Abstract
Background
Although red cell distribution width (RDW) has been linked to venous thromboembolism, its predictive value for postoperative pulmonary embolism (PE) in surgical trauma patients remains ambiguous. This study aimed to investigate the correlation between preoperative RDW and postoperative PE risk.
Methods
We incorporated 46506 surgical trauma patients from three medical institutions and the MIMIC-IV 2.2 database. We constructed receiver operating characteristic curves (ROC) utilizing preoperative (at admission) RDW and postoperative PE and classified patients into two groups. Firstly, univariate logistic regression was conducted to identify factors correlated with postoperative PE, and the variance inflation factor was computed to evaluate multicollinearity. Multivariate logistic regression analysis was subsequently conducted to identify the independent risk factors for PE. Propensity score matching (PSM) was conducted using a caliper value of 0.1, balancing 26 covariates between the two groups, including demographic features, vital signs, injury severity scores, comorbidities, and laboratory parameters. A total of 10235 pairs were successfully matched.
Results
The postoperative PE incidence was 0.56%. RDW showed significant predictive value for PE with an area under the curve (AUC) of 0.723 (0.696-0.750). Before PSM, RDW ≥14.4% was associated with increased PE risk (OR 4.70, 95% CI 3.63-6.09, P < 0.001). Multivariate analysis confirmed a 1.90–fold risk increase (95% CI 1.42-2.55, P < 0.001). After PSM, RDW ≥14.4% remained significantly associated with higher PE risk (OR 1.67, 95% CI 1.22-2.29, P = 0.002).
Conclusion
Elevated preoperative RDW is an independent risk factor for postoperative PE in trauma patients, with ≥14.4% indicating significantly increased risk.
Background
As one of the major global public health challenges, severe trauma significantly reduces life expectancy and increases the disease burden. According to the Global Burden of Disease Study 2017, trauma accounts for nearly 8% of global deaths annually. 1 Acute pulmonary embolism (PE), the most severe form of venous thromboembolism (VTE), is the third leading cause of cardiovascular disease-related death worldwide, with a mortality rate as high as 15%–20%. 2 Surgery is a vital treatment modality for trauma patients; however, as a “secondary insult,” surgery itself may worsening the systemic inflammatory response following trauma and is significantly linked to high postoperative morbidity and mortality rates. 3 The perioperative management of trauma patients presents various challenges, with potential complications such as wound infection, pneumonia, urinary tract infection, acute kidney injury, deep vein thrombosis (DVT), PE, pressure ulcers, sepsis, multiple organ dysfunction, and traumatic coagulopathy, among others.4–7
Notably, severe trauma itself can induce coagulation dysfunction, and surgical intervention together with postoperative immobilization heightens the risk of PE. 8 Following the onset of PE, trauma patients may experience several detrimental consequences, such as extended mechanical breathing, heightened susceptibility to multi-organ failure, and escalated medical expenses.9,10 Accurate identification of high-risk trauma patients for postoperative PE is crucial for implementing effective preventive strategies.
However, currently used perioperative PE risk assessment tools (such as the Wells score and Geneva score) are limited in application and controversial due to the inclusion of subjective variables.11–13 Therefore, identification of simple, easily accessible, and cost-effective predictive factors for early and effective stratification of postoperative PE risk in trauma patients during perioperative management is urgently needed. Studies have shown that elevated red cell distribution width (RDW) is associated with acute PE events in hospitalized patients.2,14,15 Celik et al 2 reported that RDW levels were significantly higher in PE patients than in non-PE patients in the emergency department. Zöller et al 14 studied a general population cohort from the Malmö Diet and Cancer Study and confirmed that elevated RDW is independently associated with the risk of first VTE events. Günay et al 15 focused on patients diagnosed with acute PE via computed tomographic pulmonary angiography (CTPA), further validating that RDW levels were significantly higher in patients with PE compared with individuals without PE. However, trauma patients were not the specific focus of the above studies, and the applicability of these findings to trauma patients remains to be further validated. The aim of this study is to utilize data from a multicenter retrospective cohort to investigate the association between preoperative RDW and the risk of postoperative PE in trauma patients.
Methods
Inclusion and Exclusion Criteria

Flow chart. ISS: injury severity score; RDW: red blood cell distribution width.
Data Collection
For trauma patients in the MIMIC-IV 2.2 database, SQL query statements were used to filter the data, and target cases were retrieved using keywords such as “fracture” and “trauma”. The other three medical centers primarily collected information through the hospital's surgical anesthesia electronic information system and clinical electronic medical records system. The basic patient information collected included: gender, age, race, emergency admission status, injury severity score (ISS) at admission, vital signs at admission[admission systolic blood pressure (SBP), admission heart rate (HR)], preoperative comorbidities [delirium, asthma, hypercholesterolemia, cerebrovascular disease, renal failure, urinary tract infection, chronic obstructive pulmonary disease (COPD), coronary heart disease, respiratory failure, anemia], preoperative laboratory parameters [RDW, preoperative hemoglobin (Hb), platelet count, alanine aminotransferase (ALT), aspartate aminotransferase (AST), albumin, sodium (Na+), potassium (K+), calcium (Ca2+)], and preoperative coagulation parameters [prothrombin time (PT), activated partial thromboplastin time (APTT)]. RDW was measured and reported as the red cell distribution width-coefficient of variation (RDW-CV) across all centers to ensure standardized reporting. The specific instrument models and manufacturers for RDW testing were as follows: the Army Medical Center used a Sysmex XN-9000 analyzer, the Chongqing Emergency Medical Center used a Mindray BC-5390 CRP analyzer, and the Rocket Force Characteristic Medical Center used a Beckman Coulter DxH 800 analyzer. Regardless the different instruments, all laboratories strictly adhered to the pertinent regulations of the National Health Commission, consistently engaged in external quality assessment (EQA) and internal quality control (IQC) protocols, thus optimizing the uniformity and comparability of results across diverse testing systems. Complications during hospitalization were primarily PE, diagnosed based on clinical symptoms and imaging studies, with CTPA serving as the gold standard for diagnosis. 16 Postoperative PE occurrences were defined as those occurring from the end of trauma surgery to the day of discharge from the initial hospitalization.
Research Method
Firstly, we computed the missing rates for each variable. For variables exhibiting a missing rate <30%, 17 multiple imputation was performed applying the mice package in R (Supplemental Material 1). Variables with a missing rate ≥30% were directly deleted. We plotted the receiver operating characteristic (ROC) curve using preoperative RDW and postoperative PE ratio (Figure 2) and calculated the cutoff value. Then, we categorized the patients into two groups according to the cutoff value: the Pre-RDW ≥ 14.4% group and the Pre-RDW < 14.4% group. Patients were divided into two groups based on postoperative PE: the postoperative PE group and the postoperative non-PE group. We confirmed the association between preoperative RDW and postoperative PE in trauma patients by univariate logistic regression, variance inflation factor (VIF) to deal with multicollinearity, 18 and multivariate logistic regression analysis. We utilized propensity score matching (PSM) for robustness analysis, defining RDW group as the dependent variable and included 26 categorical variables as covariates. These included patient gender, age group (≤ 60 years, > 60 years), admission ISS group (< 16, ≥ 16),19,20 race (Asian, Black, White, Other), admission HR group (< 60 bpm, 60–100 bpm, > 100 bpm), admission SBP group (< 120 mm Hg, ≥ 120 mm Hg),21,22 preoperative comorbidities (delirium, asthma, hypercholesterolemia, cerebrovascular disease, renal failure, urinary tract infection, COPD, coronary heart disease, respiratory failure, Hypertension, anemia), preoperative ALT group (< 40 U/L, ≥ 40 U/L),23,24 preoperative AST group (< 40 U/L, ≥ 40 U/L), 25 preoperative albumin group (< 35 g/L, ≥ 35 g/L),26,27 preoperative Na+ group (< 135 mmol/L, 135-145 mmol/L, > 145 mmol/L), preoperative K+ group (< 3.5 mmol/L, 3.5-5.0 mmol/L, > 5.0 mmol/L), preoperative Ca2+ group (< 2.1 mmol/L, 2.1-2.5 mmol/L, > 2.5 mmol/L),28–30 preoperative platelet group (< 100 × 10⁹/L, 100–300 × 10⁹/L, > 300 × 10⁹/L), preoperative PT group (< 12 s, ≥ 12 s), 31 preoperative APTT group (< 30 s, 30–40 s, > 40 s).32,33 A caliper value of 0.1 was employed for 1:1 matching. Following that, we conducted a binary logistic regression analysis to further evaluate the stability of the link between preoperative RDW and postoperative PE in trauma patients.

Receiver operating characteristic curve of preoperative RDW for predicting postoperative PE in trauma surgery patients.
To deeply validate the robustness of the study findings, we applied three statistical methods for cross-verification: first, a bootstrap resampling analysis with 500 iterations was run to examine the stability of the ROC analysis; secondly, because of to the low prevalence of postoperative PE (approximately 0.6%), Firth's penalized likelihood method was carried out to adjust for potential small-sample bias; finally, inverse probability of treatment weighting (IPTW) was conducted as a sensitivity analysis, further supporting the reliability of the PSM results. The investigation was conducted as a multicenter retrospective analysis. Ethical approval had been obtained from the institutional review boards of all participating centers, and informed consent was exempted by the ethics committees due to the retrospective character of the study. The ethical approval details are as follows: MIMIC-IV 2.2 database (ethics approval No. 63295742), Chongqing Emergency Medical Center (ethics approval No. 48, 2023), the Rocket Force Characteristic Medical Center (ethics approval No. KY 2023037), and Daping Hospital (ethics approval No. 261, 2023). Furthermore, the study was registered in the Chinese Clinical Trial Registry, with the assigned registration number ChiCTR2300078097.This study was reported in accordance with the STROBE guidelines (Supplemental Material 2).
Statistical Analysis
Data analysis was conducted using R (version 4.4.3) and SPSS 29.0 (IBM Corporation, Armonk, NY, USA). Continuous variables are summarized as mean ± standard deviation, and categorical variables as counts with percentages. Group comparisons for categorical variables were performed with the chi-square test, while non-normally distributed continuous variables were compared using the Mann-Whitney U test. The ROC curve was built using preoperative RDW values to predict PE. The best cutoff value for RDW was chosen based on the biggest Youden index (sensitivity + specificity – 1). Patients were split into two groups according to this threshold: ones above the cutoff and the ones below it.
We first applied univariate logistic regression to screen variables potentially related to postoperative PE, using a significance level of P < 0.05 and a VIF below 5. Variables meeting these criteria were then incorporated into a multivariate logistic model to further evaluate the association between preoperative RDW and postoperative PE. PSM was performed between the RDW ≥ 14.4% group and the RDW < 14.4% group, aiming to balance the distribution of baseline covariates between the two groups after matching, thereby allowing a more accurate assessment of the association between the exposure (RDW ≥ 14.4%) and the outcome (postoperative PE). Matching quality was evaluated using standardized mean differences (SMD), where an SMD < 0.1 indicated adequate balance. After matching, binary logistic regression was used to estimate adjusted odds ratios (OR) and 95% confidence intervals (CI) for the relationship of RDW to PE. A two-sided P < 0.05 was considered statistically significant.
Results
Baseline Characteristics of Patients
This study enrolled a total of 46506 trauma patients. The overall incidence of postoperative PE was 0.56% (262/46506). The area under the curve (AUC) for preoperative RDW in predicting postoperative PE in trauma patients was 0.723 (0.696-0.750), with a sensitivity of 67.20%, a specificity of 69.70%, and an accuracy of 69.70%. The optimal cut-off value for preoperative RDW in predicting postoperative PE, determined by maximizing the Youden index, was 14.4% (Figure 2). Bootstrap results showed a mean AUC of 0.723 (95% CI 0.696-0.751), a sensitivity of 67.20%, a specificity of 69.70%, and an accuracy of 69.70%, which highly overlapped with the AUC interval obtained from the conventional ROC analysis (Supplemental Material 3). The sensitivity and specificity were also consistent with the estimates from the traditional method. This indicates that the diagnostic performance evaluation of Pre-RDW is stable.
According to this threshold, 32307 patients (69.47%) were assigned to the Pre-RDW < 14.4% group, and 14199 patients (30.53%) to the Pre-RDW ≥ 14.4% group. Among all participants, 23271 individuals (50.04%) were male and 23235 (49.96%) were female. The overall mean age was 55.23 ± 19.89 years, with a mean age of 51.70 ± 19.69 years in the Pre-RDW < 14.4% group and 63.27 ± 17.88 years in the Pre-RDW ≥ 14.4% group (Table 1). Regarding preoperative comorbidities, hypertension was present in 6852 patients (14.73%), coronary heart disease in 1807 patients (3.89%), hypercholesterolemia in 1768 patients (3.80%), urinary tract infection in 1429 patients (3.07%), cerebrovascular disease in 819 patients (1.76%), COPD in 450 patients (0.97%), respiratory failure in 511 patients (1.10%), asthma in 437 patients (0.94%), renal failure in 361 patients (0.78%), and delirium in 146 patients (0.31%). Among these, 28690 patients (61.69%) were Asian, 13257 (28.51%) were White, 2002 (4.30%) were Black, and 2557 (5.50%) were other races (Table 1).
Baseline Characteristics According to Preoperative RDW Level.

RDW: red blood bell distribution width; ISS: injury severity score; HR: heart rate; SBP: systolic blood pressure; COPD: chronic obstructive pulmonary disease; Hb: hemoglobin; ALT: alanine aminotransferase; AST: aspartate aminotransferase; PT: prothrombin time; APTT: activated partial thromboplastin time; PLT: platelet count; K+: potassium; Na+: sodium; Ca2+: calcium. P < 0.05 was considered statistically significant.
Comparison of Covariates Before and After PSM
Prior to PSM, significant differences in baseline characteristics existed between the Pre-RDW <14.4% and ≥14.4% groups. The SMD > 0.1 for multiple covariates, including ISS (SMD = 0.209), age (SMD = 0.531), admission HR (SMD = 0.167), race (SMD = 1.048), hypercholesterolemia (SMD = 0.272), asthma (SMD = 0.122), renal failure (SMD = 0.153), coronary heart disease (SMD = 0.164), respiratory failure (SMD = 0.117), hypertension (SMD = 0.309), platelet count (SMD = 0.401), AST (SMD = 0.204), albumin (SMD = 0.652), PT (SMD = 0.526), APTT (SMD = 0.301), Na+ (SMD = 0.181), K+ (SMD = 0.221), Ca2+ (SMD = 0.242), anemia (SMD = 0.991), and 17 other variables.
After PSM, a total of 10235 matched pairs were formed. Only race (SMD = 0.235) and preoperative platelet count (SMD = 0.195) continued to show differences between the groups (Figure 3). The SMD values for all other variables were below 0.1. The SMD values for all remaining variables were below 0.1, indicating that the baseline characteristics between the study groups were comparable after matching (Table 2).

Variable Standardization Difference Diagram. ISS: injury severity score; HR: heart rate; SBP: systolic blood pressure; COPD: chronic obstructive pulmonary disease; PLT: platelet; ALT: alanine aminotransferase; AST: aspartate aminotransferase; PT: prothrombin time; APTT: activated partial thromboplastin time; K+: potassium; Na+: sodium; Ca2+: calcium.
Distribution Characteristics of Covariates Before and After PSM Between Pre-RDW < 14.4% Group and Pre-RDW ≥ 14.4% Group in Trauma Patients.
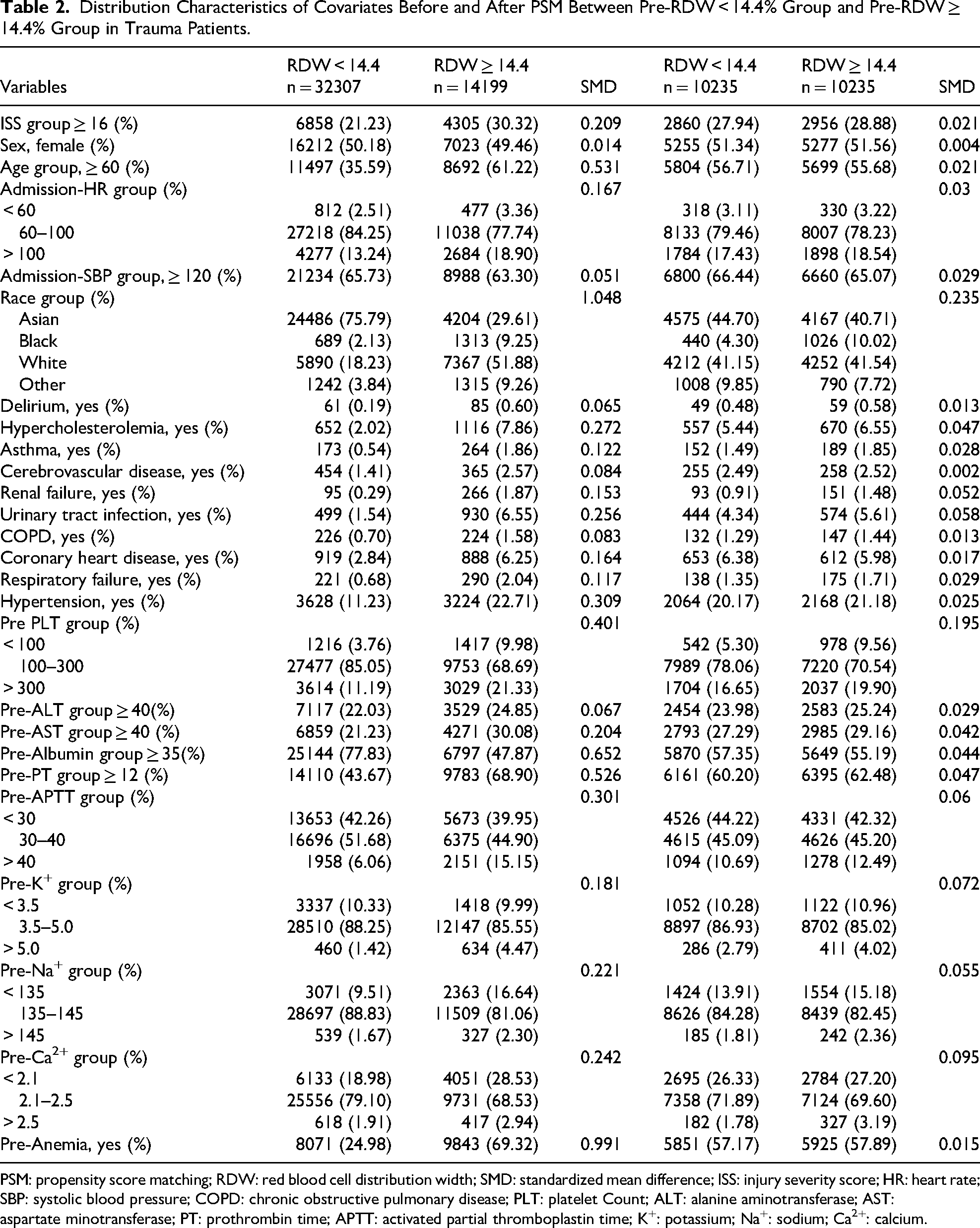
PSM: propensity score matching; RDW: red blood cell distribution width; SMD: standardized mean difference; ISS: injury severity score; HR: heart rate; SBP: systolic blood pressure; COPD: chronic obstructive pulmonary disease; PLT: platelet Count; ALT: alanine aminotransferase; AST: aspartate minotransferase; PT: prothrombin time; APTT: activated partial thromboplastin time; K+: potassium; Na+: sodium; Ca2+: calcium.
Association Between Preoperative RDW and PE
Before PSM, the overall postoperative PE rate among surgical trauma patients was 0.56% (262/46506). The postoperative PE rate in the Pre-RDW ≥ 14.4% group was significantly higher than that in the Pre-RDW < 14.4% group [1.24% (176/14199) versus 0.27% (86/32307), P < 0.001] (Table 3). Univariate logistic regression analysis showed that compared with the Pre-RDW < 14.4% group, the Pre-RDW ≥ 14.4% group had a 4.70-fold increased risk of postoperative PE (95% CI 3.63-6.09, P < 0.001) (Figure 4). Our results revealed that all the VIF values were less than 5, indicating no significant multicollinearity issues in the data (Figure 5). Our multivariable logistic regression analysis showed that compared with the Pre-RDW < 14.4% group, the Pre-RDW ≥ 14.4% group had a 1.90-fold increased risk of postoperative PE (95% CI 1.42–2.55, P < 0.001) (Figure 6). The results of Firth's analysis indicated that, before PSM, univariable Firth penalized logistic regression showed that compared to the Pre-RDW < 14.4% group, the Pre-RDW ≥ 14.4% group had a 4.69-fold increased risk of postoperative PE (95% CI 3.63–6.09, P < 0.001). Multivariable Firth penalized logistic regression analysis showed that the Pre-RDW ≥ 14.4% group had a 1.90-fold increased risk of postoperative PE (95% CI 1.42-5.54, P < 0.001) compared to the Pre-RDW < 14.4% group (Supplemental Material 4).

Univariate logistic regression analysis of risk factors for postoperative PE in trauma patients. PE: pulmonary embolism; RDW: red blood cell distribution width; OR: odds ratio; CI: confidence interval; ISS: injury severity score; HR: heart rate; SBP: systolic blood pressure; COPD: chronic obstructive pulmonary disease; PLT: platelet count; ALT: alanine aminotransferase; AST: aspartate aminotransferase; PT: prothrombin time; APTT: activated partial thromboplastin time; K+: potassium; Na+: sodium; Ca2+: calcium. P < 0.05 was considered statistically significant.
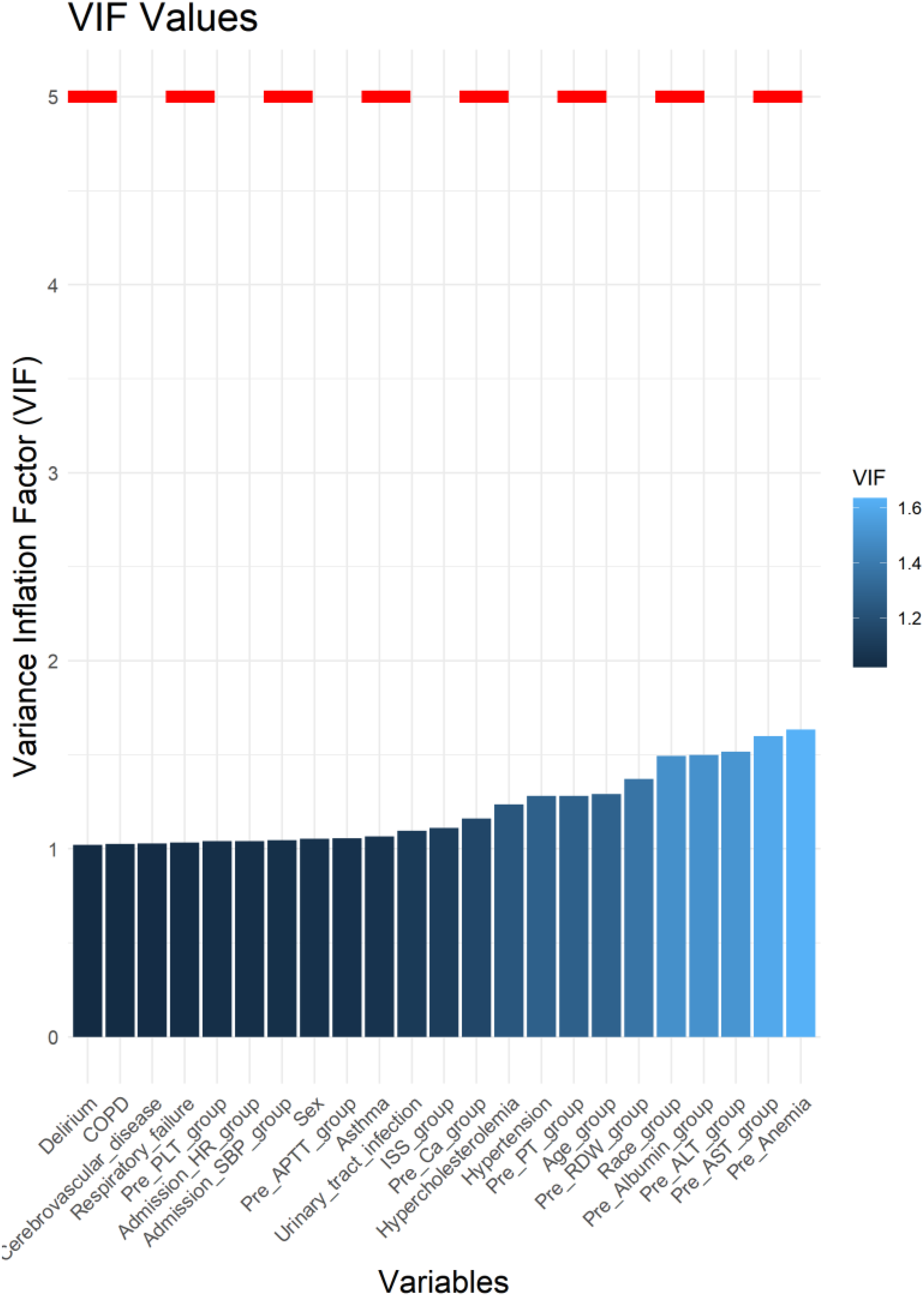
Variance inflation factor analysis of risk factors for postoperative pulmonary embolism in trauma patients. VIF: variance inflation factor; COPD: chronic obstructive pulmonary disease; PLT: platelet count; HR: heart rate; SBP: systolic blood pressure; APTT: activated partial thromboplastin time; ISS: injury severity score; Ca2+: calcium; PT: prothrombin time; RDW: red blood cell distribution width; ALT: alanine aminotransferase; AST: aspartate aminotransferase.

Multivariable logistic regression analysis of risk factors for postoperative PE in trauma patients. PE: pulmonary embolism; RDW: red blood cell distribution width; OR: odds ratio; CI: confidence interval; ISS: injury severity score; HR: heart rate; SBP: systolic blood pressure; COPD: chronic obstructive pulmonary disease; PLT: platelet count; ALT: alanine aminotransferase; AST: aspartate aminotransferase; PT: prothrombin time; APTT: activated partial thromboplastin time; Ca2+: calcium. P < 0.05 was considered statistically significant.
Comparison of Postoperative PE Between the Pre-RDW
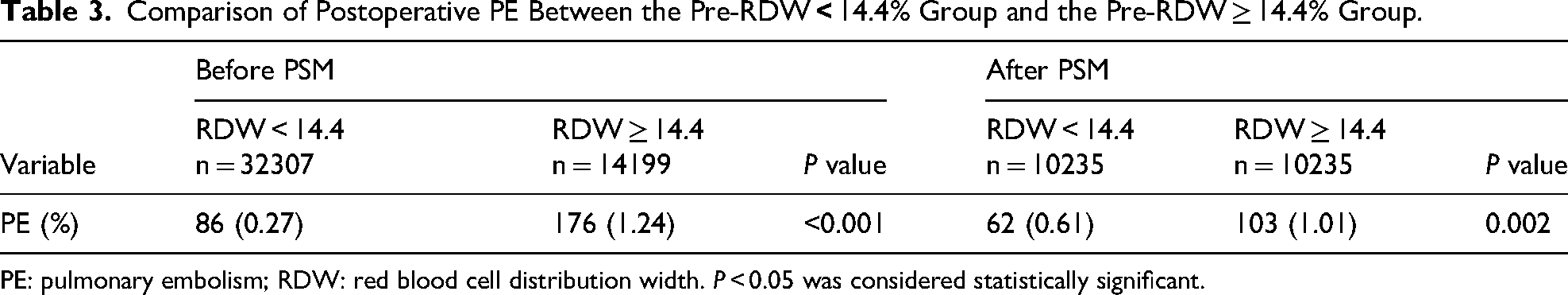
PE: pulmonary embolism; RDW: red blood cell distribution width. P < 0.05 was considered statistically significant.
After PSM, the overall postoperative PE rate was 0.81% (165/20470). The postoperative PE rates in the Pre-RDW < 14.4% group and the Pre-RDW ≥ 14.4% group were 0.61% (62/10235) and 1.01% (103/10235), respectively (P = 0.002) (Table 3). Binary logistic regression analysis showed that compared with the Pre-RDW < 14.4% group, the postoperative PE risk was 1.67 times higher in the Pre-RDW≥ 14.4% group (95% CI 1.22-2.29, P = 0.002) (Table 4). After PSM, univariable Firth penalized logistic regression analysis indicated that compared with the Pre-RDW < 14.4% group, the Pre-RDW ≥ 14.4% group has a 1.66-fold higher risk of postoperative PE (95% CI 1.22-2.29, P = 0.001) (Supplemental Material 5). This finding is consistent with the results from conventional logistic regression, supporting the robustness of the analysis.
Logistic Regression Analysis of Risk Factors for Postoperative PE in Trauma Patients After PSM.
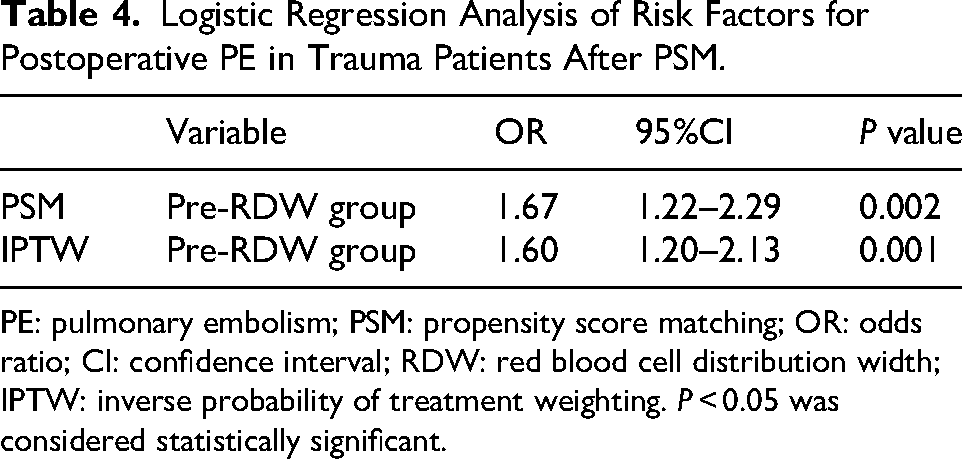
PE: pulmonary embolism; PSM: propensity score matching; OR: odds ratio; CI: confidence interval; RDW: red blood cell distribution width; IPTW: inverse probability of treatment weighting. P < 0.05 was considered statistically significant.
The effect estimate obtained from the IPTW (Supplemental Material 6) analysis (OR 1.60, 95% CI 1.20-2.13, P = 0.001) was highly consistent with the primary PSM analysis result (OR 1.67, 95% CI 1.22-2.29, P = 0.002) (Table 4). The similar conclusions reached by these two distinct propensity score analysis methods strengthen the reliability of the main study finding regarding the association between elevated preoperative RDW and an increased risk of postoperative PE.
Discussion
To the best of our knowledge, this is the first multicenter study to investigate the association between preoperative RDW and the development of postoperative PE in trauma patients. We found that the AUC for preoperative RDW in predicting postoperative PE in trauma patients was 0.723 (95% CI 0.696-0.750), with a cutoff value of 14.4%. Before PSM, univariate logistic regression analysis showed that a preoperative RDW ≥ 14.4% was a risk factor for postoperative PE in trauma patients, with a 4.70-fold increased risk. After multivariable adjustment, a preoperative RDW ≥ 14.4% remained an independent risk factor for postoperative PE, with a 1.90-fold increased risk. After PSM, binary logistic regression further confirmed that patients with a preoperative RDW ≥ 14.4% had a 1.67-fold increased risk of postoperative PE compared with those in the RDW < 14.4% group. Furthermore, the results from our bootstrap analysis, Firth's penalized logistic regression, and IPTW further validated the robustness of the findings.
Association Between RDW and Postoperative PE in Trauma Patients
Previous studies have consistently demonstrated a significant association between RDW and VTE. Zöller et al 14 identified a dose-response relationship between RDW levels and the risk of a first VTE event in a general population, noting a 1.74-fold increase in VTE risk in the highest RDW quartile. However, the predictive performance of RDW in that study was relatively limited, with AUC values of only 0.57 for men and 0.56 for women. In comparison, our investigation revealed that preoperative RDW exhibited a better predictive ability to predict postoperative PE (AUC 0.72). This discovery corresponds with the findings of Celik et al, 2 who reported that RDW at admission is an independent predictor of PE, with each 1% increase in RDW corresponding to a 17.5% rise in PE risk. However, the AUC in that study was only 0.559. Another retrospective study 34 also reported that RDW was significantly higher in VTE patients. RDW > 14.6% was an independent risk factor for isolated deep venous thrombosis and PE. In their study, RDW showed the highest diagnostic accuracy for PE at emergency department admission, with an AUC of 0.70. However, the analysis included only 431 confirmed VTE patients and lacked a non-PE control group, which may have introduced selection bias.
Additionally, multiple studies have shown that RDW is significantly associated with outcomes in trauma patients and that RDW can independently predict mortality in trauma patients.35–38 Cheng X et al reported that RDW is a valuable biomarker for predicting preoperative DVT in elderly patients with hip fractures. 39 Although RDW has demonstrated potential value in assessing the prognosis and risk of complications in trauma patients, no studies have reported on whether RDW can serve as a potential marker for postoperative PE in trauma patients.
Possible Mechanism of Preoperative RDW and Hospital Death in Trauma Patients Undergoing Surgery
RDW is a measure of the variability in red blood cell size, calculated as the standard deviation of red cell volume divided by the mean corpuscular volume (MCV).40,41 Elevated RDW is commonly observed in trauma patients, potentially attributable to factors such as acute blood loss and massive transfusion, disordered erythropoiesis, and increased destruction. Acute hemorrhage directly reduces the total red cell mass,42,43 while massive transfusion during trauma resuscitation introduces red blood cells of varying sizes and maturational stages directly into the circulation.44,45 Concurrently, the systemic stress response caused by trauma and the subsequent inflammatory cascade collaboratively impair normal erythropoiesis.42,43 Stress-induced hormone surges expedite the release of immature red cells, while inflammatory cytokines including TNF-α and IL-6 inhibit the development of erythroid precursors and induce erythropoietin resistance.46,47 These processes collectively result in anisocytosis and increased RDW in trauma patients.
The mechanism by which increased preoperative RDW in trauma patients enhances the risk of postoperative PE may be as follows: Firstly, increasing RDW causes reduced red blood cell (RBC) deformability and heightened aggregation, culminating in increased blood viscosity and microcirculatory blood stasis39,48–51. These alterations exacerbate the marginalization of leukocytes and platelets toward the vascular wall: marginalized leukocytes aggregate on the surfaces of platelet thrombi and release procoagulant substances while concurrently upregulating tissue factor expression, thereby continuously activating the coagulation cascade and facilitating VTE.39,52,53 Second, elevated RDW is closely associated with inflammatory states, and the association between RDW and VTE may also be mediated through inflammatory markers such as C-reactive protein (CRP), interleukin-6 (IL-6), interleukin-8 (IL-8), and monocyte chemotactic protein.54–56 CRP activates the complement system to promote the exposure of procoagulant membranes and induce monocytes to express tissue factor, thereby initiating coagulation; IL-6 increases platelet production and reactivity while downregulating the protein C anticoagulant pathway to promote thrombosis; and IL-8 promotes platelet release of the CD40 ligand to induce tissue factor expression.54,57
These inflammatory factors work together, increasing procoagulant activity and at the same time reducing the function of anticoagulant and fibrinolytic systems. This leads to a microenvironment that promotes blood clot formation.54,57 Furthermore, higher RDW is significantly associated with oxidative stress. Reactive oxygen species can suppress tissue factor pathway inhibitors and increase tissue factor expression, which activates coagulation. They additionally facilitate the aberrant transformation of fibrinogen into fibrin and impair critical anticoagulant processes, including the interaction between thrombin and the heparin-antithrombin III complex.39,58 Consequently, RDW, which indicates variability in red blood cell size, acts as a marker of impaired red blood cell homeostasis. This anomaly may facilitate pulmonary embolism via various mechanisms, including changes in blood rheology, activation of inflammation, and oxidative stress.
Limitations
This retrospective observational study examined the correlation between preoperative RDW and postoperative PE in trauma patients, utilizing data from hospitalizations and laboratory tests. The retrospective design is inherently constrained by the completeness of the original data, encompassing the following specific limitations: firstly, the diagnosis of postoperative PE primarily predominantly depended on clinically driven CTPA, which may lead to underdiagnosis of asymptomatic cases; secondly, as a multicenter investigation, potential discrepancies in thromboprophylaxis protocols, criteria for CTPA, and laboratory techniques for assessing RDW across participating institutions constitute sources of potential heterogeneity. The exclusion of variables with a missing data rate ≥30% may have introduced selection bias. Furthermore, the study could not systematically record details such as certain known VTE-related factors (eg, autoimmune diseases, specific thromboprophylaxis measures), lactate levels, perioperative transfusion details, specific injury characteristics, surgical information, anesthesia type, and perioperative immobilization protocols. Although we employed multiple imputation for handling missing data and used both PSM and IPTW to balance baseline characteristics between groups to control for confounding as much as possible, the aforementioned limitations may still affect the robustness of the conclusions. Future rigorously designed prospective, multicenter cohort studies that systematically collect more comprehensive clinical and surgical information are needed to further validate the findings of this study.
Conclusion
We found that an elevated preoperative RDW (at admission) was associated with postoperative PE in trauma patients and that preoperative RDW ≥ 14.4% was an independent risk factor for postoperative PE in trauma patients.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251401576 - Supplemental material for Association Betweenn Preoperative RDW and Postoperative Pulmonary Embolism in Trauma Patients: A Propensity Score-Matched Multicenter Cohort Study
Supplemental material, sj-docx-1-cat-10.1177_10760296251401576 for Association Betweenn Preoperative RDW and Postoperative Pulmonary Embolism in Trauma Patients: A Propensity Score-Matched Multicenter Cohort Study by Xiaojuan Xiong, Mi Zhou, Peng Hu, Yunqin Ren, Ye Wang and Qingxiang Mao in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-2-cat-10.1177_10760296251401576 - Supplemental material for Association Betweenn Preoperative RDW and Postoperative Pulmonary Embolism in Trauma Patients: A Propensity Score-Matched Multicenter Cohort Study
Supplemental material, sj-docx-2-cat-10.1177_10760296251401576 for Association Betweenn Preoperative RDW and Postoperative Pulmonary Embolism in Trauma Patients: A Propensity Score-Matched Multicenter Cohort Study by Xiaojuan Xiong, Mi Zhou, Peng Hu, Yunqin Ren, Ye Wang and Qingxiang Mao in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-3-cat-10.1177_10760296251401576 - Supplemental material for Association Betweenn Preoperative RDW and Postoperative Pulmonary Embolism in Trauma Patients: A Propensity Score-Matched Multicenter Cohort Study
Supplemental material, sj-docx-3-cat-10.1177_10760296251401576 for Association Betweenn Preoperative RDW and Postoperative Pulmonary Embolism in Trauma Patients: A Propensity Score-Matched Multicenter Cohort Study by Xiaojuan Xiong, Mi Zhou, Peng Hu, Yunqin Ren, Ye Wang and Qingxiang Mao in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-4-cat-10.1177_10760296251401576 - Supplemental material for Association Betweenn Preoperative RDW and Postoperative Pulmonary Embolism in Trauma Patients: A Propensity Score-Matched Multicenter Cohort Study
Supplemental material, sj-docx-4-cat-10.1177_10760296251401576 for Association Betweenn Preoperative RDW and Postoperative Pulmonary Embolism in Trauma Patients: A Propensity Score-Matched Multicenter Cohort Study by Xiaojuan Xiong, Mi Zhou, Peng Hu, Yunqin Ren, Ye Wang and Qingxiang Mao in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-5-cat-10.1177_10760296251401576 - Supplemental material for Association Betweenn Preoperative RDW and Postoperative Pulmonary Embolism in Trauma Patients: A Propensity Score-Matched Multicenter Cohort Study
Supplemental material, sj-docx-5-cat-10.1177_10760296251401576 for Association Betweenn Preoperative RDW and Postoperative Pulmonary Embolism in Trauma Patients: A Propensity Score-Matched Multicenter Cohort Study by Xiaojuan Xiong, Mi Zhou, Peng Hu, Yunqin Ren, Ye Wang and Qingxiang Mao in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-6-cat-10.1177_10760296251401576 - Supplemental material for Association Betweenn Preoperative RDW and Postoperative Pulmonary Embolism in Trauma Patients: A Propensity Score-Matched Multicenter Cohort Study
Supplemental material, sj-docx-6-cat-10.1177_10760296251401576 for Association Betweenn Preoperative RDW and Postoperative Pulmonary Embolism in Trauma Patients: A Propensity Score-Matched Multicenter Cohort Study by Xiaojuan Xiong, Mi Zhou, Peng Hu, Yunqin Ren, Ye Wang and Qingxiang Mao in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Abbreviation
Acknowledgments
This work was supported by the Chongqing Talent Program for Leading Innovation (CSTC2024YCJH-BGZXM0011), the Joint Medical Research Project of Chongqing Science and Health Commission/Health Commission (2025MSXM042), the Daping Hospital Project (ZXAIYB020), and the China Scholarship Council (CSC).
Consent for Publication
Ethical approval to report this case series was obtained from the institutional review boards of all participating centers, with the requirement for informed consent waived due to the retrospective nature of the study. The specific approval references are as follows: the MIMIC-IV v2.2 database (Approval No. 63295742), Chongqing Emergency Medical Center (Approval No. 48, 2023), Rocket Force Characteristic Medical Center (Approval No. KY 2023037), and Daping Hospital (Approval No. 261, 2023). The study was registered in the Chinese Clinical Trial Registry under the identifier ChiCTR2300078097.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the Daping Hospital Project, the China Scholarship Council (CSC), the Joint Medical Research Project of Chongqing Science and Health Commission/Health Commission, the Chongqing Talent Program for Leading Innovation, (grant number ZXAIYB020, 2025MSXM042, CSTC2024YCJH-BGZXM0011).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
Due to the protection of participants’ privacy and the constraints of ethical protocols, the data of this study has not been made public. If there is a need for academic cooperation, please provide a detailed research plan and submit a formal application through the corresponding author. After review, the anonymized data can be partially shared.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
