Abstract
Background
Pulmonary embolism (PE) is a leading cause of cardiovascular mortality. The albumin platelet product (APP), a novel biomarker integrating nutritional and inflammatory status, is associated with adverse outcomes in various diseases. However, its prognostic value for mortality in patients with PE remains unclear.
Methods
This retrospective cohort study included 2355 patients with PE from the MIMIC-IV database. The primary exposure was APP, calculated as (albumin [g/dL]×platelet [K/µL]) / 100. The primary endpoints were all-cause mortality at 28 days, 90 days, and 365 days. The association between APP and mortality was assessed using restricted cubic splines (RCS), Kaplan-Meier survival curves, and multivariable Cox proportional hazards models. Subgroup analyses were performed to evaluate consistency across patient characteristics.
Results
Using the tertile method, participants were stratified into three groups based on their APP levels (Tertile 1, Tertile 2, and Tertile 3), with Tertile1 designated as the reference group for comparative analyses against Tertile 2 and Tertile 3. RCS analysis revealed a statistically significant nonlinear relationship between APP levels and PE mortality (p for nonlinearity < 0.001). Kaplan-Meier survival analysis demonstrated a significantly higher risk of death among patients with lower APP levels (p < 0.01). Multivariable Cox proportional hazards regression models indicated an independent association between low APP levels and increased mortality risk at 28 days, 90 days, and 365 days, both before and after adjustment for potential confounders. Subgroup analyses showed no significant interaction between APP levels and mortality risk in most subgroups examined.
Conclusions
In patients diagnosed with PE, lower APP levels are significantly and independently associated with an increased risk of all-cause mortality. APP serves as a clinically useful predictor of long-term adverse outcomes in this population.
Introduction
Pulmonary embolism (PE), defined as the obstruction of pulmonary arteries by thrombo-embolism originating predominantly from deep veins, is a critical cardiovascular emergency 1 and a leading cause of cardiovascular death worldwide.2,3 Accurate risk stratification is crucial for optimizing treatment decisions and improving outcomes. While clinical scores like the Pulmonary Embolism Severity Index (PESI) exist, their complexity limits widespread use in emergency and intensive care settings.4–6 There is therefore a pressing need for simple and reliable biomarkers to aid in the early prognostic assessment of PE patients, particularly in the Intensive Care Unit (ICU).
Inflammation plays a pivotal role in the pathogenesis and progression of PE.7,8 Clot formation intensifies inflammatory responses, and dysregulated inflammatory cascades can provoke thrombosis primarily through stimulation of leukocytes, platelets, and endothelial cells. Endothelial damage in PE serves as a principal catalyst for immune system engagement. 9 On the one hand, platelets are recognized as specialized first-line inflammatory markers that interact with leukocytes and endothelial cells, thereby influencing the function of inflammatory components within these cells. 10 On the one hand, albumin, as a negative acute-phase reactant protein, also serves as an important inflammatory marker. 11 Research confirms that low serum albumin significantly increases thrombosis risk. 12 Furthermore, the study by Omar HR et al demonstrates an association between serum albumin and PE severity: for every 1 gm/dL decrease in albumin levels, the likelihood of massive PE increases by 75%. 13 Therefore, serum albumin levels and platelet counts can serve as reliable indicators of inflammatory changes and may be closely associated with PE.
The albumin platelet product (APP), as a novel biomarker integrating serum albumin levels and platelet counts, offers convenient measurement and cost-effectiveness. It has demonstrated significant prognostic relevance in patients with chronic liver disease and hepatocellular carcinoma.14,15 However, its association with clinical outcomes in PE patients is not well established. This study aimed to investigate the association between admission APP levels and all-cause mortality in patients with PE.
Methods
Data Selection
This retrospective cohort study utilized the Medical Information Mart for Intensive Care IV (MIMIC-IV, version 3.1) database, which contains de-identified clinical data from intensive care units at Beth Israel Deaconess Medical Center collected between 2008 and 2022.16,17 Author Gang Wu obtained data access by completing the Collaborative Institutional Training Initiative (CITI Program) certification (certification number: 62773844) and secured the necessary permissions to use the MIMIC-IV database. The study protocol was granted exempt status by the institutional review board due to the de-identified nature of the data.
This study identified a cohort of 5793 patients diagnosed with PE through International Classification of Diseases (ICD-9 and ICD-10) diagnostic codes who underwent non-consecutive ICU admissions within the MIMIC-IV database. The criteria for inclusion in this research were as follows: 1) the patient's first admission to the intensive care unit (ICU) within the database; 2) an age of 18 years or older; and 3) a minimum ICU stay of 24 h. We excluded patients who had incomplete information on albumin levels and platelet counts. The final analytical cohort comprised 2355 PE patients (Figure 1).
Data Collection
Data collection involved extracting baseline patient characteristics using PostgreSQL. These characteristics included demographic information (age, gender, body mass index (BMI), and ethnicity), vital signs (heart rate (HR), systolic blood pressure (SBP), diastolic blood pressure (DBP), mean arterial pressure (MAP), respiratory rate (RR), oxygen saturation, and temperature), laboratory test results (hemoglobin, erythrocyte pressure, erythrocyte distribution width, leukocytes, neutrophils, monocytes, lymphocytes, platelets, international normalized ratio (INR), prothrombin time (PT), creatinine, urea nitrogen, albumin, potassium, sodium, chloride, calcium, anion gap, and glucose), and information on ventilation usage. Additionally, Acute Physiology Score III (SAPS II) and Sequential Organ Failure Assessment (SOFA) Scores at the time of admission, as well as comorbidity information from the MIMIC-IV database, were included. Key outcome indicators comprised length of stay (LOS) in the hospital and ICU, as well as 28-, 90-, and 365-day mortality rates. Notably, all laboratory results were obtained from data recorded within the first 24 h following the patient's admission to the ICU.
APP was the primary exposure factor in this study. APP was determined according to the equation: APP = albumin × platelet / 100, 14 where albumin represents the serum albumin level (measured in g/dL), and platelet refers to blood platelet counts (expressed in K/µL). Using the tertile method, participants were divided into three groups (Tertile1, Tertile2, and Tertile3) based on their APP values, with Tertile1 designated as the reference group for comparative analysis against Tertile2 and Tertile3. This stratification approach allowed for a detailed assessment of the association between APP and other variables or health-related outcomes.
Hypertension, diabetes mellitus, coronary artery disease (CAD), congestive heart failure (CHF), cerebrovascular disease, chronic pulmonary disease, CKD, liver disease, tumor, and obesity were defined using International Classification of Diseases, 10th Revision (ICD-10) and ICD-9 codes. The follow-up period commenced on the admission date and concluded when the endpoints of interest occurred.
Endpoints of Interest
The primary outcome of this study was 28-day, 90-day and 365-day mortality rates. Mortality data were sourced from the MIMIC-IV database. We calculated the survival time for each patient by subtracting the admit-time date from the date of death.
Statistical Analysis
Continuous variables are expressed as either the mean ± standard deviation (Mean ± SD) or median and quartiles [M (Q1, Q3)], and group comparisons are made using the Mann–Whitney U test or Student's t-test, depending on data distribution. Categorical variables are presented as frequencies and percentages [N (%)], with group differences assessed via the chi-square test or Fisher's exact test.
The restricted cubic splines (RCS) model was applied to evaluate potential linear or nonlinear associations between APP levels and mortality outcomes. In the case of nonlinear relationships, potential threshold effects are identified by systematically testing all possible inflection points and selecting the most likely values. A piecewise Cox proportional hazards regression model is then applied, stratified by the identified inflection points, to investigate the association between the APP index and all-cause mortality.
The Kaplan-Meier survival analysis was used to estimate cumulative 28-day, 90-day, and 365-day mortality rates across groups stratified by APP levels.
Moreover, cox proportional hazards models were used to calculate the hazard ratio and 95% confidence interval (CI) for the association between APP levels and mortality in PE across groups, adjusting for multiple variables. The selection of confounding variables was primarily based on clinical relevance and univariate analysis results (p < 0.1). Model 1 represents an unadjusted analysis, while Model 2 adjusts for sex, age, BMI, and race. Model 3 includes the variables from Model 2 and further adjusts for factors such as HR, SBP, DBP, MBP, RR, WBC, hematocrit, hemoglobin, RDW, INR, anion gap, potassium, calcium, chloride, creatinine, HBP, CAD, CKD, liver disease, tumors, and obesity. HRs were calculated, and results are presented with 95% CIs.
Subgroup analyses were conducted to evaluate the robustness of the prognostic association between APP levels and outcomes across predefined subgroups. These included stratification by gender (female vs male), age (≤65 vs > 65 years), BMI (≤30 vs > 30 kg/m²), and clinical comorbidities (hypertension, CAD, CKD, CHF, and tumor). Likelihood ratio tests were applied to assess interaction effects between APP levels and stratification variables.
All statistical analyses were conducted using SPSS version 26.0 (IBM Corporation, USA) and R version 4.4.1 (R Foundation for Statistical Computing), with statistical significance defined as a two-sided p-value < 0.05.
Result
This study included 2355 patients with severe pulmonary embolism. The overall cohort had a median age of 65.70 years (IQR 54.51–75.58), 48.87% (n = 1151) were female, and mortality rates were 15.5%, 23.78%, and 34.1% at 28, 90, and 365 days, respectively. Stratification by APP tertiles yielded groups of 777 (Tertile 1), 801 (Tertile 2), and 777 (Tertile 3) patients. Baseline characteristics are detailed in Table 1. Significant differences (p < 0.05) were observed across tertiles for most demographics, vital signs, laboratory parameters, illness severity scores, comorbidities (hypertension, coronary artery disease, chronic kidney disease, liver disease, tumor, obesity), and all mortality outcomes. In contrast, no significant differences were found in oxygen saturation (SpO₂), temperature, sodium, blood glucose, or the prevalence of diabetes mellitus, chronic heart failure, cerebrovascular disease, chronic pulmonary disease, and mechanical ventilation use.
Baseline Characteristics of Participants Stratified by APP Tertile Levels.

SD: standard deviation, M: median, Q1: 1st quartile, Q3: 3st quartile, BMI: body mass index, HR: heart rate, bmp: beats per minute, SBP: systolic blood pressure, DBP: diastolic blood pressure, MBP: mean blood pressure, RR: respiratory rate, RDW: red cell distribution width, WBC: white blood cell, INR: international normalized ratio, PT: prothrombin time, BUN: blood urea nitrogen, SAPSII: Simplified Acute Physiology Score II, SOFA: Sequential Organ Failure Assessment, CAD: coronary artery disease, CHF: chronic heart failure, CKD: chronic kidney disease, LOS: length of stay, ICU: intensive care unit, APP: Albumin Platelet Product.
The association between APP levels and mortality risk at 28, 90, and 365 days was investigated using RCS models, which revealed a significant nonlinear relationship (p for nonlinearity < 0.001) at all time points (Figure 2). To quantify this association, we employed piecewise Cox regression models, which identified distinct risk thresholds ( Table S1 ). The risk for 28-day and 365-day mortality decreased sharply as APP levels increased up to a threshold of 0.69. Below this threshold, each 1-unit increase in the APP index was associated with a substantial 81% reduction in 28-day mortality risk (HR = 0.19, 95%CI: 0.08–0.46, p < 0.001) and a 74% reduction in 365-day mortality risk (HR = 0.26, 95%CI: 0.14–0.49, p < 0.001). However, above this threshold (APP ≥0.69), no significant protective association was observed for 28-day (HR = 1.14, 95%CI: 0.90–1.44, p = 0.276) or 365-day mortality (HR = 1.10, 95%CI: 0.94–1.30, p = 0.241), indicating a plateauing of the protective effect. For 90-day mortality, the protective effect was even more pronounced at lower APP levels, with a threshold at 0.39. Below this point, each 1-unit increase in APP was associated with a dramatic 96% reduction in risk (HR = 0.04, 95%CI: 0.01–0.30, p = 0.002), while no association was seen above it (HR = 1.02, 95%CI: 0.85–1.22, p = 0.863). The superiority of this piecewise model over a linear one was confirmed by likelihood ratio tests (p < 0.001).
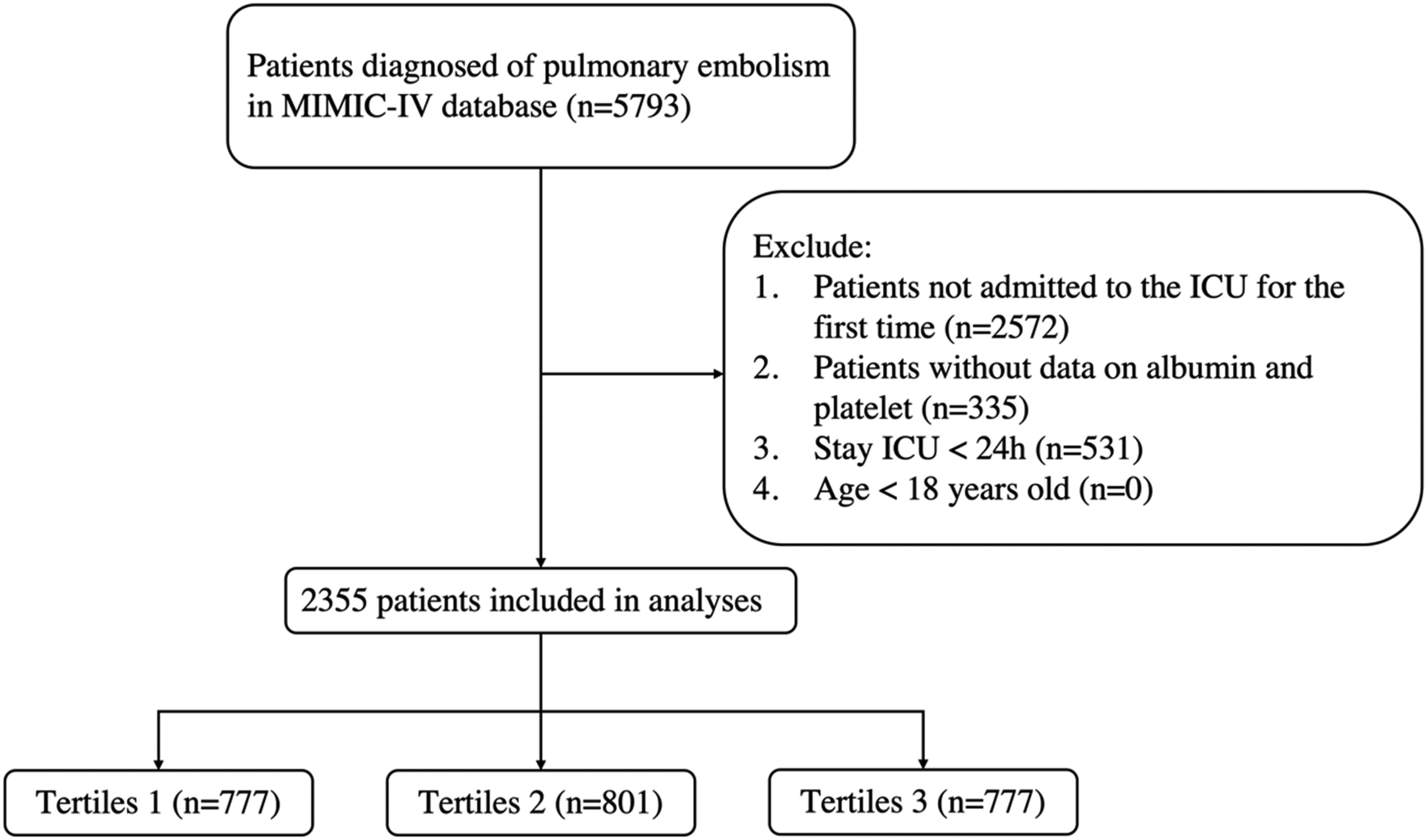
Flowchart of the study.
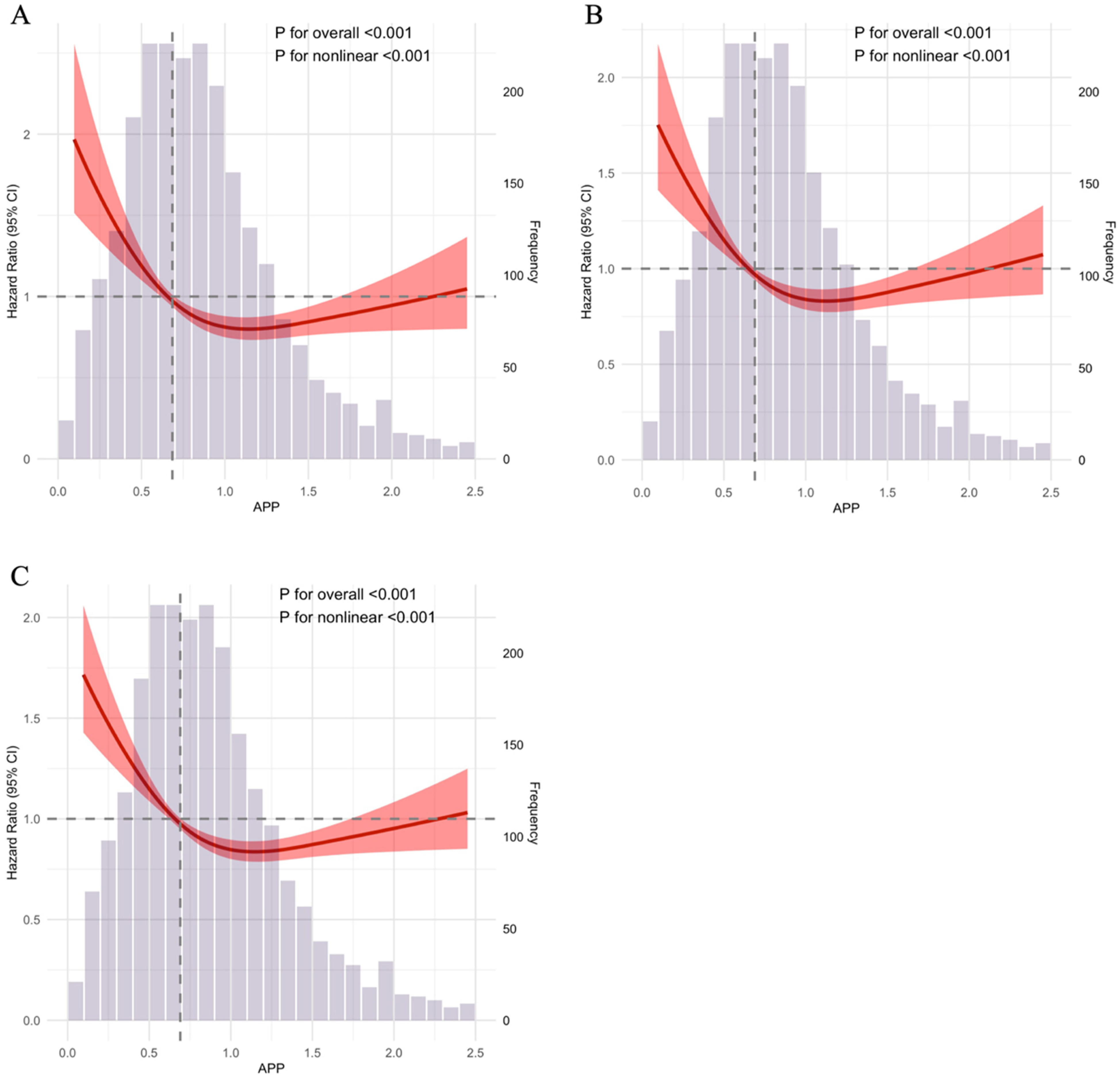
Restricted cubic spline regression analysis for 28-day (A), 90-day (B) and 365-day (C) all-cause mortality. APP: Albumin Platelet Product.
Primary Outcomes
As illustrated in Figure 3, Kaplan-Meier survival analysis was employed to assess mortality rates among pulmonary embolism patients at 28, 90, and 365 days across different study groups. The results demonstrate that patients with lower APP levels exhibit a significantly elevated mortality risk (p < 0.01).

Kaplan-meier survival analysis curves for 28-day, 90-day and 365-day all-cause mortality.
As detailed in Table 2, we constructed three sequential Cox proportional hazards models to evaluate the independent association of APP levels with 28-day, 90-day, and 365-day mortality in patients with pulmonary embolism. The multivariable analysis incorporated both statistically significant variables from univariable screening (p < 0.05) and clinically relevant covariates. After comprehensive adjustment for demographic, physiological, and comorbidity factors in Model 3, higher APP levels remained significantly associated with reduced mortality risk across all time points. Specifically, compared to the lowest APP tertile (Tertile 1), patients in Tertile 2 showed a 24% lower risk of 28-day mortality (HR = 0.76, 95%CI: 0.59-0.98), while those in Tertile 3 had a 30% risk reduction (HR = 0.70, 95%CI: 0.54-0.91). This protective trend was consistently observed at 90 days and 365 days, with risk reductions ranging from 19% to 29% in the highest APP tertile relative to the lowest. The graded inverse relationship, where progressively lower APP levels predicted incrementally higher mortality, was consistently observed in all models, underscoring APP as a robust and independent prognostic marker in PE.
Cox Proportional Hazard Ratios for PE at 28 Days, 90 Days and 365 Days.

HR: Hazard Ratio, CI: Confidence Interval.
Model1: Crude.
Model2: Adjust: gender, age, BMI, race.
Model3: Adjust: gender, age, BMI, race, HR, SBP, DBP, MBP, RR, WBC, hematocrit, hemoglobin, RDW, INR, anion gap, potassium, calcium, chloride, creatinine, HBP, CAD, CKD, liver disease, tumor, obesity.
As shown in Figure 4, to investigate the relationship between APP levels and mortality in patients with pulmonary embolism, we evaluated the association between APP levels and all-cause mortality at 28 days, 90 days, and 365 days across subgroups, including sex, age, BMI, HBP, CAD, CKD, CHF, and tumor. The results showed that at 28 days, no significant interaction was observed between APP and the other subgroups (interaction p-values: 0.168–0.929) except for the tumor (interaction p-values = 0.012) group; at 90 days, no significant interaction (interaction p-values: 0.081–0.736) was observed except for the BMI (interaction p-values = 0.036) and tumor (interaction p-values = 0.035) groups; and at 365 days, no significant interaction (interaction p-values: 0.098–0.994) was observed except for the BMI (interaction p-values = 0.016)group.

Forest plots of hazard ratios for the primary endpoint in different subgroups. (A) 28-day mortality, (B) 90-day mortality, (C) 365-day mortality. BMI: Body Mass index, HBP: High Blood Pressure, CAD: Coronary Artery Disease, CKD: Chronic Kidney Disease, CHF: Chronic Heart Failure.
Discussion
This retrospective analysis of 2355 PE patients revealed a significant nonlinear relationship between the Albumin-Platelet Product (APP) and all-cause mortality, with inflection points at 0.69 for 28-day and 365-day and 0.39 for 90-day mortality. Patients with lower APP levels at admission had significantly poorer survival rates at 28, 90, and 365 days. This inverse association remained robust after extensive multivariable adjustment and was consistent across subgroups, establishing APP as a novel and independent prognostic biomarker in PE. Our findings underscore the clinical value of routine APP tracking for risk stratification and timely intervention to improve outcomes.
The threshold effect observed at APP = 0.69 (for 28-day and 365-day mortality) and APP = 0.39 (for 90-day mortality) represents a critical inflection point below which patients experience substantially elevated mortality risk. Below these thresholds, each unit increase in APP confers profound protective effects (HR = 0.19 for 28-day mortality and HR = 0.04 for 90-day mortality, both p < 0.01), while above these values, the protective effect plateaus with no additional benefit from further increases (HR≈1.0, p > 0.05 for all time points). The likelihood ratio test confirmed that the piecewise model was superior to standard linear regression (p < 0.001), indicating that this threshold approach better captured the true nature of the APP-mortality relationship. This pattern likely reflects a minimum protective threshold below which physiological reserve capacity becomes critically insufficient. When APP falls below the threshold, it signals inadequate functional reserve to counteract PE-induced endothelial damage, coagulopathy, and systemic inflammatory responses.3,18 The nonlinear dose-response relationship identified through restricted cubic spline analysis suggests that once adequate reserve capacity is achieved above the threshold, further increases provide diminishing marginal benefit, a phenomenon well-described as the saturation effect in biomarker research.19,20 Notably, the median APP values across tertiles were 0.45 (Q1 0.31, Q3 0.54) for Tertile 1, 0.80 (Q1 0.71, Q3 0.89) for Tertile 2, and 1.28 (Q1 1.12, Q3 1.57) for Tertile 3 (p < 0.001), clearly demonstrating distinct risk stratification around the identified thresholds. The different threshold values for 90-day versus 28/365-day mortality (0.39 vs 0.69) may reflect temporal variations in pathophysiological demands. 21 The lower 90-day threshold might capture a subacute phase where sustained metabolic and reparative processes require moderate but prolonged reserve capacity, whereas the higher threshold at 28 and 365 days may represent acute critical illness and long-term complications requiring greater immediate physiological reserves to overcome the initial hemodynamic insult and prevent late sequelae. 22 This temporal heterogeneity in threshold values provides insights into the dynamic nature of PE pathophysiology and suggests that risk stratification strategies may need to be tailored to different prognostic timeframes.
PE is a serious condition characterized by blockage of the pulmonary arteries, often leading to an increased afterload on the right ventricle and potential right ventricular failure. 3 The cascade from PE to right ventricular dysfunction creates multiple pathways affecting APP components. Contemporary understanding emphasizes the role of venous congestion and inflammatory activation in driving both hypoalbuminemia and platelet consumption. 13 Right heart failure causes plasma volume expansion, diluting the plasma pool and thereby leading to decline in albumin levels. 11 Our findings corroborate this mechanism, with albumin levels of 3.20 ± 0.79 mmol/L in Tertile 1 significantly lower than 3.60 ± 0.69 mmol/L in Tertile 2 and 3.89 ± 0.61 mmol/L in Tertile 3 (p < 0.001). Additionally, PE and right heart failure reduce protein intake, thereby slowing albumin synthesis, which ultimately lowers albumin levels. 11
Recent mechanistic studies have elucidated the bidirectional relationship between albumin and thrombosis. Beyond its role as a negative acute-phase reactant, albumin exhibits direct anticoagulant properties through multiple pathways: binding of factor Xa, inhibition of von Willebrand factor-mediated platelet adhesion, and scavenging of reactive oxygen species that promote platelet activation.23–26 Serum albumin reduces thrombus formation through multiple mechanisms: inhibiting platelet aggregation, exerting anti-inflammatory and antioxidant effects, stabilizing endothelial cells, and serving as a molecular carrier.27,28 Hypoalbuminemia compromises these protective functions, consequently elevating the risk of coronary atherosclerosis and thrombosis. This relationship is clinically evidenced by the increased incidence of restenosis following coronary stent implantation. 29 More significantly, earlier research has directly established an association between serum albumin levels and PE severity: each 1 gm/dL decrease in albumin concentration corresponds to a 75% increase in the likelihood of massive PE. 13
The platelet component of APP reflects complex hemostatic perturbations in PE. he gradient in platelet counts across tertiles was striking: median values of 137 K/µL (Q1 99, Q3 170) in Tertile 1, 221 K/µL (Q1 193, Q3 259) in Tertile 2, and 339 K/µL (Q1 284, Q3 429) in Tertile 3 (p < 0.001), demonstrating the profound platelet consumption characteristic of severe PE. Recent investigations published in the Journal of Thrombosis and Haemostasis have demonstrated that platelet indices in PE extend beyond simple numerical changes. 30 The phenomenon of “platelet exhaustion” in severe PE involves not only quantitative depletion but also qualitative dysfunction, with circulating platelets showing reduced aggregation responses despite increased baseline activation markers. 31 Multiple studies have found that platelet counts were significantly lower in the PE group compared to the control group, with excessive platelet consumption postulated as the underlying cause of thrombocytopenia.32,33 Furthermore, Varol et al observed that thrombocytopenia in PE patients was accompanied by a significant increase in mean platelet volume (MPV). 33 Elevated MPV, serving as a biomarker of platelet activation, exhibits greater metabolic and enzymatic activity than smaller platelets, consequently possessing heightened prothrombotic potential.34–36
The inflammatory milieu in PE creates a self-perpetuating cycle affecting both APP components. Supporting this inflammatory paradigm, our data revealed significantly elevated white blood cell counts in lower APP tertiles: median 10.70 K/µL in Tertile 1 versus 11.10 K/µL in Tertile 2 and 12.40 K/µL in Tertile 3 (p < 0.001), with corresponding increases in neutrophils (7.79 vs 8.20 vs 8.97 K/µL, p < 0.001). Novel research has identified specific inflammatory mediators that simultaneously suppress albumin synthesis and promote platelet activation, including interleukin-6, tumor necrosis factor-α, and damage-associated molecular patterns released from hypoxic lung tissue.37,38 Research indicates significantly elevated C-reactive protein (CRP) concentrations in acute PE. 39 CRP remains persistently elevated throughout the inflammatory phase until resolution of inflammation. 40 This acute inflammatory response promotes P-selectin expression on activated platelets, thereby amplifying thrombus formation. 41 Our study observed heightened mortality risk in PE patients with thrombocytopenia, likely attributable to concurrent platelet activation and exacerbated thrombus formation during thrombocytopenic states.
The APP, a novel composite biomarker of fibrosis integrating serum albumin levels and platelet counts, has demonstrated significant prognostic relevance in patients with chronic liver diseases and hepatocellular carcinoma.14,15 The clinical utility of APP is further evidenced by its association with disease severity markers: patients in Tertile 1 exhibited higher SAPS II scores (median 38 vs 33 vs 34, p < 0.001) and SOFA scores (median 4 vs 2 vs 2, p < 0.001), along with prolonged hospital length of stay (median 11.94 days vs 10.07 vs 10.61, p < 0.001). The pathophysiological basis linking reduced APP to increased mortality in PE involves interconnected pathways: Albumin, functioning as the primary endogenous inhibitor of platelet aggregation (EIPA) in plasma, suppresses platelet aggregation and serotonin (5-HT) release by binding free arachidonic acid (AA) and modulating its metabolic pathway.42,43 Consequently, hypoalbuminemia promotes aberrant platelet activation, accelerates aggregation, and significantly increases thrombotic risk. 44 Furthermore, activated platelets expressing P-selectin bind leukocytes, forming platelet-leukocyte aggregates.45,46 This interaction potentiates endothelial inflammation and endothelial glycocalyx (eGCX) shedding. 47 Crucially, eGCX integrity is vital for regulating vascular permeability, inflammation, and elasticity 48 ; its damage impairs the endothelial barrier, leading to increased albumin extravasation. This creates a self-perpetuating cycle where diminished albumin levels further exacerbate platelet hyperactivity and vascular dysfunction.
This study has several strengths. First, it targets a clinically important but commonly neglected cohort with PE. The simplicity and accessibility of the APP index further enhance its practical utility. APP can be calculated from routine laboratory tests (albumin and platelet counts) without additional blood draws or specialized equipment. The identified threshold values (APP <0.69 for 28-day and 365-day mortality, < 0.39 for 90-day mortality) provide actionable cutoffs for clinical decision-making. APP could be integrated into existing risk stratification tools such as PESI or sPESI to enhance risk discrimination and guide therapeutic intensity. Serial APP monitoring during hospitalization may signal clinical deterioration or improvement, supporting timely adjustments in patient management. Implementation through electronic health record systems with automated calculation would facilitate real-time risk assessment without increasing clinician workload. This work provides the first exploratory assessment of the APP index's potential association with mortality risk in PE patients, contributing new clinical information and serving as a foundation for future validation studies. Secondly, the data analyzed in the current study were derived from a nationally representative MIMIC database survey that selected 2355 eligible participants with pulmonary embolism using a sophisticated multi-stage probability sampling method. Large sample size ensures strong representativeness. Finally, the study examines the relationship between the APP index and all-cause mortality in PE patients, adding to the existing knowledge in this area.
However, several limitations should be acknowledged. First, this study is fundamentally exploratory in nature, aimed at establishing whether APP has an independent association with PE mortality. APP is not proposed as a standalone clinical decision tool ready for immediate implementation, but rather as a readily available marker warranting validation in larger, multicenter cohorts where its incremental value over existing risk scores (such as sPESI or ESC risk stratification) can be rigorously evaluated through analyses including decision curve analysis and reclassification metrics. Second, participants were predominantly from the United States, potentially limiting generalizability to other populations. Third, although we adjusted for multiple confounders, residual confounding remains possible. Fourth, the robustness of our findings could be further strengthened; due to constraints in data availability, we were unable to perform sensitivity analyses, such as using multiple imputation for missing data, to fully assess the impact of our methodological assumptions on the primary results. Fifth, APP was assessed only at baseline; dynamic changes during follow-up weren't evaluated. Future research should investigate whether serial APP measurements improve prognostic accuracy and whether APP-guided therapeutic interventions improve outcomes. Finally, absence of genetic data prevented Mendelian randomization analysis.
Conclusions
Our findings indicate that the APP index is not only cost-effective but also demonstrates potential clinical utility in assessing all-cause mortality risk among individuals with PE. Consequently, we suggest incorporating APP index monitoring into clinical practice as a practical tool for risk stratification.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251401575 - Supplemental material for Prognostic Significance of the Albumin Platelet Product for Mortality Risk in Pulmonary Embolism: A Retrospective Analysis Based on the MIMIC-IV Database
Supplemental material, sj-docx-1-cat-10.1177_10760296251401575 for Prognostic Significance of the Albumin Platelet Product for Mortality Risk in Pulmonary Embolism: A Retrospective Analysis Based on the MIMIC-IV Database by Gang Wu, Huanya Ke, Zijia Tong and Zhengjun Shen in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-2-cat-10.1177_10760296251401575 - Supplemental material for Prognostic Significance of the Albumin Platelet Product for Mortality Risk in Pulmonary Embolism: A Retrospective Analysis Based on the MIMIC-IV Database
Supplemental material, sj-docx-2-cat-10.1177_10760296251401575 for Prognostic Significance of the Albumin Platelet Product for Mortality Risk in Pulmonary Embolism: A Retrospective Analysis Based on the MIMIC-IV Database by Gang Wu, Huanya Ke, Zijia Tong and Zhengjun Shen in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
We would like to sincerely thank the Free Statistics team for their indispensable technical assistance and provision of tools that are necessary for data analysis and visualization.
Ethics statement
Research involving human subjects received ethical approval after thorough review from both the Massachusetts Institute of Technology and the Beth Israel Deaconess Medical Center. The studies adhered to local regulations and institutional guidelines. Participants provided their written informed consent to take part in this research. Additionally, written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
