Abstract
Acute pulmonary embolism (PE) remains a life-threatening condition. A critical therapeutic dilemma exists for a significant proportion of patients at high bleeding risk(approximately 30%-50%). For these patients, anticoagulation is the mainstay, but the use of conventional agents (warfarin, DOACs, heparins) is severely limited by the risk of major bleeding, creating a substantial unmet medical need. Abelacimab addresses this unmet need through a novel mechanism: dual inhibition of factor XI (FXI) and activated FXI (FXIa). This paradigm shift uncouples potent antithrombotic efficacy from hemostasis, selectively suppressing contact pathway-driven thrombosis while preserving tissue factor-mediated hemostasis. We synthesize its unique mechanism of action, pharmacokinetic profile, and existing clinical evidence to evaluate its role in addressing this clinical gap. Abelacimab's mechanism selectively inhibits the contact activation pathway—crucially overactive in the RBC-rich thrombi characteristic of PE—while preserving tissue factor-mediated physiological hemostasis. Clinically, abelacimab demonstrated significantly lower rates of major bleeding or clinically relevant non-major bleeding compared to rivaroxaban in atrial fibrillation prophylaxis (AZALEA-TIMI 71 trial) and to enoxaparin in post-arthroplasty venous thromboembolism prevention (ANT-005 trial), establishing a favorable clinical safety profile. Its favorable pharmacokinetics (long half-life supporting monthly subcutaneous administration, with minimal renal/hepatic clearance) further simplify management and enhance its suitability for patients with PE. However, current evidence is extrapolated from AF/VTE prophylaxis studies, lacking direct human data for acute PE management. Urgent phase III trials (eg, PE-FOCUS) are therefore essential to validate efficacy and safety in acute PE, potentially redefining the anticoagulation paradigm.
Introduction
Pulmonary embolism (PE) encompasses a spectrum of clinical syndromes characterized by the obstruction of the pulmonary artery or its branches by endogenous or exogenous embolism. 1 This obstruction leads to pulmonary circulatory and right heart dysfunction, the most prevalent and severe manifestation of which is pulmonary thromboembolism. Acute PE, a leading cause of cardiovascular mortality, poses a significant public health burden due to high morbidity, mortality, and resource utilization.2,3 Although recent studies indicate declining acute PE mortality rates in China and Western nations,4–6 certain demographics remain vulnerable, underscoring disparities in access to optimal care.7–11 These disparities likely reflect limited access to advanced therapies in resource-poor settings.
Current PE guidelines stratify patients by risk (high/intermediate/low) to guide interventions such as anticoagulation, systemic thrombolysis, catheter-directed therapy, or embolectomy. However, a critical therapeutic challenge remains for a significant proportion (30-50%) of intermediate- and high-risk patients who are ineligible for systemic thrombolysis due to absolute contraindications (eg, active bleeding, recent stroke, intracranial pathology).12–14 For these high-risk individuals, anticoagulation remains the sole therapeutic option, but presents a formidable dilemma: balancing thromboprophylaxis against bleeding risk. Current clinical guidelines recommend anticoagulation therapy with oral agents or heparins as the primary treatment for suspected acute PE. While direct oral anticoagulants (DOACs) demonstrate improved safety profiles compared to Vitamin K antagonists (VKAs), they still significantly increase major bleeding risk by 30%-40% in cancer patients relative to low-molecular-weight heparin (LMWH),15–17 severely limiting their utility in this high-thrombosis-risk subgroup, complicating treatment, and rendering them unsuitable for long-term outpatient care. Consequently, a pressing unmet need exists for safer anticoagulants that provide potent thromboprophylaxis without compromising hemostasis in this high-risk, thrombolysis-ineligible cohort.
Abelacimab, a dual inhibitor of coagulation factor XI (FXI) and its activated form (FXIa), stabilizes FXI in an inactive zymogen state while concurrently blocking FXIa activity.18,19 This unique mechanism selectively suppresses contact pathway-driven thrombosis while preserving tissue factor (TF)-mediated hemostasis. Preliminary clinical trials have demonstrated the safety and efficacy of abelacimab in patients with atrial fibrillation (AF), 20 for VTE prevention, 21 and in individuals requiring extended dual antiplatelet therapy. 22 However, its potential in acute PE—particularly for thrombolysis-ineligible patients—remains unexplored, representing a pivotal evidence gap.
Therefore, this review aims to bridge this gap by critically examining the translational potential of abelacimab for acute PE. We analyze its unique mechanism of action, pharmacokinetic(PK) characteristics, and convincing clinical safety evidence to evaluate its role in addressing the critical challenge of bleeding-free anticoagulation. By doing this, we provide a potential therapeutic solution for acute PE patients at elevated bleeding risk and offer evidence-based support for ongoing clinical trials.
Mechanism and PK of Abelacimab
Abelacimab is a novel targeted anticoagulant that selectively inhibits coagulation FXI/XIa. Its dual mechanism of action suppresses pathological thrombus amplification while preserving physiological hemostasis. This approach overcomes a major limitation of conventional anticoagulants by decoupling antithrombotic efficacy from bleeding risk. Furthermore, abelacimab's distinct PK properties and favorable bleeding safety profile may offer significant advantages for acute PE patients at elevated bleeding risk, including elderly populations and those requiring dual antithrombotic therapy. As depicted in Table 1.
Comparison of the Mechanism and Properties of Abelacimab with Conventional Anticoagulants.

Abbreviations: DOACs, direct oral anticoagulants; LMWH, low-molecular-weight heparin; eGFR, estimated glomerular filtration rate.
Targeted Dual Mechanism of FXI/XIa
Abelacimab is a monoclonal antibody that specifically targets both coagulation FXI and its activated form (FXIa), exerting anticoagulant effects by inhibiting FXI activation and/or FXIa activity. FXI, initially characterized as a precursor of plasma S1 serine protease, 27 is a homodimeric glycoprotein composed of two identical 80 kDa subunits. Each subunit contains four apple domains (A1-A4) and a single protease domain. This dimeric structure facilitates simultaneous binding to platelet surfaces via the apple domains and to substrate factor IX (FIX) via the protease domain, thereby amplifying the coagulation cascade. 28
The coagulation cascade (Figure 1) is traditionally conceptualized as comprising two initiation pathways: the extrinsic pathway, activated by TF, and the intrinsic pathway, initiated by contact activation. These converge into a common path, catalyzing fibrinogen (Fg) conversion to fibrin (Fn). FXI activation to FXIa occurs via thrombin or activated factor XII (FXIIa). Crucially, physiological hemostasis driven by the TF pathway functions largely independently of FXI/XIa. This pathway initiates at vascular injury sites through the binding of FVII/VIIa to exposed TF in the vascular adventitia, forming the VIIa-TF complex. This complex directly activates factor X (FX), generating FXa, which catalyzes the thrombin burst and fibrin deposition. A minor fraction of thrombin activates FXI through platelet GPIb receptors, creating a feedback loop essential for thrombus propagation.29–32 In the contact activation pathway (eg, triggered by artificial surfaces), FXIIa activates FXI, amplifying coagulation and promoting thrombosis.
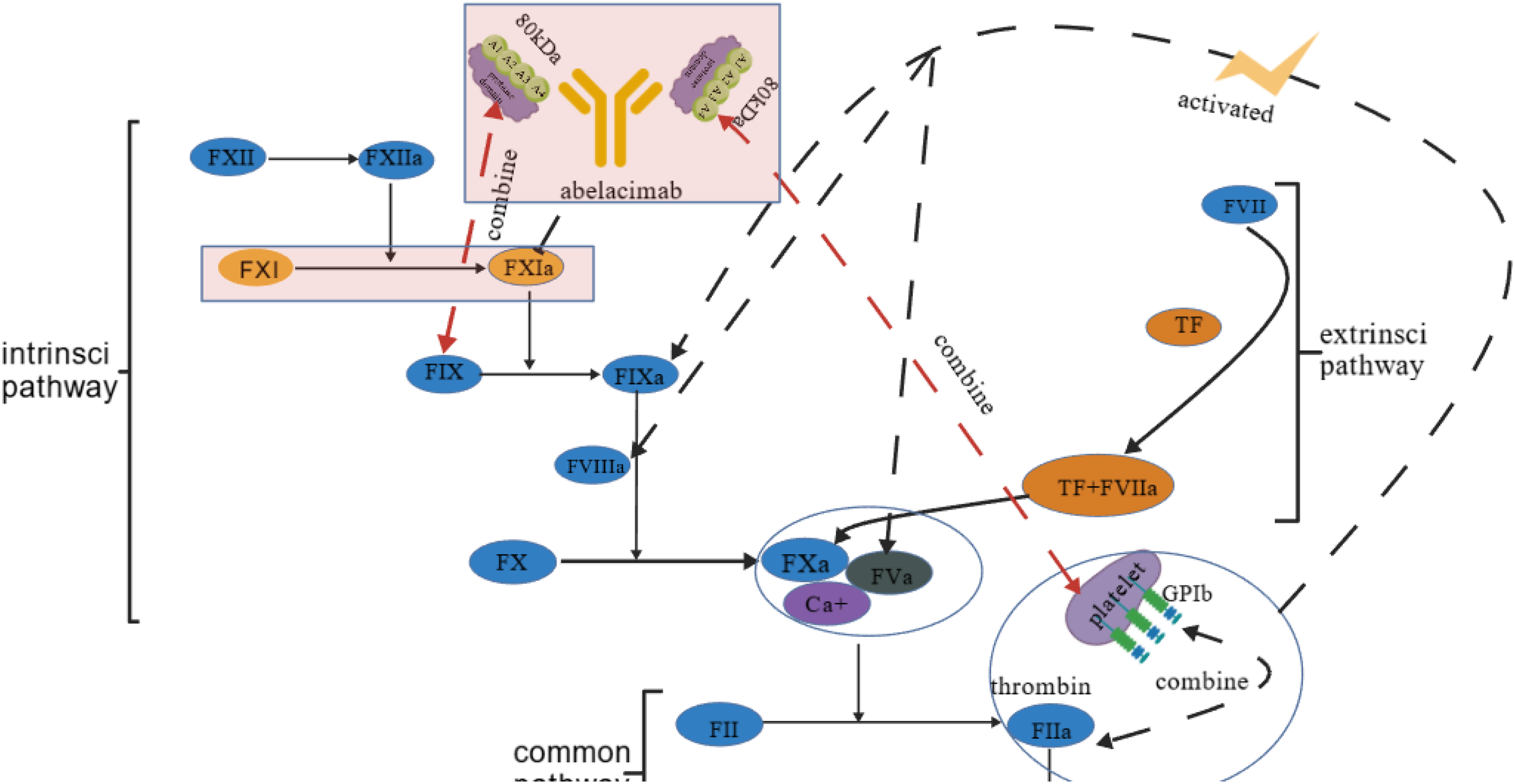
Blood Coagulation Enzyme Processes. Created with BioGDP.com. 33
This differential involvement indicates that FXI/XIa plays a more prominent role in thrombosis than in general hemostasis. Abelacimab binds the inactive FXI zymogen and inhibits FXIa, stabilizing FXI in its inactive conformation and preventing activation by FXIIa or thrombin. 19 Its mechanism features a unique dual activity against both FXI and FXIa, effectively uncoupling pathological thrombosis from physiological hemostasis.
Pathological Mechanisms of VTE and PE
VTE encompasses deep vein thrombosis (DVT) and PE, representing manifestations of the same disease continuum with shared pathophysiology and comparable thrombotic mechanisms. PE, characterized by obstruction of pulmonary arterial blood flow primarily due to embolization of thrombi originating from lower extremity deep veins, is associated with significant morbidity and mortality.34–39 Approximately 70%-80% of PE cases arise from lower extremity DVT, with 50% of patients with proximal DVT developing concurrent PE. 34 Thrombogenesis in VTE is classically described by Virchow's triad – endothelial injury, hypercoagulability, and venous stasis – which retains fundamental clinical relevance. Endothelial dysfunction disrupts key vascular homeostatic processes, including the regulation of tone, inflammation, and coagulation.40,41 Consequently, endothelial homeostasis is essential for general vascular health. While inflammation is not a part of Virchow's triad, it is now understood to play a significant role in thrombosis. 42 Inflammatory mediators cause platelet activation and endothelial damage. 43 Activated platelets then bind to neutrophils, promoting the release of neutrophil extracellular traps (NETs) and the auto-activation of FXI into FXIa, which initiates the coagulation cascade and promotes thrombus aggregation.
The fundamental mechanism of venous thrombosis is characterized by the unchecked activation of the pathological coagulation cascade within the vasculature. But abelacimab can specifically target FXIa in the contact activation route and prevent it from activating FIX, 44 which stops thrombin from being produced. This unique approach to blocking the contact pathway may have less of an effect on hemostatic function than conventional anticoagulants (such as direct inhibitors of FIIa or FXa). This lowers the risk of bleeding, which is important for thrombus prevention and offers a new approach to treating PE.
Thrombus Composition Heterogeneity
PE thrombosis primarily originates from fragmented lower-extremity DVTs. The excessive activation of the coagulation cascade within the venous system causes a large amount of thrombin to be produced, which encourages the development of a fibrin network entangled with red blood cells, 45 ultimately leading to the production of red thrombi. Because of their loose structure and propensity for tail detachment, these thrombi may cause PE. They have different compositional characteristics compared to arterial thrombi. While arterial thrombi are platelet-rich (31%) and fibrin-dense (43%) due to high shear stress, PE thrombi retain venous characteristics: predominantly red blood cells (RBCs, 63%) with loosely organized fibrin strands. 46 This unique architecture generates extensive procoagulant surfaces and is a critical determinant of therapeutic response.
The Rationale for FXI/XIa Inhibition in Acute PE
The RBC-rich, fibrin-based composition of PE thrombi creates a pathological environment uniquely susceptible to FXI/XIa-driven thrombogenesis and impaired fibrinolysis, thereby presenting a compelling therapeutic target for abelacimab.
The extensive surface of exposed phosphatidylserine on erythrocyte membranes within PE thrombi provides potent anionic phospholipid surfaces that drive contact pathway activation by potentiating FXII autoactivation 47 and subsequent cleavage of FXI to FXIa.30,31,46 This FXIa-dependent amplification cascade is a key mechanism for localized thrombin burst generation within the pulmonary vasculature.
And most critically, this thrombin burst has a dual detrimental effect: it enhances thrombin-activatable fibrinolysis inhibitor (TAFI) generation, 48 suppressing plasmin-mediated clot dissolution within the pulmonary vasculature (Figure 2). TAFI is a potent inhibitor of the endogenous fibrinolytic, systemsynthesized as a zymogen in the liver, inhibits fibrinolysis upon activation: (1) Plasminogen (PLG) and tissue plasminogen activator (t-PA) bind to fibrin via lysine-binding sites and nascent C-terminal lysine residues, forming a ternary complex that promotes plasmin generation; (2) Plasmin cleaves fibrin, generating new C-terminal lysines that further amplify plasmin formation; (3) The thrombin-thrombomodulin complex activates TAFI, which potently inhibits fibrinolysis by cleaving these essential C-terminal lysine residues on fibrin, thereby disrupting the positive feedback loop for plasmin generation.49–52 The suppression of this fibrinolytic activity is a major reason why large PE thrombi persist and cause ongoing obstruction.

PE Blood Clots Promote the Activation of Contact Pathway and Abelacimab Blocks Coagulation Circulation. Abelacimab Inhibits FXIa, Reducing TAFI Generation and Restoring Fibrinolysis:49–52 ①PLG/t-PA Bind to the Surface of Fibrin; ② PL Generated; ③ Activated TAFI Inhibits Fibrinolysis. t-PA: tissue-type plasminogen activator, PLG: plasminogen, PL: plasmin, TM: thrombomodulin. Created with BioGDP.com. 33
Abelacimab disrupts this vicious cycle through a precision mechanism highly relevant to acute PE: 53
By directly inhibiting FXIa, it dampens the contact pathway-amplified thrombin generation specifically at the site of the RBC-rich thrombus. By attenuating the thrombin burst, it subsequently reduces the activation of TAFI. The reduction in active TAFI levels restores the endogenous fibrinolytic capacity by preserving the C-terminal lysine residues on fibrin, allowing the body's own t-PA and plasminogen to effectively bind and degrade the thrombus.48,52 This mechanism—suppressing pathological thrombosis while promoting endogenous fibrinolysis—is a paradigm shift from conventional anticoagulants (eg, FXa or FIIa inhibitors) that solely inhibit clot formation and may even impair fibrinolysis. The anticoagulant efficacy of abelacimab has been demonstrated in preclinical studies. In murine arterial thrombosis models (FeCl -induced carotid injury), abelacimab dose-dependently inhibited occlusive thrombus formation in FXI-deficient mice reconstituted with human FXI/XIa when achieving a molar excess over FXI/XIa, concurrently prolonging activated partial thromboplastin time (APTT). 19 Crucially, in baboon venous graft thrombosis models, abelacimab prevented downstream propagation of platelet and fibrin deposits within collagen/TF-coated grafts. 54 Although direct evidence from PE-specific thrombus resolution models remains limited, this consistent demonstration of enhanced fibrinolysis in venous-type thrombi, structurally analogous to RBC-rich PE emboli, provides a compelling translational rationale for abelacimab's therapeutic efficacy in pulmonary vascular beds.
PK Characteristics of Abelacimab
Abelacimab is a monoclonal antibody that is entirely human and can be injected intravenously (IV) or subcutaneously. In healthy volunteers (Study ANT-003), peak plasma concentrations occurred 1.75-2 h post-IV infusion across doses. The antibody exhibits a prolonged terminal elimination t½. Dose proportionality was observed for the area under the curve (AUC) within the 30-150 mg range. Notably, obese subjects (BMI ≥35 kg/m²) demonstrated 30%-45% reductions in initial concentration and AUC, likely attributable to increased volume of distribution. 23 These PK properties support the feasibility of monthly SC dosing, potentially enhancing adherence and facilitating long-term anticoagulation management.
Abelacimab undergoes metabolism primarily via endocytosis and proteolytic degradation by the reticuloendothelial system (eg, hepatic Kupffer cells), independent of the cytochrome P450 system, hepatocyte uptake, or biliary excretion. Phase I data indicated no significant differences in maximum concentration(Cmax), AUC, or t½ between healthy volunteers and renally/hepatically impaired patients following a single 150 mg IV dose (P > .05), suggesting no dose adjustment is necessary.23,24 No drug-induced liver injury or clinically significant elevations in liver enzymes or bilirubin were observed. Its non-hepatic clearance pathway minimizes accumulation risks in cirrhosis—particularly with portal hypertension or coagulopathy—compared to conventional anticoagulants (eg, warfarin, rivaroxaban). 25
Abelacimab, as a macromolecule, has a poor renal clearance of the total clearance and is unaffected by glomerular filtration rate, indicating favorable safety in end-stage renal disease (ESRD). 26 24 ESRD patients receiving heparin-free dialysis participated in a small phase 2, randomized, double-blind study to assess the pharmacological and clinical effects of a single predialysis dosage of the targeted FXI/XIa analogue xisomab 3G 3 (0.25 or 0.5 mg/kg) in comparison to a placebo. 55 The findings demonstrated that there were no adverse drug reactions following the administration of xisomab 3G 3, and the frequency of occlusion events necessitating the replacement of hemodialysis circuits was minimal. These findings suggest that abelacimab may have little effect on the kidneys of hemodialysis patients, given its macromolecular structure, 56 though model limitations warrant cautious extrapolation to abelacimab. The abelacimab hemodialysis phase II trial is now underway, and the outcomes of the subsequent clinical trial are anticipated.
Clinically Validated
Abelacimab has demonstrated significant bleeding risk reduction while maintaining antithrombotic efficacy across multiple clinical settings, including AF (AZALEA-TIMI 71 trial), 57 VTE prophylaxis (ANT-005 trial), 58 and special populations. Clinical evidence consistently shows reductions in the international society on thrombosis and haemostasis (ISTH) major bleeding or clinically relevant non-major bleeding (CRNMB) without compromising efficacy, with no reported fatal bleeding events. The studies’ outcomes are detailed in Table 2.
Efficacy and Safety Outcomes Between Abelacimab and Other Anticoagulants.

Abbreviations: ISTH, International Society on Thrombosis and Haemostasis; CRNMB, clinically relevant non-major bleeding; AF, atrial fibrillation; VTE, venous thromboembolism; SC, subcutaneous; IV, intravenous injection.
Clinical Safety and Efficacy Data: Implications for Acute PE
In the AZALEA-TIMI 71 trial, abelacimab significantly reduced the risk of ISTH major bleeding or CRNMB compared to rivaroxaban, including >80% reduction in gastrointestinal bleeding risk. 57 This profound reduction in bleeding, particularly at a site of major concern in anticoagulated patients, provides a strong safety signal that is directly relevant to acute PE management. For the high-bleeding-risk PE population—which often includes the elderly, those with cancer, or individuals with comorbidities—this safety advantage could be transformative. Furthermore, despite 55.8% undergoing invasive procedures within 29 days post-dosing, the rate of perioperative bleeding occurred in only 0.9% of patients. 60 This is of great significance for pulmonary embolism patients who may require urgent interventions, such as catheter-directed therapy or thrombectomy.
Beyond its safety profile, the ANT-005 trial in patients undergoing total knee arthroplasty also demonstrated abelacimab's potent efficacy in preventing venous thrombosis. 58 Compared to enoxaparin, a single IV of abelacimab (30 mg, 75 mg, and 150 mg) dramatically decreased the incidence of VTE, establishing proof-of-concept for its powerful antithrombotic effect in the venous circulation. Importantly, this efficacy was achieved without an increase in major or CRNMB bleeding. For acute PE, this suggests that abelacimab could achieve therapeutic anticoagulation without exacerbating bleeding risk, a critical balance often difficult to attain with current therapies. The extended half-life enabling single-dose efficacy also hints at a potential for simplified treatment regimens, which could facilitate earlier discharge and transition to outpatient care for stable PE patients.
Analyses of special populations further bolster its potential for acute PE. Both dosages of abelacimab significantly decreased the risk of major bleeding or CRNMB when compared to rivaroxaban, according to an age-stratified analysis from the AZALEA-TIMI 71 trial, which revealed consistent bleeding risk reduction with abelacimab versus rivaroxaban across age groups, with greater absolute benefit in patients ≥75 years. 59 This is directly applicable to the elderly PE population, which is disproportionately affected by bleeding complications from conventional anticoagulants. Moreover, abelacimab significantly reduced bleeding risk compared to rivaroxaban in patients requiring concomitant antiplatelet therapy (APT), highlighting a key safety advantage for PE patients with underlying coronary artery disease. 22
This suggests that abelacimab could be a safer option for patients requiring dual pathway inhibition. Abelacimab is currently being evaluated in pivotal trials for cancer-associated VTE (NCT05171049, NCT05171075),61,62 a patient group with exceptionally high rates of both thrombosis and bleeding. Positive results here would directly support its use in cancer-associated PE, a major clinical challenge. Collectively, while direct evidence from acute PE studies is awaited, the consistent demonstration of potent antithrombotic efficacy coupled with an exceptionally favorable bleeding profile across these indications provides a compelling rationale for its investigation in acute PE populations. This safety-efficacy profile positions abelacimab as a promising candidate to address the critical unmet need in high-bleeding-risk PE patients.
Translational Pathway and Future Perspectives for Abelacimab in Acute PE
A paucity of clinical evidence specific to acute PE now limits the application of abelacimab, a dual FXI/XIa inhibitor. It is crucial to close this gap by conducting specialized trials like PE-FOCUS to confirm its effectiveness and safety in PE populations. Prospects for the future depend on its capacity to revolutionize PE care through easier dosage, decreased risk of bleeding, altered course of CTEPH, and enhanced cost-effectiveness.
Clinical Trials Design
The absence of dedicated trials in acute PE remains a pivotal evidence gap for abelacimab, despite its established anticoagulant efficacy in VTE prophylaxis and atrial fibrillation. To definitively establish its role in acute PE, a future phase III trial must incorporate design elements that are specifically tailored to address the unique pathophysiology and clinical challenges of this condition.
An ideal PE-focused trial should employ a primary efficacy endpoint that captures thrombus reduction—such as objective regression of thrombus burden assessed by imaging—which would be mechanistically aligned with abelacimab's potential pro-fibrinolytic properties and its capacity to alleviate PE-specific vascular obstruction. On the safety side, the use of ISTH major bleeding as the primary safety endpoint is essential, as it reflects the critical importance of hemorrhagic risk in treatment decision-making for acute PE.
Furthermore, prospective enrichment for high-risk and clinically complex subgroups—including patients with cancer-associated PE, renal impairment, and advanced age—would ensure that the trial generates meaningful evidence for populations most vulnerable to both thrombotic and bleeding complications. Such a design would not only evaluate antithrombotic efficacy but also delineate the benefit-risk profile of abelacimab across the heterogeneous spectrum of acute PE.
High-risk Populations and Bleeding Safety
The evaluation of abelacimab in high-bleeding-risk acute PE populations requires focused attention on clinically relevant subgroups. Patients with cancer-associated PE represent a critical population due to their elevated thrombosis and bleeding risks. Insights from studies of FXI/XIa inhibitors, including bleeding safety signals from an in vitro hemodialysis model, 63 help support the biologic rationale for abelacimab's use in cancer-related PE, though direct evidence remains investigational. The significantly reduced bleeding rates observed with abelacimab in the AZALEA-TIMI 71 trial, particularly the >80% reduction in gastrointestinal bleeding, provide a compelling rationale for its investigation in this vulnerable subgroup. Similarly, elderly PE patients—who experience disproportionate bleeding complications with conventional anticoagulants—may derive particular benefit, as evidenced by consistent bleeding risk reduction across age groups in AZALEA-TIMI 71, with enhanced absolute benefit in those ≥75 years. While abelacimab's minimal renal clearance and low hepatic metabolism suggest theoretical advantages for patients with renal or hepatic impairment, PE-specific safety and efficacy data in these populations are awaited. Future studies should prioritize these high-risk PE subgroups to address unmet needs in those most vulnerable to bleeding complications.
Potential for Disease Modification and Long-Term Safety Considerations
Beyond its immediate antithrombotic effect, abelacimab may offer transformative benefits in altering acute PE sequelae, particularly chronic thromboembolic pulmonary hypertension (CTEPH) and recurrent PE. By suppressing FXIa-mediated TAFI generation, abelacimab restores endogenous fibrinolytic capacity, thereby promoting acute thrombus resolution and potentially preventing inadequate clot dissolution and organization. Thus, abelacimab represents a paradigm shift from mere recurrence prevention towards active disease modification.
However, this potential requires validation in dedicated long-term studies. While current guidelines recommend long-term anticoagulation for secondary prevention,1,64 no clinical trials have specifically evaluated abelacimab's efficacy in preventing CTEPH or recurrent acute PE. Therefore, the PE-FOCUS trial still needs to validate the CTEPH prophylactic effect in order to evaluate long-term safety. Additionally, regarding immunogenicity, phase I studies detected no treatment-related anti-drug antibodies with either intravenous or subcutaneous administration; 23 nevertheless, long-term monitoring (>1 year) in larger PE cohorts remains warranted to fully characterize its safety profile in this population.
Practical Implementation and Health Economic Considerations
Abelacimab's pharmacological profile offers significant advantages for implementing streamlined care pathways in acute PE management. Its monthly subcutaneous dosing regimen could simplify the transition from inpatient to outpatient care, potentially facilitating earlier hospital discharge for stabilized intermediate-risk patients while maintaining effective anticoagulation. This dosing strategy is particularly valuable for elderly patients and those with renal impairment who face challenges with frequent dosing or monitoring of conventional anticoagulants. Furthermore, abelacimab's hemostasis-sparing mechanism may reduce bleeding-related complications that often necessitate rehospitalization and increase resource utilization.
From a health economic perspective, the reduced bleeding rates demonstrated in the AZALEA-TIMI 71 trial—which showed substantial cost reductions per patient (>$50 000 in the US; €392-693/£7347 in EU4/UK) and gains of 1.3-1.5 quality-adjusted life years65,66—suggest similar or greater benefits might be achievable in acute PE management where bleeding events carry significant clinical and economic consequences. Although anticipated pricing in China suggests favorable cost-effectiveness, 67 local reimbursement policies and interprovincial variations must be considered. Future empirical research should confirm whether the combination of simplified management and reduced bleeding complications translates into meaningful cost savings within Chinese PE treatment pathways.
Summary
Abelacimab is a novel, fully human monoclonal antibody targeting FXI/XIa, represents a promising therapeutic strategy for addressing the critical unmet need of bleeding-free anticoagulation in high-risk patients with acute PE. Its mechanism of action involves potent inhibition of the contact activation pathway, while preserving physiological hemostasis—a key safety advantage. This confers a significant safety advantage that may position abelacimab as an optimal alternative for patients ineligible for thrombolysis or those with heightened bleeding risk. Additionally, its extended half-life supports once-monthly subcutaneous administration, potentially simplifying long-term management and facilitating outpatient care transitions. Preclinical data also suggest potential disease-modifying effects through enhanced fibrinolysis, possibly attenuating the progression to chronic thromboembolic pulmonary hypertension. However, current evidence supporting its efficacy in acute PE is primarily derived from in vitro models and molecular studies. Robust clinical validation, particularly through trials such as PE-FOCUS, remains essential to substantiate its translation into effective acute PE management.
Footnotes
Authors’ Contributions
ML and JW conceived the review. ML collected literatures and wrote the initial draft, while JW revised and proofread. Both authors collaboratively decided on the final manuscript, revised drafts, and approved the final version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
