Abstract
Cardiovascular disease and infection are the leading causes of mortality in patients with stage 5 chronic kidney disease on hemodialysis (CKD5-HD). Inflammation is a large component in the pathogenesis of both atrial fibrillation (AF) and sepsis and may link these conditions in CKD5-HD. Procalcitonin (PCT) is an inflammatory biomarker elevated in systemic infection and CKD5-HD, yet its value with regard to comorbid AF has not been thoroughly investigated. The aim of this study sought to evaluate circulating inflammatory markers, including PCT, Angiopoietin-1, Angiopoetin-2, CD40-L, C-reactive protein,
Introduction
Following cardiovascular disease, infection is the next leading cause of death in patients with stage 5 chronic kidney disease on hemodialysis (CKD5-HD). 1 Most hemodialysis patients are hospitalized at least once per year due to infection alone, and the progression of CKD is strongly associated with increased mortality due to infection. 1,2 Sepsis, a severe form of systemic infection, exerts a serious risk of morbidity and mortality in this patient population. Sarnak et al demonstrated sepsis to increase the mortality risk of hemodialysis patients 100- to 300-fold, and Dagher et al found the mortality rate of hemodialysis patients hospitalized for sepsis to reach 26.7%. 3,4 Iatrogenic sources of infection simply due to the routine process of hemodialysis places this population at a great risk; therefore, the increased burden of infection in this population necessitates the need to further evaluate the role of infection and inflammation.
Procalcitonin (PCT) is a 116-amino acid propeptide expressed in non-neuroendocrine tissues in response to inflammation and cellular injury, particularly in the setting of bacterial sepsis. 5,6 Elevated serum levels of PCT upregulated during sepsis correlates with infection severity. 7 Procalcitonin is becoming increasingly useful as a diagnostic and prognostic indicator in multiple types of infections, and recent studies have demonstrated PCT-based algorithms to be useful in guiding antimicrobial therapy in sepsis. 8,9 Additionally, PCT is elevated in CKD5-HD, but its relevance with inflammatory cardiac conditions, such as atrial fibrillation (AF), has not been established. 10,11
Atrial fibrillation is one of the most common cardiac conditions in the hemodialysis population, affecting over 25% of patients and increasing patient mortality by 1.72-fold. 12,13 As AF is the most encountered arrhythmia in severely ill patients, studies suggest sepsis may be a predisposing factor to AF, yet this relationship has not been evaluated long-term among sepsis survivors. The presence of inflammation in the development of AF is widely supported, yet its exact role in the pathogenesis of AF remains largely unknown.
Due to the strong relationship between PCT and inflammatory conditions, monitoring levels of this biomarker in CKD5-HD patients may have a dual predictive role in the evaluation of AF and sepsis. This study aimed to profile conventional and nonconventional inflammatory biomarkers, including PCT, in hemodialysis patients and demonstrate their value in relation to comorbid AF and a prior history of sepsis.
It was hypothesized that comorbid AF and a positive sepsis history amplifies circulating levels of inflammatory markers, including PCT, which are upregulated in CKD5-HD.
Materials and Methods
Patient Selection
Patients 18 years or older with CKD5-HD undergoing maintenance hemodialysis through the Loyola University Medical Center Hemodialysis Clinic were selected to participate in this study. Those in critically ill condition, on mechanical ventilation, and pregnant or nursing females were excluded.
Methods
Whole blood samples were drawn from CKD5-HD patients prior to dialysis. Samples were collected in 3.2% (0.109 mol/L) sodium citrate tubes and within 2 hours were centrifuged at 1100g for 15 minutes. The platelet-poor plasma supernatant was then separated, aliquoted, and stored at −80 °C until analysis. Commercially available control citrated plasma from healthy, nonsmoking, drug-free volunteers (n = 50; age range = 19-54 years, mean = 33 years) was purchased through George King Biomedical Inc.
Electronic Medical Record Chart Review
Review of all participants’ electronic medical records established history of sepsis, duration of hospitalization, time from sepsis resolution, and mortality. Age, sex, medications, complete blood counts, and the presence of comorbid cardiac conditions were analyzed. Cardiac comorbidities of interest included AF, left ventricular hypertrophy, heart failure, coronary artery disease, and myocardial infarction, as indicated by International Classification of Diseases diagnostic codes, cardiac catheterization reports, and physician documentation. Patients with prior cardiac catheter ablation for the treatment of AF were excluded from analysis.
Biomarker and Statistical Analysis
Plasma concentrations of PCT, Angiopoietin-1 (Ang-1), Angiopoetin-2 (Ang-2), CD40-L, C-reactive protein (CRP),
Statistical analysis utilized Microsoft Excel version 16.0 and GraphPad Prism version 8.3.0 software. Applicable analysis including nonparametric Mann-Whitney t tests and Kruskal-Wallis analysis of variance were used to compare groups. Results were expressed as mean ± standard error of the mean (SEM). P values less than .05 were considered statically significant.
Results
Ninety-seven CKD5-HD patients were enrolled in this study. Participant ages ranged from 19 to 87 years (mean age = 60 years), while sex distribution was nearly equal (female = 50.5%, male = 49.5%). Comorbid AF was present in 23 patients, while 35 patients had a history of sepsis (mean time from resolution = 762 days, SD = 730 days). One patient was excluded for previous AF catheter ablation.
Biomarker Levels Compared to Controls
Procalcitonin concentration in CKD5-HD patient plasma (121.10 ± 12.05 pg/mL) was significantly higher (P < .001) than in controls (17.64 ± 3.20 pg/mL). Plasma concentrations of Ang-1, Ang-2, CD40-L, CRP,
Biomarker Levels in Controls, CKD5-HD (+) AF, and CKD5-HD (−)AF Patients.a

Abbreviations: AF, atrial fibrillation; Ang-1, angiopoietin-1; Ang-2, angiopoetin-2; CKD5-HD, stage 5 chronic kidney disease on hemodialysis; CRP, C-reactive protein; PCT, procalcitonin; TNF-α, tumor necrosis factor-α; vWF, von Willebrand factor.
a Concentrations are reported as mean ± SEM for controls (n = 50), CKD5-HD (+) AF (n = 24), and CKD5-HD (−) AF patients (n = 72).
b P value represents comparison between CKD5-HD (−) AF and CKD5-HD (+) AF groups.
Biomarker Levels With Comorbid AF
In CKD5-HD patient plasma, levels of PCT were significantly higher (P = .0270) among those with comorbid AF (143.6 ± 25.3 pg/mL) than those without AF (113.8 ± 13.9 pg/mL; Table 1, Figure 1). Levels of Ang-2 were also significantly higher (P = .0196) in patients with comorbid AF (13.0 ± 1.55 pg/mL) compared to those without AF (9.61 ± 0.70 pg/mL), as previously reported. 14

Comparison of PCT levels in CKD5-HD patients without AF versus CKD5-HD patients with comorbid AF. AF indicates atrial fibrillation; CKD5-HD, stage 5 chronic kidney disease on hemodialysis; PCT, procalcitonin
Biomarker Levels With Sepsis History
History of sepsis correlated with elevated plasma Ang-2 (P = .0138), yet did not alter PCT levels (P = .3441), as shown in Table 2. Evaluation of sepsis survivors demonstrated PCT levels to be significantly higher (P = .0405) among those with AF (160.7 ± 39.5 pg/mL) compared to those without AF (117.4 ± 25.3 pg/mL), as shown in Table 3 and Figure 2. Analysis of variance revealed significantly higher levels of PCT in all CKD5-HD patients compared to controls, regardless of sepsis history or comorbid AF (P < .0001; Figure 3).
Comparison of Biomarker Levels Between CKD5-HD (+) Sepsis and CKD5-HD (−) Sepsis Patients.a

Abbreviations: Ang-1, angiopoietin-1; Ang-2, angiopoetin-2; CRP, C-reactive protein; CKD5-HD, stage 5 chronic kidney disease on hemodialysis; PCT, procalcitonin; TNF-α, tumor necrosis factor-α; vWF, von Willebrand factor.
a Concentrations are reported as mean ± SEM.
Comparison of Biomarker Levels Between (+) AF and (−) AF in CKD5-HD Patients With Sepsis History.a
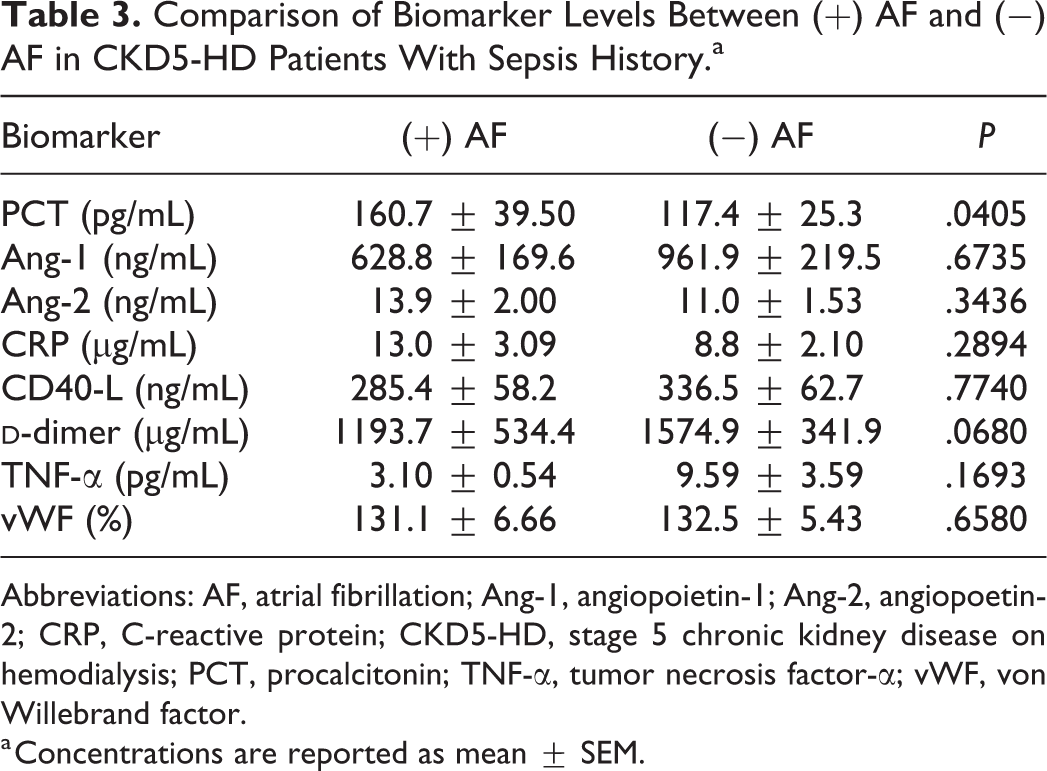
Abbreviations: AF, atrial fibrillation; Ang-1, angiopoietin-1; Ang-2, angiopoetin-2; CRP, C-reactive protein; CKD5-HD, stage 5 chronic kidney disease on hemodialysis; PCT, procalcitonin; TNF-α, tumor necrosis factor-α; vWF, von Willebrand factor.
a Concentrations are reported as mean ± SEM.
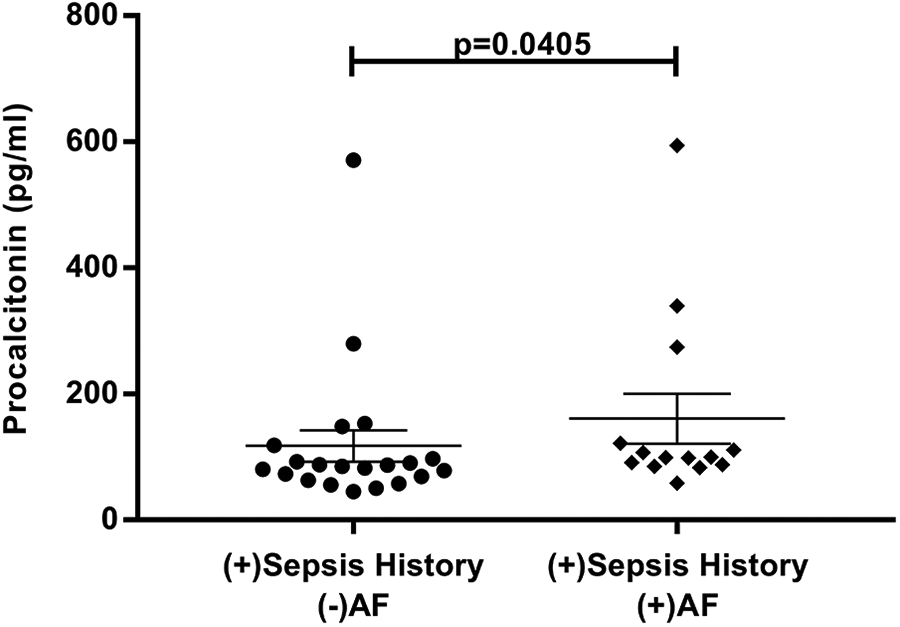
Effect of AF diagnosis on PCT levels in CKD5-HD patients with positive sepsis history. AF indicates atrial fibrillation; CKD5-HD, stage 5 chronic kidney disease on hemodialysis; PCT, procalcitonin.

Levels of procalcitonin in controls (n = 50) and CKD5-HD patients without sepsis history or AF (n = 49), with sepsis history only (n = 23), with AF only (n = 9), and patients with both sepsis history and AF (n = 15). AF indicates atrial fibrillation; CKD5-HD, stage 5 chronic kidney disease on hemodialysis.
There was no correlation between the duration of time since resolution of sepsis nor duration of sepsis and PCT levels. Baseline characteristics, including sex and age, medications, and additional cardiac comorbidities did not impact biomarker levels.
Discussion
In CKD5-HD patients, the high prevalence of comorbid AF has been speculated to originate from sustained inflammation associated with this condition. The prevalence of additional chronic inflammatory disease states, such as sepsis as demonstrated by substantial increases in PCT and Ang-2 levels seen in this study, may provide further explanation for the increased burden of AF in this population. Additionally, the presence of AF in sepsis survivors may also explain the adverse health outcomes seen in this population. As PCT is currently useful in the evaluation of sepsis, analysis of CKD5-HD patients with a prior history of sepsis revealed significantly elevated PCT levels only in those patients with comorbid AF. Therefore, measurement of this biomarker may be useful in the evaluation of AF in postseptic CKD5-HD patients.
Sustained systemic inflammation as a result of sepsis has been well-documented. A recent study evaluating a cohort of nearly 500 sepsis survivors demonstrated persistently elevated levels of inflammatory biomarkers, such as CRP and soluble programmed death ligand 1 (sPDL1), for 1 year in two-thirds of their patients. 15 Severe chronic inflammation following sepsis resolution can lead to organ failure, immunosuppression, and metabolic wasting and thus contribute to the poor long-term health outcomes known to the sepsis survivor population. 16
Although conventionally thought of as a byproduct of inflammation and cellular damage, PCT may also play a role in the propagation of the inflammatory response. Studies have demonstrated dose-dependent increases in inflammatory markers such as tumor necrosis factor α (TNF-α) and interleukin 6, as well as alterations in white blood cell function in response to exogenous recombinant PCT. 17 Therefore, PCT may be a marker of acute and sustained inflammation and serve as a potential target in the reduction of the inflammatory response.
This study also demonstrated elevated levels of Ang-2 in hemodialysis patients with a positive sepsis history. Angiopoetin-2 plays a role in endothelial dysregulation and vascular remodeling by competing with growth factor glycoproteins involved in endothelium stability. 18 The elevation of this biomarkers is likely secondary to the increased inflammation associated when both CKD5 and sepsis history are present and may possibly be due to nonclassical alternative inflammatory pathways.
Sepsis, and the resultant chronic inflammation mediated by biomarkers such as PCT, may place sepsis survivors in the hemodialysis patient population at a greater risk of developing AF. Evaluation of this relationship may further elucidate the role of inflammation and the long-term effect of endothelial damage in the pathogenesis of AF. We suspect PCT and Ang-2 to play a role in complex sustained inflammatory pathways not yet fully understood. Future prospective studies analyzing PCT levels in patients with sepsis would be useful in determining the predictive value of this biomarker in relation to AF progression.
Although this study found correlation between elevated PCT levels and comorbid AF, there was no correlation in biomarker concentrations and time since sepsis resolution. This limitation is likely influenced by the sample size of hemodialysis patients who had a history of sepsis (n = 35) and the large variation in their times since resolution (mean resolution = 762 days, SD = 730 days). Categorizing patients according to their sepsis history allowed for statistical significance to be achieved and additionally demonstrated the chronicity of this relationship with AF over time. While statistically significant, these results remain nondiscriminatory, which may be due to sample size, and therefore limit the clinical differentiation of patients among the 2 groups. Other limitations of this study included a lack of age-matched controls as the patient population was significantly more elderly than the control population and the retrospective design of this study thus restricting the ability to predict AF development. Strengths of this study include the large hemodialysis patient cohort and the prevalence of comorbid AF that closely reflects rates reported in the literature.
Conclusion
This study demonstrated that PCT is significantly elevated in hemodialysis patients with AF and in CKD5-HD patients with a history of sepsis with comorbid AF. Inflammation is prominent in CKD5-HD, which may be the reason for the increased incidence of AF in this patient population. As hemodialysis patients are at a high risk for infection complicated by significant mortality rates, further evaluation of diagnostic and predictive biomarkers is warranted. Both Ang-2 and PCT are elevated in CKD5-HD patients with comorbid AF. The pro-inflammatory biomarker, PCT, is elevated in hemodialysis patients with a history of sepsis and comorbid AF, suggesting that the resultant chronic inflammatory state following sepsis resolution may place sepsis survivors at a greater risk of developing AF. Procalcitonin and Ang-2 are nonconventional mediators of inflammation which at a cellular level may disrupt cellular integrity and endothelial stability and lead to development of comorbid conditions such as AF. It is well noted that the relative changes in conventional biomarkers of inflammation were not remarkable, which may additionally suggest the specific nature of nonconventional inflammatory pathways in the pathogenesis of these 2 conditions. Further evaluation on the prognostic value of PCT in patients with AF alone would be of interest.
Footnotes
Authors’ Note
Ethical approval for this study was obtained through the Loyola University Chicago Institutional Review Board. Written informed consent was obtained from the participants for the use of their anonymized information for this publication.
Acknowledgments
The authors are grateful for the guidance and support from the faculty and staff of the Thrombosis and Hemostasis Laboratories, Cardiovascular Research Institute (CVRI), and the Departments of Pathology and Medicine at the Loyola University Medical Center Chicago. The authors are thankful to Dr Eva Wojcik, Chairperson of the Department of Pathology, Dr Gail Hecht through the Student Training in Approaches to Research (STAR) and Research Honors programs, and Dr Seth Robia of the CVRI for their continued support throughout this project. The authors are also thankful to Mr Jonas Kingo of Aniara Diagnostics, Inc for providing some of the kits used in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Heart, Lung and Blood Institute of the National Institutes of Health under award number T35HL120835. The data in this manuscript were presented at the American Heart Association Scientific Sessions 2018.
