Abstract
Background
There is conflicting evidence of the correlation between fatty acid binding protein 4 (FABP4) and cardiovascular disease (CVD) in previous observational studies.
Objective
This study aims to explore the genetic causal association between FABP4 and adverse cardiovascular (CV) events.
Methods
A two-sample bidirectional Mendelian randomization (MR) analysis was performed using summary statistics from GWAS. The primary method used for MR analysis was the inverse-variance weighted (IVW) method, complemented by MR-Egger, weighted median, and weighted mode methods to explore the causal association between FABP4 and adverse CV events. For sensitivity analysis assessing heterogeneity and pleiotropy, MR-Egger and MR-PRESSO were employed to address horizontal pleiotropy, while Cochran's Q test was used to assess heterogeneity between instrumental variables (IVs). The leave-one-out analysis was used to detect outliers.
Results
IVW suggested that FABP4 showed no genetic causal effect on stroke (OR = 1.01, 95% CI = 0.98–1.04, p = 0.52), angina (OR = 1.0003, 95% CI = 0.9994–1.0012, p = 0.53), arrhythmia (OR = 1.0004, 95% CI = 0.9998–1.0009, p = 0.25), heart failure (OR = 0.99, 95% CI = 0.96–1.02, p = 0.53) or myocardial infarction (OR = 0.99, 95% CI = 0.97–1.01, p = 0.21). In the reverse MR analysis, IVW analysis showed no genetic causal effect of adverse CV events on FABP4. The results of other methods were consistent with the IVW method. Sensitivity analysis indicated the results was robust.
Conclusion
Our study did not find evidence to support a causal relationship between FABP4 and adverse CV events. Further studies are needed to comprehensively assess this potential association.
Keywords
Introduction
Cardiovascular disease (CVD) encompasses a complex array of pathophysiological states, including coronary artery disease (CAD), cerebrovascular disease, atrial fibrillation (AF), heart failure (HF), thrombotic disease and others. 1 CVD remains the leading cause of morbidity and premature death worldwide, accounting for nearly 19 million deaths in 2020 and 37% of non-communicable disease deaths under the age of 70.2‐4 Although mortality varies substantially across regions, particularly with higher rates in Eastern Europe and Central Asia, CVD continues to represent a critical global health burden. 3
The etiology of CVD is multifactorial, involving a constellation of genetic, metabolic, and environmental factors. 5 Recent decades have seen significant progress in identifying genetic variants associated with cardiovascular risk through genome-wide association studies (GWAS), particularly revealing the complex interactions between genetic susceptibility and metabolic factors in the pathogenesis of cardiovascular diseases. 6 Fatty acid binding protein 4 (FABP4), also known as adipocyte FABP (A-FABP) or aP2, belongs to the family of lipid chaperone proteins. 7 Predominantly expressed in adipocytes and macrophages, FABP4 is implicated in the regulation of insulin resistance and atherosclerosis, which may contribute to the metabolic and inflammatory underpinnings of CVD.7,8
Previous observational studies have shown that elevated circulating levels of FABP4 are associated with obesity, metabolic syndrome, hyperlipidaemia, left ventricular diastolic dysfunction and heart failure,9‐13 highlighting its possible role in cardiovascular disease and metabolic disorders. In addition, genetic variants in the FABP4 locus associated with reduced expression appear to reduce the risk of cardiovascular disease, 14 suggesting a possible genetic causal association between them. However, others studies have failed to establish a significant relationship between FABP4 concentrations and systolic dysfunction or heart failure prognosis, particularly in populations without pre-existing cardiac disease.15,16 These conflicting findings highlight the need for further research to clarify the role of FABP4 in cardiovascular disease.
Mendelian randomization (MR) provides a powerful method for elucidating causal relationships between modifiable risk factors and health outcomes. 17 By using genetic variation as an instrumental variable (IV), MR can overcome the limitations of traditional observational studies, such as confounding and reverse causation. 18 This approach provides a quasi-experimental framework to assess the causality of exposure on disease outcomes, thereby providing insight into the lifelong effects of genetic predisposition on CVD. 6
Our study aimed to investigate the causal relationship between FABP4 and adverse CV events using a two-sample bidirectional MR design. We hypothesize that FABP4 is associated with the onset and progression of cardiovascular disease, and that MR analysis may provide clearer evidence to support this association. With this study, we hope to deepen the understanding of the role of FABP4 in cardiovascular disease and potentially inform future therapeutic strategies targeting this pathway.
Methods
Study Design
The methodology employed in this study was a two-sample bidirectional MR analysis, as depicted in Central Figure. Reverse MR analysis was used to observe the causal effect of CVD on FABP4. The conceptual framework of this study was founded on three core tenets integral to MR methodology: (a) the relevance criterion, which necessitates a demonstrable and potent association between the IVs and the exposure of interest; (b) the independence criterion, which mandates that the IVs are not correlated with any confounders that may concurrently influence the exposure and the outcome; and (c) the exclusion-restriction criterion, which stipulates that the IVs exert their influence on the outcome solely through their effect on the exposure, without initiating any other causal mechanisms. 17 All data were derived from published studies or publicly available Genome-Wide Association Studies (GWAS) abstract data, for which ethical approval and informed consent had already been provided. Therefore, no separate ethical approval was required for this study.

Flow chart of the study design.
Data Sources
The outcomes of this study included stroke, angina, arrhythmia, HF and Myocardial infarction (MI). Stroke outcomes included ischemic stroke, intracerebral hemorrhage, undetermined stroke types, and detailed ischemic stroke subtypes. 19 HF was defined as a clinical diagnosis of HF of any aetiology with no inclusion criteria based on left ventricle ejection fraction. 20 MI cases were defined as positive for International Classification of Diseases version-10 (ICD10) codes I21, I22, I23, I25.2, which included MI, and complications following acute MI. Doctor-diagnosed and self-reported MI were also included in the definition of MI. 21 Following datasets form IEU Open GWAS were selected as outcome data: 1) stroke (ebi-a-GCST006906) with 40 585 European ancestry cases, 406 111 European ancestry controls 19 ; 2) angina (ebi-a-GCST90038609) with 15 527 European ancestry cases, 469 071 European ancestry controls 22 ; 3) arrhythmia (ebi-a-GCST90038611) with 7207 European ancestry cases, 477 391 European ancestry controls 22 ; 4) HF (ebi-a-GCST009541) with 47 309 European ancestry cases, 9041 European ancestry controls 20 ; 5) MI (ebi-a-GCST011364) with 395 795 European ancestry cases. 21 GWAS summary-level data for circulating FABP4 protein levels were used as the exposure. These data were obtained from the IEU Open GWAS database (ID: prot-b-71), encompassing 3394 individuals of European ancestry. Supplementary Table 1 systematically presents more details of the exposure factor and cardiovascular outcomes datasets.
Instrumental variable Selection
The selection of instrumental variables (IVs) for this study adhered to a meticulously structured process to ensure the relevance. First, we selected single nucleotide polymorphisms (SNPs) associated with circulating FABP4 (forward MR) at a threshold of P < 5 × 10−6 to secure a sufficient number of IVs for sensitivity analyses, whereas for the reverse MR (adverse CV events as the exposure) we applied the conventional genome-wide significance threshold of P < 5 × 10−8 23 Second, only SNPs with a minor allele frequency (MAF) > 0.01 were retained. 23 Third, linkage disequilibrium (LD) was pruned using a threshold of R2 < 0.001 and a window size of 10 000 kb, ensuring independence among selected SNPs.6,24 Fourth, if a candidate IV was not available in the outcome summary statistics, a proxy SNP with high LD (R2 > 0.8) was used as a substitute.6,24 Finally, to ascertain the robustness of each SNP within the IV and to minimize the risk of weak instrumental variable bias, the F statistic for each SNP was calculated using the formula: F = R2*(N−2)/(1−R2). R2 refers to the proportion of variance in exposure explained by the SNPs in IV, and the requirement for the F value is >10 25
MR Analysis
The foundational analytical approach adopted in this study was the inverse variance weighted (IVW) method, which calculates a weighted average of the effect sizes and utilizes the IVs of each SNP as the weights. 26 This approach allows to assess the causal relationship between exposure and risk of outcomes, producing estimates of the odds ratio (OR) and 95% confidence intervals (CI). To ensure the robustness of the results, supplementary analyses were conducted employing the MR-Egger, weighted median, and weighted mode methods. The MR-Egger approach, accounting for the potential presence of pleiotropy through an intercept term, offers an unbiased estimate of causal effects even in its presence. 27 The weighted median method, predicated on the validity of at least half of the IVs, was applied to examine the causal link between exposure and outcome. 28
Sensitivity Analysis
Sensitivity analyses were employed to identify potential heterogeneity and horizontal pleiotropy which could bias MR estimates. Heterogeneity among IVs was evaluated using Cochran's Q test with P > 0.05 indicating low heterogeneity, suggesting random variation among IVs with minimal impact on IVW results. 29 MR-Egger regression was utilized to assess the presence of horizontal pleiotropy, with a non-significant intercept term indicating its absence. 30 Furthermore, the MR pleiotropy residual sum and outlier (MR-PRESSO) method was applied to detect and exclude potential outliers (SNPs with p < 0.05), thereby correcting for horizontal pleiotropy and refining causal estimates. 31
Statistical Analysis
The analysis was executed using R version 4.0.5 along with the “Two-sample MR” package. Interpretative visual aids such as forest plots, scatter plots, and funnel plots were utilized for graphical representation of the data. The statistical power of the MR analyses was calculated using the mRnd online tool (https://shiny.cnsgenomics.com/mRnd/). 32
Results
SNP Selection
In this study, eleven IVs relevant to FABP4 were selected (p < 5 × 10−6). A total of 89 IVs relevant to adverse CV events (stoke, angina, arrhythmia, HF and MI) were selected (p < 5 × 10−6). The detailed information of the included IVs is summarized in Supplementary Table 2. All selected IVs exceeded the F-statistic threshold of 10.
MR Analysis
Causal Effects of FABP4 on Adverse CV Events
IVW suggested that FABP4 showed no genetic causal effect on stroke (OR = 1.01, 95% CI = 0.98–1.04, p = 0.52), angina (OR = 1.0003, 95% CI = 0.9994–1.0012, p = 0.53), arrhythmia(OR = 1.0004, 95% CI = 0.9998–1.0009, p = 0.25), HF (OR = 0.99, 95% CI = 0.96–1.02, p = 0.53) or MI (OR = 0.99, 95% CI = 0.97–1.01, p = 0.21) (Table 1 and Figures 1 and 2). The result of weighted median analysis, MR Egger and weighted mode analysis were consistent with the IVW method (Supplementary Table 3).
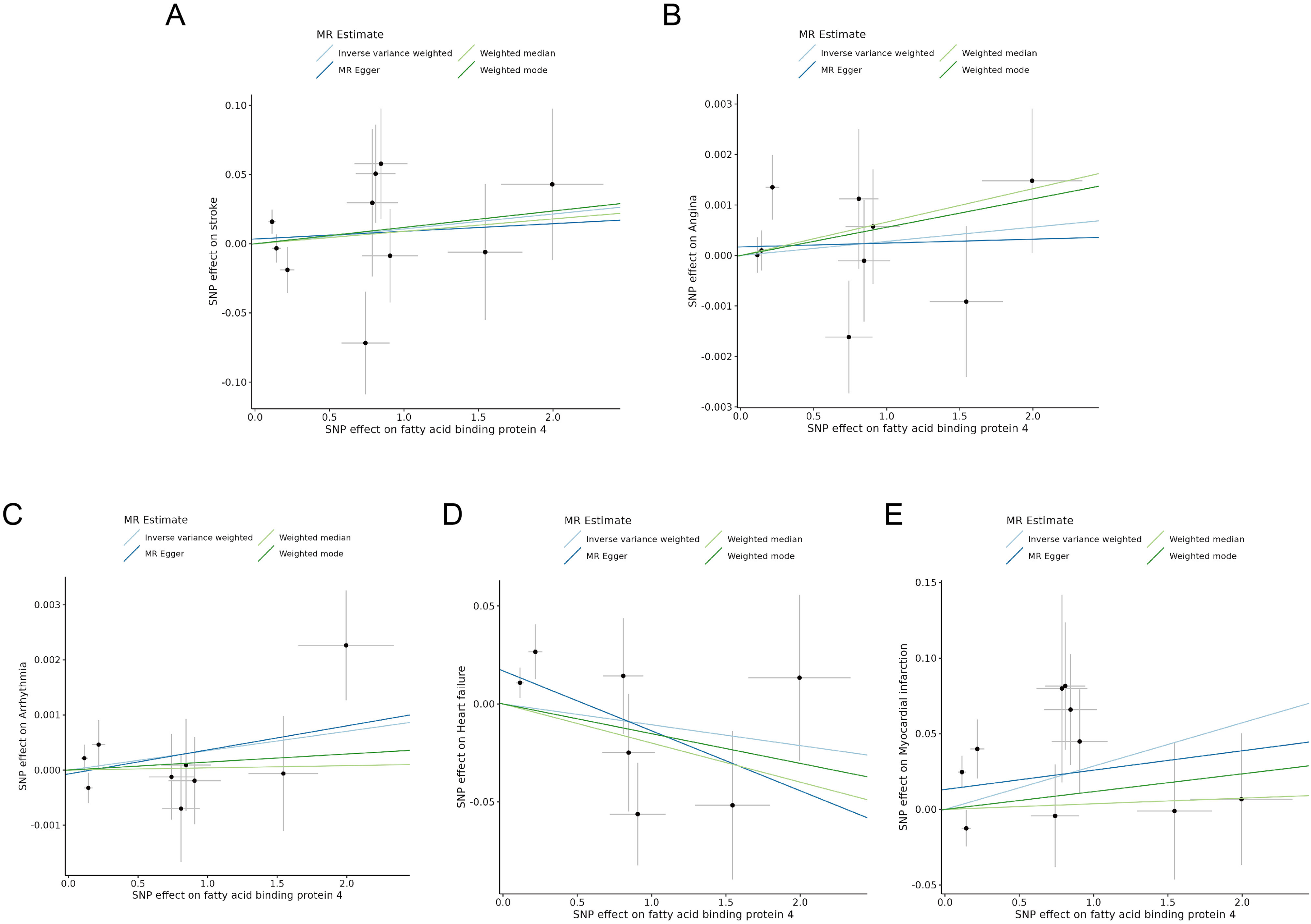
Scatter plots visualize the causal effects of fatty acid binding protein 4 (FABP4) on adverse cardiovascular (CV) Events: (A) Stroke; (B) Angina; (C) Arrhythmia; (D) Heart failure (HF); (E) Myocardial infarction (MI).

Forest plot visualize the causal effects of FABP4 on adverse CV events: (A) Stroke; (B) Angina; (C) Arrhythmia; (D) HF; (E) MI.
Genetic Prediction of Causal Association Between FABP4 and Risk of Adverse CV Events Using IVW and MR-PRESSO Analysis.
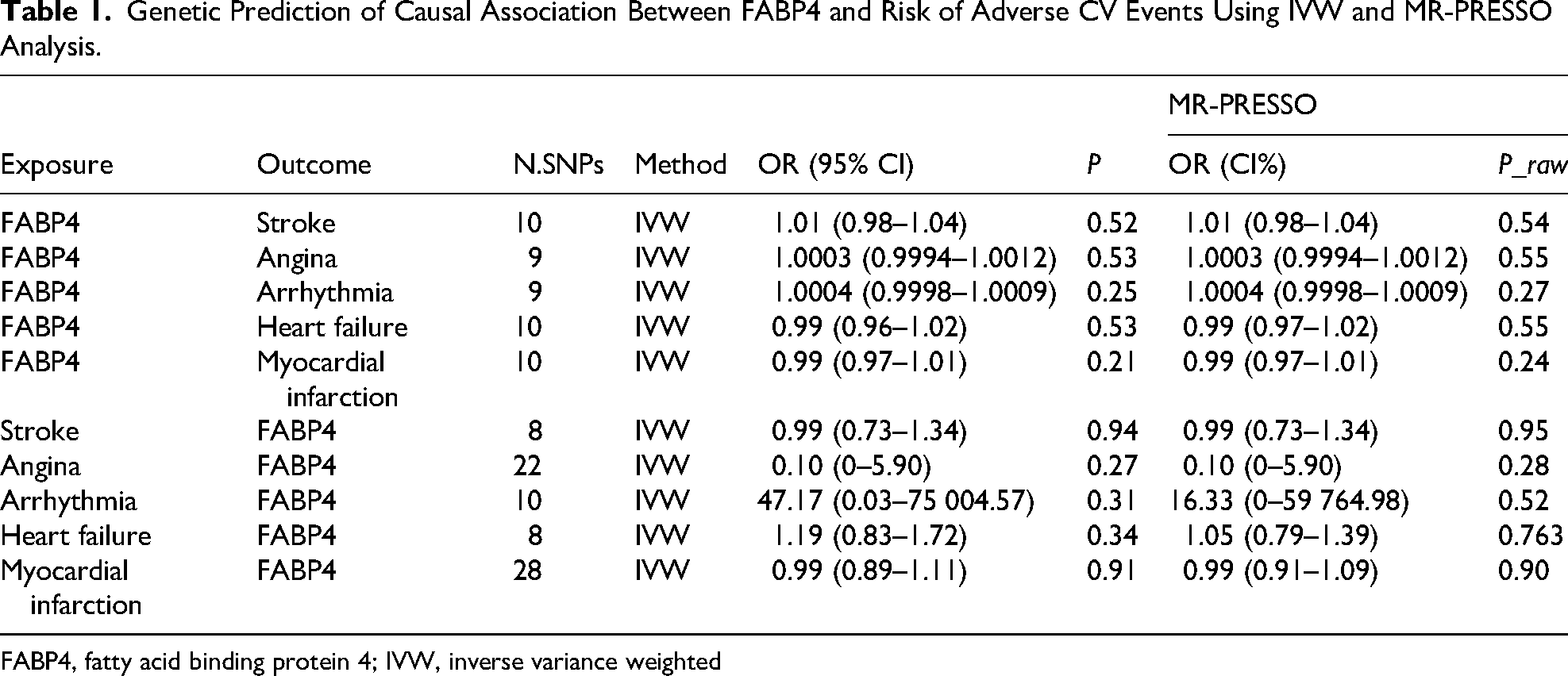
FABP4, fatty acid binding protein 4; IVW, inverse variance weighted
Causal Effects of Adverse CV Events on FABP4
No significant genetic causal effect of adverse CV events on FABP4 was revealed (stroke: OR = 0.99, 95% CI: 0.73–1.34, p = 0.94; angina: OR = 0.10, 95% CI: 0.00–5.90, p = 0.27; arrhythmia: OR = 47.17, 95% CI: 0.03–75 004.57, p = 0.31; heart failure: OR = 1.19, 95% CI: 0.83–1.72, p = 0.34; MI: OR = 0.99, 95% CI: 0.89–1.11, p = 0.91) (Table 1, Figures 3 and 4). Consistently, other MR methods corroborated the null findings from IVW (see Supplementary Table 3). In summary, genetic prediction showed no statistical association between FABP4 and adverse CV events.
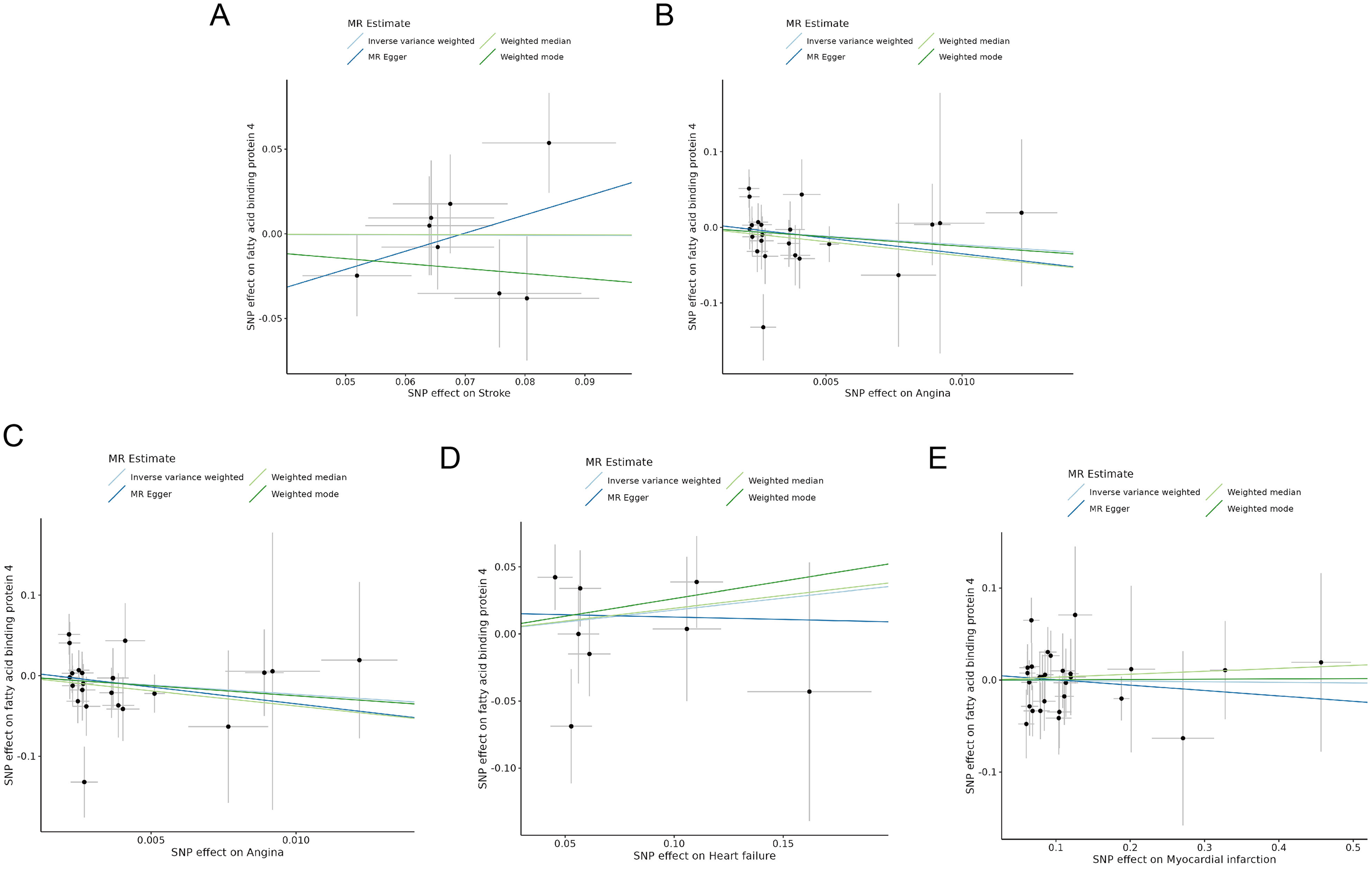
Scatter plots visualize the causal effects of adverse CV events on FABP4: (A) Stroke; (B) Angina; (C) Arrhythmia; (D) HF; (E) MI.

Forest plot visualize the causal effects of adverse CV events on FABP4: (A) Stroke; (B) Angina; (C) Arrhythmia; (D) HF; (E) MI.
Sensitivity Analysis
IVW test for heterogeneity showed that there was no heterogeneity in MR analysis results between FABP4 and adverse CV events (all P > 0.05) (Table 2). MR PRESSO results showed no outlier. (Table 1) In the MR-Egger intercept test, which investigates potential directional pleiotropy, no substantial evidence of significant horizontal pleiotropy was observed (Table 2, all P > 0.05). Visual inspection of the funnel plots for the causal effect of FABP4 on adverse CV events (Supplementary Figure 1) and the reverse effect (Supplementary Figure 2) showed general symmetry, suggesting no significant heterogeneity in either direction of the MR analysis, consistent with the Cochran's Q test. To further explore the potential impact of outlying genetic variants, a leave-one-out analysis was performed, systematically removing each instrumental variable and recalculating the causal estimate. This analysis did not demonstrate any significant changes in the overall causal estimates for FABP4 or adverse CV events (Supplementary Figures 3 and 4), indicating that the null findings were not influenced by any individual influential IV.
Heterogeneity Tests and Multiple Validity Tests for Instrumental Variables.

FABP4, fatty acid binding protein 4; IVW, inverse variance weighted
Power Calculations
The calculated statistical power for the forward MR analyses is presented in Supplementary Table 4. Across all tested cardiovascular outcomes, the statistical power range from 5.00% to 5.09%. For the reverse MR analyses, with FABP4 as the outcome, only the total sample size was available without information on the number of cases and controls; therefore, power calculations could not be performed.
Discussion
In this two-sample MR study, we explored the bidirectional genetic causal association between FABP4 and the occurrence of adverse CV events. The findings reveal no statistical association between genetic variations influencing FABP4 expression and the risk of adverse CV outcomes in individuals of European descent. Similarly, the reverse MR analysis indicated no evidence that genetic liability to CVD was causally related to FABP4. Our study leverages the robustness of MR to mitigate confounding factors typical of observational research, thereby providing a more accurate reflection of genetic causality. This result contrasts with the hypothesis derived from previous observational studies that FABP4 is a potential biomarker of increased cardiovascular risk and enriches the genetic basis of cardiovascular disease.
The role of FABP4 in CVD has been extensively explored in observational studies with varying results. The protein helps regulate inflammasome activation in macrophages and may be involved in disease processes such as atherosclerosis, which is a precursor to many cardiovascular diseases. 33 Höbaus et al demonstrated that elevated serum FABP4 levels significantly associated with major adverse CV events in patients with peripheral arterial disease (PAD). 34 FABP4 levels were elevated during the early hours after the onset of AMI. 35 Aleksandrova et al reported a higher risk of stroke associated with elevated FABP4 levels in men. 36 Moreover, Tso et al and Tu et al found high serum FABP4 levels associated with increased three-month mortality post-stroke and with poor functional outcomes and mortality in acute stroke patients, respectively.37‐39 Our bidirectional two-sample MR study finds no genetic evidence to support a causal relationship between FABP4 and these adverse outcomes. The potential reason for this inconsistency might be the acute phase reaction in observational studies, where FABP4 levels increase in response to systemic inflammation or tissue injury rather than causing the cardiovascular event. Similarly, there are observational studies that highlighted a nuanced aspect of FABP4's relationship with cardiovascular health, particularly emphasizing its differential impact in asymptomatic versus symptomatic populations. Reiser et al noted that while circulating FABP4 is a prognostic biomarker in patients with acute coronary syndrome, it does not serve as a predictive marker in asymptomatic individuals, suggesting that FABP4 levels become significant only in the presence of underlying cardiovascular conditions. 40 It indicates that FABP4 may not directly cause cardiovascular events but may instead be more relevant as a marker of disease progression or severity. Previous studies have indicated that ectopic expression of FABP4 under pathological conditions contributes to the development of atherosclerosis and endothelial injury.7,8 In contrast, our findings did not establish a causal relationship between FABP4 and major cardiovascular events, such as ischemic stroke and myocardial infarction. This observation may imply that genetically determined elevations in FABP4 do not directly drive systemic thrombogenic processes. However, this hypothesis warrants further validation through MR analyses focusing on specific hematologic outcomes, including venous thromboembolism. Taken together, these observations indicate that FABP4 may function more as a biomarker of disease severity or progression, rather than a direct contributor to cardiovascular events. This reinforces the importance of context when evaluating biomarkers, and further research is therefore necessary.
The role of FABP4 in arrhythmia has been investigated in several observational studies. For instance, Wang et al reported that higher plasma FABP4 levels were significantly associated with QTc prolongation in patients with stable angina and chronic kidney disease, indicating a relationship between FABP4 and electrical disturbances in the heart. 41 López-Canoa et al further observed that FABP4 levels were predictive of atrial fibrillation recurrence post-catheter ablation. 42 Also, De With et al found that FABP4 levels were notably higher in women within cohorts evaluating risk profiles for atrial fibrillation therapy, suggesting FABP4 as a potential sex-specific biomarker role in arrhythmia. 43 Our study mitigate s the effect of gender on causality through MR, which again supports the interpretation that FABP4 reflects metabolic or inflammatory states instead of directly mediating arrhythmogenesis.
In previous studies, Pandhi et al identified FABP4 among the most significantly up-regulated proteins in severely congested HF patients. 44 Similarly, Cabré et al found that FABP4 levels in HF patients were associated with clinical symptoms and other biomarkers like NT-proBNP. 45 These studies suggest a possible correlation between FABP4 and the manifestation of HF. However, not all findings have been consistent across different cohorts. Some studies have failed to demonstrate a significant relationship between FABP4 levels and systolic dysfunction or HF prognosis, especially in populations without existing cardiac disease.15,16,46 Thus, FABP4 may still serve as a valuable biomarker in acutely decompensated patients, but its role as a causal driver of HF is unconfirmed.
A recent systematic review and meta-analysis by Agbaedeng et al highlighted that FABP4 was independently associated with poor functional outcomes and increased mortality at 6 months in stroke patients. 47 Saksi et al have shown that variants in FABP4 associated with its low expression could lead to favorable cardiovascular profiles, including reduced cholesterol levels, thinner carotid intima-media, and a lower prevalence of MI in a meta-analysis. 48 These findings suggest that while genetic variants influencing FABP4 expression can have significant effects on disease markers and outcomes, the direct causative roles of FABP4 levels themselves in disease development may be more complex. This perspective emphasizes the need for further studies that differentiate between causal and consequential roles of biomarkers in disease, integrating genetic data with biochemical and clinical insights to provide a more comprehensive understanding of their functions in cardiovascular health.
The application of MR in this context advances our understanding of the genetic foundations of cardiovascular disease. To ensure the robustness and c used, we applied strict selection criteria for IVs and conducted multiple sensitivity analyses to validate the stability of the findings. Although our results do not support a direct genetic link between FABP4 and adverse CV events, they contribute to the existing literature by highlighting the potential disconnect between biomarker expression and genetic predisposition to disease. By employing a methodologically sound approach free from the biases inherent in traditional observational studies, this research not only broadens the scope of genetic epidemiology in cardiovascular conditions but also underscores the complexity and multifactorial nature of CVD etiology. Although our study suggests that FABP4 may not have a direct causal role in CVD, it may still serve as a valuable clinical biomarker. FABP4 levels have been observed to be associated with metabolic dysfunction, inflammation, atherosclerosis, and adverse cardiovascular outcomes in observational studies. Elevated FABP4 may still provide important prognostic information for CVD risk assessment and disease monitoring. Notably, we observed that certain cardiovascular outcomes, such as arrhythmia, yielded extreme odds ratios with very wide confidence intervals in the reverse MR analysis. These unstable estimates are most likely statistical artifacts resulting from the limited number of SNPs available as IVs and the potential influence of weak instrument bias. Therefore, such findings should be interpreted with caution. It should also be noted that MR analysis primarily reflects the lifelong effect of genetically determined FABP4 levels. This approach may not fully capture the impact of acute fluctuations in FABP4 concentrations caused by events such as myocardial infarction or short-term physiological changes, which could also be clinically relevant. Regarding therapeutic implications, FABP4 should not be entirely deprioritized as a drug target based on our causality findings. Given its involvement in cardiometabolic pathways, targeting FABP4 may offer therapeutic benefits, particularly in patients with metabolic comorbidities. Therefore, further mechanistic and clinical studies are needed to better investigate its potential as a biomarker or a therapeutic target.
Despite employing a robust MR design, several limitations warrant consideration. First, our findings are primarily derived from European ancestry populations; thus, replication in diverse groups is necessary to confirm generalizability. Second, the instruments were mainly derived from circulating plasma protein quantitative trait loci (pQTL) data, which enabled evaluation of systemic FABP4 exposure but may differ from eQTLs regulating adipose tissue expression, where FABP4 is predominantly expressed. Utilizing pQTL data for circulating FABP4 levels might not fully capture potential tissue-specific functions, nor fully elucidate the complex pathway from genetic variant to protein expression. Future integration of tissue-specific eQTL data is needed to clarify local versus systemic effects. Also, the exposure dataset for FABP4 was relatively small, which may have limited statistical power and increased the risk of negative findings. Finally, MR assesses lifelong genetic predisposition, which may differ from FABP4's potential role as a dynamic biomarker reflecting acute cardiovascular states, potentially explaining discrepancies with some observational findings where FABP4 might act more as a marker than a causal factor. These factors highlight the need for cautious interpretation. Moreover, since this analysis was restricted to European ancestry, future MR and observational studies in non-European populations are needed to assess the generalizability of the findings. This would help clarify whether the observed associations hold across diverse genetic backgrounds.
Conclusion
Analysis using bidirectional two-sample MR did not find evidence to support a causal relationship between FABP4 and adverse CV events, which could provide new insights into the etiology of cardiovascular diseases.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251401588 - Supplemental material for Genetic Association of FABP4 with Cardiovascular Events: A Mendelian Randomization Study
Supplemental material, sj-docx-1-cat-10.1177_10760296251401588 for Genetic Association of FABP4 with Cardiovascular Events: A Mendelian Randomization Study by Pei Zhang, Wenbin Tian, Ye Tian, Ning Yu, Xuefang Liu, Chao Liu, Shuxian Ma and Huaihai Lu in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-6-cat-10.1177_10760296251401588 - Supplemental material for Genetic Association of FABP4 with Cardiovascular Events: A Mendelian Randomization Study
Supplemental material, sj-docx-6-cat-10.1177_10760296251401588 for Genetic Association of FABP4 with Cardiovascular Events: A Mendelian Randomization Study by Pei Zhang, Wenbin Tian, Ye Tian, Ning Yu, Xuefang Liu, Chao Liu, Shuxian Ma and Huaihai Lu in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations
The data for this study were obtained from publicly available databases and published literature data, which were previously approved by the Ethics Committee.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Authors’ Contributions
Pei Zhang and Huaihai Lu carried out the studies, participated in collecting data, and drafted the manuscript. Wenbin Tian, Ye Tian, Ning Yu, Xuefang Liu, Chao Liu and Shuxian Ma performed the statistical analysis and participated in its design. Huaihai Lu participated in acquisition, analysis, or interpretation of data and draft the manuscript. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:This research fund is supported by the Hebei Medical Science Research Project Plan for 2024, project number 20241377.
Hebei Medical Science Research Project Plan for 2024, (grant number 20241377).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analysed during this study are included in this published article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
