Abstract
Objectives:
Genes related to endothelial function are responsible for the regulation of vascular functions.
Aim:
The aim of this study is to investigate whether endothelial gene-associated polymorphism and their plasma levels can be used to predict the risk for venous thromboembolism (VTE).
Methods:
We studied 133 patients with VTE and 164 healthy controls. Endothelin (EDN) G8002A, EDN T1370G, EDN 3A/4A, eNOSG894T, angiotensin-converting enzyme I/D, vascular endothelial growth factor C936T, and endothelial cell protein C receptor A6936G polymorphism was genotyped by restriction fragment length polymorphism. Plasma levels of endothelin 1 (EDN1), endothelial nitric oxide synthase, and angiotensin-converting enzyme were measured by enzyme-linked immunoassay kit.
Results:
The genotype and allele frequency between control and patients with VTE were significantly altered only for EDN T1370G polymorphism. The plasma EDN1 concentration was relatively higher in patients with VTE (P = .0017) compared to healthy controls and showed an association with the EDN1 gene polymorphism in male Indian population. Logistic regression model analysis for EDN T1370G indicated a significant association between EDN G allele and occurrence of VTE.
Conclusion:
The EDN1 gene polymorphism may play a significant role in predicting individual’s susceptibility toward VTE and its clinical progression.
Introduction
Venous thromboembolism (VTE) is a widespread multifactorial disease associated with inherited and acquired predisposing factors. 1 –4 The global scenario about the occurrence of VTE depicts that it has an annual incidence of 6 to 29 persons per 100 000 in distinct populations 5 ; however, the reported incidence is much lower in the Indian populations as compared to other counterpart due to the limited epidemiological records of VTE for this specific geographical region. 6 It is a well-known fact, the genetic risk factors for VTE comprise of mutations in the genes related to endothelial function along with those involved in coagulation and fibrinolytic pathway, where mutations in either procoagulant or anticoagulant have clearly been related to the risk of VTE, and the role of endothelial-associated genes in determining risk of VTE still lies in infancy. 7 –10
Endothelium maintains vascular hemostasis by providing a conductive surface for interactions of several coagulant and anticoagulant molecules, 11,12 mediates vasodilatation, prevents platelet adhesion, activation, and modulates smooth muscle cell proliferation and atherogenesis. 13,14 Endothelium performs a wide array of functions by producing a variety of molecules, such as endothelial nitric oxide synthase (eNOS), prostacyclins, and release of adhesion molecules and chemotactic factors. Among various others, the vascular endothelial cells exclusively produce endothelin 1 (EDN1), an endothelial vasoconstrictor, which plays complex and multidimensional roles in cardiovascular physiology besides its distinct action in neural, pulmonary, reproductive, and renal physiology. 15 –17 The primary mechanisms pertaining to control endothelin bioavailability are mediated by the key transcription factors that govern EDN1 expression. 18 Study reports suggest that in healthy human being, the production of EDN1 brings a rise in mean arterial blood pressure and reduced heart rate, thereby resulting in potent and long-lasting vasoconstriction. 19 Nonetheless, the upregulation of EDN1 expression is identified as one of the reasons behind the altered response to endothelial receptor blockade in cardiovascular disease. More importantly, studies related to genetic polymorphisms in the EDN1 promoter region and altered EDN1 expression patterns have been reported in the pathogenesis and progression of CVDs. 20,21 It is also interesting to note that EDN1 also shows important interactions with other endothelium-derived substances such as nitric oxide (NO) and angiotensin-converting enzyme (ACE). Apart from the stimulating effect of EDN1 on NO release through the endothelin B receptor, NO also inhibits the production of EDN1 by inhibiting superoxide, 22 whereas EDN1 impairs NO production and downregulates the expression of eNOS in endothelial cells in cardiovascular pathologies. 23 In line with the fact that EDN1 interacts with eNOS and ACE, the present study aims to identify endothelial risk factors predisposing individuals to VTE by studying common genetic polymorphisms in candidate genes of endothelial function by genotyping analysis of mutations in EDN1, 24 endothelial cell protein C receptor (EPCR), 25 vascular endothelial growth factor (VEGF), 26 ACE, 27 and eNOS. 28 Furthermore, we also measured the plasma levels of endothelin (EDN), ACE, and eNOS and analyzed if they were associated with an increased risk of VTE.
Materials and Methods
Study Population
One hundred thirty-three male patients with VTE less than 45 years of age, hospitalized at the Department of Hematology, Army Hospital Research and Referral (AHRR), Delhi (a tertiary care unit), and 164 healthy, age- and sex-matched volunteers were approached for consent to participate in the study. All the study participants were interviewed about medical history. The persons themselves or their near relatives with history of any risk for VTE were excluded from the control group. For the patients, clinical profile, date, and site of venous thrombosis (VT) episode along with the presence of predisposing factors (such as surgery, trauma, prolonged immobilization, hypertension, diabetes, familial history of bleeding) were documented. Patients with a record of systemic diseases were excluded from the study. All the patients had their diagnosis confirmed by at least one of the neurological or radiological imaging methods such as color Doppler/contrast-enhanced computed tomography/computed tomography angiography/magnetic resonance imaging.
Blood Collection
Peripheral blood samples were collected from all participants in vacutainer tubes (Becton and Dickinson, NJ, USA) containing K2EDTA and Na-citrate as anticoagulant. Blood collected in K2EDTA tubes was used for genotyping analysis, whereas vacutainers with Na-citrated blood samples were centrifuged at 2000g at 4°C for 20 minutes, and the obtained plasma was stored at −80°C until analysis. Multiple freeze–thaw was avoided.
Genotyping
High-molecular-weight DNA was extracted from peripheral blood collected in EDTA vacutainers by QIAamp DNA isolation kit (Qiagen, Germany), according to the manufacturer’s protocol. Genomic DNA concentration was determined using NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, USA). For qualitative analysis, DNA samples (100 ng/µL) were loaded on 0.7% agarose gel containing ethidium bromide and electrophoresed for ∼20 minutes and visualized using a gel imaging system (Fusion FX5, Vilber Lourmat, France).
Specific gene sequences for EPCR A6936G (rs 867186), VEGF C936T (rs 3025039), ACE I/D (rs 1799752), eNOSG894T (rs 1799983), EDN G8002A (rs 2071942), EDN 3A/4A (rs10478694), and EDN T1370G (rs1800541) regions were amplified using specific primer sequences. The final polymerase chain reaction (PCR) contained 100 ng of DNA, 200 mmol/L deoxy ribonucleotide triphosphates, 10 pmol of each forward and reverse primer, and 0.6 U Taq polymerase in a total volume of 25 μL reaction buffer (50 mmol/L KCl, 20 mmol/L Tris-HCl, pH 8.3). The PCR products thus obtained were digested with specific restriction enzymes at optimized temperature. Digested PCR products were subjected to gel electrophoresis on 2% to 3% agarose gel containing ethidium bromide to obtain band of different size. Electrophoresis was performed at room temperature, and the bands were visualized under UV. Allelic sizes were determined by comparison of bands with molecular-weight markers. The presence or absence of digested bands confirmed the presence of specific alleles. Single-nucleotide polymorphism (SNP) positive (sample with known profile) and SNP negative (blank) were used for verifying results in each experiment. Complete details of PCR conditions and genotypes screened for different genes are listed in Supplementary Table S1.
Angiotensin-Converting Enzyme, eNOS, and Endothelin Plasma Level
Plasma concentration of ACE, eNOS, and endothelin was determined in both the control and thrombotic groups by commercial enzyme-linked immunoassay kits according to the manufacturer’s protocol. Samples were diluted to maintain plasma concentration well within the accurate boundaries of the kits. Absorbance was measured by Synergy H4 hybrid reader (Biotek, VT, USA).
Statistical Analysis
Hardy-Weinberg equilibrium (HWE; 1 degree of freedom) from the observed distribution of genotypes in the population was calculated. Genotypic and allelic frequencies were determined by gene counting and compared by 3 × 2 and 2 × 2 contingency table, respectively. SNPStat software was used to estimate the statistical significance of differences in categorical variables between the controls and patients by χ2 and Fisher exact test with odds ratio (OR) and 95% confidence interval (CI). Odds ratios were calculated as a measure of genotypic distribution for mutations and association of SNPs with the VTE, with effect of respective allele assumed to be dominant, recessive, overdominant, and log additive. For each, OR, 2-tailed P values, and 95% CIs was calculated for the genetic models. Difference between expected and observed heterozygosity was measured and shown as percentage heterozygosity. Linkage disequilibrium among these SNPs was calculated. Haplotype frequency estimation and association of these haplotypes with the response were evaluated using SNPStat software. Plasma levels of EDN1, eNOS3, and ACE I in controls and patients with VTE were presented as mean ± standard error of the mean (SEM) and were tested using 2-sample t test, Mann-Whitney test for unpaired independent data, and Pearson ρ correlation using SPSS 21.0 software. Statistical significance criteria was P < .05 for all tests.
Results
Characteristics of the Study Population
The study cohort comprised of males from Indian Army. The mean age of patients was 32 years (standard deviation [SD] ±3.96). There was no significant difference in the mean age of patients with thrombosis and that of healthy controls. All participants (patients and controls) were physically active, and the body mass index for each of them was in normal range (<29.9 kg/m2; data not shown).
Plasma EDN Concentrations and EDN Gene Polymorphisms Are Associated With VTE
The plasma EDN concentration was measured in the patient and control groups (Figure 1A). The EDN levels were significantly higher in the patient group than in the control group. The mean plasma EDN level in patients was 963.2 ± 151.2 pg/mL (median, 714.9 pg/mL; range 526.4-1023 pg/mL) and in the control group was 412.5 ± 76.04 pg/mL (median, 217.1 pg/mL; range, 26.43-821.4 pg/mL; P = .0017). Data from patient samples show 2.3-fold increment in the plasma EDN concentration.
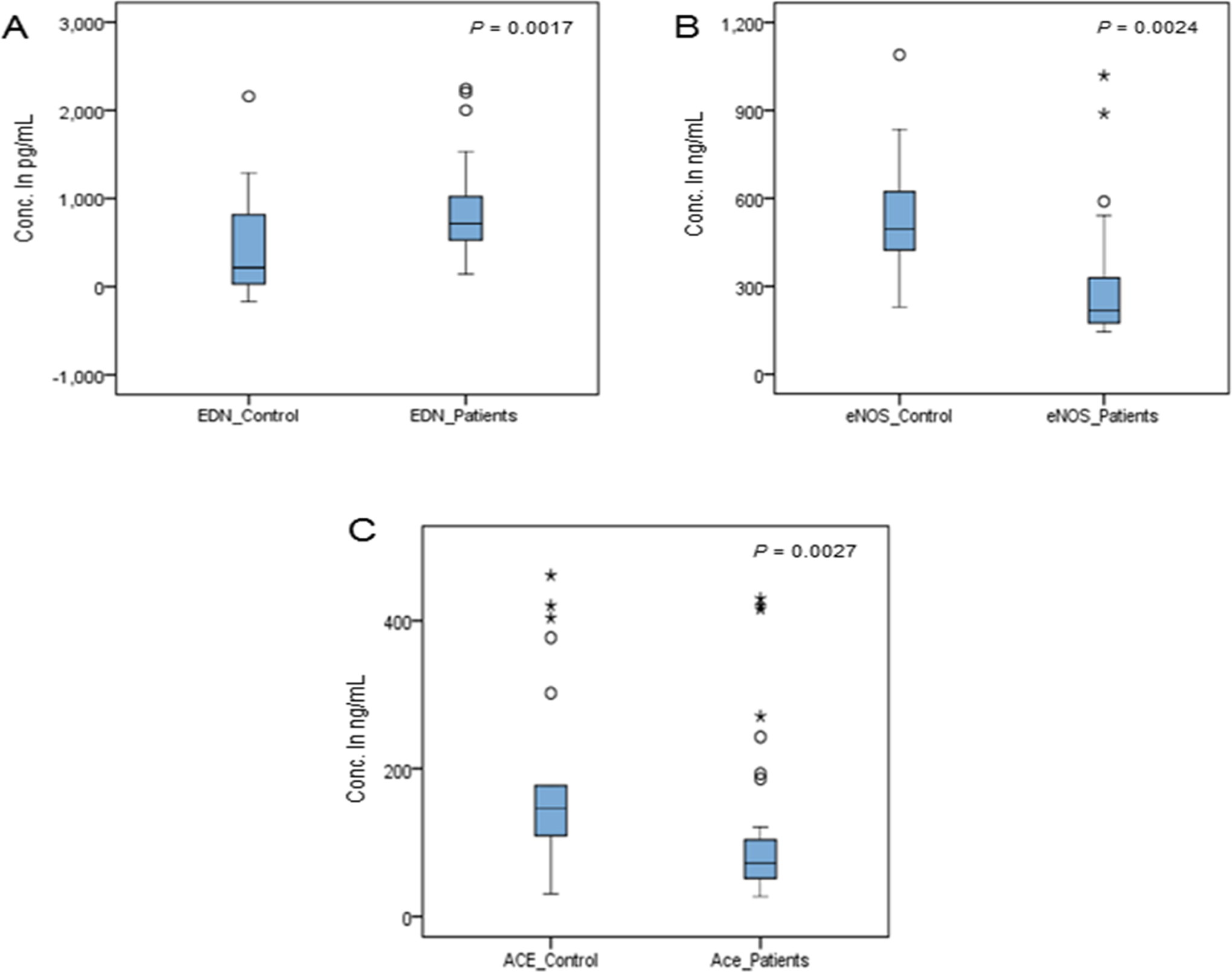
Plasma level of endothelin (EDN; A), endothelial nitric oxide synthase (eNOS; B), and angiotensin-converting enzyme (ACE; C) in both the control and venous thromboembolism (VTE) patient groups. Statistical significance was at P = .05.
The genotype and allele frequencies of the 3 SNPs (rs 2071942, rs10478694, and rs1800541) in the EDN gene are listed in Table 1. The genotype frequencies of EDN in patients with VTE and healthy controls satisfy the HWE (P > .05) with significant allele frequency, except EDN G8002A (HWE P < .05). Thus, EDN G8002A was excluded for further analysis. No significant difference between patients with VTE and controls was observed in the genotype and allele frequencies of the EDN 3A/4A polymorphism (P > .05). EDNT1370G polymorphism was significantly associated with VTE and showed a higher percentage of GG genotype in patients (33.83%) compared to controls (22.56%; χ2 = 7.04, P = .02), as shown in Table 2. Also at the allelic level, percentage occurrence of T allele was 48.49% in VTE compared to 56.09% in controls, whereas percentage occurrence of G allele was 51.50% in VTE compared to 43.90% in controls (P = .06, OR = 1.35, CI = 0.98-1.87; Table 1). Table 2 shows the unadjusted OR and 95% CI from multiple logistic regression analysis for the association between EDN T1370G and VTE risk according to codominant, dominant, recessive, overdominant, and additive models. The patients carrying the EDN G allele were significantly associated with an increased risk of VTE compared with the persons with homozygous TT genotype in recessive (OR = 0.50; CI = 0.30-0.83; P = .007) and overdominant (OR = 1.79; CI = 1.11-2.91; P = .017) models.
Observed Genotypic and Allelic Frequencies of Gene Polymorphisms in All Study Groups.a
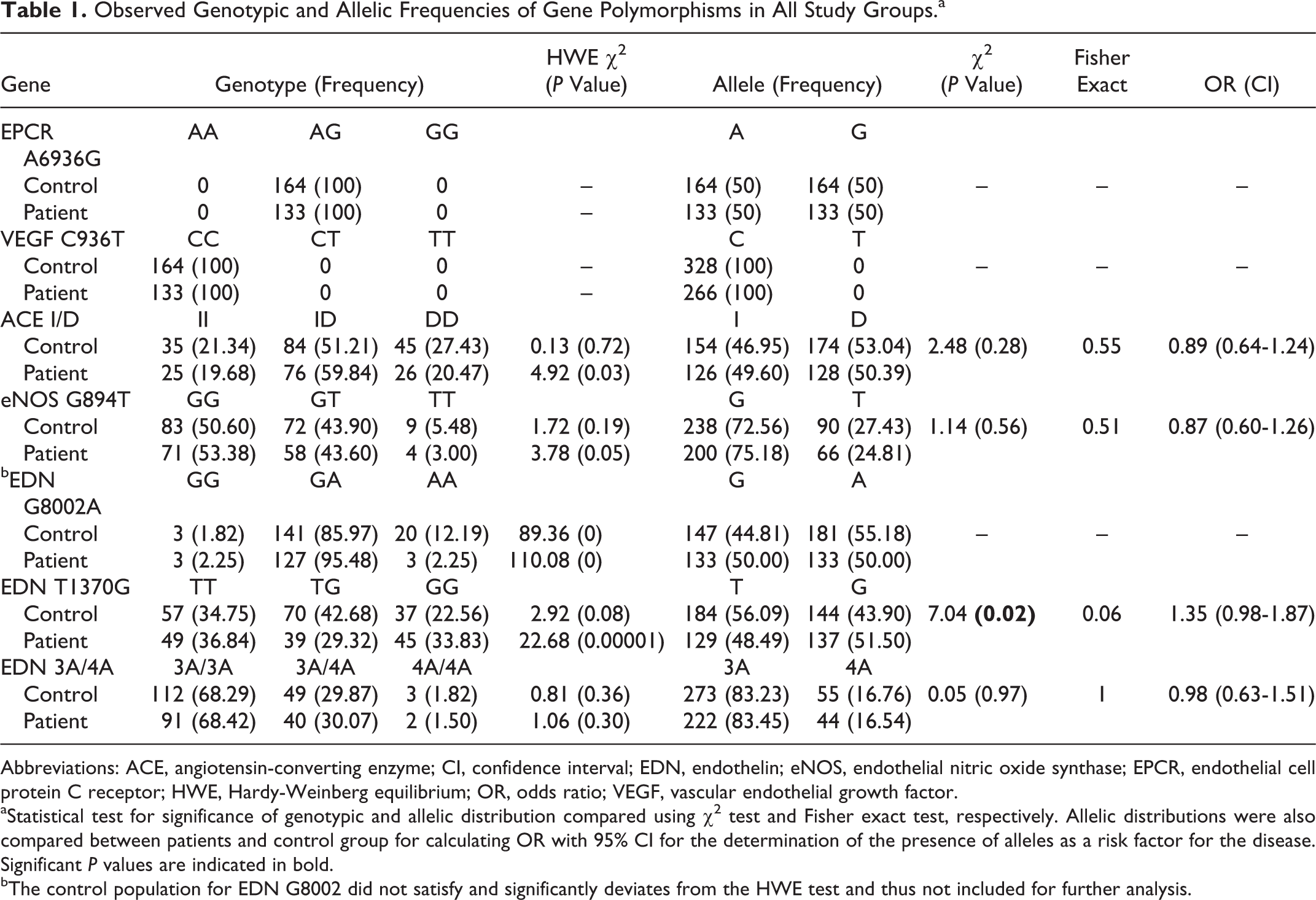
Abbreviations: ACE, angiotensin-converting enzyme; CI, confidence interval; EDN, endothelin; eNOS, endothelial nitric oxide synthase; EPCR, endothelial cell protein C receptor; HWE, Hardy-Weinberg equilibrium; OR, odds ratio; VEGF, vascular endothelial growth factor.
aStatistical test for significance of genotypic and allelic distribution compared using χ2 test and Fisher exact test, respectively. Allelic distributions were also compared between patients and control group for calculating OR with 95% CI for the determination of the presence of alleles as a risk factor for the disease. Significant P values are indicated in bold.
bThe control population for EDN G8002 did not satisfy and significantly deviates from the HWE test and thus not included for further analysis.
SNP Association Chart Showing Codominant, Dominant, Recessive, Overdominant, and Additive Genetic Model for the Polymorphism Under Study.a
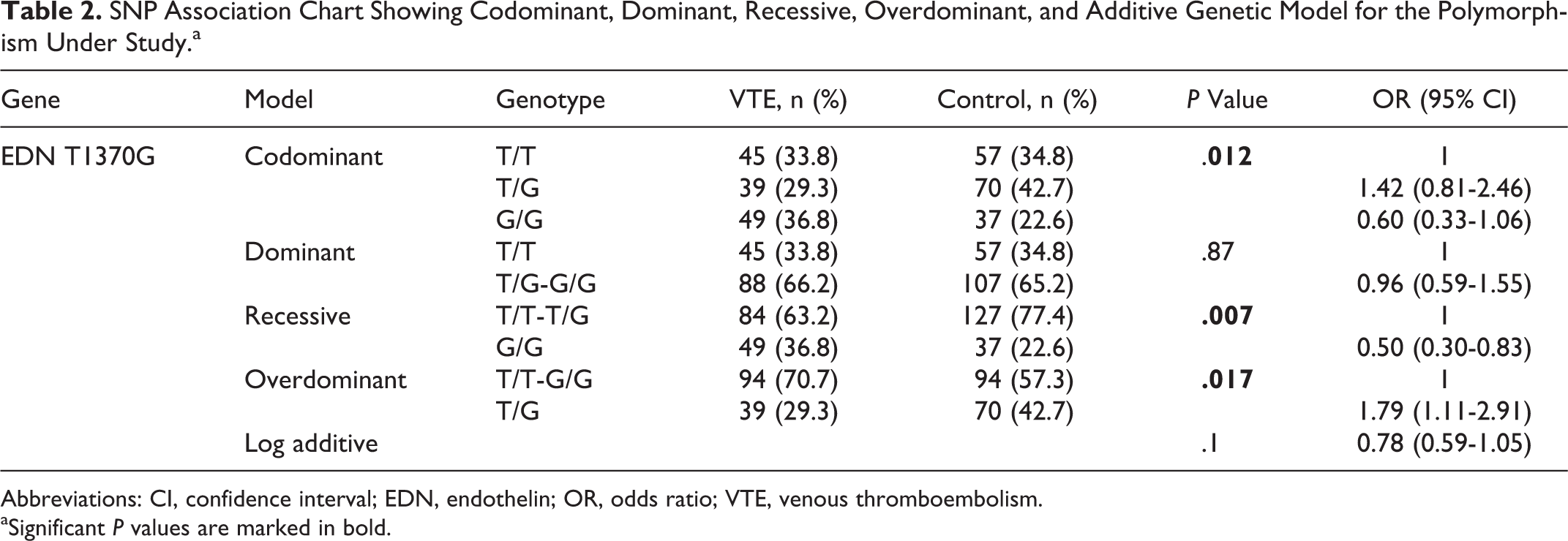
Abbreviations: CI, confidence interval; EDN, endothelin; OR, odds ratio; VTE, venous thromboembolism.
aSignificant P values are marked in bold.
Endothelial Nitric Oxide Synthase, ACE Plasma Level, and SNPs Associated With Endothelial Function-Related Genes
We evaluated the mean plasma level of eNOS and ACE I in patients with VTE and healthy controls. The mean plasma levels for eNOS and ACE I were significantly lower in the VTE patient group (345.2 ± 53.05 ng/mL, 107.30 ± 15.85 ng/mL) than in the control group (592.3 ± 58.48 ng/mL, 293.20 ± 57.86 ng/mL). The difference between the 2 was highly significant (P = .002 and P = .003, respectively). More importantly, there was a significant reduction (42% and 63%) in patient’s plasma level of eNOS3 and ACE I, respectively, as compared to controls (Figure 1B and C), and in line with this evidence, plasma eNOS and ACE levels were found to be a significant predictor of thrombosis. Furthermore, ACE and eNOS plasma levels showed a strong positive correlation between their expression levels in patients with VTE (r = .904, P < .001) and controls (r = .739, P < .001; Figure 2). Whereas, nonsignificant positive correlation was observed between EDN plasma level and ACE level in patients (r = .352, P = .128) and control group (r = .160, P = .501). Similar result was also observed between EDN and eNOS plasma levels (Figure 2).
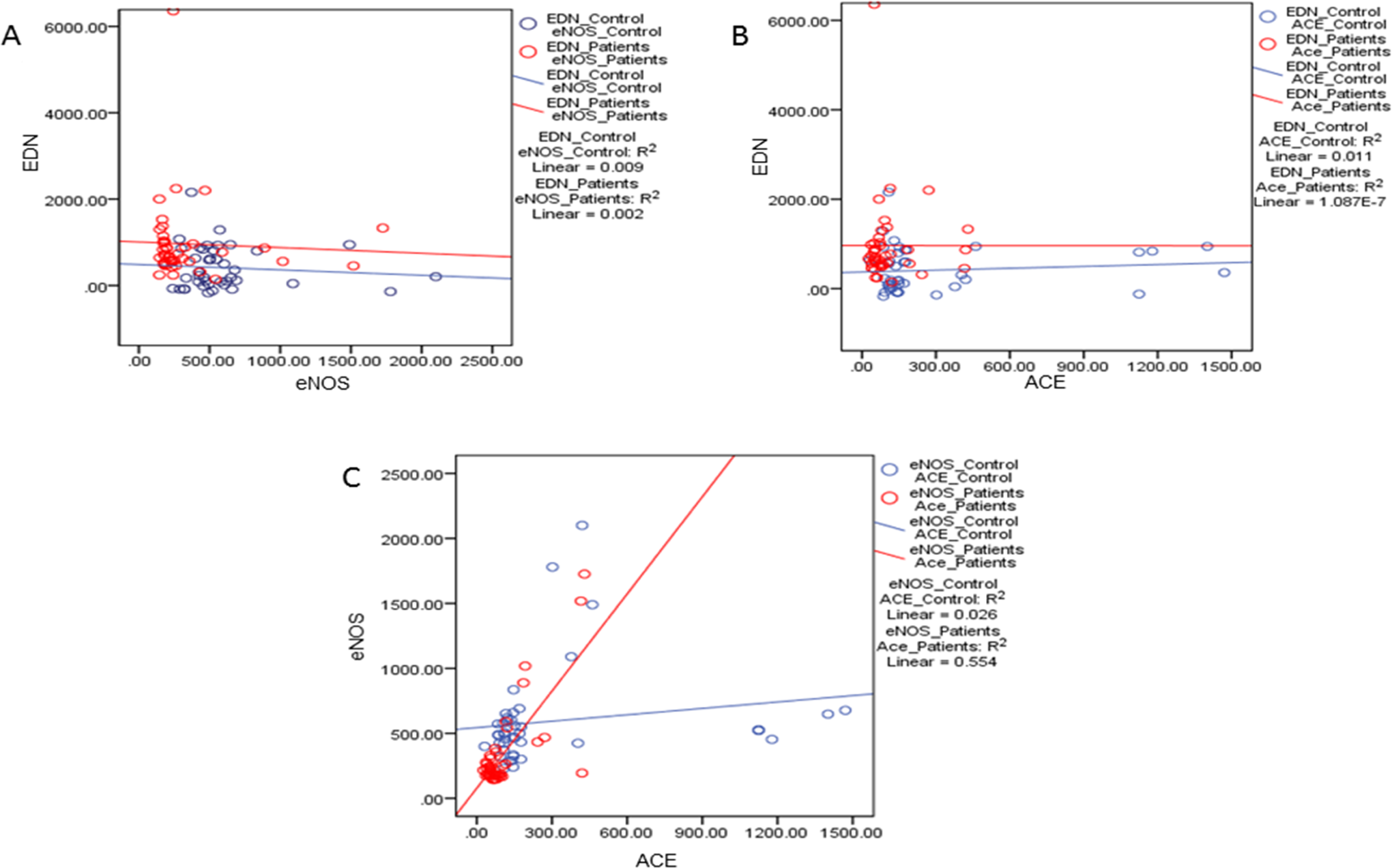
Correlation between plasma level of endothelin (EDN), angiotensin-converting enzyme (ACE), and endothelial nitric oxide synthase (eNOS) in both the control and venous thromboembolism (VTE) patient groups. Graphs represent correlation between (A) EDN and eNOS; (B) EDN and ACE; and (C) eNOS and ACE. Statistical significance was at P = .05.
The SNPs studied in EPCR gene (A6936G) and VEGF gene (C936T) showed similar profile in all the samples (monomorphic) of both the patient and the control groups under study. Both groups were similarly genotyped as A6936G and C936T for EPCR and VEGF gene, respectively. Moreover, we did not find any statistically significant difference between patient with thrombosis and control group in case of ACE I/D (χ2 = 2.48, P = .28, CI = 0.64-1.24) and eNOS G894T (χ2 = 1.14, P = .56, CI = 0.60-1.26) both at genotypic and allelic level (Table 1). There was no significant association between ACE I/D and eNOS G894T SNPs with VTE risk under genetic models.
Heterozygosity and Linkage Disequilibrium Analysis
The observed heterozygosity was found to be much higher than expected both in normal controls and patients with VTE (Figure 3A). This difference was minimal in case of ACE I/D (−2.3; −12.5), eNOS G894T (−6.7; −8.4), and EDN 3A/4A (−3.2; −3.3). In case of ACE I/D, eNOS G894T, and EDN 3A/4A SNPs, since all the values of graph are negative, it can be inferred that the observed heterozygosity were much higher than expected, both in the control and patient groups. Of note, EDN T1370G showed a positive heterozygosity. Observed frequency was lower than expected frequency for heterozygosity. These differences could be due to ascertainment bias in biological sample, which may in turn lead to gains or losses in observed counts. Multiple SNP linkage disequilibrium analysis was done among 4 candidate genes (ACE I/D, eNOS G894T, EDN T1370G, and EDN 3A/4A). D statistic, D′ statistic, r statistic, and P values were calculated. Association of EDN T1370G was observed with ACE and EDN 3A/4A, although the association was not found to be significant at P < .05. Angiotensin-converting enzyme was found to be significantly associated with eNOS G894T (Figure 3B).
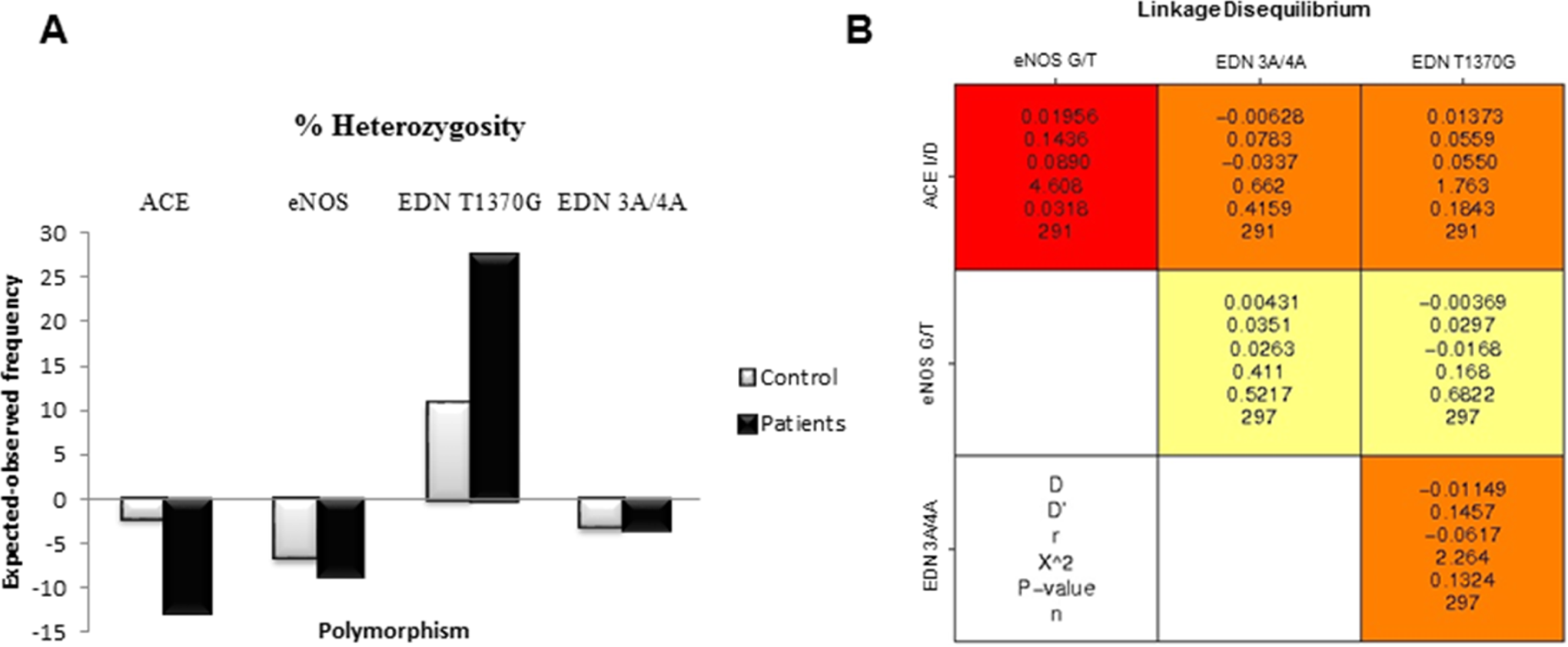
(A) Percentage heterozygosity (expected heterozygosity-observed heterozygosity) in both control and patient groups for angiotensin-converting enzyme (ACE) I/D, endothelial nitric oxide synthase (eNOS) G894T, EDN 3A/4A, and endothelin (EDN) T1370G polymorphism. (B) Single-nucleotide polymorphisms (SNPs) linkage disequilibrium analysis among SNPs. The red and orange boxes are the loci pairs, which are significantly linked to each other. Relevant variables for multivariate analysis were selected on the basis of their association found in univariate analysis and linkage of variables.
Haplotype Analysis
Frequency occurrence of 14 different haplotypes of 4 candidate genes in control and patients with VTE was analyzed. The association of 2 haplotypes, D-T-3A-T (P = .009) and I-G-4A-T (P < .0001), was found to be significant with responses (Table 3). The other observed haplotypes in these 4 genes did not show a significant difference in the 2 groups under study.
Haplotype Association With Response (Crude Analysis).

Abbreviations: ACE, angiotensin-converting enzyme; CI, confidence interval; EDN, endothelin; eNOS, endothelial nitric oxide synthase; OR, odds ratio.
Discussion
The endothelium has emerged as one of the pivotal regulators of hemostasis through its ability to express anticoagulant and vasodilatory molecules in healthy conditions, to release vasoconstrictors, and to express procoagulant, cell adhesion molecules, and cytokines in diseased. 29,30 Based upon the fact, the endothelial dysfunction has been conclusively shown to be an early event in the progression of cardiovascular diseases. 14,31
This study attempts to evaluate the role of mutations in genes related to endothelium functions in determining the susceptibility toward VTE and the influence of these genetic polymorphisms on clinical phases of the disease and prognostic factors in Indian population. Previous studies have linked eNOS G894T, EPCR A6936G, VEGF C936T, and ACE I/D polymorphism to VTE occurrence. 32 –38 Whereas, we could not find their association with VTE occurrence in male Indian population. However, significant change in eNOS and ACE plasma level in patients with VTE has been observed. Reduced eNOS plasma level leading to impaired NO production and increased ACE plasma levels in patients with VTE suggest their role in the increased risk of thrombosis as discussed in previous studies. 39,40
All 3 EDN gene SNPs (rs1800541, rs2071942, and rs10478694) included in the present study have been involved in previous studies. 41,42 According to Zouridakis et al, 43 plasma endothelin was raised in patients with coronary artery disease progression and may be a marker for risk of rapid stenosis progression. Increased plasma endothelin level may indicate early disturbances of endothelial function in vascular diseases. It has also been demonstrated to be associated with increased oxidative stress and endothelial dysfunction in humans. 44
The most prominent finding of this study is the association of EDN T1370G mutation toward VTE. Our data showed statistically significant difference between the patient and control groups in case of EDN T1370G polymorphism. The EDN T1370G polymorphism is located in the 5′-flanking promoter region of the gene and thus may be involved in its differential transcriptional regulation and in increased level of EDN. In the present study, we found that the EDN plasma concentration seems to correlate with VTE, although the difference between these patients and controls did not reach the level of statistical significance. High plasma levels of EDN in patients compared to controls suggest the role of EDN in the prevalence of VT. However, there is a scope for further exploration of this SNP with larger sample size to obtain conclusive results. Endothelin also regulates messenger RNA levels, protein levels, and activity of plasminogen activator inhibitor 1, which inhibits fibrinolysis. 45 Thus, higher concentrations of EDN might cause prothrombotic state. An elevated EDN level also has been associated with carotid atherosclerosis and asymptomatic cerebrovascular lesions in patients with essential hypertension. 46 Our current findings indicate that of the 2 SNPs tested in EDN gene, EDN T1370G was associated with VTE; however, we failed to demonstrate an association between the EDN 3A/4A polymorphism and VTE.
Correlation study between plasma levels of ACE, eNOS, and EDN showed association between ACE and eNOS expression; however, no significant correlation was observed between EDN and ACE, EDN, and eNOS plasma level. This suggests that EDN may have an individual functional role in VTE, whereas ACE and eNOS may have a combined effect in VTE. Our result showed an increased EDN level and decreased eNOS level in patients with VTE; however, no significant correlation was found. Therefore, endothelial dysfunction accompanied by overproduction of EDN and decreased eNOS levels may play a key role in the occurrence or progression of VTE.
This study does not link EPCR A6936G, VEGF C936T, ACEI/D, eNOSG894T, EDN G8002A, and EDN 3A/4A SNPs gene to VTE susceptibility. Similarly, in our previous study, the established genetic variants FVL 1691G/A, prothrombin 20210G/A, and TFPI-536C/T were not found to be associated with VTE in Indian population. 47 These differences could be due to different ethnic backgrounds and differences in gene–gene and environment–gene interactions. The 1000 GENOME Project included the population from 26 different nations including nonresident Gujarati and Telugu peoples from India in the study 48 ; however, there are absence of extensive and large studies on endothelin gene polymorphism in North Indian population We found a trend toward differential expression of EDN in VTE but no statistical difference between the patient and control groups. In addition, the relationship between the plasma levels and genotype frequency has no statistical significance in our study. We considered that the reason was an insufficient sample size. Since this study has a relatively small number of participants, large number of participants would be needed to achieve a more definitive conclusion regarding involvement of the endothelial function-related gene polymorphisms and plasma expression levels in the risk of VTE. Besides these, there are 2 different haplotype associations that were found to be significant to the VTE response.
We screened endothelial function-related gene variants to evaluate their role in VTE. Here, the association of SNPs in the EDN gene and increased plasma level of EDN with increased risk of VTE has been reported for the first time and predicts the role of emerging biomarkers of endothelial dysfunction that may increase the predictability of a VTE event in male Indian population.
Footnotes
Authors’ Note
Babita Kumari and Amit Prabhakar contributed equally to this article.
Acknowledgments
The authors are extremely thankful to all the volunteers who participated in the study from Army Hospital Research and Referral (AHRR), Delhi, that was a great support for us in collection of samples. Also, the authors are grateful to Director, DIPAS, Defence Research and Development Organization (DRDO), India, for funding this project (SLT-09/DIP-255) and Director General of Armed Forces Medical Sciences, India, for approving the human study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
The supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
