Abstract
Purpose:
To compare the incidence of 90-day venous thromboembolism (VTE) in obese critically ill medical patients receiving VTE chemoprophylaxis with nonobese patients of similar illness severity. We also identified other VTE risk factors.
Methods:
Eligible patients spent ≥24 hours in an intensive care unit between November 2007 and November 2013 and received VTE chemoprophylaxis within 48 hours of admission. The primary outcome was 90-day VTE.
Results:
A total of 11 111 patients were evaluated, of which 1732 obese and 1831 nonobese patients were enrolled with mean BMIs of 38.9 ± 9.2 kg/m2 and 24.5 ± 3.1 kg/m2 and mean Acute Physiology and Chronic Health Evaluation II scores of 28.4 ± 11.8 and 26.6 ± 11.7, respectively. The rate of 90-day VTE for the total cohort, obese, and nonobese patients was 6.5%, 7.5%, and 5.5%, respectively. Obese patients were more likely to develop VTE compared with nonobese patients (odds ratio [OR]: 1.41; 95% confidence interval [CI]: 1.03 -1.93). Other risk factors significantly associated with 90-day VTE included prior VTE (OR: 3.93; 95% CI: 1.83-8.48), trauma with surgery in the previous 30 days (OR: 3.70; 95% CI: 1.39-9.86), central venous catheters (OR: 2.64; 95% CI: 1.87-3.72), surgery within 90 days (OR: 2.40; 95% CI: 1.61-3.58), mechanical ventilation (OR: 1.94; 95% CI: 1.39-2.71), male sex (OR: 1.55; 95% CI: 1.13-2.14), and increasing age using 1-year increments (OR: 1.02; 95% CI: 1.01 -1.03).
Conclusions:
The rate of VTE in critically ill medical patients remains high despite standard chemoprophylaxis. Obesity is among 8 risk factors independently associated with 90-day VTE.
Keywords
Introduction
Venous thromboembolism (VTE) includes deep vein thrombosis (DVT) and pulmonary embolism (PE) and is associated with increased patient morbidity, mortality, hospital length of stay, and cost. 1 –7 The reported rates of hospital-acquired VTE vary widely from 0.5% to 60% depending on patient characteristics, method of diagnosis, and follow-up duration. 8 –10 For critically ill medical patients, the rate of VTE is estimated at 10% to 31%. 1 –4,11 Accurate assessment of thrombotic risk and appropriate chemoprophylaxis is paramount to the reduction of overall hospital-associated VTE. However, many hospitalized patients may fail to receive VTE chemoprophylaxis despite guideline recommendations. 12 –14 Irrespective of chemoprophylaxis, VTE may still occur in up to 15% of critically ill patients depending on a multitude of factors including etiology of critical illness. 4,15 –18
Known risk factors contributing to the development of VTE in hospitalized medical patients include critical illness, obesity, prior VTE, advanced age, cancer, central venous catheterization (CVC), heart failure, recent surgery, myocardial infarction (MI), respiratory failure, estrogen treatment, stroke, and prolonged immobilization. 1,2,10,19 –23 Among hospitalized medical patients, the Padua Risk score allots a single point for obesity though does not take critical illness into consideration. 19 Former studies have also shown a greater incidence of VTE in obese patients. 22 On the other hand, the Immediate Management of Patients with Rupture: Open Versus Endovascular Repair (IMPROVE) investigators did not find an increased risk of VTE in obese medical patients, 23 although neither of these studies followed an entirely critically ill patient population. Currently, no validated risk assessment models stratify VTE risk among critically ill medical patients. 21 Only 5 randomized controlled studies have examined chemoprophylaxis among critically ill patients, 16,24 –27 and even less is known about the risk of VTE in obese critically ill patients. 2,28
Chemoprophylaxis with unfractionated heparin (UFH) or low-molecular-weight heparin (LMWH) is effective in preventing VTE in most patients. 2,21 In addition, chemoprophylaxis has shown to decrease mortality when compared with mechanical or no VTE prophylaxis 29 and therefore should be utilized in critically ill patients who lack a contraindication. 2,30
Current guidelines recommend critically ill patients receive either LMWH or UFH (Grade 2C), and although some evidence supports considering dose adjustments for VTE prophylaxis among the obese, 31 little guidance exists. 30 In nonorthopedic surgical patients, guidelines suggest consulting with a pharmacist for dosing recommendations in patients who may require higher doses of UFH or LMWH (eg, obese patients) but otherwise following the manufacturer’s recommendations. 32
A recent study evaluated risk factors associated with incident VTE during intensive care unit (ICU) stay among critically ill patients receiving anticoagulant thromboprophylaxis. 18 However, to our knowledge, ours is the first study reporting risk of 90-day VTE among critically ill adults. The objective of this study was to compare the rate of VTE at 90 days among critically ill obese medical patients receiving standard VTE chemoprophylaxis to the rate in nonobese patients with similar illness severity. We also sought to elucidate other risk factors associated with 90-day VTE. We hypothesized the incidence of 90-day VTE would be higher in critically ill obese medical patients receiving standard VTE chemoprophylaxis.
Materials and Methods
Study Design
This 6-year (November 1, 2007, to November 30, 2013) retrospective cohort study was conducted at Intermountain Medical Center, a 452-bed tertiary care university-affiliated hospital. All data were retrieved from Intermountain Healthcare’s Enterprise Data Warehouse (EDW), a central data repository of all Intermountain Healthcare’s patient encounter data, including inpatient, outpatient, pharmacy, clinic, and imaging data. No research funding was provided. The protocol included a waiver of informed consent and was approved by the Intermountain Medical Center Institutional Review Board.
Study Population
Patients with a body mass index (BMI) of 17 to 90 kg/m2 spending ≥24 hours in 1 of 2 ICUs (the Shock Trauma ICU-STICU or Respiratory ICU-RICU) were eligible for inclusion. Typical admitting diagnoses of patients in the STICU and RICU include severe sepsis and septic shock, decompensated liver disease, drug overdoses, chronic obstructive pulmonary disease and asthma exacerbations, other causes of respiratory failure, diabetic ketoacidosis, and trauma requiring mechanical ventilation or surgery, among others. However, patients presenting after trauma were excluded. Obese and nonobese patients had BMI ranges ≥30 to 90 kg/m2 and 17 to 29.9 kg/m2, respectively. Patients had to receive their first dose of appropriate VTE chemoprophylaxis within 48 hours of hospital admission. Appropriate VTE prophylaxis was defined as UFH 5000 units subcutaneously (SQ) every 8 or 12 hours, enoxaparin 30 mg SQ every 12 hours, or enoxaparin 40 mg SQ daily. The majority of patients received either UFH 5000 units SQ every 8 hours or enoxaparin 40 mg SQ daily, as these are the protocolized VTE prophylactic regimens in our medical population. Patients were excluded if they were diagnosed with a PE or DVT within 48 hours of admission, had a hypercoagulable disorder (eg, factor V Leiden mutation, antiphospholipid antibody syndrome, and protein C and S deficiencies) as identified by a representative International Classification of Diseases, Ninth Revision (ICD-9) code, received therapeutic anticoagulation as an outpatient or within 48 hours of admission (eg, therapeutic UFH, therapeutic enoxaparin, therapeutic fondaparinux, warfarin, dabigatran, rivaroxaban, or apixaban), were admitted due to trauma, or underwent surgery within 7 days prior to or following admission. Surgery was defined as an admission into the operating room through the operating room management system (ORMS), which is a comprehensive repository of all surgical procedures. Additional exclusion criteria were pregnancy, age younger than 18 years, and enoxaparin doses other than those specified a priori.
We collected baseline demographics and variables previously shown to increase the risk of VTE. The binary logistic regression model included age, sex, Charlson Comorbidity Index score, trauma and/or surgery 30 days prior to admission (but not within 7 days of admission as these patients were excluded), any prior or active cancer, prior VTE, congestive heart failure (CHF), MI, cerebrovascular accident, active or historical hormone therapy (either hormone replacement therapy or oral contraceptives) as defined by an outpatient prescription provided and filled using insurance, surgery in the 90 days following admission, mechanical ventilator support at any time during ICU stay, CVC in place during hospitalization, Acute Physiology and Chronic Health Evaluation (APACHE) II score upon hospital admission, and BMI.
Outcomes and Variables
The primary outcome, the difference in 90-day VTE between obese and nonobese patients, was defined as DVT or PE using natural language processing (NLP) and electronic charts utilizing ICD-9 codes for DVT and PE, confirming the NLP methodology (Appendix 1). Secondary outcomes included 30-day and in-hospital VTE, 90-day mortality, defined as in-hospital mortality documented in the EDW or through the Utah State Death Registry, hospital and ICU length of stay, and the incidence of major bleeding and heparin-induced thrombocytopenia (HIT). Major bleeding was defined as a composite end point of bleeding into a critical organ or organ space, including intracranial, intraspinal, intraocular, intra-articular, peritoneal, and pericardial, or other overt bleeding with transfusion of ≥2 units packed red blood cells identified by requisite ICD-9 code or upon electronic medical record interrogation for administration of blood products as we have previously described, 33–34 which is derived from the definition of major bleeding proposed by Schulman and Kearon. 35 Heparin-induced thrombocytopenia was defined as a positive serotonin release assay characterized as release of serotonin from target platelets in the presence of patient serum and low-dose (0.1 units/mL) heparin of >20%, together with inhibition of release (<20%) with high-dose (100 units/mL) heparin. All outcomes were analyzed using a binomial BMI cutoff of ≥30 kg/m2 unless otherwise specified. Additional analyses of the primary outcome used BMI as a continuous variable with 1-point increments. We included 16 other risk factors as covariates so that we could best determine the influence of obesity on 90-day VTE, independent of other risk factors.
Statistical Analysis
For our primary outcome, binary logistic regression analysis was used to evaluate the influence of obesity status on 90-day VTE. Covariates were included to assess for risk factors that would significantly influence 90-day VTE. For the logistic regression, we calculated P values and odds ratios (ORs) with 95% confidence intervals (CIs). The binary logistic regression was repeated with actual BMI scores inserted as the main predictor in place of the usual binomial BMI cutoff of ≥30 kg/m2 to determine whether a change in BMI influenced 90-day VTE across the entire range.
For secondary outcomes we used χ2 tests for independence to compare the proportion of VTE during ICU stay and within 30 days between obese and nonobese patients. This univariate analysis was performed separately from the multivariable analysis used for the primary outcome of 90-day VTE. T tests were used to compare continuous measures for uncorrected baseline characteristics between obese and nonobese patients. Where applicable, descriptive statistics (eg, means, medians, standard deviations) are presented. P values <.05 were considered statistically significant. Statistical analyses were performed using SPSS for Windows, Version 19.0 (SPSS Inc, Chicago, Illinois).
Results
Patients
There were 11 111 evaluable patients admitted to either the STICU or RICU between November 1, 2007, and November 30, 2013. The final cohort included 1732 obese and 1831 nonobese patients after applying exclusion criteria (Figure 1). The mean age of the population was 53 ± 18.6 years, and 47.8% were male. The mean BMI of the entire cohort was 31.5 ± 9.9 kg/m2, and the mean APACHE II score was 27.5 ± 11.8. The mean BMIs of the obese and nonobese cohorts were 38.9 ± 9.2 kg/m2 and 24.5 ± 3.1 kg/m2, and the mean APACHE II scores on hospital day 1 were 28.4 ± 11.8 and 26.6 ± 11.6, respectively. The average time to first administration of VTE chemoprophylaxis was 10 ± 10.2 hours. Baseline patient characteristics and VTE risk factors studied are found in Table 1. The obese cohort consisted of older female patients, with a higher proportion of CHF. In addition, the obese cohort was more likely to receive invasive mechanical ventilation and surgery in the 90 days following admission when compared with the nonobese cohort.
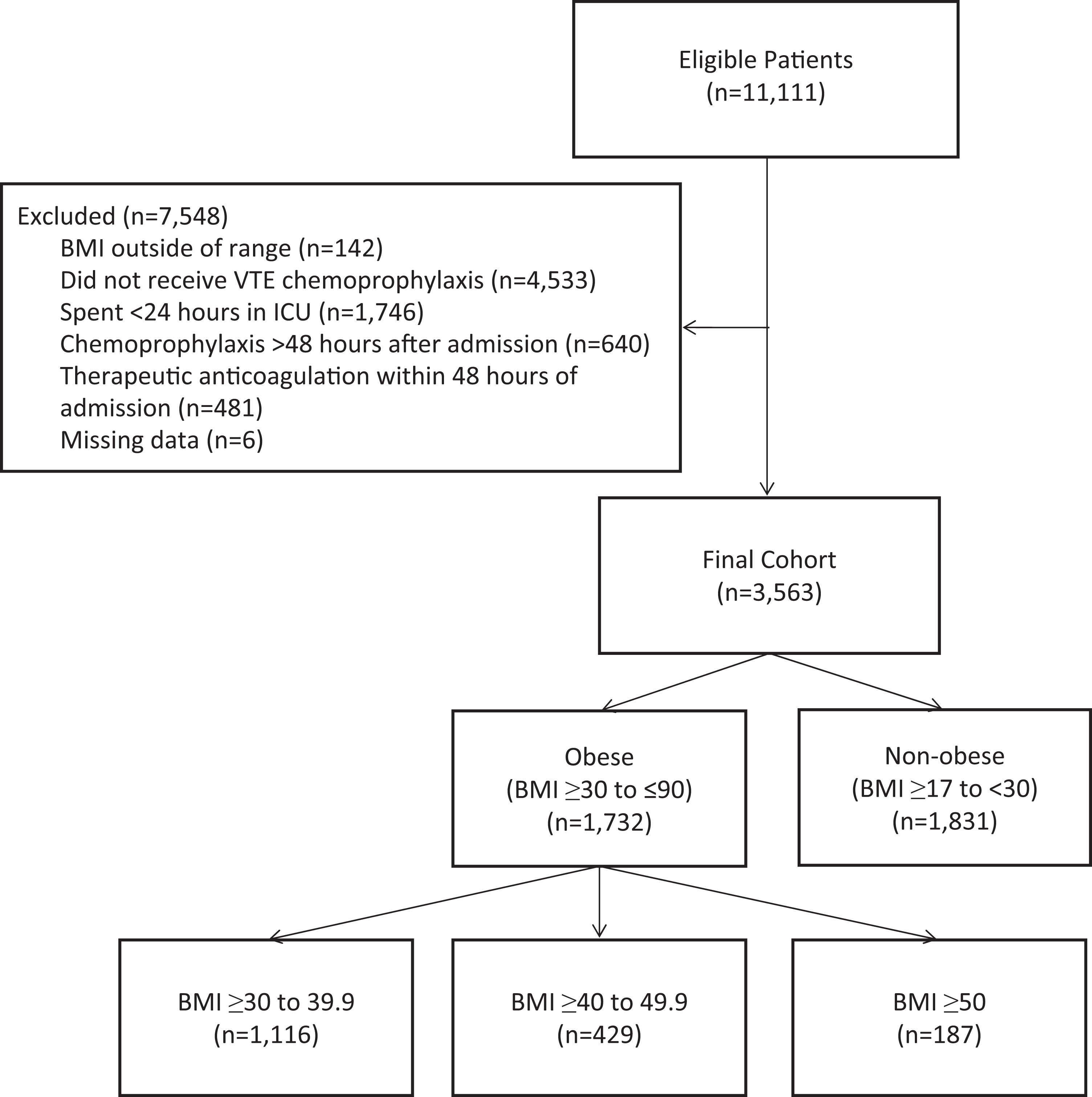
Creation of the study cohorts after applying inclusion and exclusion criteria. BMI indicates body mass index; VTE, venous thromboembolism; ICU, intensive care unit.
Baseline Demographics and VTE Risk Factors.
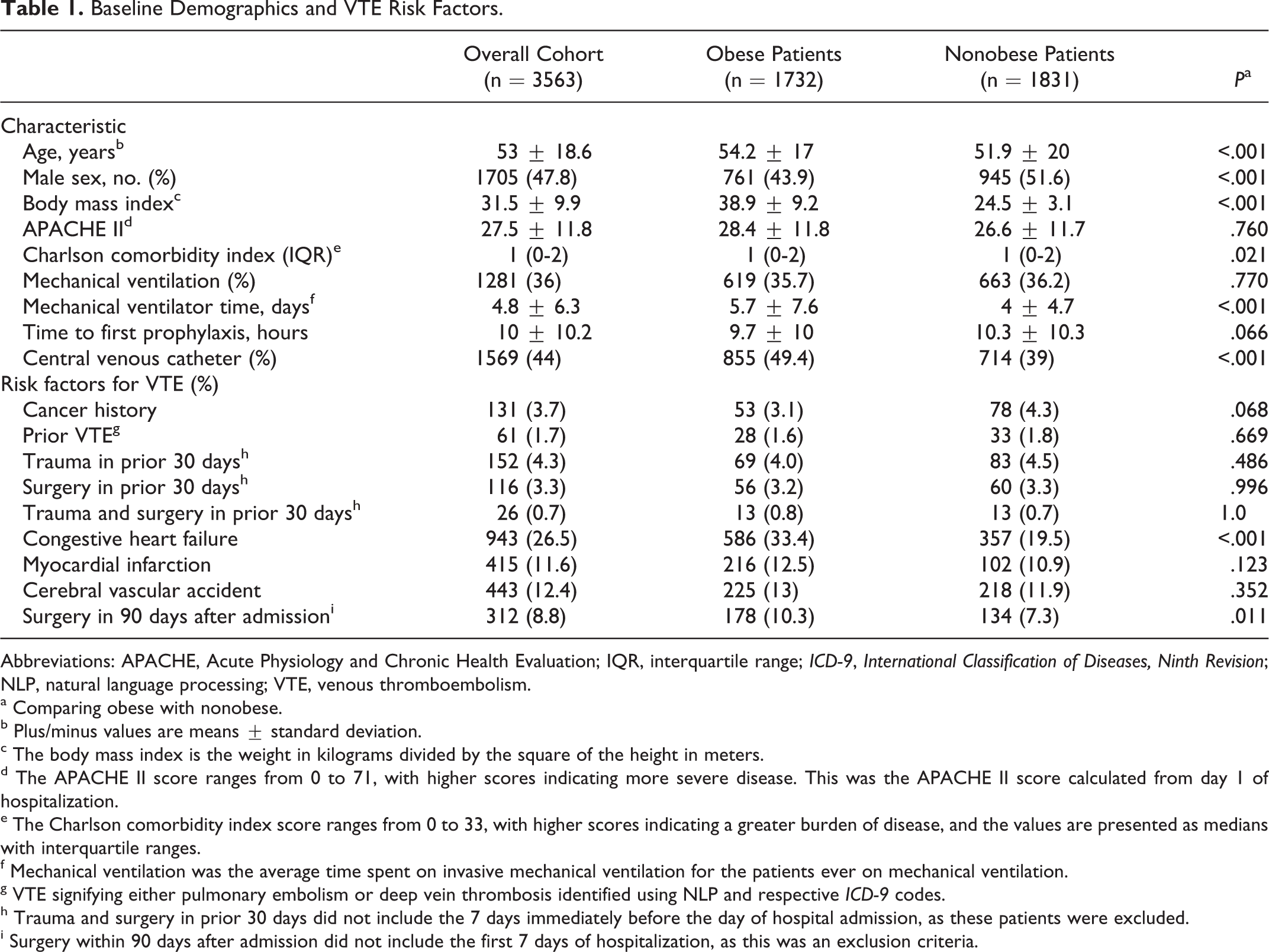
Abbreviations: APACHE, Acute Physiology and Chronic Health Evaluation; IQR, interquartile range; ICD-9, International Classification of Diseases, Ninth Revision; NLP, natural language processing; VTE, venous thromboembolism.
a Comparing obese with nonobese.
b Plus/minus values are means ± standard deviation.
c The body mass index is the weight in kilograms divided by the square of the height in meters.
d The APACHE II score ranges from 0 to 71, with higher scores indicating more severe disease. This was the APACHE II score calculated from day 1 of hospitalization.
e The Charlson comorbidity index score ranges from 0 to 33, with higher scores indicating a greater burden of disease, and the values are presented as medians with interquartile ranges.
f Mechanical ventilation was the average time spent on invasive mechanical ventilation for the patients ever on mechanical ventilation.
g VTE signifying either pulmonary embolism or deep vein thrombosis identified using NLP and respective ICD-9 codes.
h Trauma and surgery in prior 30 days did not include the 7 days immediately before the day of hospital admission, as these patients were excluded.
i Surgery within 90 days after admission did not include the first 7 days of hospitalization, as this was an exclusion criteria.
Primary and Secondary Outcomes.
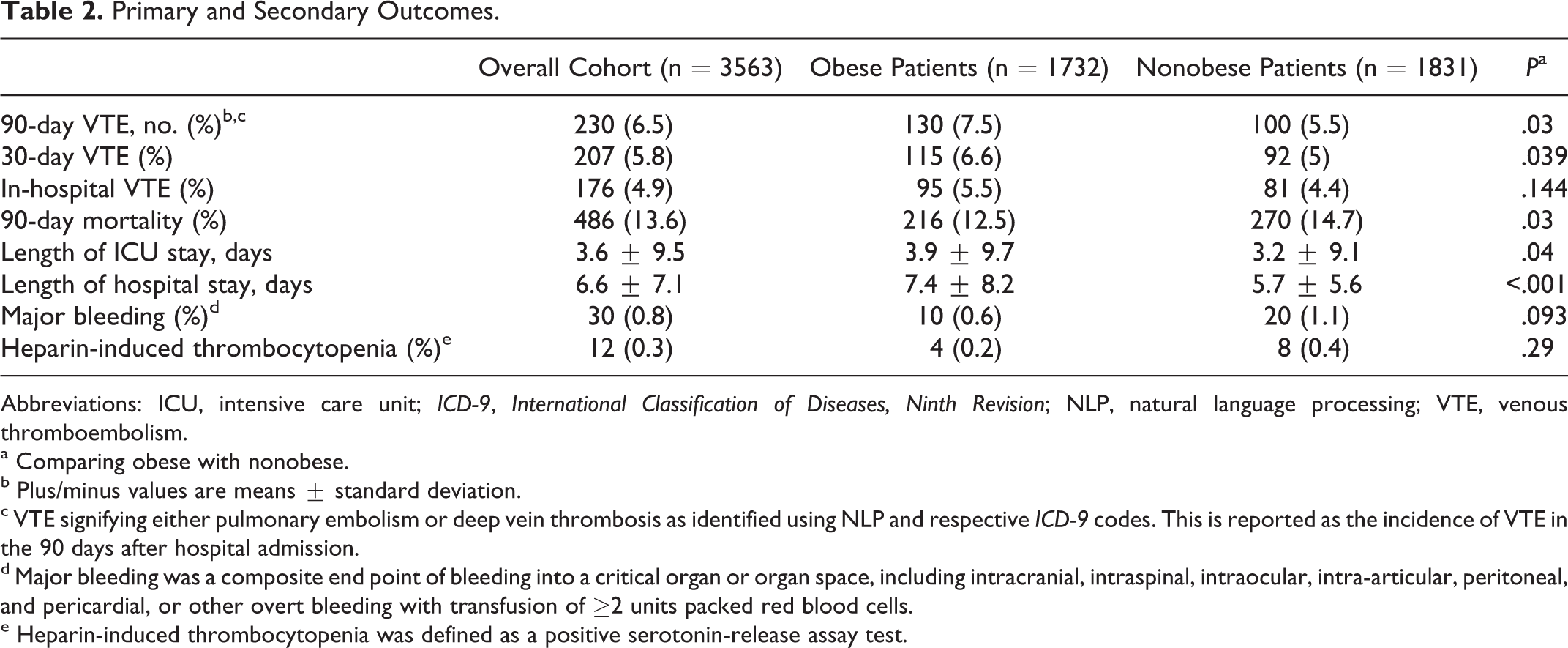
Abbreviations: ICU, intensive care unit; ICD-9, International Classification of Diseases, Ninth Revision; NLP, natural language processing; VTE, venous thromboembolism.
a Comparing obese with nonobese.
b Plus/minus values are means ± standard deviation.
c VTE signifying either pulmonary embolism or deep vein thrombosis as identified using NLP and respective ICD-9 codes. This is reported as the incidence of VTE in the 90 days after hospital admission.
d Major bleeding was a composite end point of bleeding into a critical organ or organ space, including intracranial, intraspinal, intraocular, intra-articular, peritoneal, and pericardial, or other overt bleeding with transfusion of ≥2 units packed red blood cells.
e Heparin-induced thrombocytopenia was defined as a positive serotonin-release assay test.
Outcomes
The 90-day incidence of VTE in obese and nonobese patients was 7.5% and 5.5%, respectively, with an overall incidence of 6.5% (Table 2). Our logistic regression identified obese patients were significantly more likely to have a 90-day VTE than nonobese patients (P = .03; OR: 1.41; 95% CI: 1.03-1.93). Moreover, when the traditional binomial cutoff of BMI ≥30 kg/m2 was replaced with continuous BMI values, the influence of BMI remained a significant association with 90-day VTE (P = .047, OR: 1.02; 95% CI: 1.01-1.03). Our regression also identified 7 additional variables that significantly influenced 90-day VTE: prior VTE (P < .001; OR: 3.93; 95% CI: 1.83-8.48), trauma with surgery within the 30 days prior to admission—excluding the 7 days prior to admission (P = .009; OR: 3.70; 95% CI: 1.39-9.86), presence of a CVC (P < .001; OR: 2.64; 95% CI: 1.87-3.72), surgery within 90 days of admission—excluding the 7 days prior to and following admission (P < .001; OR: 2.40; 95% CI: 1.61-3.58), mechanical ventilation (P < .001; OR: 1.94; 95% CI: 1.39-2.71), male sex (P = .007; OR: 1.55; 95% CI: 1.13-2.14), and increasing age using 1-year increments beginning at 18 years of age (P < .001; OR: 1.02; 95% CI: 1.01-1.03; Table 3).
Multiple Logistic Regression of Risk Factors on the Primary Outcome of 90-Day Venous Thromboembolism.
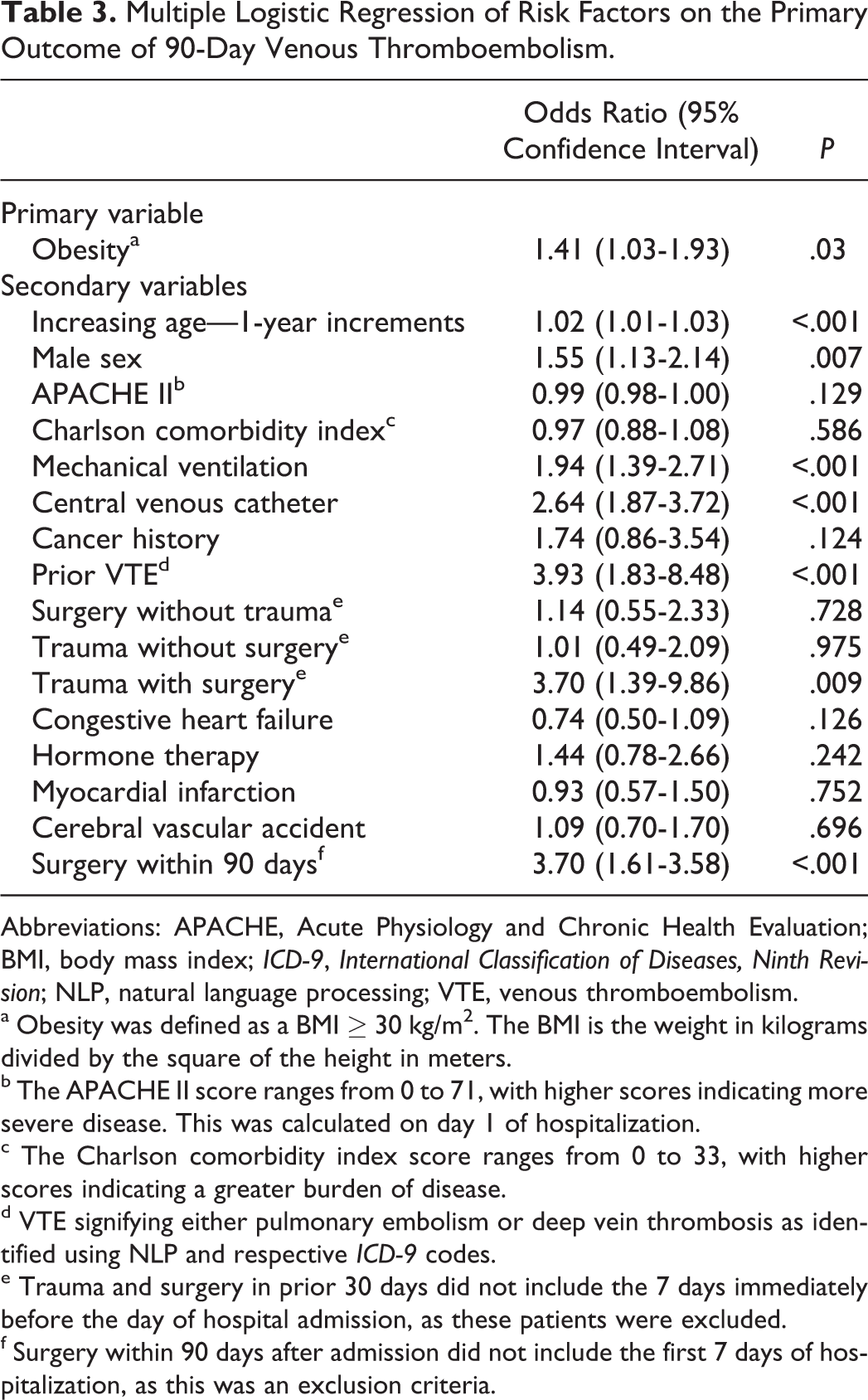
Abbreviations: APACHE, Acute Physiology and Chronic Health Evaluation; BMI, body mass index; ICD-9, International Classification of Diseases, Ninth Revision; NLP, natural language processing; VTE, venous thromboembolism.
a Obesity was defined as a BMI ≥ 30 kg/m2. The BMI is the weight in kilograms divided by the square of the height in meters.
b The APACHE II score ranges from 0 to 71, with higher scores indicating more severe disease. This was calculated on day 1 of hospitalization.
c The Charlson comorbidity index score ranges from 0 to 33, with higher scores indicating a greater burden of disease.
d VTE signifying either pulmonary embolism or deep vein thrombosis as identified using NLP and respective ICD-9 codes.
e Trauma and surgery in prior 30 days did not include the 7 days immediately before the day of hospital admission, as these patients were excluded.
f Surgery within 90 days after admission did not include the first 7 days of hospitalization, as this was an exclusion criteria.
The rates of 30-day VTE (6.6% vs 5.0%; P = .04) and in-hospital VTE (5.5% vs 4.5%; P = .14) upon univariate analysis were higher among obese patients, although only the former was statistically significant. The incidence of HIT (0.2% vs 0.4%; P = .29) was similar between groups. Obese patients had significantly longer ICU length of stay (3.94 ± 9.7 days vs 3.18 ± 9.14 days; P = .04) and hospital length of stay (7.4 ± 8.2 days vs 5.7 ± 5.6 days; P < .001) compared with nonobese patients. Major bleeding was numerically lower (0.7% vs 1.1%; P = .09), and 90-day mortality was significantly lower (12.5% vs 14.7%; P = .027) in obese patients (Table 2).
Discussion
In this retrospective cohort study of over 3500 critically ill medical patients, we found an increase in the incidence of 90-day VTE among obese patients receiving chemoprophylaxis when compared with nonobese patients of similar illness severity. Our results support previous studies reporting an increased risk of VTE in hospitalized obese patients. 18,19,22 In contrast to our study, however, former studies evaluated hospitalized medical and bariatric surgery patients, rather than critically ill medical patients. 19,22 Similar to the most recent study describing the rate of VTE in patients receiving VTE chemoprophylaxis, our cohort consisted of critically ill patients; however, we studied a medical population with a primary outcome of 90-day VTE, whereas Lim et al described a mixed medical–surgical population using ICU-associated VTE. 18 By excluding patients with surgery within 7 days of admission, we refrained from including primary surgical patients but likely included some medical patients receiving surgery during their hospital stay. We believe our cohort to be more representative of “real-world” medical ICU care, and therefore, our observations likely have more broad applicability. We studied only patients receiving guideline-recommended UFH and LMWH for VTE chemoprophylaxis 30 and did not assess the effect of variable dosing among the obese, such as enoxaparin 0.5 mg/kg weight-based dosing or 40 mg twice daily. Notably, the rate of VTE increased with time in both groups, and the differences became more significant between the groups as time progressed. The rate of major bleeding in our cohort was low. Similarly, the overall rate of major hemorrhage reportedly associated with VTE chemoprophylaxis is low and varies among population case mix. 21,36 Uncertainty exists regarding the degree obesity may be protective among patients receiving a fixed dose of UFH or LMWH due to a comparatively lower dose to body weight ratio.
Statistical differences were found in most baseline characteristics when comparing obese with nonobese patients. Given the large sample size, these small differences in many demographics, although statistically dissimilar, were not likely clinically relevant. Of note, there were more obese patients with CVCs, CHF, and surgery in the 90 days following admission. There was also a higher proportion of females in the obese group. Our binary logistic regression included all of these variables, and thus, differences between the groups would have been mitigated in our analysis. However, residual confounding of unmeasured characteristics cannot be completely alleviated without randomization.
Multiple logistic regression identified 8 risk factors associated with increased 90-day VTE, including previous VTE, surgery within 90 days of admission (excluding the 7 days prior to and following admission), CVC, trauma with surgery in the 30 days prior to admission (excluding the 7 days prior to admission), invasive mechanical ventilation at any time during hospitalization, male sex, obesity, and increasing age. Increased risk of VTE in medical–surgical ICU patients with personal or family history of VTE, 4 mechanically ventilated patients, 3 obese patients, elderly patients, and those receiving vasopressors 18 has been reported previously. However, in a recent study, no difference in VTE incidence was observed in males or in those with central venous catheters or receiving mechanical ventilation. 18
Strengths and Limitations
This is the first study, to our knowledge, evaluating VTE risk factors in a broad population of solely critically ill medical patients receiving appropriate VTE chemoprophylaxis. It is also the largest retrospective cohort study of its type to date. Day 1 mean APACHE II score of 27.5 indicates our study population consisted of high-acuity critically ill patients. We have a high level of certainty that a meaningful difference in obesity existed given the divergence of BMI between the groups. This allowed for a true evaluation of the role of increasing BMI on 90-day VTE rates. Interestingly, the risk of 90-day VTE began to rise when BMI increased above 17 kg/m2, well below the classic obesity definition of 30 kg/m2. We did not directly compare obese patients (BMI ≥ 30 kg/m2) with normal-weight patients (BMI: 17-25 kg/m2), although our results remained robust when using BMI as a continuous variable in our logistic regression model. Thus, one could postulate an increased risk of VTE in obese patients when compared to normal-weight patients. The large patient population and robust data set allowed us to appropriately incorporate many risk factors into the multiple logistic regression analysis including risk factors previously shown to increase VTE in hospitalized medical patients. The use of strict inclusion and exclusion criteria meant we were able to identify a homogenous, critically ill, medical population. We included only patients with an ICU stay ≥24 hours and excluded patients receiving therapeutic anticoagulation within the first 48 hours and those admitted into the operating room within the first 7 days of hospitalization. Therefore, we excluded patients with severe trauma or need for immediate surgery and those with less severe illness (spending <24 hours in the ICU). Excluding patients who did not receive a prophylactic dose of UFH or enoxaparin within 48 hours of admission ensured prompt VTE chemoprophylaxis, another strength in our identification of the study cohort. Data have shown the omission of early thromboprophylaxis is associated with an increased risk of mortality in critically ill patients. 37
The retrospective design of our study begets inherent limitations including outcome ascertainment, patient selection, lack of randomization, and the possibility of unbalanced cohorts. However, analyzing the data with multiple logistic regression should mitigate many of these imbalances. Second, certain variables were not included in our final analysis, such as vasopressor use, renal replacement therapy, or acute kidney injury (AKI). With greatly varying doses, intensity, and duration of vasopressors, we felt it best to exclude this variable given the retrospective design of our study. Retrospectively, it is also difficult to diagnose AKI based upon changes in serum creatinine. End-stage renal disease (ESRD) has shown a variable role in VTE development, with former studies describing an increased risk of VTE 4 and more recent studies showing no increase in VTE in patients with ESRD and AKI. 18,38 Emerging risk factors for thrombosis including testosterone have been described. 39,40 While testosterone use is not routine among ICU patients we cannot exclude effects given we did not query for use. Severe sepsis and septic shock have recently shown to be a greater risk of on-therapy VTE than described previously. 41 We did not however separate these diagnoses from other medical diagnoses. We also did not differentiate between minor and major surgeries in patients admitted through the ORMS. Third, due to inconsistent documentation and variation in patient compliance, we were unable to verify the use of mechanical VTE prophylaxis such as sequential compression devices or graduated compression stockings (GCS). Studies suggest that mechanical thromboprophylaxis variably affects clinically important outcomes. Although some studies have shown no decrease in mortality, 29 others have demonstrated a decrease in symptomatic and proximal VTE. 42 Recent data report a numerical decrease in early VTE in patients receiving mechanical prophylaxis 43 and a greater decrease in the incidence of VTE with intermittent pneumatic compression devices compared with GCS. 44 At our institution, intermittent pneumatic compression devices are used in nearly all critically ill patients, barring relative contraindications such as limb ischemia or local orthopedic injury. Fourth, patients in this study on average received their first dose of VTE chemoprophylaxis within 10 hours of hospital admission; however, the ongoing use of chemoprophylaxis was not retrospectively verified, and neither was the duration of therapy. Barring contraindications, such as active bleeding or HIT, our institution universally utilizes chemoprophylaxis in critically ill medical patients. Critical care pharmacists located within the ICUs assess the need for VTE prophylaxis twice daily. Thus, the majority of patients should have received chemoprophylaxis throughout their ICU and hospital stay. Fifth, retrospectively identifying the primary outcome may have led to the inclusion of asymptomatic distal DVTs. This inclusion would increase incident VTE at 90 days. We were unable to further clarify the specific VTE event (PE vs DVT). However, the incidence of VTE in our study is similar to the results in a recent randomized controlled trial of critically ill patients receiving dalteparin or twice-daily UFH (8.2% and 9.9%, respectively) 16 and also with the results of Lim et al (7.7% VTE incidence during ICU stay). 18 Moreover, the NLP methodology of VTE data extraction has been validated in previously published work. 45,46
Although not addressed in our study, the use of increased doses of heparin (eg, 7500 units SQ 3 times daily) or enoxaparin (eg, 40 mg SQ 2 times daily or weight-based dosing strategies) should be evaluated in large clinical trials. At the present time, these strategies are often utilized with very little supporting evidence. However reasonable these empiric dose increases may seem in obese critically ill medical patients, uncertainty exists regarding the relative risk-to-benefit ratio, and further validation is needed. 47,48
Conclusion
The rate of VTE in critically ill medical patients remains high, despite the use of standard VTE chemoprophylaxis. Obesity is independently associated with 90-day VTE, along with 7 other risk factors, including prior VTE, trauma with surgery in the 30 days prior to admission, surgery within the 90 days following admission, presence of a central venous catheter, mechanical ventilation, male sex, and older age. Given the significant morbidity, mortality, and cost associated with developing VTE, it is crucial that future studies identify ideal chemoprophylactic regimens for critically ill patients at highest risk of VTE.
Footnotes
Appendix 1. International Classification of Diseases,Ninth Revision ( ICD-9 ) Codes for Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE) Used in Data Abstraction
Deep vein thrombosis: 451.1, 451.11, 451.19, 451.2, 451.81, 451.83, 451.89, 451.9, 453.2, 453.3, 453.40 to 453.42, 453.50 to 453.52, 453.6, 453.71 to 453.79, 453.8 to 453.89, 453.9, 671.30, 671.31, 671.33, 671.40, 671.42, 671.44, 673.20 to 673.24.
Pulmonary embolism: 415.1, 415.11, 415.19, 673.2.
Acknowledgments
The authors thank Dr Courtney McKinney, PharmD, BCPS, for her editorial assistance.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S. C. Woller and S. M. Stevens disclose grant support from Bristol-Myers-Squibb, Iverson Genetics, and Twine Clinical LLC paid to Intermountain Healthcare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
