Abstract
Objectives
This study aimed to investigate the clinical characteristics and risk factors associated with secondary infection in patients diagnosed with sepsis.
Methods
Clinical data of patients diagnosed with sepsis on the first day of admission to the Intensive Care Unit (ICU) were extracted from the MIMIC-IV version 2.0 database. Univariate and multivariate logistic regression analyses were employed to analyze the influencing factors. A nomogram for predicting the risk of secondary infection was constructed using R software.
Results
A total of 2247 patients met the eligibility criteria for this study. Lower oxygen saturation, reduced lymphocyte proportion, decreased platelet count, higher neutrophil proportion, elevated lactic acid levels within 24 h after ICU admission, and the use of invasive mechanical ventilation were identified as independent risk factors for secondary infection in sepsis patients during their ICU stay. These predictors were integrated into a nomogram to estimate the risk of secondary infection in sepsis patients, and R software was employed to construct this Nomogram.
Conclusions
The Nomogram, incorporating oxygen saturation, lymphocyte ratio, platelet count, neutrophil ratio, lactic acid levels, and mechanical ventilation, can provide individualized risk predictions for secondary infection in patients with sepsis.
Introduction
Sepsis, a life-threatening condition characterized by dysregulated host responses to infection, 1 constitutes a prevalent clinical syndrome within the critical care medicine domain. In recent years, owing to advancements in diagnostic and therapeutic modalities, while the mortality rate of sepsis has shown a decline, with the increase in the number of clinical patients, the absolute count of deceased patients remains substantially elevated. 2 A predominant focus of contemporary research endeavors has been directed towards elucidating the mechanisms underlying early mortality among septic patients, often attributable to excessive activation of the innate immune system during acute infection. 3 Nonetheless, a fraction of fatalities among septic patients transpires beyond the first week following admission to the intensive care unit (ICU). 4 The principal contributors to these delayed mortalities encompass a constellation of complications, including secondary superinfections, multiple organ dysfunction syndrome, septic shock, and the persistent inflammatory immunosuppression catabolic syndrome, culminating in ultimate demise. 5
Notably, the incidence of secondary infections during the hospitalization of septic patients markedly surpasses that observed in non-septic counterparts, concomitant with a commensurate rise in mortality rates. 6 The crux of sepsis lies not solely in the inflammatory response but also encompasses immune dysfunction. 7 The underlying rationale for secondary infections in sepsis lies in the induction of a state of immunosuppression within afflicted patients, characterized by immune cell hyperreactivity, exhaustion, apoptosis, and an augmented presence of myeloid-derived suppressor cells. 8 Furthermore, mounting evidence underscores the pivotal role of dysbiosis within the intestinal microbiota in facilitating secondary infections in this context. 9 Sepsis precipitates alterations in intestinal integrity, resulting in heightened intestinal permeability. This perturbation potentially instigates bacterial translocation via the mesenteric lymph nodes or the portal venous system, subsequently instigating concomitant inflammatory cascades. 10
While secondary infections in septic patients within the ICU have garnered considerable attention, there remains a paucity of comprehensive clinical data. The principal objective of this investigation is to scrutinize the incidence and determinants of secondary infections among hospitalized septic patients, thereby furnishing an evidence-based foundation for mitigating secondary infection rates and overall mortality within this patient cohort. Furthermore, within the realm of exploratory analysis, we endeavor to construct a predictive model that comprehensively encompasses independent risk factors for secondary infections.
Methods
Data Sources
The data utilized in this investigation were sourced from the extensive American Intensive Care repository known as the Medical Information Mark for Intensive Care (MIMIC). The database was acquired with the approval of the Institutional Review Board (IRB) at both the Massachusetts Institute of Technology and Beth Israel Deaconess Medical Center. As a result, the need for obtaining informed consent from patients was waived. Access to this repository is predicated upon the successful completion of an institution-specific training program examination.
Patient Information
The PostgreSQL 9.6 software was employed for the retrieval of fundamental demographic data, clinical particulars, diagnostic information, and outcome metrics pertaining to the cohort under examination. Specifically, individuals diagnosed with sepsis within the initial 24 h of their admission to the ICU were selectively extracted from the MIMIC-IV version 2.0 database. The criteria for exclusion were as follows: (1) Patients whose age upon ICU admission was either < 18 years or > 80 years; (2) Patients whose duration of ICU sojourn was < 48 h; (3) In cases involving multiple ICU admissions, solely data from the maiden hospitalization and inaugural ICU admittance were considered, with information regarding subsequent re-admissions to the ICU being omitted; (4) Pregnant women; (5) Patients displaying conspicuous immunodeficiency post-organ transplantation, acquired immunodeficiency syndrome (AIDS), neoplastic disorders, or hematological malignancies; (6) Patients featuring substantial gaps in clinical dataset (patients with more than 10% missing clinical data).
Definition and Diagnostic Criteria
The categorization of diseases underwent a progressive refinement in accordance with the International Classification of Diseases code ICD-9. Subsequently, an exploratory data analysis was executed based on the disease diagnoses. Sepsis is a life-threatening organ dysfunction caused by a dysregulated body response to infection. Diagnostic criteria: patients with infection or suspected infection, and Sequential Organ Failure Assessment (SOFA) score ≥2 points or SOFA score change ≥2 points. 1 Secondary infection was defined as the emergence of novel pathogens or fresh infections at distinct anatomical sites during the course of sepsis treatment. 11
Data Collection
In this research study, an extensive dataset was curated encompassing diverse aspects of demographic, clinical characteristics, laboratory findings, and clinical diagnoses pertinent to patients admitted to the Intensive Care Unit (ICU). In adherence to the predefined exclusion criteria and research objectives, specific data were meticulously gathered from the cohort of eligible subjects. The key variables under scrutiny included: age, gender, SOFA Score within 24 h after admission to ICU, Simplified Acute Physiology Score II (SAPS II), Mean Artery Pressure (MAP), Heart Rate, Respiratory Rate, Body Temperature, Pulse Oxygen Saturation (SPO2), The White Blood Cell Count (WBC), Neutrophil Count (NEUT), and Percentage of Neutrophils (NEU) in the first 24 h of ICU admission, Monocyte Count (MONO), Monocyte Percentage (MONO%), Lymphocyte Count (LYMPH), Lymphocyte Percentage (LY), Eosinophil Count, Eosinophil Percentage, Basophils Count, Basophils Percentage, Red Blood Cell count (RBC), Hemoglobin (HGB), Hematocrit (HCT), Mean Corpuscular Hemoglobin (MCH), Mean Corpuscular Hemoglobin Concentration (MCHC), Mean Corpuscular Volume (MCV), Red Blood Cell Distribution Width (RDW), red Blood Cell Distribution Width-Standard Deviation (RDW-SD), Platelet (PLT), the First Blood Lactate (Lac) within 24 h after ICU admission, Potential of Hydrogen (PH), Alanine transaminase (ALT) within 24 h after ICU admission, Aspartate Transaminase (AST), Total Bilirubin (TB), Serum Creatinine (SCR), Blood Urea Nitrogen (BUN), Prothrombin Time (
Construction, Validation, and Evaluation of the Prediction Model
Employing a random sampling methodology, the incorporated dataset was partitioned into a training set and a validation set, in adherence to a 7:3 ratio. Subsequently, the process of developing the prediction model was exclusively conducted within the confines of the training set. The validation set was used for model verification to further judge the feasibility and verification performance of the model. Continuous variables with normal distribution were expressed as mean ± standard deviation, and continuous variables with skewed distribution were expressed as median and interquartile range. The characteristics of the two groups of patients were compared using t-test, chi-square test, or Wilcoxon rank-sum test, as appropriate.
Building upon the results of this multivariate analysis, we harnessed the R software to craft a nomogram predictive model predicated on independent risk factors. Following the model's construction, a rigorous process of verification and evaluation was undertaken. First and foremost, model accuracy was meticulously scrutinized by evaluating the area under the curve (AUC) derived from the receiver operating characteristic (ROC) curve. Furthermore, we subjected the model to 200 iterations of repeated sampling verification. This comprehensive validation process encompassed the generation of calibration curves. These curves facilitated an assessment of the alignment between the predicted probabilities and the observed outcomes, establishing the model's calibration performance and its conformity to established standards. In addition, the clinical effectiveness of the model was evaluated by analyzing the clinical influence curve and decision curve.
The analysis software used in this study was IBM SPSS Statistics 24.0 (IBM Corporation, Armonk, New York, USA) and R version 3.3.3 (The R Foundation for Statistical Computing, Austria, Vienna).
Results
Patient Characteristics
The dataset comprised a total of 257,366 patients, with 15,566 patients receiving a sepsis diagnosis within the initial 24 h of admission to ICU2. Following the application of inclusion and exclusion criteria, a final cohort of 2247 patients was eligible for inclusion in this investigation. The study flow chart is shown in Figure 1, which includes the training cohort (n = 1573) and the validation cohort (n = 674). Subsequently, we compiled a summary of the patients’ demographic and clinical attributes, as outlined in Table 1.

Research Flowchart.
Baseline Characteristics of Patients with Sepsis.

Abbreviations: RR, Respiratory Rate; HR, Heart Rate; MAP, Mean Artery Pressure; SPO2, Pulse Oxygen Saturation; SOFA Score, Sequential Organ Failure Assessment Score; SAPS, Simplified Acute Physiology Score; WBC, White Blood Cell Count; NEUT, Neutrophil Count; NEU, Percentage of neutrophils; MONO, Monocyte Count; MONO %, Monocyte Percentage; LYMPH, Lymphocyte Count; LY, Lymphocyte Percentage; EOS, Eosinophil Count; EOS%, Eosinophils Percentage; BAS, Basophil Count; BAS%, Basophils Ratio; RBC, Red Blood Cell Count; HGB, Hemoglobin; HCT, Hematocrit; MCH, Mean Corpuscular Hemoglobin; MCHC, Mean Corpuscular Hemoglobin Concentration; MCV, Mean Corpuscular Volume; PLT, Platelet RDW, Red Blood Cell Distribution Width; RDW-SD, Red Blood Cell Distribution Width-Standard Deviation; Lac, Lactate; PH, Potential of Hydrogen; ALT, Alanine Transaminase; AST, Aspartate Transaminase; TB, Total Bilirubin; SCR, Serum Creatinine; BUN, Blood Urea Nitrogen; PT, Prothrombin Time; APTT, Activated Partial Thromboplastin Time; CRRT, Renal Replacement Therapy; MV, Mechanical Ventilation.
The diagnostic procedure involved initially extracting the diagnostic data of patients upon their first day of admission to the ICU from the database. Patients diagnosed with “sepsis” were then identified and subsequently screened based on predefined exclusion criteria. Ultimately, a total of 2247 patients were deemed eligible for inclusion in the study (Table 1). Subsequently, the etiological and diagnostic information pertaining to the patients was extracted for further analysis. A total of 2247 patients were enrolled in our study (Table 1), the patient cohort exhibited a mean age of 63 years (range: 53-71), with 1256 (55.9%) being male. Notably, 31.6% of septic patients experienced the development of secondary infections. Additionally, our investigation revealed that the average time interval between ICU admission and the occurrence of secondary infection was approximately 9.0 days (4.6-17.8). Patients were subjected to random allocation into distinct training and validation cohorts, adhering to rigorous randomization protocols. In order to bolster the robustness and credibility of our analytical outcomes concerning these two cohorts, we conducted a comprehensive comparative examination of the datasets involved. A total of 41 clinical variables underwent meticulous analysis. Following a rigorous scrutiny and comparison of the two dataset groups, it was discerned that both datasets exhibited a remarkable degree of uniform distribution.
Upon the analysis of the training cohort comprising 1573 patients (Table 2), it was observed that the mean age of these patients stood at 63 years, with a range of 53 to 71 years. Among this cohort, 890 individuals, constituting 56.6% of the population, were of male gender, and 490 patients (31.2%) were afflicted with secondary infections. In the comparative analysis of sepsis patients with and without secondary infections, noteworthy differences emerged. Specifically, patients devoid of secondary infections exhibited a higher initial oxygen saturation compared to their counterparts with secondary infections, with respective values of 96.6% ± 2.3% and 96.2% ± 3.4%, this disparity achieved statistical significance (
Characteristics of Patients and Diseases.

Abbreviations: S_ infection, Secondary infection; RR, Respiratory Rate; HR, Heart Rate; MAP, Mean Artery Pressure; SPO2, Pulse Oxygen Saturation; SOFA Score, Sequential Organ Failure Assessment Score; SAPS, Simplified Acute Physiology Score; WBC, White Blood Cell Count; NEUT, Neutrophil Count; NEU, Percentage of neutrophils; MONO, Monocyte Count; MONO %, Monocyte Percentage; LYMPH, Lymphocyte Count; LY, Lymphocyte Percentage; EOS, Eosinophil Count; EOS%, Eosinophils Percentage; BAS, Basophil Count; BAS%, Basophils Ratio; RBC, Red Blood Cell Count; HGB, Hemoglobin; HCT, Hematocrit; MCH, Mean Corpuscular Hemoglobin; MCHC, Mean Corpuscular Hemoglobin Concentration; MCV, Mean Corpuscular Volume; PLT, Platelet; RDW, Red Blood Cell Distribution Width; RDW-SD, Red Blood Cell Distribution Width-Standard Deviation; Lac, Lactate; PH, Potential of Hydrogen; ALT, Alanine Transaminase; AST, Aspartate Transaminase; TB, Total Bilirubin; SCR, Serum Creatinine; BUN, Blood Urea Nitrogen; PT, Prothrombin Time; APTT, Activated Partial Thromboplastin Time; CRRT, Renal Replacement Therapy; MV, Mechanical Ventilation.
Predictors of Secondary Infection in Sepsis Patients
Univariate and multivariate Logistic regression analyses were systematically executed to ascertain pertinent predictors. Variables exhibiting a significance level of P < .1 during univariate binary Logistic regression were selected for further evaluation. Statistically significant variables thus identified were subsequently incorporated into the multivariate Logistic regression model. Finally, these selected variables were introduced into a predictive model designed for this purpose. The outcomes of both univariate and multivariate Logistic regression analyses, conducted within the confines of the training cohort, have been meticulously presented in Table 3. Our analysis unveiled six variables that exhibited potential association with the risk of secondary infections in sepsis patients: the oxygen saturation level at the time of initial admission to the ICU, percentages of neutrophils, percentages of lymphocytes, platelet count, blood lactate concentration, and the utilization of mechanical ventilation as adjunctive therapy (Table 3). Consequently, we conducted a multivariate analysis to confirm the independent role of oxygen saturation, neutrophil percentage, lymphocyte percentage, platelet count, blood lactic acid concentration, and mechanical ventilation at initial ICU admission as risk factors for secondary infection in sepsis patients. These aforementioned factors were included in the development of a prediction model, and we utilized R software to establish a nomogram representing the predictive model (Figure 2).

Nomogram for Predicting the Risk of Secondary Infection in Patients with Sepsis.
Univariate and Multivariate Analysis of Secondary Infection Factors in Patients with Sepsis.
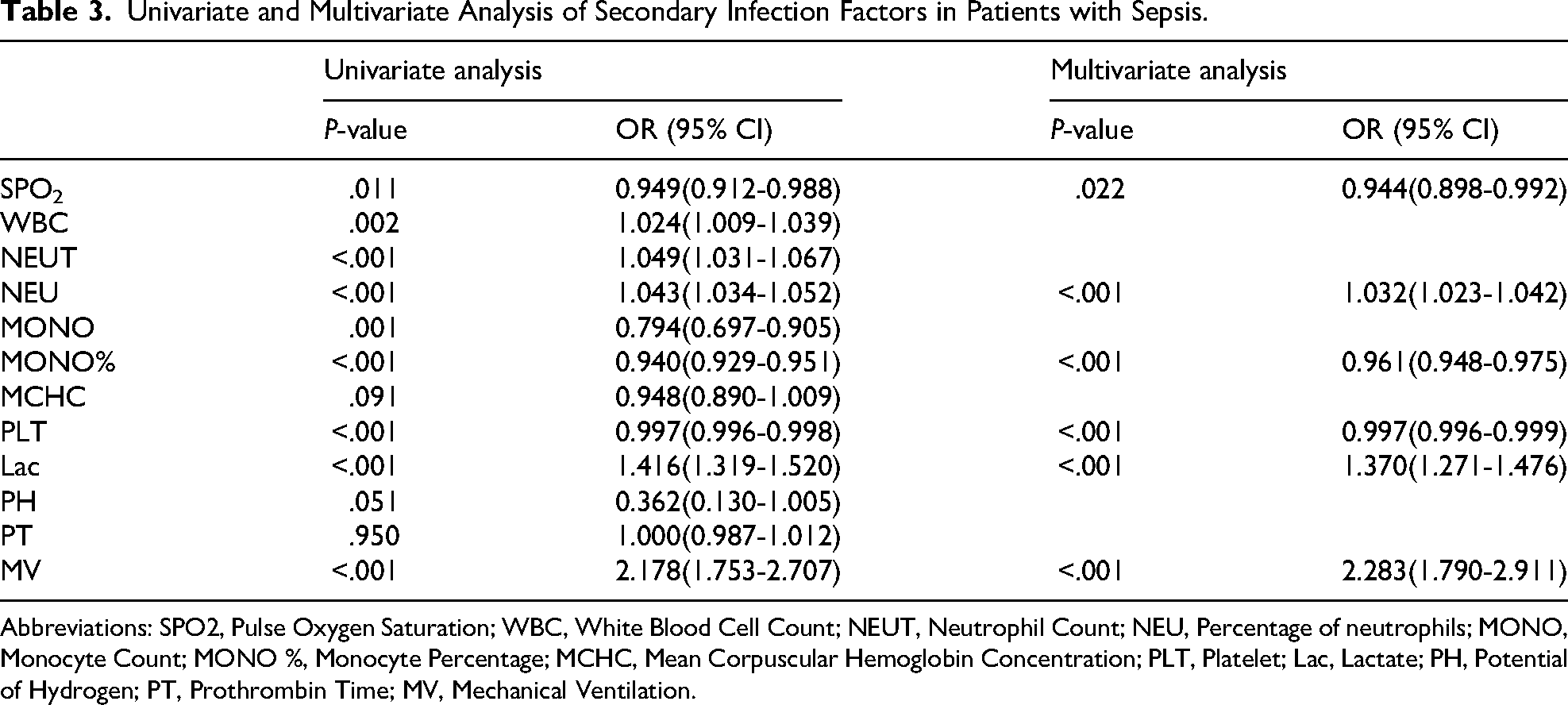
Abbreviations: SPO2, Pulse Oxygen Saturation; WBC, White Blood Cell Count; NEUT, Neutrophil Count; NEU, Percentage of neutrophils; MONO, Monocyte Count; MONO %, Monocyte Percentage; MCHC, Mean Corpuscular Hemoglobin Concentration; PLT, Platelet; Lac, Lactate; PH, Potential of Hydrogen; PT, Prothrombin Time; MV, Mechanical Ventilation.
Interpretation and Analysis of the Nomogram for the Prediction Model
Nomogram is constructed through the application of multivariate regression analysis, allowing for the amalgamation of diverse predictors. The nomogram (Figure 2) provides a comprehensive depiction of essential information crucial to the predictive model. Firstly, it prominently displays the variables integrated into the prediction model, including SPO2, NEU, LY, PLT, Lac, and MV, all of which are meticulously marked on line segments corresponding to their respective ranges of values. Additionally, the nomogram features a summative score, referred to as ‘Total Points,’ which signifies the cumulative score obtained by aggregating individual item scores across all variables. Finally, the nomogram culminates in the estimation of the anticipated probability, denoted as “S_infection Risk” within the figure. This probability signifies the likelihood of experiencing a secondary infection.
Determination of the Critical Value of Secondary Infection in the Prediction Model
After conducting the modeling process, the accuracy of the prediction model was assessed by utilizing the area under the receiver operating characteristic (ROC) curve (AUC). The nomogram was internally validated in the training cohort (Figure 3a) and externally validated in the validation cohort (Figure 3b) using ROC analysis. The AUC value for internal validation was determined to be 0.7685 (95% CI 0.7437-0.7932), indicating a favorable performance of the model. Similarly, the external validation ROC curve yielded an AUC value of 0.8182 (95% CI 0.7844-0.852), further confirming the model's advantageous properties. To further ascertain the model's validity, a calibration curve was established using R. Calibration was conducted both within the training cohort (Figure 4a) and outside the validation cohort (Figure 4b). The calibration curve demonstrated a satisfactory degree of fit to the standard ideal curve, signifying that the predicted probability of occurrence by the model closely aligned with the actual probability of occurrence. Finally, the clinical efficacy of the nomogram was further assessed through the generation of a decision curve (Figure 5) and a clinical impact curve (Figure 6) utilizing R statistical software.

The ROC Curve Verifies the Accuracy of the Training Dataset (a) and Validation Dataset (b) Models.

Calibration Curves of the Nomogram in the Training Dataset (a) and Validation Dataset (b).

Decision Curve Analysis of the Nomogram in the Training Dataset (a) and Validation Dataset (b).
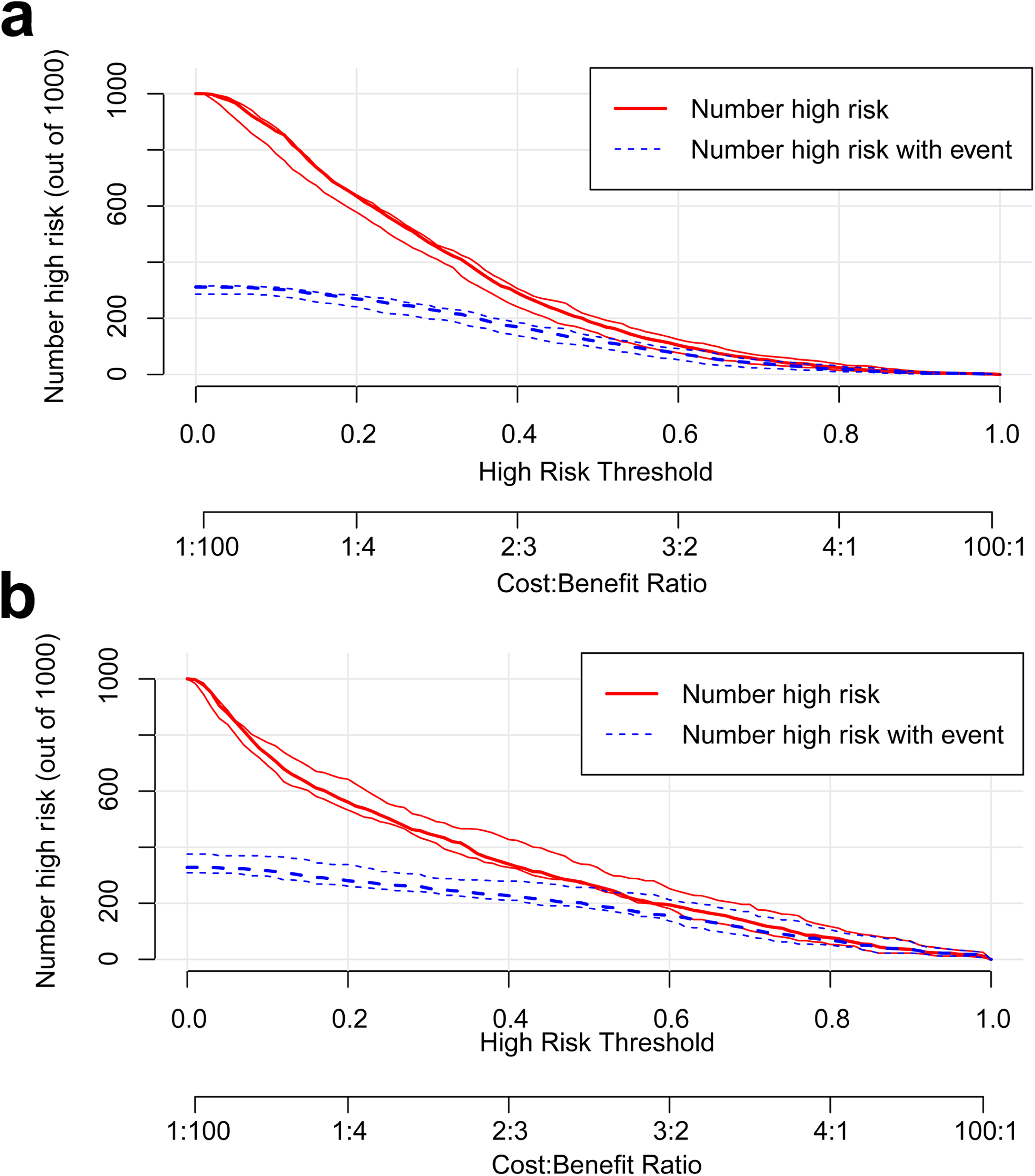
Clinical Impact Curves of the Nomograms in the Training Dataset (a) and Validation Dataset (b).
Discussion
Sepsis claims the lives of over 10 million individuals annually, and the mortality rate demonstrates a decline with an augmented proportion and count of sepsis survivors. 12 A retrospective cohort study conducted by Sun et al 13 unveiled the prevalence of ICU readmissions subsequent to sepsis episodes, with secondary infection being the primary cause for such readmissions. The inherent pathogen colonization in sepsis patients, coupled with immunosuppression brought about by therapeutical agents, may heighten the vulnerability to secondary infections. Furthermore, an investigation focused on the host's genomic attributes pertaining to secondary infections in ICU patients afflicted by sepsis revealed leukocyte gene expression profiles indicative of disrupted glycolysis and gluconeogenesis, fundamental processes governing energy supply and the inflammatory response of immune cells. As we all know, secondary infection has a negative impact on the prognosis of patients with sepsis. An investigation elucidated that the hazard of delayed mortality in sepsis patients following secondary infection escalates by an approximate factor of 5.8 in comparison to their counterparts devoid of secondary infections. 14 Studies have also shown that the early demise of sepsis patients primarily ensues from recalcitrant multiple organ failure, with secondary infection emerging as the second foremost causative factor for delayed mortality in individuals grappling with severe sepsis and septic shock. 15 In a comprehensive study encompassing 778 critically ill patients within the ICU milieu, it was discerned that nosocomial infections contributed to a substantial extension of hospital stay, amounting to an average augmentation of 18.2 days per patient. 16
In this investigation, the incidence of secondary infections in sepsis-afflicted patients stood at 31.6%, and the median time of secondary infection was 9.0 days after ICU admission. While secondary infections following sepsis have been a subject of considerable attention, it is noteworthy that the number of clinical studies addressing this phenomenon remains relatively limited within the medical literature. A clinical inquiry highlighted a noteworthy temporal pattern in secondary infection development among patients with sepsis or septic shock. Those in the late stage (≥ 15 days post-admission) exhibited a markedly elevated propensity for secondary infections, frequently attributed to common opportunistic pathogens and fungal agents, in contrast to their early-stage counterparts (< 6 days). 17 Concordantly, the outcomes of a retrospective analysis mirrored our own findings, showcasing a substantial occurrence of secondary infections in 39% of septic patients within the ICU, with the diagnosis of secondary infections typically occurring at a median time of 9 days (6 to 12 days) post-admission. 14 Furthermore, our findings align with other pertinent investigations, collectively reinforcing the notion of a high incidence rate (31.0%) of secondary infections in patients grappling with sepsis. Predominantly, the respiratory tract emerged as the most common site of secondary infections, and the median onset for the initial secondary infection was consistently observed around the eighth day. 18
The development of the nomogram model to predict the risk of secondary infection in patients with sepsis is highly significant. The model demonstrated satisfactory predictive performance, as indicated by an AUC ranging from 0.7437 to 0.7932. This suggests that the model has the ability to effectively identify patients who are at higher risk of developing secondary infections. Previous studies have also examined the risk factors associated with secondary infection in sepsis patients. These studies have identified several factors, including age, severity of the primary disease, length of ICU stay, and invasive surgery, as having an impact on the occurrence of secondary infections. 14 However, our study observed that patients with secondary infection did not have higher SAPS II and SOFA scores upon admission compared to patients without secondary infection. This finding differs from previous studies. 19 One potential explanation for this observation could be that patients with a higher burden of disease severity may experience early mortality attributable to the primary disease before the manifestation of secondary infections, thereby potentially influencing the veritable association between risk factors and the severity of the underlying condition. Additionally, studies have identified indwelling urinary catheterization as an independent risk factor for secondary infection in sepsis patients.18,20 Similarly, multiple studies have confirmed that deep vein catheterization is also an independent risk factor for secondary infection.
The proportion of neutrophils, recognized as a risk factor for secondary infection in septic patients, demonstrates an elevation. Following the onset of sepsis, a substantial accumulation of neutrophils occurs at the septic focus. 21 This influx is advantageous during the initial phase of sepsis, as neutrophils fulfill their phagocytic role in eliminating pathogenic bacteria. Nevertheless, the regulatory role of neutrophils in sepsis is intricate, and the impaired migration and shortened lifespan of neutrophils in severe sepsis have been extensively documented. 22 In the context of sepsis, alterations in the percentage of neutrophils occur, and excessive neutrophil activation can inflict damage upon organ parenchymal cells, leading to multiple organ dysfunction. 23 By employing this established model, our attention can be directed towards sepsis patients presenting with an elevated percentage of neutrophils upon admission to the ICU, thereby ensuring vigilance towards the occurrence of secondary infections.
The diminished lymphocyte proportion, recognized as a pivotal risk factor contributing to secondary infections among sepsis patients, tends to exhibit depletion. Lymphocytopenia stands as a prevailing aberrant parameter often observed within the population of individuals admitted to the ICU. Nevertheless, a limited body of research has concentrated on the comprehensive assessment of peripheral blood lymphocyte proportions among sepsis patients predisposed to secondary infections. Consequently, the present investigation delved into the examination of risk factors associated with secondary infections in sepsis-afflicted individuals. The findings of this inquiry underscored that a diminished lymphocyte proportion emerged as a discernible predictor of secondary infections. In the context of sepsis, the reduced lymphocyte count precipitates an immunosuppressive state within the host, thereby hampering the effective regulation of specific immune responses aimed at combatting pathogenic intruders. This immunological impairment consequently fosters the progression of infections, instigates the onset of MODS, and in severe instances, culminates in fatal outcomes. 23 Analogous investigations conducted on patients afflicted with severe COVID-19 have corroborated the association between lymphocytopenia and susceptibility to secondary infections.24,25 Therefore, when assessing sepsis patients undergoing admission to the ICU, meticulous attention must be devoted to scrutinizing the status of immune cell.
The risk factors associated with secondary infection in septic patients include the utilization of mechanical ventilation. For individuals with sepsis, the invasive procedure of ventilator-assisted ventilation can significantly impact various barrier functions and bacterial colonization. Concurrently, potential immunosuppression also contributes to this effect, thereby increasing the risk of secondary infection. This study demonstrates that patients undergoing mechanical ventilation following admission to the ICU have a higher likelihood of experiencing secondary infections. Prolonged immune dysfunction in the body, coupled with the presence of inflammation, creates an immunosuppressive state characterized by an ongoing pro-inflammatory response concomitant with immunosuppression. This state facilitates bacterial colonization and infection, thereby elevating the risk of secondary infections. Previous research has also established a strong association between the need for mechanical ventilation in critically ill patients and a high incidence of ventilator-associated pneumonia, which accounts for nearly half of nosocomial infections. 26 However, some studies have reported conflicting results, wherein mechanical ventilation was deemed a risk factor in univariate analysis but did not achieve statistical significance in multivariate analysis. 18 Consequently, it is crucial to monitor the immune status of patients and provide appropriate nursing care.
In addition to the previously mentioned risk factors, our study identified several other factors associated with an increased risk of secondary infection in patients with sepsis. Notably, elevated levels of lactic acid, decreased platelet count, and diminished pulse oxygen saturation emerged as significant risk factors. The lactic acid levels serve as both a marker of disease severity and a robust predictor of patient mortality, as reported in prior literature. 27 Furthermore, the Surviving Sepsis Campaign (SSC) recommends the use of lactate levels as an indicator of sepsis-induced hypoperfusion to guide prompt fluid resuscitation strategies. 28 Platelets hold a crucial role as pivotal mediators that promptly release diverse bioactive substances within the circulatory system for the regulation of inflammation and hemostasis. In the context of sepsis, the infectious process directly inhibits the megakaryocyte system within the bone marrow, consequently leading to a decrease in platelet production originating from this vital hematopoietic site. 29 Additionally, this study further corroborated the association between decreased platelet count and the occurrence of secondary infections in patients. Prior investigations have examined the comparative efficacy of a conservative oxygen strategy, characterized by maintaining a target SpO2 range of 88% to 92%, and a liberal oxygen strategy, striving for a target SpO2 of ≥96%, for the maintenance of pulsatile saturation levels. 30 Remarkably, patients subjected to the lower target oxygen levels exhibited escalated mortality rates of 8% and 14% at days 28 and 90, respectively. Hence, while respiratory support stands as a pivotal therapeutic approach to reduce sepsis-related mortality, concomitant attention must be directed towards the timely identification and management of secondary infections to improve patient prognosis.
This study possesses several limitations. Firstly, it relies on data derived from a single-center observational database, thus the findings presented in our study can only be regarded as a point of reference and necessitate further validation. To comprehensively investigate the clinical characteristics, risk factors, and impact on prognosis of secondary infection in sepsis, additional high-quality randomized trials with larger sample sizes are indispensable. Secondly, this article lacks a specific analysis of the localization of secondary infection, the specific microbial flora involved, and its implications on patient outcomes. For instance, factors such as the duration of hospitalization following a subsequent infection, rates of ICU readmission, and mortality rates have not been thoroughly examined. Given the current focus on delineating the risk factors associated with secondary infection in patients, conducting more detailed research to evaluate key aspects of secondary infection is imperative.
Conclusion
The nomogram devised in this study has affirmed that septic individuals exhibiting diminished oxygen saturation, decreased lymphocyte proportions, reduced platelet counts, elevated neutrophil proportions, heightened lactic acid levels, and necessitating invasive mechanical ventilation within the initial 24 h of ICU admission manifest a heightened susceptibility to secondary infections during their ICU stay in comparison to their counterparts. Consequently, following the establishment of this personalized predictive model, the potential for secondary infections among sepsis patients admitted to the ICU can be individually assessed.
Footnotes
Ethical Considerations
All the data were obtained from The Medical Information Mark for Intensive Care (MIMIC) database. The database was acquired with the approval of the Institutional Review Board (IRB) at both the Massachusetts Institute of Technology and Beth Israel Deaconess Medical Center. . It is an open access database containing no personal information of the registrants. As a result, the need for obtaining informed consent from patients was waived. Access to this repository is predicated upon the successful completion of an institution-specific training program examination, and we extracted data in strict accordance with its regulations. Therefore, ethical approval and informed consent are not required.
Author Contribution
Writing - original draft preparation: Jiaqi Lu, Guangzhi Shi; Writing - review and editing: Jiaheng Zhang, Shuai Liu, Guangzhi Shi; Conceptualization: Shuai Liu; Methodology: Jiaqi Lu, Jiaheng Zhang; Formal analysis and investigation: Jiaqi Lu, Shuai Liu; Resources: Guangzhi Shi; Supervision: Guangzhi Shi. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
