Abstract
Background and Objective
Su He Xiang Pill (SHXP) is a traditional Chinese medicine widely used for treating acute stroke. This study systematically evaluates the efficacy and safety of SHXP to provide evidence for integrated Traditional Chinese and Western Medicine treatment approaches for acute stroke.
Methods
Randomized controlled trials (RCTs) on SHXP for acute stroke were retrieved from CNKI, Wanfang, VIP, PubMed, the Cochrane Library, and other databases up to December 2024. Two reviewers independently screened literature, selected eligible studies, extracted data, and performed meta-analysis using RevMan 5.4. The primary outcome was the National Institutes of Health Stroke Scale (NIHSS); secondary outcomes included China Stroke Scale (CSS), Barthel Index (BI), Modified Rankin Scale (mRS), and adverse drug reactions (ADR).
Results
A total of 26 RCTs involving 2537 patients were included. Compared with conventional treatment alone: ① SHXP significantly reduced NIHSS scores (19 studies, MD = -2.37, 95% CI: −3.05 to −1.70, p < 0.00001) ; ② SHXP significantly improved BI scores (5 studies, MD = 9.79, 95% CI: 8.71 to 10.86, p < 0.00001); ③ No significant increase in ADR was observed (17 studies, OR = 0.56, 95% CI: 0.39 to 0.82, p = 0.002).
Conclusions
SHXP exhibits significant efficacy and safety in treating acute stroke (onset within 2 weeks), supporting its use in integrated Traditional Chinese and Western Medicine to improve clinical outcomes.
Keywords
Introduction
Stroke is a high-burden disease characterized by sudden loss of consciousness, paralysis, facial drooping, and slurred speech. 1 It is the second leading cause of global death and the top cause of disability/death in China, with high incidence, disability, mortality, recurrence, and economic burden.2,3 Early intervention in the acute phase (within 2 weeks of onset, 4 extendable to 1 month if organ involvement occurs) is critical for improving prognosis. 5
Current Western medical treatment for acute stroke mainly relies on symptomatic support (eg, blood pressure regulation, intracranial pressure reduction, infection prevention 6 ), but lacks targeted therapies and significant efficacy. In contrast, Su He Xiang Pill (SHXP), a classic traditional Chinese medicine, has been widely used in the acute phase of stroke, 7 making it necessary to explore an integrative Traditional Chinese and Western Medicine plan.
SHXP has a long clinical history: first documented in the Tang Dynasty's Wai Tai Mi Yao as “Qiliqia Wan,” it was later renamed and included in the Taiping Huimin Heji Ju Fang. 8 Its formula consists of 15 traditional Chinese medicinal materials (see Table 1), including aromatic ingredients (Styrax, Benzoin, Musk) for resuscitation, qi-regulating herbs (Sandalwood, Aquilaria) for promoting blood circulation, and sedative components (Cinnabaris) for relieving anxiety.9,10 These effects align with the Traditional Chinese Medicine pathogenesis of acute stroke (“phlegm turbidity obstructing orifices, qi-blood stagnation”), providing a theoretical basis for its clinical application.
Components of Su He Xiang Pill.

To our knowledge, while several primary clinical studies on SHXP for acute stroke have been published, there is a scarcity of systematic reviews and meta-analyses synthesizing this evidence. This study aims to provide an up-to-date and comprehensive systematic review and meta-analysis to quantitatively evaluate the efficacy and safety of SHXP in acute stroke treatment, incorporating the latest clinical evidence available up to December 2024, aiming to provide a basis for clinical decision-making.
Method
This review adhered to the PRISMA Statement11,12 to present the results, and all the process was conducted according to the guidance of the Cochrane Handbook for Systematic Reviews.
Search Strategy
Databases (CNKI, Wanfang, VIP, China Biomedical Literature Database, PubMed, Cochrane Library, EMbase, Web of Science) were searched for RCTs on SHXP for acute stroke, covering publications from database establishment to December 31, 2024.
PubMed Search Strategy
(“Su He Xiang Pill” OR “Suhe Xiang Pill” OR “SHXP”) AND (“Acute Stroke” OR “Acute Cerebral Infarction” OR “Acute Cerebral Hemorrhage”) AND (“Randomized Controlled Trial” OR “RCT”);
Other Databases
Keywords (Su He Xiang Pill, stroke, acute cerebral infarction, acute cerebral hemorrhage) were adjusted based on retrieval volume, combining subject terms and free terms;
Supplementary Steps
Manual tracing of references of included studies.The two-step search (first comprehensive to ensure coverage, then precise to ensure accuracy) aimed to avoid missing eligible studies.
Literature Selection
Two researchers (Zhang and Liao) independently conducted the screening process according to the inclusion and exclusion criteria, extracted data, and cross-checked it. They first read the titles and abstracts of the retrieved literature to exclude irrelevant articles, and then read the full texts to determine whether to include them. In instances of discrepancies, the researchers engaged in a discussion to reach a consensus. Should disagreements persist, a third investigator(Li) was consulted for resolution。
Inclusion Criteria
The inclusion Criteria of this study is as follows:
Study Design: Randomized controlled trials (RCTs) focusing on the use of integrated Traditional Chinese and Western medicine in the treatment of acute stroke. The language is limited to Chinese and English. Participants: Patients diagnosed with acute stroke (within 2 weeks from onset) according to the World Health Organization (WHO) diagnostic criteria for stroke or the Chinese Guidelines for the Prevention and Treatment of Cerebrovascular Diseases. Intervention: Control group: Receives standard treatment(ie, conventional treatment, CT), including blood pressure control, improvement of microcirculation, brain vasodilation, the use of neurotrophic factors, and physical therapy. Experimental group: Receives standard treatment along with SHXP (a specific Traditional Chinese Medicine formulation). Outcome Measures: The study includes the National Institutes of Health Stroke Scale (NIHSS), Glasgow Coma Scale (GCS), Barthel Index (BI), and Adverse Drug Reactions (ADR).
Exclusion Criteria
Non-randomized controlled trials or studies that do not clearly specify the research design.
Studies where only partial raw data can be extracted, making it impossible to gather complete data.
Studies in which patients’ onset time exceeds 2 weeks.
Animal studies, literature reviews, or conference abstracts.
Studies where the outcome measures are unclear or do not include NIHSS, GCS, BI, or AD.Studies where the intervention does not meet the specified criteria.
Participant Characteristics
Age, gender, and race were not limiting criteria for inclusion. Patients with acute stroke who met the aforementioned criteria were considered eligible for inclusion. This study included a total of 2537 patients, with 1270 in the experimental group and 1267 in the control group. The included studies were all patients in the acute phase of stroke. The experimental group received SHXP combined with conventional treatment, while the control group received conventional treatment. Twenty studies reported the efficacy rate. Twenty studies reported NIHSS, five studies reported the Barthel Index, two studies reported GCS, two studies reported mRS, two studies reported ESS, two studies reported MOCA, and seventeen studies reported adverse reactions to SHXP or their incidence rates. The basic characteristics of the included articles are shown in Table 2. Preferably, relevant information was directly acquired from publications, with data indirectly obtained from provided figures and datasets, when necessary.
Characteristics of Studies Included in the meta-Analysis.

Intervention Measures
This study included all randomized controlled trials comparing SHXP with conventional methods for the treatment of acute stroke, regardless of language, publication status, or blinding. Non-randomized trials, reviews, case reports, and animal studies were excluded. The experimental group received both SHXP and CT, while the control group received CT alone for comparison. Both groups received the same conventional treatment methods.
Outcome Measures
The NIHSS (National Institute of Health Stroke Scale, the National Institutes of Health Stroke Scale) score is the primary outcome measure, while BI, GCS, mRS, ESS, MoCA, and ADR are secondary outcomes.
NIHSS
The NIHSS score is a quantitative indicator for assessing the severity of AIS (acute ischemic stroke) and is commonly used as a surrogate endpoint in clinical research. 39 Patients are stratified based on NIHSS scores to guide clinical practice.
BI
The Barthel Index (BI) is used to assess the activities of daily living in stroke patients, covering aspects such as feeding, bathing, dressing, toileting, transferring between bed and wheelchair, walking, climbing stairs, personal hygiene, bowel control, and bladder control. The total score is 100 points. When the total score is ≤40 points: severe dependence, requiring complete care from others; when the total score is 41–60 points: moderate dependence, requiring care for most activities; when the total score is 61–99 points: mild dependence, requiring care for some activities; when the total score is 100 points: no dependence, able to perform daily activities independently. The BI score can effectively evaluate the recovery status of patients before and after treatment. 40
GCS
The Glasgow Coma Scale (GCS) is a widely used tool in medicine to objectively assess the level of impaired consciousness in patients with acute medical conditions or trauma. It evaluates patients based on three key aspects of responsiveness: eye-opening, verbal, and motor responses. The GCS is widely used in clinical settings to assess the severity of brain injuries, guide early management decisions (such as the need for neuroimaging or intubation), and monitor changes in a patient's condition. 41
mRS
The Modified Rankin Scale (mRS) is a widely used clinical and research tool for assessing the degree of disability and recovery in stroke patients. By quantifying a patient's ability to perform activities of daily living, it helps clinicians determine the prognosis of patients.he mRS is an important tool for predicting outcomes in stroke patients. It is not only used to assess the recovery of neurological function but also to evaluate treatment efficacy in clinical trials. For example, scores of 3–5 are often defined as a state of disability. Additionally, the mRS is used to guide rehabilitation and tailor individualized treatment plans. 42
ESS
The European Stroke Scale (ESS) was designed by Hantson et al in 1994, primarily for clinical trials involving middle cerebral artery stroke. The scale includes 14 items, covering multiple aspects such as level of consciousness, orientation, language, visual fields, gaze, and facial palsy. Higher scores indicate milder stroke severity.
MoCA
The Montreal Cognitive Assessment (MoCA) is a rapid screening tool for mild cognitive impairment (MCI), developed by Dr Nasreddine in 2004. The scale includes 11 items covering eight cognitive domains, such as attention and concentration, executive function, memory, language, visuospatial skills, abstract thinking, calculation, and orientation. 43
ADR
According to the definition by the World Health Organization (WHO), ADR refers to any harmful and unintended reaction that occurs when a qualified drug is used at a normal dose for the prevention, diagnosis, treatment of diseases, or regulation of physiological functions. 44 In this study, the adverse reactions mainly refer to the clinical symptoms unrelated to the therapeutic effects of the drug that occur in patients with acute stroke after taking Suhe Xiang Pill, such as vomiting, rash, headache, etc.
Data Extraction
Two researchers (Zhang and Liao) independently screened the literature, extracted data, and cross-checked the information. Any discrepancies were resolved through discussion or consultation with a third party (Li). The extracted content included: ① Basic study information: author, publication year, country, sample size; Patient characteristics: age, gender; ② Interventions: drug dosage, duration of treatment, route of administration; ③ Outcome measures: total effective rate, NIHSS, BI, GCS, mRS, ESS, MoCA, and incidence of adverse reactions.
Methodological Quality Assessment
Two researchers (Zhang and Liao) independently evaluated the risk of bias in the included studies and cross-checked the results. The quality of the included studies was assessed using the Cochrane Risk of Bias Assessment Tool, which includes evaluation of random sequence generation, allocation concealment, blinding, completeness of data, and selective reporting.
Data Synthesis and Analysis
Review Manager 5.4 software was used for statistical analysis. Count data were expressed as relative risk (RR), and continuous data were expressed as mean difference (MD), both with 95% confidence intervals (CI). 45 A p-value of less than 0.05 was considered statistically significant. In this study, participants were grouped and subgroup analyses were performed. The I² statistic was used to assess heterogeneity among studies. If heterogeneity was low (P > 0.1, I² ≤ 50%), a fixed-effect model (FEM) was used. If heterogeneity was high (P < 0.1, I² > 50%), sensitivity analysis was conducted to identify the sources of heterogeneity. After excluding the influence of significant clinical heterogeneity, a random-effects model (REM) was applied. If quantitative meta-analysis was not appropriate, qualitative descriptions were used instead. 46
Risk of Publication Bias
For outcome measures that include more than 10 studies, funnel plots were used to analyze the presence of publication bias. 47
Quality of Evidence
Two independent reviewers (Zhang and Liao) used the GRADE (Grading of Recommendations, Assessment, Development, and Evaluations) approach to assess the risk of bias for each included study. In case of disagreements regarding downgrading or upgrading assessments, a third party (Li and Wang) would conduct the evaluation and make the final decision. The quality of evidence was graded into four levels: “high,” “moderate,” “low,” and “very low.”
Results
Study Identification and Selection
A total of 883 records (879 articles + 4 registered records) were initially identified. After excluding 169 duplicates, 714 articles were screened: 655 were excluded by title/abstract (irrelevant topics, non-RCTs), and 59 full-text articles were assessed. Finally, 33 articles were excluded (4 with non-SHXP interventions, 1 with unreported outcomes, 28 others), leaving 26 RCTs for inclusion (see Figure 1). Table 2 summarizes the general characteristics of the 26 studies.
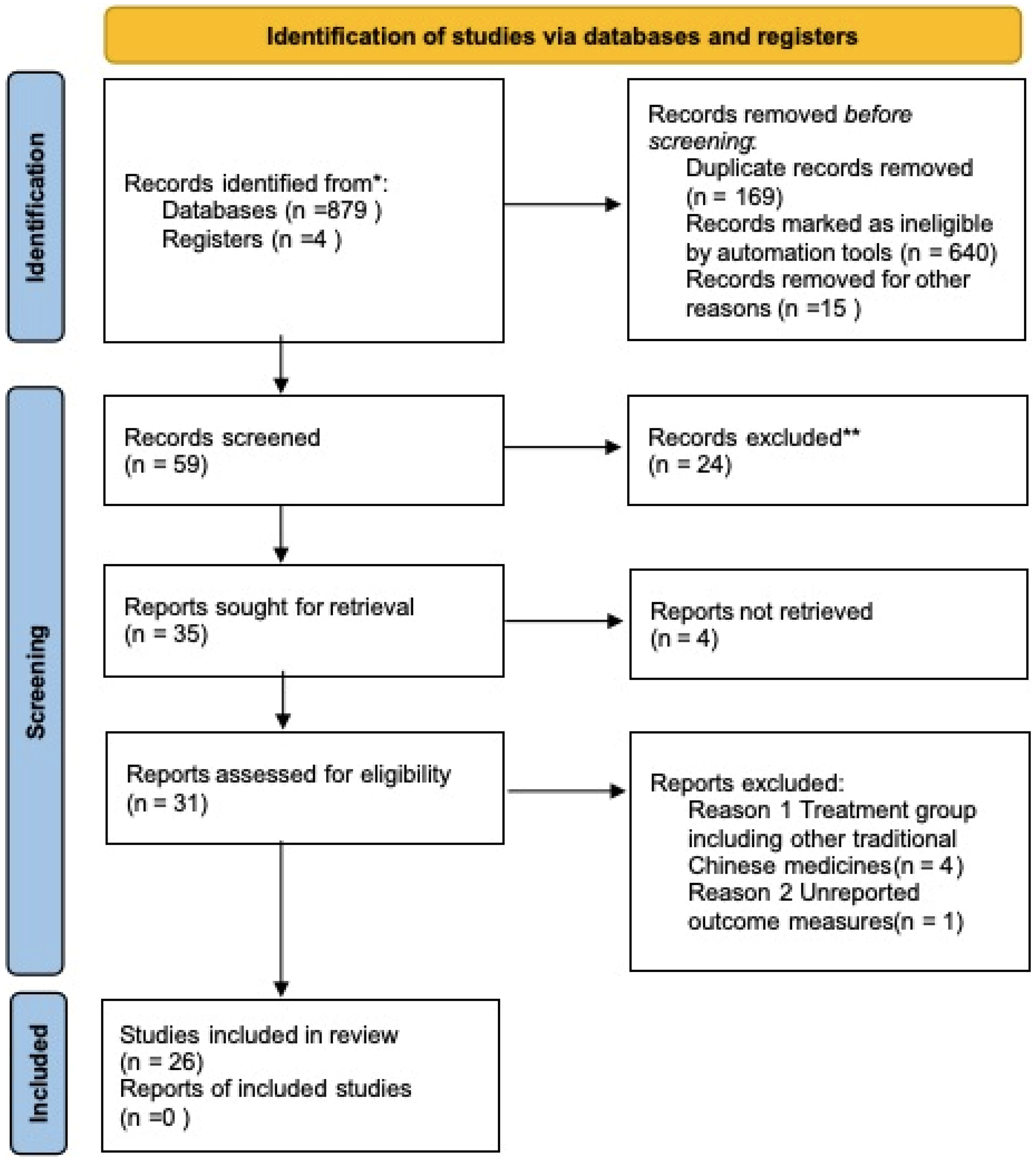
PRISMA Diagram of Searching.
Risk of Bias
The risk of bias in the trials is presented in Figure 2 and Figure 3. All 26 trials included in this study clearly used randomization methods for grouping. Among them, 19 trials provided detailed descriptions of the randomization methods: 15 trials used random number tables, 1 trial used the envelope method, one trial used the odd-even number method, one trial used the double-color ball method, and one trial used time-based grouping. The remaining 7 trials mentioned the use of randomization but did not provide detailed information on the randomization methods.Among the 26 included studies, 15 studies had low risk of bias in random sequence generation (using random number tables), 1 study had low risk (using envelope method), and 7 studies had unclear risk (no detailed randomization method reported); all studies had unclear risk of bias in allocation concealment (no relevant information reported).

Risk of Bias Summary.

Risk of bias summary.
Based on the information from the included studies, none of the studies mentioned allocation concealment or blinding of participants, personnel, or outcome assessors. This widespread lack of concealment and blinding introduces a high risk of performance biasand detection bias, as knowledge of treatment allocation could systematically influence the care provided (performance bias) and the assessment of outcomes (detection bias).
Primary Outcome
Clinical Efficacy
Evaluated according to the Guiding Principles for Clinical Research of New Chinese Medicines, with criteria as follows: Markedly effective: NIHSS score decreased by ≥46%, or BI score increased by ≥40 points; Effective: NIHSS score decreased by 18%-45%, or BI score increased by 20%-39 points; Invalid: Failure to meet the above criteria. The total effective rate = (number of markedly effective cases + number of effective cases)/total number of cases × 100%. Nineteen articles reported the clinical efficacy of patients after treatment with SXHP combined with conventional therapy. See Figure 4, the heterogeneity test among studies showed no significant heterogeneity (p = 0.49, I² = 0%), and thus a fixed-effect model was used. The meta-analysis results indicated that the clinical efficacy in the experimental group was significantly higher than that in the control group (RR = 1.21, 95% CI: 1.16,1.25, p < 0.00001).

The Forest plot Shows that the Clinical Efficacy in the Experimental Group was Better Compared with the Control Group.
NIHSS
Nineteen articles reported the NIHSS scores of patients after treatment with SXHP plus conventional therapy. Given the results of the heterogeneity test among studies (p < 0.01, I² = 97%), a random-effects model was used. The meta-analysis results showed that the NIHSS score in the experimental group was significantly lower than that in the control group (MD = -2.37, 95% CI: −3.05,-1.70, p < 0.00001) as depicted in Figure 5. Subgroup analyses were also conducted based on stroke type,(a) dosage regimen(b), and treatment duration(c), but all demonstrated considerable heterogeneity (I² > 90%) as shown in Figure 6.
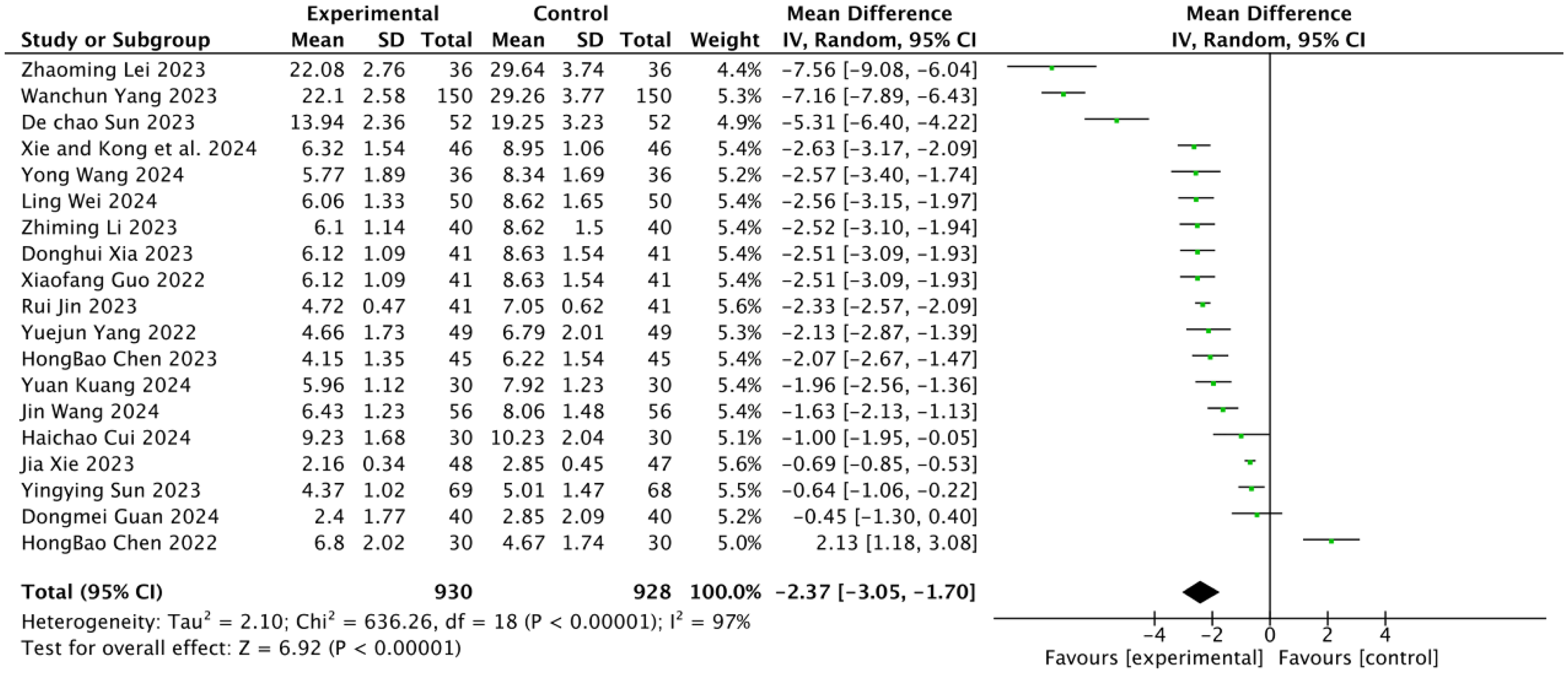
Forest Plots Showed that the NIHSS Score of the Experimental Group Decreased Compared with that of the Control Group.
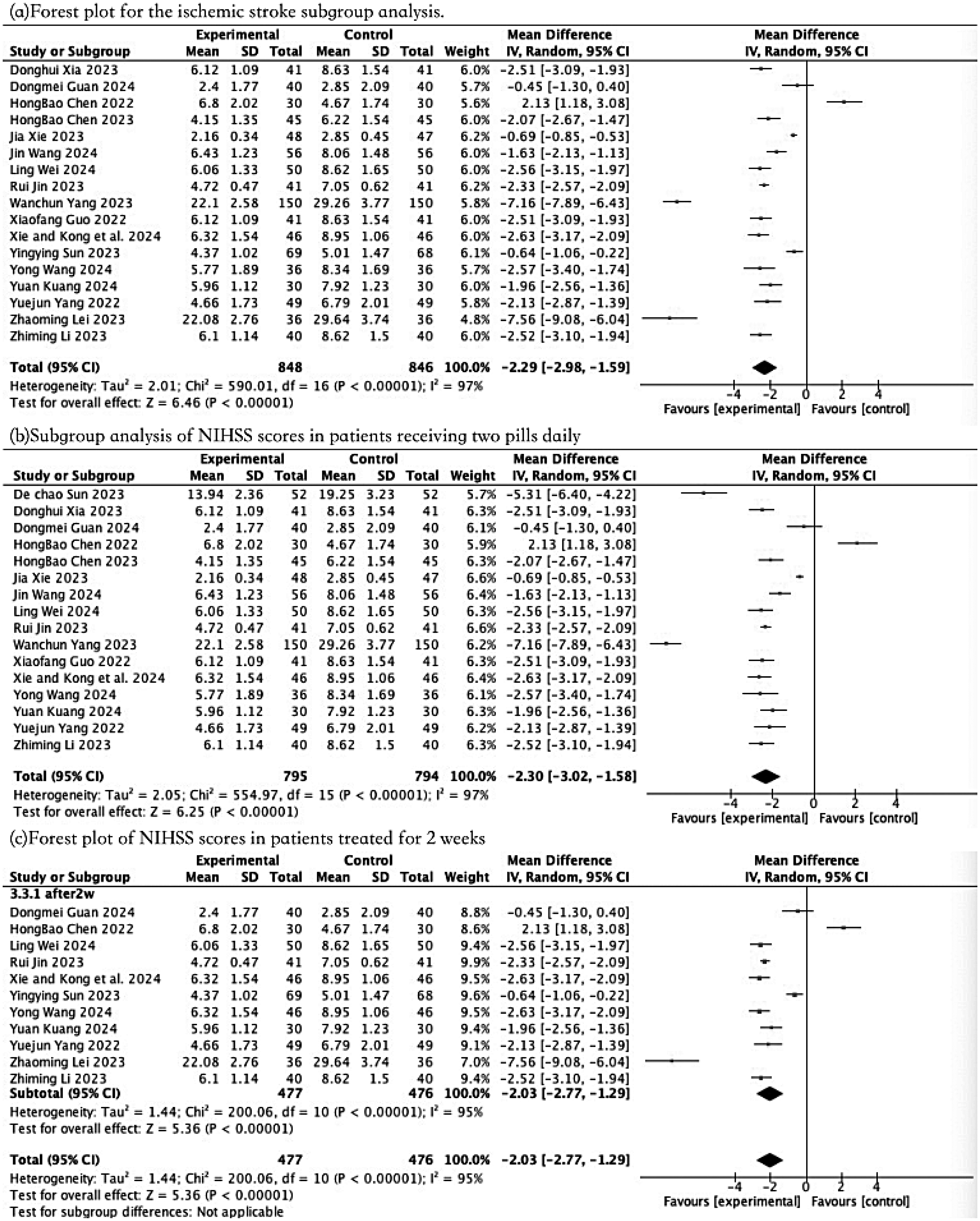
Forest Plot of Subgroup Analyses by Stroke Type (a), Treatment Regimen (b) and Duration of Therapy (c) Based on NIHSS Scores.
Secondary Outcomes
BI
The meta-analysis results showed that, compared with the control group, the ADL (Activities of Daily Living) of patients in the experimental group was significantly improved (MD = 9.79, 95% CI: 8.71,10.87, p < 0.00001) as shown in Figure 7.
Other Secondary Outcome Measures
Other outcome measures had insufficient sample sizes and were not statistically analysed.
ADR
Compared with conventional treatment alone, SHXP plus conventional treatment did not increase adverse reactions in patients (OR = 0.56, 95% CI: 0.39, 0.82, p = 0.002) refer to Figure 8. A total of 48 ADR cases were reported in the experimental group (5.93%, 48/809) and 81 cases in the control group (10.02%, 81/808) as summarised in Table 3. The main ADRs included vomiting (9 cases in experimental group vs 14 cases in control group), rash (8 cases vs 22 cases), and headache (5 cases vs 12 cases). These studies all report that adverse reactions in patients resolved following discontinuation of the medication. All ADRs were mild to moderate, and no severe ADRs or deaths related to SHXP were reported.

The BI (Barthel Index) Score in the Experimental Group was Significantly Higher than that in the Control Group.
Adverse Reactions of Studies Included in the meta-Analysis.

Publication Bias
The funnel plot for the Clinical efficacy showed potential publication bias in small trials (Figure 9).
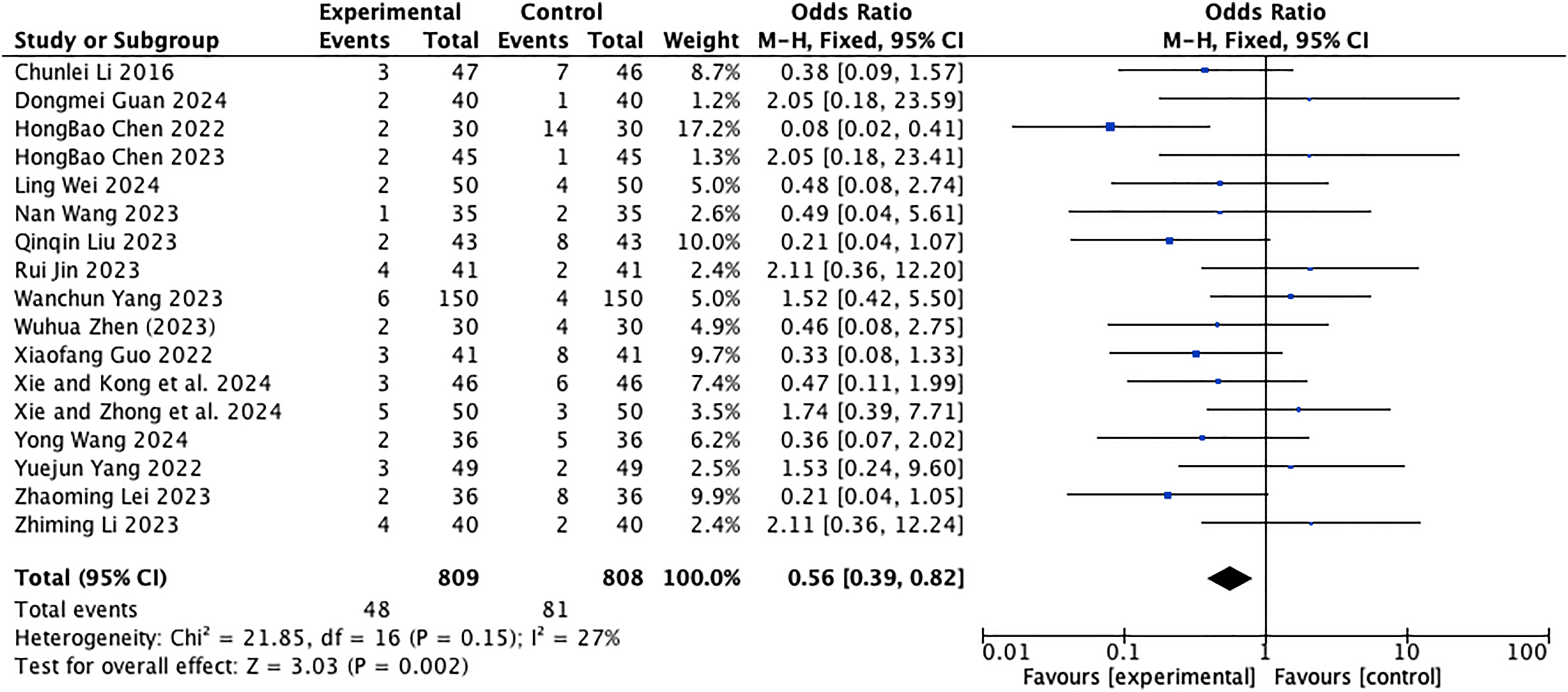
Forest Plots Showed that the Increase in ADR in the Experimental Group was not Obvious Compared with that of the Control Group.

Funnel Plots of clinical Effiency Scores Showed that there was no Obvious Publication Bias.
Discussion
Correlation Analysis of Research Results with Existing Evidence
Stroke is the second leading cause of death worldwide, and treatment during the acute phase is closely related to patient mortality and later quality of life. The combined approach of traditional Chinese and Western medicine plays a crucial role in the treatment of acute stroke. Therefore, this meta-analysis aims to evaluate the clinical efficacy and safety of SHXP in the recovery phase of acute stroke patients. This study, through a meta-analysis of 26 randomized controlled trials (RCTs) involving 2537 patients, confirmed that Su He Xiang Pill (SHXP) combined with conventional treatment is significantly superior to conventional treatment alone in improving clinical efficacy, neurological function, and activities of daily living in patients with acute stroke, without increasing the risk of adverse reactions. This result is highly consistent with the conclusions of multiple previous studies. For instance, Zhou et al (2021) found in a rat stroke model that styrax, the main component of SHXP, could improve long-term prognosis after stroke by inhibiting neuroinflammatory responses and reducing cerebral infarction volume 7 ; meanwhile, a clinical study by Guo et al (2022) showed that SHXP combined with argatroban in the treatment of acute ischemic stroke could reduce patients’ National Institutes of Health Stroke Scale (NIHSS) scores by 2.1–2.5 points and increase Barthel Index (BI) scores by 8.3–9.5 points, which is similar to the results of this study (a 2.37-point reduction in NIHSS scores and a 9.79-point increase in BI scores). 18
Interpretation from the Perspective of Traditional Chinese Medicine Theory
The therapeutic principle of SHXP, “aromatic resuscitation and warming meridians,” corresponds closely to the TCM pathogenesis of acute stroke, which is characterized by “phlegm turbidity obstructing the orifices and stagnation of qi and blood.” The formula's composition directly addresses this pathogenesis: components such as styrax, benzoin, and musk can resuscitate the mind and improve consciousness disorders caused by cerebral orifice obstruction 48 ; qi-regulating herbs like sandalwood, agarwood, and aucklandia root can soothe qi movement, promote qi-blood circulation, and alleviate cerebral ischemia and hypoxia.9,10 In addition, cinnabaris has a sedative effect, which can relieve symptoms such as restlessness and anxiety commonly seen in stroke patients, preventing emotional fluctuations from exacerbating brain damage. This multi-component strategy provides a coherent TCM theoretical foundation for the clinical application of SHXP in acute stroke.
Elucidation Based on Modern Pharmacological Mechanisms
Modern research has begun to elucidate the scientific basis underlying SHXP's effects. Studies suggest its mechanisms are multi-faceted and complementary to modern therapeutic concepts of improving cerebral circulation, protecting neurons, and regulating neurotransmitters. For example, the essential oil of Su He Xiang Pill was found to alleviate β-amyloid-induced memory impairment in mice by inhibiting tau protein phosphorylation in mice. 48 Further studies suggest Su He Xiang Pill may suppress neuroinflammation and neuronal apoptosis in acute-phase cerebral infarction rats by activating the cAMP/PKA pathway. 49 Moreover, through network pharmacology, molecular docking and clinical observation, research has revealed that Su He Xiang Pill exerts therapeutic effects against stroke by regulating neuroactive ligand-receptor interactions and calcium signalling pathways via similar core targets such as PTGS2 and ESR1. This multi-target action is predicted to inhibit apoptosis, promote neural repair, and exert anti-inflammatory effects 50 providing a mechanistic rationale for its potential broad application in the acute phase of stroke across different syndromes.
These multi-component and multi-target mechanisms of action complements the modern medical treatment concept of “improving cerebral circulation, protecting nerve cells, and regulating neurotransmitters”, providing theoretical and practical basis for the integrated Traditional Chinese and Western medicine treatment of acute stroke.
Notably, the meta-analysis of NIHSS scores and Subgroup analyses in this study showed high heterogeneity (I²=97%). It is speculated that heterogeneity may stem from the following factors: (a) Concomitant medication: ‘Differences in dosage and concomitant medication regimens may represent core factors contributing to heterogeneity. For instance, studies A and B both targeted ischaemic stroke, yet study A combined with dimethindene while study B did not, yielding significantly divergent outcomes (effect sizes). This suggests concomitant medication may serve as a critical effect-modifying factor.’ (b) Baseline severity: ‘The baseline NIHSS scores at patient enrolment varied considerably (ranging from 5 to 20). Su He Xiang Pills may exhibit markedly different efficacy for mild versus severe patients.’ (c) Dose-course interaction: Only the influence of ‘course duration’ was excluded, yet dose and course duration may interact. For instance, administering 2 pills per dose for 2 weeks versus 1 pill per dose for 4 weeks could yield differing outcomes—a complexity not fully captured by simple subgroup analyses. (d) Residual confounding factors: Even after stratification by ‘stroke type,’ studies within each subgroup may still exhibit significant differences in patient baseline characteristics (eg, baseline NIHSS score, age, time from onset to treatment), research methodology (eg, randomisation method, blinding), and outcome measurement details.
Interpretation of the Clinical Significance of Research Results
Clinical Value in Efficacy
This study showed that SHXP combined treatment increased the total clinical effective rate of acute stroke patients by 21% (RR = 1.21), reduced NIHSS scores by 2.37 points, and increased BI scores by 9.79 points. From the perspective of clinical practice, a reduction of more than 2 points in NIHSS scores is generally considered clinically significant, indicating a significant reduction in the degree of neurological impairment in patients—for example, limb muscle strength can be improved by 1–2 grades, and language or swallowing disorders can be alleviated 39 ; an increase of 9.79 points in BI scores can help patients transition from “moderate dependence” (41-60 points) to “mild dependence” (61-99 points), reducing the need for family care and lowering long-term care costs. 40 For acute stroke, a disease with high disability rates, such improvements not only enhance the short-term treatment effect for patients but also reduce the risk of long-term disability and improve their quality of life.
Clinical Guarantee in Safety
Patients with acute stroke often have comorbidities such as hypertension, diabetes, and coronary heart disease, and need to take multiple drugs simultaneously. Therefore, the safety of the treatment regimen is particularly important. This study found that the incidence of adverse reactions in the SHXP combined treatment group was 5.93% (48/809), which was lower than that in the conventional treatment alone group (10.02%, 81/808). All adverse reactions were mild to moderate (mainly vomiting, rash, and headache), with no severe adverse reactions or drug-related deaths reported; only 5 patients discontinued treatment due to adverse reactions. This result indicates that SHXP has good tolerability in the treatment of acute stroke, making it particularly suitable for elderly high-risk patients with multiple comorbidities. In addition, as a traditional Chinese patent medicine, SHXP has clear ingredients and controllable quality (modern studies have established a supercritical fluid extraction detection method for volatile components in SHXP 8 ), which further reduces the risk of medication.
In-Depth Analysis of Research Limitations and Improvement Directions
Limitations in Methodological Quality of Included Studies
Although all 26 included studies were labeled as RCTs, their overall methodological quality was low. Specific manifestations include: 7 studies did not elaborate on the random sequence generation method, which may lead to selection bias; none of the studies reported the allocation concealment scheme, making it impossible to rule out the risk of researchers selectively enrolling patients due to knowing the grouping; and no study mentioned the implementation of blinding for participants, researchers, or outcome assessors, which may result in performance bias (eg, researchers subjectively overestimating the efficacy of the treatment group) or detection bias (eg, more rigorous outcome assessment for patients in the treatment group). In addition, only 17 studies that reported adverse reactions and detailed the management measures for adverse reactions (eg, whether symptoms relieved after drug withdrawal, whether symptomatic treatment drugs were used), while the remaining studies only mentioned “no obvious adverse reactions” without specific data support, which may underestimate potential risks.
Limitations in Study Design and Result Generalization
First, the geographical distribution of the included studies was highly concentrated (all were East Asian populations, mainly Chinese patients), and there was a lack of research data from other regions such as Europe, America, and Africa, making it difficult to generalize the results to populations of different ethnicities. Second, subgroup analysis of stroke types was insufficient—among the RCTs included in this study, 23 focused on acute ischemic stroke and 2 included patients with acute cerebral hemorrhage, but the study did not separately analyze the efficacy differences of SHXP on different types of stroke. However, there are essential differences in the pathological mechanisms between ischemic and hemorrhagic stroke (ischemia-hypoxia vs hemorrhagic compression). The “warming and unblocking” effect of SHXP may be more suitable for ischemic stroke, and whether it has the potential risk of increasing bleeding in patients with hemorrhagic stroke still needs further verification. Third, the outcome indicators of this study mainly focused on short-term efficacy (2-4 weeks of treatment), and there was a lack of long-term follow-up data (eg, modified Rankin Scale [mRS] scores, recurrence rates at 3 months or 6 months), making it impossible to evaluate the impact of SHXP on the long-term prognosis of stroke patients. Furthermore, while our inclusion criteria specified patients within 2 weeks of stroke onset, we were unable to perform a subgroup analysis based on the precise timing of intervention (eg, hyper-acute phase <24–48 h vs acute phase 3–14 days). Consequently, the optimal time window for initiating SHXP therapy remains undefined and warrants investigation in future studies designed to address this question.
Specific Recommendations for Future Research
Based on the above limitations, future research should be improved in the following three aspects: ① Conduct multi-center, large-sample RCTs: Design trials in strict accordance with the CONSORT statement, clearly report random sequence generation (eg, central randomization), allocation concealment (eg, sealed envelope method), and double-blinding schemes (eg, using placebos with consistent appearance), and register through international clinical trial registration platforms (eg, ClinicalTrials.gov) to improve research transparency and reproducibility. ② Refine subgroup study design: Design separate trials for ischemic stroke (classified by infarction location and whether thrombolysis is performed) and hemorrhagic stroke (classified by bleeding volume and bleeding location) to analyze the efficacy differences of SHXP in different subtypes. Meanwhile, explore the optimal administration dosage (eg, 1 pill/time vs 2 pills/time) and course of treatment (eg, 2 weeks vs 4 weeks). ③ Extend follow-up time and increase outcome indicators: In addition to short-term indicators such as NIHSS and BI scores, include long-term outcomes (eg, disability rate, recurrence rate, mortality at 6 months) and patient-reported outcomes (eg, SF-36 quality of life score). At the same time, monitor the interaction between SHXP and other drugs (eg, antiplatelet drugs, anticoagulants) to provide more comprehensive evidence for clinical medication.
Research Shortcomings and Objective Positioning
It is necessary to objectively acknowledge that as a systematic review based on existing literature, the conclusions of this study are limited by the quality of the included studies. Due to the lack of high-quality, multi-center RCTs, the evidence strength of this study is still classified as “low” (GRADE classification) and cannot fully replace the conclusions of prospective clinical trials. In addition, the original data (eg, baseline blood glucose and lipid levels, post-treatment imaging results) in some of the included studies were incompletely reported, which may affect the comprehensive evaluation of efficacy and safety. Therefore, when clinicians use SHXP to treat acute stroke, they should formulate individualized treatment plans based on the patient's specific condition (eg, onset time, stroke type, comorbidities), drug tolerance, and local medical conditions, avoiding blind medication.
In summary, this study provides evidence-based basis for the application of SHXP in the integrated Traditional Chinese and Western medicine treatment of acute stroke, but more high-quality studies are still needed to further verify its efficacy and safety. In the future, through a stepwise research design of “basic research to clarify mechanisms—clinical research to verify efficacy—real-world research to observe long-term effects”, it is expected to give full play to the clinical value of SHXP and promote the standardization of integrated Traditional Chinese and Western medicine treatment for acute stroke.
Conclusion
Compared with conventional treatment alone, the combination of Su He Xiang Pills and conventional treatment significantly improved the total clinical effective rate, reduced the National Institutes of Health Stroke Scale (NIHSS) score (a marker of neurological function), and increased the Barthel Index (BI) score (a marker of activities of daily living) in patients with acute stroke, without increasing the incidence of adverse drug reactions (ADR)—this finding provides preliminary evidence for the application of integrated Traditional Chinese and Western medicine in acute stroke management. Notably, this conclusion applies only to patients with acute stroke within 2 weeks of onset (consistent with the inclusion criteria of this study), and its efficacy in patients with stroke beyond the acute phase remains to be verified.
However, due to the limitations of this study —including unclear randomization methods in some included trials, lack of reporting on allocation concealment and blinding of participants/personnel, and insufficient details on adverse reaction management—the included trials generally have low methodological quality, the above conclusions should be interpreted with caution. Future research requires more rigorously designed, multi-center, double-blind, randomized controlled trials with sufficient sample sizes and long-term follow-up to further verify the clinical efficacy and safety of Su He Xiang Pills in the treatment of acute stroke, especially focusing on subgroup analyses by stroke type (eg, acute cerebral infarction vs acute cerebral hemorrhage) and treatment course to further verify the clinical efficacy and safety of Su He Xiang Pills in the treatment of acute stroke, thereby providing more reliable evidence-based support for its clinical application.
Footnotes
Ethical Approval
Ethical Approval and consent to participate: not applicable.
Author Contributor Statement
The first author, Zhang, and Liao are responsible for the collection, extraction, and analysis of data. Li ensures that the data processing by both is unbiased and discusses any parts where consensus cannot be reached. Bai and Fang reviewed and revised the article.
Funding
This work was supported by the Sichuan Science and Technology Progect (2022YFS0613), Luzhou Science and Technology Project (2024YF151,2024YF138), Integrated Traditional Chinese and Western Medicine Special Project at Southwest Medical University (2023ZYYJ01, 2023ZYYJ09), and the Innovation Team of Affiliated Traditional Chinese Medicine Hospital of Southwest Medical University (2022-CXTD-05).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Data Availability
Availability of data and materials: not applicable.
