CI]) medical cost reductions associated with NOAC use versus warfarin were as follows: apixaban: −US$4443 [95% CI: −US$6525 to −US$2341], rivaroxaban: −US$2979 [95% CI: −US$5014 to −US$980], edoxaban: −US$1964 [95% CI: −US$4359 to US$362), and dabigatran: −US$567; (95% CI: −US$3721 to US$2604). Of the 10 000 random Monte Carlo simulation cycles, 100.0%, 99.8%, 95.1%, and 64.5% had a cost reduction <US$0 for apixaban, rivaroxaban, edoxaban, and dabigatran, respectively. The means of such 10 000 random cycles of Monte Carlo simulations were very close to the estimated cost avoidances in the default model analysis. This showed that the results of the original estimated cost avoidances are relatively robust to random variations.
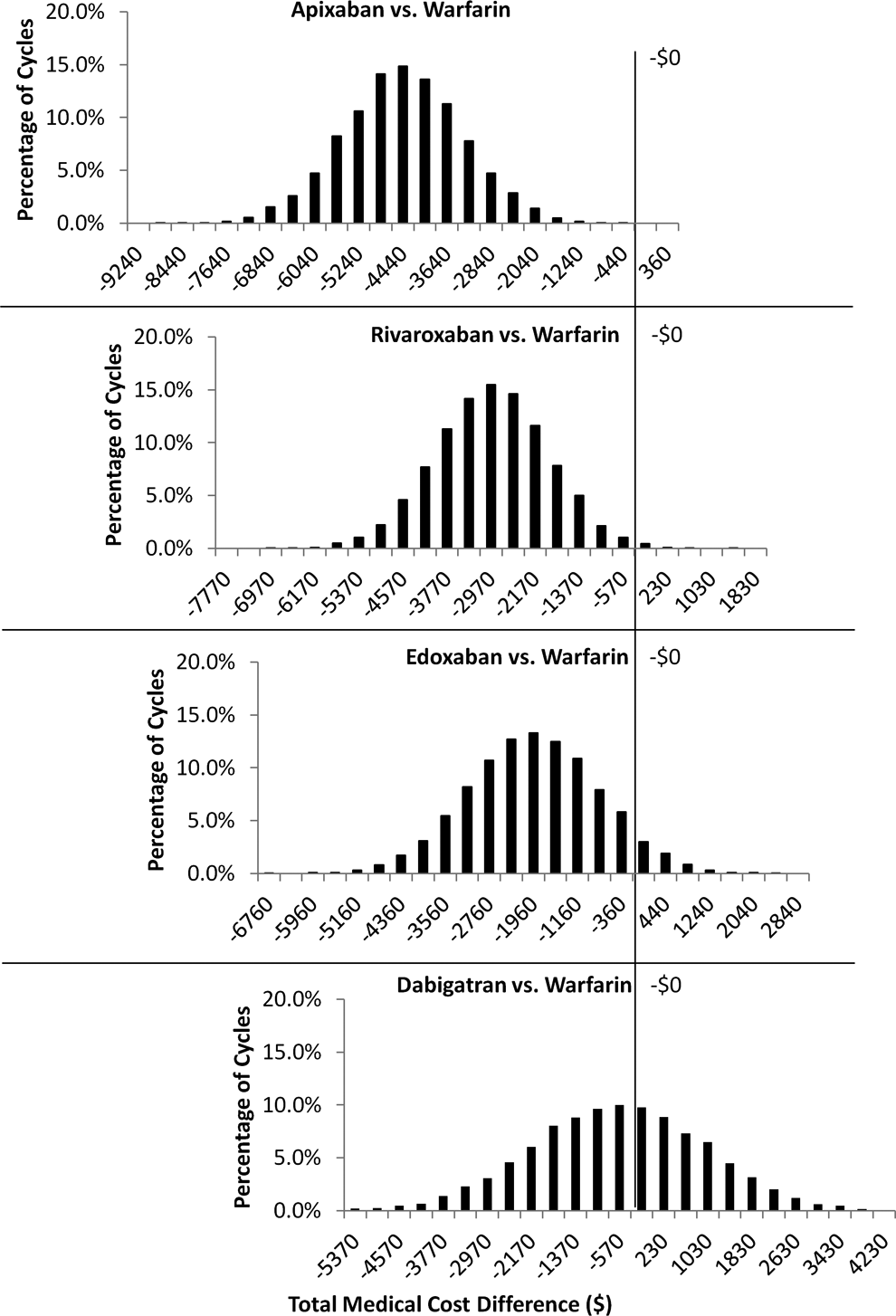
Distribution of total medical cost differences from 10 000 cycles of Monte Carlo simulations: new oral anticoagulants (NOACs) versus warfarin.
Discussion
Based on our economic analysis using real-world clinical event rates and costs, annual total medical cost avoidances versus warfarin were estimated to be greatest for patients with VTE treated with apixaban (−US$4440 ppy), followed by those treated with rivaroxaban (−US$2971 ppy), edoxaban (−US$1957 ppy), and dabigatran (−US$572 ppy). Our estimates of the medical cost differences between NOACs and warfarin were consistent under additionally evaluated sensitivity analyses. From the 10 000 random variations in each of the Monte Carlo multivariate analyses, the mean cost reductions associated with use of the NOACs versus warfarin were similar to that estimated in the default analysis, with use of apixaban associated with the greatest medical cost reduction versus warfarin. Apixaban was associated with a cost reduction in 100% of the 10 000 Monte Carlo iterations, while approximately one-third of the iterations for dabigatran showed a medical cost difference >US$0 (cost increase). Rivaroxaban and edoxaban showed a medical cost difference <US$0 in 99% and 95% of the 10 000 Monte Carlo iterations, respectively.
Other bleeding measurements such as clinically relevant nonmajor bleeding (CRNMB) and minor bleedings were measured in the clinical trials of NOACs but not measured in the Worcester VTE study. However, in the previous study in which the medical cost avoidances of patients treated with NOACs versus warfarin were estimated based on clinical trial data, the event rates of CRNMB and minor bleeding had only very minor impacts on the medical cost avoidances. 15 Thus, the lack of such data in the Worcester study is not likely to have caused major differences in the estimated real-world medical cost avoidances.
Since the differences in VTE and MB rates were substantially greater among patients treated with NOACs versus warfarin in the community setting in comparison to patients who participated in clinical trials, the medical cost avoidances associated with NOAC use were also much greater than previously estimated using clinical trial data. 15 The differences in recurrent VTE and MB rates among patients in the clinical trials and those in the Worcester study can be attributed to several factors. First, patients who participate in clinical trials receive carefully planned treatments and monitoring and therefore, their quality of care may be greater than that in real-world settings. Second, the clinical characteristics of the Worcester study population were different from that of the NOAC trial populations. 6 –13 The patient population in the Worcester study had a greater mean age, (65 years vs 54-58 years in NOAC trial populations) and approximately 50% had a provoked initial VTE event, while the proportions of patients in the clinical trials with a provoked VTE event were lower. 6 –13 Other real-world observational studies have also reported high VTE recurrence rates, with 5-year cumulative incidence rates ranging between 21.5% and 29.1%. 14,19 The higher MB rate in the Worcester study, relative to that of the NOAC clinical trials, may also be related to the greater age and high prevalence of comorbidities of the Worcester study population. 13 In our recent retrospective database analysis of 112 885 patients with VTE, we observed a combined annual rate of 28% and individual rates of 14% for MB and CRNMB. 17 These rates are also substantially higher than that reported in clinical trials of patients with VTE, which ranged from <2% for MB and 4% to 9% for CRNMB. 6 –12 In the retrospective database study, in which we compared the annual health care burden among patients who had VTE with MB, CRNMB, and no bleedings, the mean age of patients with VTE who had either MB or CRNMB was 63.6 years and ∼20% were older than the age of 80. 17 The mean age of patients having VTE with no bleedings was 59.6 years and more similar to that observed for patients in the NOAC versus warfarin acute VTE clinical trials. 6 –12,17 As real-world observational studies have shown patient populations with VTE markedly differ from those participating in clinical trials, it is important to predict medical cost differences among patients with VTE receiving different treatments in real-world settings. 6 –14,17,19
The study of Lefebvre et al, from which the cost of recurrent VTE was obtained for the current analysis, reported that in 2008 dollars the total annual health care costs among patients with recurrent VTE were US$86 744 per patient versus US$37 525 for patients who had an initial VTE but no recurrent VTE. 16 These high health care costs of patients with recurrent VTE are reflective of much greater health care utilization and poor outcomes and are pointedly suggestive of the need for better management of patients with VTE. Since none of the NOACs require frequent monitoring and dose adjustments and are administered orally they may simplify the treatment of VTE and potentially improve patient outcomes. Furthermore, the risk of MB is an important factor and concern for patients receiving VTE treatments.
Currently, the Wholesale Acquisition Costs of apixaban, rivaroxaban, edoxaban, and dabigatran in the U.S. are similar. 20 Thus, the drug prices of NOACs are not likely to affect the relative cost differences among the 4 NOACs vs. warfarin. Warfarin for VTE treatment in comparison to NOACs is less expensive when only drug costs are taken into account. However, the avoidance of VTE-related clinical events and hence lower health care costs should assist clinicians and health policy makers in determining the most cost-effective pharmacotherapies for prevention of VTE. Having the medical costs for clinical events avoided as a component of overall drug cost is highly relevant, given the fact that hospitalizations and physician/clinical services account for 51% of all US health expenditures. 21
This economic analysis has limitations in that monitoring related expenses, as well as the long-term burden of clinical events, indirect costs, and quality of life, all of which may be impacted by more efficacious pharmacotherapy for VTE were not taken into account. Further studies incorporating all of these factors may provide valuable information for health care providers, patients, and health policy makers and will likely be best accomplished by also using a real-world observational study design. Additionally, as claims, electronic health records, and marketing surveillance data become available on the outcomes and adverse event occurrences of patients with VTE treated with the different NOACs in real-world settings, it will be beneficial to conduct further future analyses to provide evidence of the most suitable NOAC treatments for particular patient populations. As mentioned, the Worcester patient population with VTE differed from the clinical trial study populations in VTE recurrence risk, preexisting conditions, and other characteristics, which may have influenced the clinical event rates and hence the estimates of medical cost differences. Therefore, the validity of the estimates of the differences in medical costs between the NOACs and warfarin may need to be further assessed when applying the study findings to other geographic regions in the United States. The incremental annual medical costs for patients with clinical events were based upon those obtained from published studies or database claims, which estimated costs from different US health insurers. Therefore, the medical costs are generalized and may not apply to specific payers of US health plans.
Conclusions
When any of the 4 NOACs are used instead of warfarin for VTE treatment, medical costs are reduced in the real-world setting. Apixaban is associated with the greatest reduction in medical costs, which is driven by medical cost reductions associated with both efficacy and safety end points. The estimated medical cost reductions associated with NOAC use relative to warfarin may be helpful in determining the overall value of the use of NOACs for patients with VTE in the United States in the real-world setting.
Footnotes
Authors’ Note
AA, AB, JT, and JL designed the study, contributed to the interpretation of data and revising of the intellectual content of the manuscript, and approved the final version to be published. JL, ML, and AA additionally assisted in the development of the cost model and contributed to the writing of the manuscript. ML also assisted in the interpretation of data.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AA is a consultant for Novosys Health which received funding from Bristol-Myers Squibb and Pfizer in connection with conducting this study and development of this manuscript. AB is an employee of Bristol-Myers Squibb and owns stock in the company. JT is an employee of Pfizer and owns stock in the company. JL and ML are employees of Novosys Health, which has received research funds from Bristol-Myers Squibb and Pfizer in connection with conducting this study and development of this manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Sponsorship for this study and article processing charges were funded by Bristol-Myers Squibb and Pfizer.
