Abstract
Objectives
To investigate the correlation between red blood cell distribution width-to-platelet ratio (RPR) and 30-day mortality in patients with postoperative acute pulmonary embolism (PAPE).
Methods
This retrospective cohort study included patients diagnosed with PAPE who were hospitalized in the general surgery ward of our hospital from January 2019 to January 2024. We collected general clinical parameters and laboratory test results at admission and on the day of PAPE diagnosis. Multivariate analysis was performed to identify independent risk factors associated with 30-day mortality. A nomogram was constructed, and its predictive performance was evaluated.
Results
132 patients with PAPE were included in this study, comprising 36 males and 96 females. The median age was 67 years, PAPE occurred on average 3.2 days after primary disease surgery, 18 patients experienced 30-day mortality. Concurrently with DVT (OR: 0.15, 95% CI: 0.04-0.64, P = .011) and RPR ≥ 0.08 on the diagnosis of PAPE (OR: 9.19, 95% CI: 2.91-29.05, P < .001) were independently associated with 30-day mortality in PAPE patients. The AUC of the multivariate model was 0.77 (95% CI: 0.63-0.91). The Internal validation of nomogram showed the bootstrap-corrected AUC was 0.82 (95%CI 0.76-0.88).
Conclusions
RPR on the diagnosis of PAPE is independently associated with the prognosis of PAPE patients. The 30-day mortality prediction model constructed based on independent risk factors demonstrated good predictive performance.
Keywords
Introduction
Postoperative acute pulmonary embolism (PAPE) is a severe postoperative complication with an overall incidence of 0.9% to 3%.1-4 PAPE can reduce the prognosis quality of the primary disease. Some studies have reported that the short-term mortality rate of PAPE is approximately 10% to 23%.3,5-7 This condition significantly increases the burden on both patients and national healthcare systems. Timely identification of high-risk patients and aggressive intervention are critical for improving clinical outcomes in patients with PAPE. Previous studies have confirmed that common inflammatory markers,8,9 such as the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and systemic immune-inflammation index (SII), are associated with the prognosis of pulmonary embolism (PE). These findings suggest that the inflammatory response plays a crucial role in the pathogenesis and progression of PE. The red blood cell distribution width-to-platelet ratio (RPR), as a novel, readily available and cost-effective marker of inflammation, can be easily derived from routine complete blood count (CBC) data, existing studies have demonstrated its association with the prognosis of various diseases.10-13 However, due to the relatively low incidence of PAPE, no studies have yet investigated the correlation between RPR and the short-term prognosis of PAPE.
In this study, we utilized data from a large medical center in China to investigate the correlation between RPR and 30-day mortality in patients with PAPE. Furthermore, we innovatively examined which stage of hospitalization yielded RPR values derived from CBC data with greater predictive power.
Methods
Inclusion and Exclusion Criteria
This is a retrospective cohort study. All clinical data were obtained from the hospital's medical record system. This study was reviewed and approved by the Ethics Committee of Beijing Friendship Hospital, Capital Medical University. The inclusion criteria for this study were as follows: patients admitted to the Department of General Surgery at Beijing Friendship Hospital, Capital Medical University, between January 2019 and December 2024; those who underwent standard surgery recommended by guidelines for their primary diseases; admission diagnoses that did not include pulmonary embolism (PE) or deep vein thrombosis (DVT); and confirmation of PE after surgery for the primary disease, with discharge diagnoses including PE. The exclusion criteria included: any anticoagulant therapy received before surgery for the primary disease, and previous inferior vena cava filter (IVCF) placement without subsequent removal. A total of 132 PAPE patients were enrolled in this study.
Data Collection
Clinical baseline characteristics, laboratory test results, and imaging findings related to PE were collected from the enrolled patients. The special clinical parameters are defined as follows: chronic cardiopulmonary disease (CCPD) includes heart failure and chronic pulmonary disease. All blood gas analysis data were collected within 2 h after the diagnosis of PAPE and measured under room air conditions without supplemental oxygen, and the other laboratory test results were obtained on the day of PAPE diagnosis. Additionally, we also collected the CBC results of the patients upon admission to calculate the RPR at different periods. RPR was calculated as RPR = red blood cell distribution width (RDW, %) / platelet count (PLT, 109/L). DVT was diagnosed using color Doppler ultrasound, and pulmonary embolism was confirmed via computed tomography pulmonary angiography (CTPA). Inferior vena cava filter (IVCF) placement was indicated for PAPE patients with concurrent DVT and contraindications to anticoagulation therapy. The primary endpoint of this study was all-cause mortality within 30 days of PAPE diagnosis.
Statistical Analysis
Continuous variables were presented as mean (standard deviation) or medians (interquartile ranges), depending on their distribution. Categorical variables were expressed as percentages (%). Associations between categorical variables and outcomes were evaluated using the chi-square test, while continuous variables were analyzed using either the t-test or the Mann–Whitney U test, based on their distribution characteristics. The receiver operating characteristic (ROC) curve was used to assess the predictive performance of risk factors for 30-day mortality. The area under the curve (AUC), obtained from the ROC analysis, with higher values indicating better discriminatory ability. The optimal cut-off point was determined using the Youden Index, which is defined as the maximum value of (sensitivity + specificity - 1).
Multivariate logistic regression analysis with a stepwise regression approach was conducted to identify independent risk factors associated with 30-day mortality. The results were visualized using a nomogram, and the predictive performance of the nomogram was evaluated using ROC curve with AUC and calibration curves. To evaluate the clinical utility of the nomogram, decision curve analysis (DCA) was performed. The internal validation of the nomogram was performed to evaluate its robustness against overfitting, we employed “Bootstrap resampling” with 1000 iterations to estimate the optimism-corrected performance metrics, and provided the bootstrap-corrected AUC and the bootstrap AUC distribution. All statistical tests were two-sided, with P-values less than .05 indicating statistical significance. Statistical analyses were performed using SPSS 26.0 (IBM) and R (version 4.4.2).
Results
Patient Baseline Characteristics
In this study, a total of 132 patients with PAPE were included, comprising 36 males and 96 females. The median age was 67years (interquartile range: 60-76 years). Of these patients, 98 had malignant tumors as their primary disease, and all underwent the guideline-recommended radical surgical protocol for their primary disease. The average length of hospital stay was 20.6 days (standard deviation: 8.9 days), and the average time from surgery to diagnosis of PAPE was 3.2 days (standard deviation: 2.1 days). Baseline clinical characteristics of the PAPE patients are summarized in Table 1.
Comparison of Clinical Baseline Characteristics, Laboratory Test Results, and Imaging Findings Between Survival and Death Group.

Abbreviations: MetS, metabolic syndrome; CCPD, chronic cardiopulmonary disease; DVT, deep vein thrombosis; PAPE, postoperative acute pulmonary embolism; PA, pulmonary artery; MPA, main pulmonary artery; IVCF, inferior vena cava filter; RDW, red blood cell distribution width; PLT, platelet count; RPR, red blood cell distribution width-to-platelet ratio; TNT, cardiac troponin T (normal range: 0.010-0.017 ng/ml); BNP, N-terminal pro-B-type natriuretic peptide (normal range: 0-900 ng/l); TNI, cardiac troponin I (normal range: 0.000-0.030 ng/ml).
Among the 90 patients with pulmonary embolism-related symptoms, which included sudden chest tightness, chest pain, profuse sweating, and syncope, 42 patients exhibited no obvious clinical symptoms. Of these asymptomatic patients, 35 patients had abnormal laboratory findings, including hypoxemia and significantly elevated D-dimer levels, 7 patients incidentally discovered pulmonary embolism during postoperative enhanced CT reexamination. A total of 120 patients were concurrently diagnosed with DVT; among them, 41 received inferior vena cava filter (IVCF) placement. The remaining patients received standard-dose low-molecular-weight heparin (LMWH) anticoagulation therapy, with no major bleeding events related to anticoagulation. None of the patients underwent thrombolysis or pulmonary artery thrombectomy.
Risk Factor of 30-day Mortality
A total of 18 patients experienced 30-day mortality, resulting in a 30-day mortality rate of 13.6%. All patients had a high-risk sPESI score. Patients were divided into the survival group and the death group based on whether they experienced 30-day mortality. Detailed comparisons between the two groups are presented in Table 1. Significant differences were observed between the groups in terms of gender (P < .001), concurrently with DVT (P = .001), PLT on the diagnosis of PAPE (P = .004), and RPR on the diagnosis of PAPE (P = .001).
The AUC for RDW, PLT, and RPR at admission and on the diagnosis of PAPE for predicting 30-day mortality are presented in Table 2. Among these, the PLT and RPR on the diagnosis of PAPE showed statistically significant predictive power for 30-day mortality. According to the Youden index, the optimal cut-off points for PLT and RPR on the diagnosis of PAPE were 159 and 0.08, respectively.
the AUC for RDW, PLT, and RPR at Admission and on the day of PAPE Diagnosis for Predicting 30-day Mortality.

Abbreviations: PAPE, postoperative acute pulmonary embolism; RDW, red blood cell distribution width; PLT, platelet count; RPR, red blood cell distribution width-to-platelet ratio.
the Multivariate Regression Results of 30-Days Mortality for PAPE Patients.
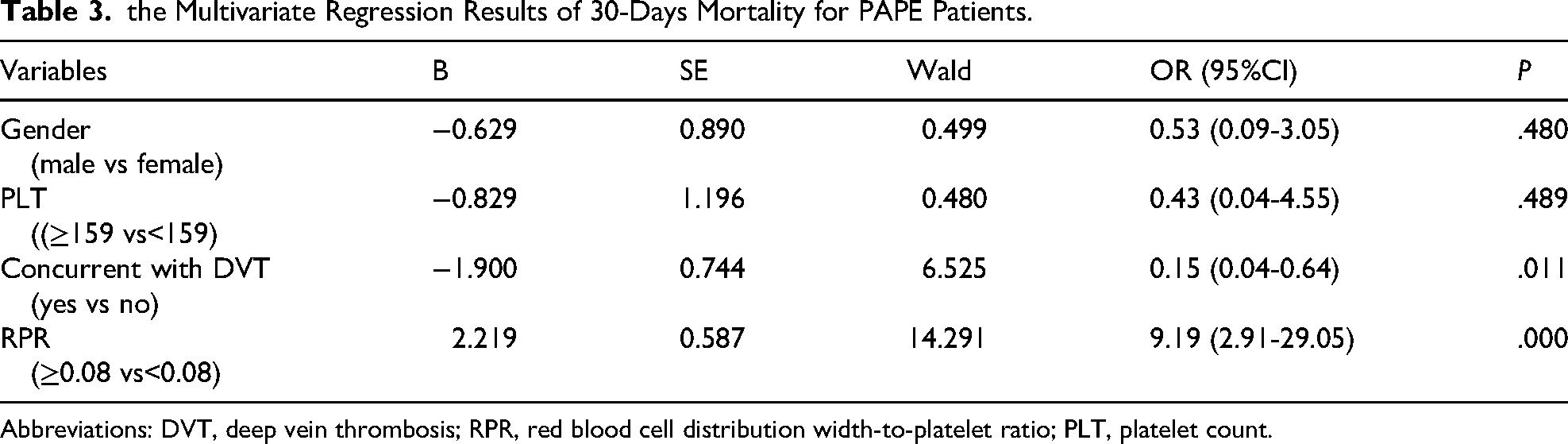
Abbreviations: DVT, deep vein thrombosis; RPR, red blood cell distribution width-to-platelet ratio; PLT, platelet count.
Gender (male vs female), concurrently with DVT (yes vs no), PLT on the diagnosis of PAPE (≥159 vs <159), and RPR on the diagnosis of PAPE (≥0.08 vs < 0.08) were included in the multivariate logistic regression analysis. The results indicated that concurrently with DVT (OR: 0.15, 95% CI: 0.04-0.64, P = .011) and RPR on the diagnosis of PAPE ≥ 0.08 (OR: 9.19, 95% CI: 2.91-29.05, P < .001) were independently associated with 30-day mortality in PAPE patients (Table 3). The AUC of the multivariate model was 0.77 (95% CI: 0.63-0.91). Based on these results, a nomogram was developed (Figure 1). The ROC curve and calibration curve of nomogram for predicting 30-day mortality in patients with PAPE was presented in Figures 2 and 3, respectively.
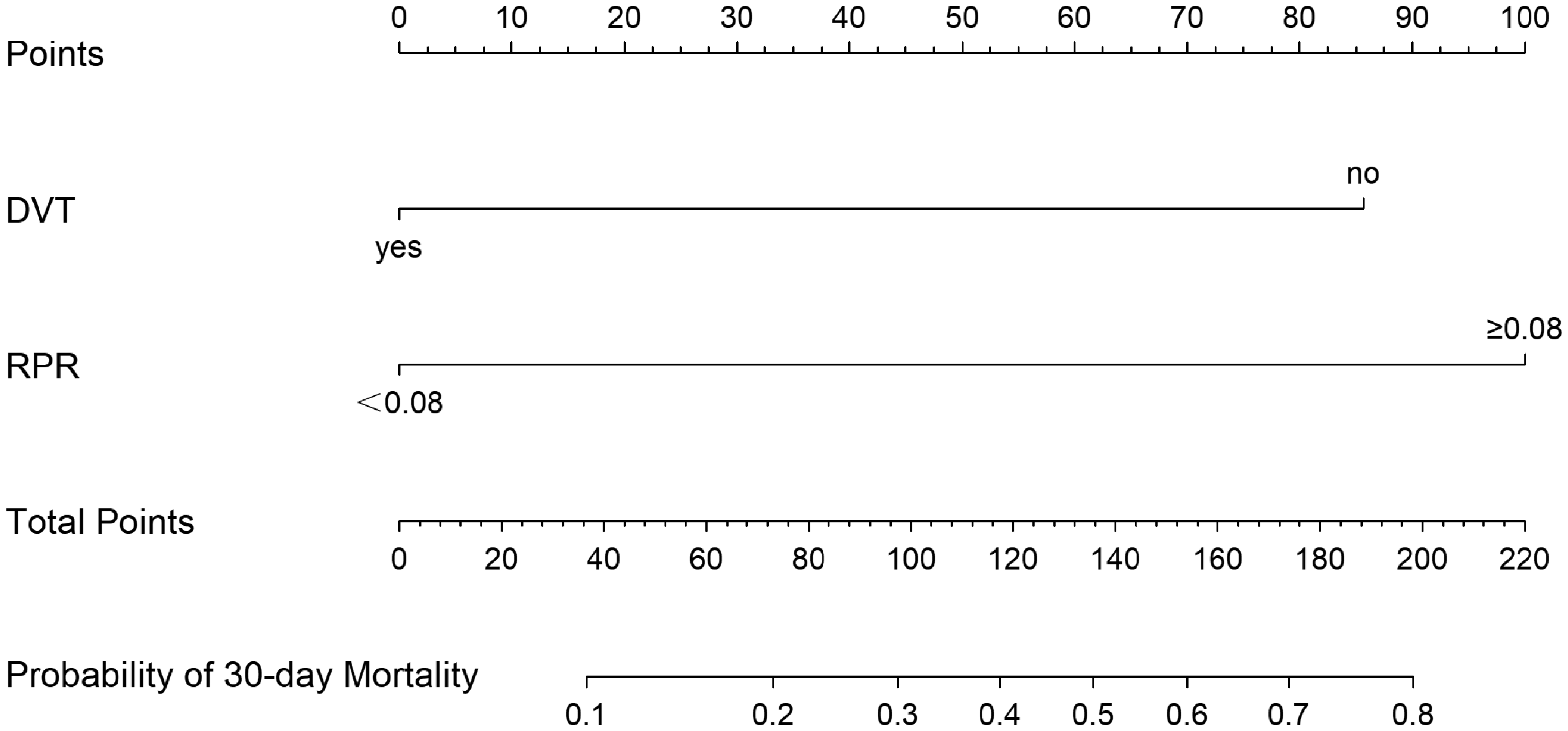
the Nomogram for Predicting 30-day Mortality in Patients with PAPE.

The ROC Curves of Nomogram for Predicting 30-Day Mortality.

the Calibration Curve for the Nomogram for Predicting 30-Day Mortality.
The decision curve analysis (DCA) of nomogram for predicting 30-day mortality was presented in Figure 4, and DCA showed that the nomogram provided a net benefit of 0.22–0.58 across clinically relevant threshold probabilities (8%-50%). The Internal validation was performed using 1000 bootstrap resamples to correct for optimism. The bootstrap-corrected AUC was 0.82 (95%CI 0.76-0.88), and the bootstrap AUC distribution was presented in Figure 5.

the Decision Curve Analysis of Nomogram for Predicting 30-Day Mortality.

the Bootstrap AUC Distribution for the Internal Validation of Nomogram.
Discussion
PAPE is a complex condition with a poor prognosis. Due to its low incidence, however, there has been limited research on this disease. In this study, we integrated the clinical baseline characteristics and laboratory test results of PAPE patients to develop a clinical prediction model for 30-day mortality. This model aims to enhance our understanding of PAPE and provide valuable insights into its prognosis.
Most pulmonary artery thrombi are believed to originate from the detachment of DVT in the lower extremities. However, the prognostic role of DVT in PE remains controversial. A prospective study involving 707 PE patients found that compared with patients without DVT, those with DVT had a twofold increase in all-cause mortality and a fourfold increase in PE-related mortality at 3 months. 14 Conversely, some studies have reached different conclusions. For instance, Slobodan D et al demonstrated that DVT was associated with reduced in-hospital mortality exclusively in patients with intermediate-high PE (OR 0.320, 95%CI 0.164-0.627; P = .001). They proposed that symptomatic DVT often results from complete venous occlusion without residual flow, which may displace thrombus fragments and consequently play a protective role against life-threatening embolization. 15 Another 2020 study, which included 392 PE patients and classified them into high-risk, intermediate-risk, and low-risk groups according to ESC guidelines, found that the proportion of DVT was lower in high-risk PE patients compared to low-risk and intermediate-risk groups. 16 In our study, we also observed a similar trend: PAPE patients with DVT had a lower risk of 30-day mortality. We propose the following explanations for this phenomenon. First, in some PAPE patients, most of the deep vein thrombi in the lower extremities may have already completely detached and migrated to the pulmonary artery. The sudden blockage of the pulmonary artery by a large amount of thrombi can increase the risk of death, while thrombi in the deep veins of the lower extremities become undetectable. Additionally, if DVT occurs first after surgery of primary disease, it can heighten clinical vigilance and prompt early anticoagulant therapy before the occurrence of PE, potentially reducing PE-related mortality.
RDW is an indicator that measures the variability in red blood cell size. Several observational studies have found that elevated RDW is associated with adverse outcomes in various diseases,17-19 such as a higher incidence of venous thromboembolism (VTE) and increased overall mortality. However, some researchers argue that high RDW may merely reflect other adverse prognostic factors, including age, comorbidities, or physiological stress. In our study, RDW alone was not significantly associated with 30-day mortality in PAPE patients. RPR has been proposed as a novel systemic inflammation marker. Multiple studies have demonstrated that RPR is associated with the prognosis of various diseases.20-22 For instance, a retrospective analysis based on MIMIC data, which included 1922 patients, found that RPR was associated with in-hospital mortality in patients with acute exacerbations of chronic obstructive pulmonary disease, with an odds ratio (OR) of 1.36. 20 Another study involving 257 glioblastoma patients found that elevated RPR was associated with poorer overall survival, with an optimal cut-off point of 0.05. 21 To date, no studies have specifically examined the association between RPR and the prognosis of PAPE patients.
Given the previous multiple studies on RPR, we aimed to investigate the association between RPR and 30-day mortality in PAPE patients using our center's clinical data. we also found that previous RPR-related studies have been inconsistent in selecting the timing of CBC data for calculating RPR. Some studies utilized admission CBC data, while others used data collected at different time points. To comprehensively assess the prognostic value of RPR in PAPE, we innovatively collected CBC data from PAPE patients both at admission and on the day of PAPE diagnosis. Our findings revealed that RPR calculated from the CBC data on the day of PAPE diagnosis was independently associated with 30-day mortality, whereas RPR derived from admission data showed no significant correlation with 30-day mortality. RPR is primarily considered an inflammatory marker. The hemodynamic changes and tissue hypoxia induced by PAPE can exacerbate systemic inflammation, leading to elevated RPR levels. This may explain why RPR calculated from admission data, which may fail to fully capture the acute inflammatory response elicited by PAPE, shows no correlation with prognosis. Several widely accepted prognostic models for PE have been established, with the simplified Pulmonary Embolism Severity Index (sPESI) being the most commonly used in clinical practice.3,23-25 However, in our study, all PAPE patients had high-risk sPESI scores, limiting its utility for further risk stratification. To address this limitation, we developed a prediction model for 30-day mortality in PAPE patients by integrating RPR with general clinical parameters (concurrently with DVT). This model demonstrated superior predictive performance.
This study also has certain limitations. Firstly, PAPE can occur in any surgical department; however, we only included patients from the general surgery department in this study. Secondly, our analysis is based on data from a single center with a relatively small sample size, the AUC of the predictive model developed in this study was 0.77, falling short of high predictive performance (AUC > 0.8), Moreover, this study exclusively enrolled sPESI high-risk patients, which may constrain the generalizability of the predictive model. Future studies with larger, multi-center samples are needed to further validate the association between RPR and the prognosis of PAPE.
Conclusion
RPR demonstrates good predictive value for the short-term prognosis of PAPE patients. As a novel, easily accessible, and cost-effective inflammatory marker, RPR holds significant potential for clinical application.
Footnotes
Ethical Considerations
The study was approved by the Beijing Friendship Hospital, Capital Medical University, Beijing, China (Approval No: BFHHZS20240188).
Consent to Participate
Informed consents were taken from all patients included in the study.
Author Contributions
Yunchao Xing: Writing-original draft, Writing-review and editing, Data curation, Methodology.
Wenrui Li: Writing-review and editing, Data curation.
Xueming Chen: Writing-review and editing, Supervision, Investigation.
Hai Feng: Writing-review and editing, Supervision, Validation, Methodology.
All authors approved the final version of the manuscript to be published.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Beijing Municipal Science and Technology Program “Unveiling the List and Appointing the Leader” Military Order Pledge Project (BMSTP-ULAL-MOPP, Grant No. Z231100004823006).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request and with permission from Beijing Friendship Hospital, Capital Medical University, Beijing, China.
