Abstract
Background
Red blood cell distribution width (RDW) was a risk factor for poor prognosis in acute myocardial infarction (AMI). Recent reports suggested that combining RDW with other laboratory metrics could provide a better prediction. This retrospective study aimed to investigate whether the RDW-albumin ratio (RAR) may be associated with mortality after an AMI.
Methods
This cohort study was conducted among adults (over 16 years old) with AMI in the Medical Information Mart for Intensive Care Database III V1.4 (MIMIC-III). The primary outcome was 30-day mortality, and the secondary outcome was 1-year and 3-year mortality. Cox hazard regression model and Kaplan–Meier survival curves were constructed to estimate the effect of biomarkers on mortality. We used three models to adjust for potential bias. Receiver operating characteristic (ROC) curves and the area under the curve (AUC) were analyzed for the excellent performance of RAR on prognosis.
Results
A total of 826 patients were eventually enrolled in our study. In multivariate analysis, RAR was found to be associated with 30-day mortality (Model 3: HR = 1.23, 95% CI = 1.09-1.39, P < .001). In addition, Subgroup analysis showed that the effect of RAR was higher in female patients than in male patients (P for interaction = .026). Kaplan-Meier survival curves showed that patients in the lower RAR quartile tended to have higher survival rates in the short and long term. AMI patients with RAR ≥ 4 had a 122% increase in 3-year mortality. Results of ROC and AUC showed that the prognostic performance of RAR for mortality was the best (30-day mortality: 0.703; 1-year mortality: 0.729; 3-year mortality: 0.737).
Conclusions
RAR is a simple and stable predictor of prognosis in AMI patients. Our results support RAR = 4.0 as a criterion for prognostic risk stratification of AMI patients.
Keywords
Introduction
Acute myocardial infarction (AMI) is a frequent cause of admission to the intensive care unit (ICU). Bora Nadlacki et al stated an all-cause mortality rate of 23.8% up to three years after an AMI. 1 Therefore, sensitive determination of the prognosis with AMI has important implications for treatment. In the past few years, highly sensitive biomarkers for rapid detection of AMI have been emerging. However, with their quick and comprehensive advantages, hematological parameters remain indispensable in the diagnosis and prognostic assessment of AMI.
Among the hematological indicators, the red blood cell distribution width (RDW), a parameter of the volumetric of peripheral red blood cells (RBC), has also emerged as a prognostic marker in various cardiovascular disorders. 2 The associations between RDW and AMI have no definitive evidence but may involve higher pro-inflammatory substance levels. It leads us to think of indicators of inflammatory response spontaneously. As is known to all, serum albumin is a conventional indicator used to measure body nutritional status or inflammatory reaction. According to reports, low serum albumin on admission for AMI was an independent predictor of cardiovascular death events. 3 RDW-albumin ratio (RAR) was associated with the prognosis of aortic aneurysms, 4 diabetic ketoacidosis, 5 cancer 6 and heart failure (HF). 7 It could be a critical metric for assessing criticality scores in cardiovascular disease. Thus, we planned a retrospective analysis to verify the connection between RAR and mortality within adult AMI patients admitted to ICU.
Materials and Methods
Data Source
Our study is based on a publicly available critical care database known as Multiparameter Intelligent Monitoring in Intensive Care III (MIMIC-III). 8 More than 50 000 patients admitted to the ICU of Beth Israel Deaconess Medical Center (BIDMC) between 2001 and 2012 made up this enormous and complete database. The demographic characteristics, laboratory index, and medical history of patients with AMI were screened to participate in this research. One of our authors obtained approval for using the database (certification number 43453324). Meanwhile, the patients in the database are not identified to protect their privacy. The Institutional Review Boards (IRB) of the Massachusetts Institute of Technology (MIT, Cambridge, MA, USA) and BIDMC approved MIMIC-III. The informed consent of all patients was not required for this study.
Data Extraction
Database management tools were used to filter data from MIMIC-III. We enrolled adult patients (over 16 years old) with AMI. According to the International Classification of Diseases (ICD)-9 code, AMI was defined from 410.00 to 410.92. The extracted data for study mainly included demographics (age and sex), vital signs [heart rate (HR), systolic blood pressure (SBP), diastolic blood pressure (DBP), mean blood pressure (MBP), respiratory rate (RR), pulse oxygen saturation (SpO2), and temperature], comorbidities (atrial fibrillation (AF), coronary artery disease (CAD), congestive heart failure (CHF), hypertension, diabetes, renal failure and malignancy). In addition to RDW and serum albumin, we extracted laboratory parameters such as creatinine, blood urea nitrogen (BUN), potassium, sodium, hemoglobin, platelets, and white blood cell (WBC) from patients within 24 h after admission. For patients with multiple hospitalizations, only data of initial admission were used. Only the initial test results were included if patients received a laboratory test more than once during their hospitalization.
Statistical Analysis
The sample was categorized into quantity-specific quartiles of RAR. 30-day, 1-year, and 3-year mortality were extracted as outcome variables. Basic vital signs and laboratory parameters were fitted as continuous variables, whereas relevant cardiovascular disease was fitted as dichotomous variables. Quantitative data were analyzed with the X2 test: quantitative data in a normal distribution were expressed as x ± s, and quantitative data in a non-normal distribution were expressed as the rank-mean. The inter-group difference was assessed using the t-test. We used the Kruskal-Wallis test to examine differences in continuous variables. The prognostic values of laboratory tests were evaluated using the Cox hazard regression model and Kaplan-Meier curve analysis. Cox proportional hazards regression with backward stepwise elimination was used to estimate hazard ratios (HRs) adjusted for potential confounding factors. Receiver operating characteristic (ROC) curves and the area under the curve (AUC) were used to analyze the sensitivity and specificity of mortality among AMI. Three different models were designed to adjust for potential confounders. Model 1 was adjusted for nothing; Model 2 adjusted for age, sex, and vital signs. Model 3 adjusted for comorbidities and Laboratory parameters based on model 2.
The analyzes were performed with the statistical software package R v3.3.2 (http://www.R-project.org, The R Foundation). A P value less than .05 was considered statistically significant.
Results
Baseline Characteristics
The flow chart is presented in Figure 1. The study population's baseline demographics and clinical characteristics concerning quantity-specific quartiles of RAR (< 3.5 ml/g, 3.5-4.0 ml/g, 4.0-4.8 ml/g and > 4.8 ml/g) are shown in Table 1. Patients in the lowest quartile RAR (RAR < 3.5 ml/g) displayed a younger age and proportionally larger female. As RAR rises, so does the incidence of AF, CHF, hypertension, diabetes, renal failure, and malignancy was high. Patients in high tertile were more likely to have lower serum albumin, hemoglobin and platelet levels. As of the third year, a total of 305 (36.9%) patients had died.

The flow chart of the study.
Baseline Characteristics of the Study Population.

Abbreviations: SBP, systolic blood pressure; DBP, diastolic blood pressure; MBP, mean blood pressure; SpO2, pulse oxygen saturation; AF, Atrial fibrillation; CAD, Coronary artery disease; CHF, Congestive heart failure; RAR, ratio of red blood cell volume distribution width to albumin; RDW, red cell distribution width; BUN, blood urea nitrogen; WBC, white blood cell.
Predictive Value of RAR for Mortality
Table 2 lists cox proportional hazard analysis for all-cause death. In univariable cox proportional hazards regression, hazard ratios (HRs) for RAR respectively were 1.32 (95% CI, 1.22-1.44; P < 0.001), 1.34 (95% CI, 1.26-1.42; P < .001), 1.33 (95% CI, 1.26-1.41; P < .001). Multivariate-adjusted HRs showed that RAR was an independent predictor of short and long-term mortality in patients with first-onset AMI [30-day mortality (Model 3: HR = 1.23, 95% CI = 1.09-1.39, P < .001), 1-year mortality (Model 3: HR = 1.24, 95% CI = 1.13-1.37, P < .001), 3-year mortality (Model 3: HR = 1.25, 95% CI = 1.15-1.37, P < .001)]. In addition, the HR in the highest RAR group (>4.8 ml/g) in tertiles had a significant difference compared with the reference group in all models (P < .001). Subgroup analysis (Table 3) showed that the effect of RAR was significantly higher in female patients than in male patients (P for interaction = .026) The Kaplan-Meier survival curved comparing patients within different RAR were shown in Figure 2, which showed that patients in the higher RAR quartile tended to have lower short-term survival. For 30-day mortality, the prognostic performance of RAR was significantly better than the other two groups (RAR AUG = 0.703; albumin AUG = 0.665; RDW AUG = 0.657) (Figure 3A). For 1-year and 3-year mortality, the prognostic performance of RAR was also the best among them (Figure 3B: RAR AUG = 0.729; albumin AUG = 0.671; RDW AUG = 0.701. Figure 3C: RAR AUG = 0.737; albumin AUG = 0.666; RDW AUG = 0.725).

ROC curves for RAR in 30-day mortality, 1-year mortality and 3-year mortality of the AMI patients. (A) 30-day mortality; (B) 1-year mortality; (C) 3-year mortality.

Kaplan-Meier survival analysis plot for 3-year overall survival within different quartiles. (A) represents 30-day mortality; (B) represents 1-year mortality; (C) represents 3-year mortality.
Multivariate Cox Regression for Prognosis of Patients with RAR and AMI.
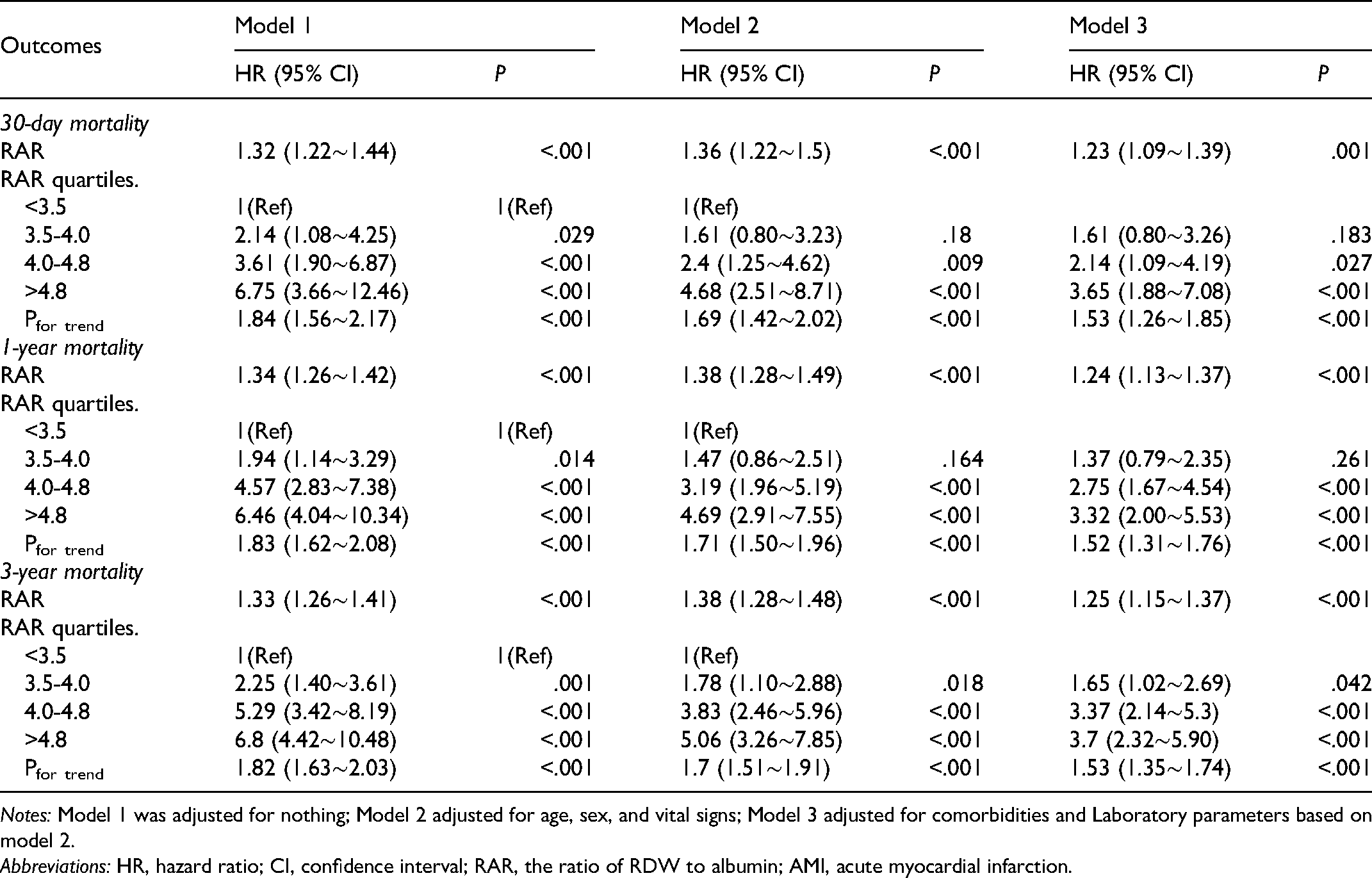
Notes: Model 1 was adjusted for nothing; Model 2 adjusted for age, sex, and vital signs; Model 3 adjusted for comorbidities and Laboratory parameters based on model 2.
Abbreviations: HR, hazard ratio; CI, confidence interval; RAR, the ratio of RDW to albumin; AMI, acute myocardial infarction.
Subgroup Analysis of the Relationship Between 30-day Mortality and RAR.

Abbreviations: AF, Atrial fibrillation; CAD, Coronary artery disease; CHF, Congestive heart failure; HR, hazard ratio; No. event_%, 30-day mortality in subgroup; CI, confidence interval; RAR, the ratio of RDW to albumin.
Discussion
To our knowledge, the study was the first to evaluate the relationship between RAR and the prognosis of AMI. Its significant finding was that RAR was an independent predictor for short-term and long-term mortality among patients with first-onset AMI. Moreover, the RAR is more sensitive than RDW and serum albumin values to predict mortality in AMI patients. Based on Cox regression and Kaplan-Meier survival curves, we divided the RAR values into two groups with a 4.0 threshold (Table S1). The results showed that patients with RAR greater than 4.0 had significantly higher mortality at 30-day, 1-year and 3-year than the other group. The fitted curves (Figure S1) also show significantly higher mortality in AMI patients when the RAR is ≥ 4.0. Patients with RAR ≥ 4.0 on admission should be focused on, as the group of patients often has a poor prognosis for death. Table S2 shows a 122% increased risk of 3-year mortality in AMI patients with RAR ≥ 4. As a stable and readily available hematological indicator, the RAR can also help clinicians assess the long-term prognosis of AMI patients after discharge from the hospital.
It is well known that RDW reflects changes in RBC volume and function. The all-cause mortality of cardiovascular and thrombotic diseases, including coronary artery disease, acute and chronic heart failure and peripheral arterial disease, is often associated with a high degree of anisocytosis. 2 A study of 2304 patients with the acute coronary syndrome (ACS) showed a significant sensitivity (79%) of RDW for the diagnosis of ACS. 9 In addition, Sánchez-Chaparro et al 10 reported that patients with high RDW values are more likely to develop metabolic syndrome, including multiple risk factors for cardiovascular disease. Physiologically, erythrocytes deliver oxygen to tissue cells and release mediators for cardiovascular regulation. 11 Thus, alterations in red blood cells have a predisposing and exacerbating effect on cardiovascular disease. Persistent increased RDW was related to pathophysiological processes with oxygen deficit and inflammation. 12 A hormone called erythropoietin (EPO) secreted during the hypoxic condition, promoting the release of enlarged RBCs, leads to the increased RDW abnormally in cardiovascular diseases. 13 For another, chronic inflammation is often accompanied by oxidative stress and ineffective erythropoiesis. The release of immature erythrocytes from the bone marrow into the blood leads to a variation in the size of circulating erythrocytes. 12 Pro-inflammatory cytokines regulate erythrocytes by effectively inhibiting EPO release and erythroid maturation. Finally, malnutrition and dyslipidemia are other factors associated with high RDW through effects on erythropoiesis. 14
Serum albumin is a major protein whose primary function is to maintain cellular osmotic pressure by transporting substances. In addition, the albumin value produced by the liver inevitably decreases in disease states. Previous studies showed low serum albumin levels are an independent predictor for long-term all-cause mortality in patients with AMI.15, 16 Further studies have suggested that low albumin values are associated with cardiogenic ischemic stroke. 17 Atherosclerosis, an inflammatory disease, is involved in the process of cardiovascular disease. 18 Serum albumin plays an anti-inflammatory function in this process, and once serum albumin decreases, the disease process worsens. Besides, serum albumin dissolves tiny amounts of sterol and promotes cholesterol efflux from cells, 19 exacerbating cardiovascular disease risk factors. Finally, it was reported that serum albumin within normal concentrations might scavenge peroxyl radicals, 20 inhibit platelet activation and aggregation, 21 relieve vasospasm, 22 and improve blood viscosity 23
A study showed that the RAR values are independent correlates of all-cause mortality. The stroke patients in the ICU with high levels of RAR (RAR > 4.03 ml/g) had a higher risk of all-cause mortality. 24 Exploring based on the existing putative mechanisms, we may respond to the association between RAR and AMI. First, AMI is a related inflammatory disease, leading to increased elevation of RDW and decreased albumin. Second, aberrant RDW and albumin values are associated with accelerated disease progression. On the one hand, abnormalities in hematological indicators are predisposing factors for disease. On the other hand, RAR is a useful prognostic progression indicator. Meanwhile, our study indicates that the RAR showed a better expression of inflammatory response outcomes than either marker alone. It is considered that the RAR value with a cut-off value of 4.0 is easy to calculate and simple, the reliable parameter for risk stratification of AMI patients admitted to the ICU.
Some limitations must be taken into account. Firstly, this was a retrospective analysis; potential bias may be involved. In this regard, we adjusted different variables to ensure that the results were as accurate as possible. Secondly, it should be noted that the subjects enrolled in this study were admitted to ICU. Thus, the conclusions of this study cannot be extended to the AMI patients admitted to other departments. Thirdly, due to the lack of relevant surgical information, the model of this study is imperfect. Further prospective studies are needed to validate our conclusions. Despite these drawbacks, our research is meaningful in exploring the relationship between RAR and AMI.
Conclusion
In summary, RAR is a prognosis-related risk factor for patients with AMI. We support physicians in using the RAR value of 4.0 as a criterion for prognostic risk stratification of patients.
Supplemental Material
sj-zip-1-cat-10.1177_10760296221121286 - Supplemental material for Association of Red Blood Cell Distribution Width-Albumin Ratio for Acute Myocardial Infarction Patients with Mortality: A Retrospective Cohort Study
Supplemental material, sj-zip-1-cat-10.1177_10760296221121286 for Association of Red Blood Cell Distribution Width-Albumin Ratio for Acute Myocardial Infarction Patients with Mortality: A Retrospective Cohort Study by Dan Li, MD, Zhishen Ruan, MD, and Bo Wu, MD in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgements
There are no additional acknowledgements.
Author Contributions
Dan Li participated in the study design, analyzed the results, and edited the manuscript. Zhishen Ruan for data mining and cleaning. Bo Wu participated in the research design and editor of the manuscript.
Availability of Data and Materials
Data in the article can be obtained from the MIMIC-III database (https://mimic.physionet.org/).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by the Shandong Science and Technology Development Program of Traditional Chinese Medicine (2021Z044) and National Natural Science Foundation of China (82174177).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
