Abstract
Background
ICU patients have a high incidence of VTE. The American College of Chest Physicians antithrombotic practice guidelines recommend assessing the risk of VTE in all ICU patients. Although several VTE risk assessment tools exist to evaluate the risk factors among hospitalized patients, there is no validated tool specifically for assessing the risk of VTE in ICU patients.
Methods
A retrospective corhort study was conducted between June 2018 and October 2022. We obtained data from the electronic medical records of patients with a variety of diagnoses admitted to a mixed ICU. Multivariable logistic regression analysis was used to evaluate the independent risk factors of VTE. Receiver operating characteristic (ROC) curves were used to analyse the predictive accuracy of different tools.
Results
A total of 566 patients were included, and VTE occurred in 89 patients (15.7%), 62.9% was asymptomatic VTE. A prediction model (the ICU-VTE prediction model) was derived from the independent risk factors identified using multivariate analysis. The ICU-VTE prediction model included eight independent risk factors: history of VTE (3 points), immobilization ≥4 days (3 points), multiple trauma (3 points), age ≥70 years (2 points), platelet count >250 × 103/μL (2 points), central venous catheterization (1 point), invasive mechanical ventilation (1 point), and respiratory failure or heart failure (1 point). Patients with a score of 0–4 points had a low (1.81%) risk of VTE. Patients were at intermediate risk, scoring 5–6 points, and the overall incidence of VTE in the intermediate-risk category was 17.1% (odds ratio [OR], 11.1; 95% confidence interval [CI], 4.2-29.4). Those with a score ≥7 points had a high (44.1%) risk of VTE (OR, 42.6; 95% CI, 16.4-110.3). The area under the curve (AUC) of the ICU-VTE prediction model was 0.838, and the differences in the AUCs were statistically significant between the ICU-VTE prediction model and the other three tools (ICU-VTE score, Z = 3.723, P < 0.001; Caprini risk assessment model, Z = 6.212, P < 0.001; Padua prediction score, Z = 7.120, P < 0.001).
Conclusions
We identified eight independent risk factors for acquired VTE among hospitalized patients in the ICU, deriving a new ICU-VTE risk assessment model. The model aims to predict asymptomatic VTE in ICU patients. The new model has higher predictive accuracy than the current tools. A prospective study is required for external validation of the tool and risk stratification in ICU patients.
Keywords
Introduction
Venous thromboembolism (VTE) is a group of diseases, including deep vein thrombosis (DVT) and pulmonary embolism (PE). It is the third most common vascular disease after acute coronary syndrome and stroke, and the fatality rate is second only to tumors and myocardial infarction.1,2 Intensive care unit (ICU) patients are at high risk for VTE due to many risk factors, such as immobilization, vascular injury, and hypercoagulability. The risk of VTE was significantly increased in ICU patients, especially those receiving invasive mechanical ventilation, compared to that in general inpatients. 3 The incidence of VTE occurrence within ICU as high as 1.4% to 10.5% from 2007 to 2018.4–6 Due to the lack of specific clinical manifestations, the missed diagnosis rate of VTE is high, especially in asymptomatic VTE. 7 The American College of Chest Physicians antithrombotic practice guideline for the prevention of VTE clearly states that all critically ill patients should be assessed for the risk of VTE, and those at high risk should receive preventive treatment.8,9 Therefore, the selection of effective risk assessment tools to screen high-risk patients and taking preventive measures to reduce the incidence of VTE is critical.
The commonly used VTE risk assessment tools among hospitalized patients include the International Medical Prevention Registry on Venous Thromboembolism (IMPROVE), 10 Caprini risk assessment model (Caprini RAM), 11 and Padua prediction score (PPS). 12 However, there is no validated tool specifically for assessing the risk of VTE in ICU patients. The risk of VTE in medical patients, both during hospitalization and up to 90 days after discharge, can be assessed using the IMPROVE. The PPS is used to assess the risk of VTE in medical patients within 90 days of admission. The Caprini RAM is mainly used for surgical patients and its use in ICU patients has been reported in previous studies.13,14 However, its scoring category is not suitable for ICU patients, as based on this scoring category, the majority of ICU patients will be classified as high-risk Viarasilpa et al 4 developed an ICU-VTE score in 2020, which mainly predicted symptomatic VTE in critically ill patients. Although the ICU-VTE score is based on a large sample and special assessment tool for ICU patients, there may be some limitations. Asymptomatic VTE was not considered, ICU-specific risk factors such as the Acute Physiology and Chronic Health Evaluation II (APACHE II) score, sepsis, and vasoactive medication were not considered as potential risk factors during the development process, and external verification was not carried out. Therefore, the purpose of this study was to combine the risk factors in the current VTE assessment tools with an evidence-based study on risk factors in ICU patients, identify the independent risk factors, and develop a specific and easy-to-use ICU-VTE prediction model for patients during ICU admission.
Methods
Study Design
A retrospective corhort study was conducted to identify the risk factors for VTE among hospitalized patients in the ICU and to develop a predictive model.
Setting and Participants
All data of patients admitted to one ICU (30 beds) between June 2018 and October 2022 were collected at the First Affiliated Hospital of Chongqing Medical University. Inclusion criteria were age ≥18 years and ICU length of stay ≥24 h. Exclusion criteria were acute PE or DVT occurring in the hospital before or ICU admission, and incomplete patient data (eg, lack of color Doppler ultrasound). In the present study, VTE included acute incident DVT (either in the upper or lower extremity), PE, or both, and was diagnosed with duplex venous ultrasonography, venous angiography, or computed tomography pulmonary angiography. These approaches have been reported in our previous study. 15 Doppler ultrasounds was part of routine care during ICU stay and performed within 48 h of ICU admission, 7 days apart, the patients were discharged from ICU or the patients had significant symptoms. The lack of color Doppler ultrasound meant that patients who were not screened within 48 h of admission, within a 7-day interval, or at the time of discharge before being diagnosed with VTE.
Data collected included demographic information (age, sex, diagnosis, APACHE II score, and type 2 diabetes); laboratory examination results (activated partial thromboplastin time, creatinine, and D-dimer); ICU-specific factors reported in a high-quality systematic review and meta-analysis 16 (sepsis, central venous catheterization [CVC], invasive mechanical ventilation, and vasoactive medication), PPS (including 11 risk factors), 12 Caprini RAM (including 40 risk factors), 11 and ICU-VTE score (including 6 risk factors). 4 Other risk factors included the duration of mechanical ventilation, and ICU length of stay. Outcome assessments included VTE, and mortality. Past history of VTE was defined as VTE that occurred before hospital admission. Immobilization was defined as patients who were confined to bed, had severely limited ability to walk, or could not bear their own weight and must be assisted in a chair or wheelchair (Braden activity score <3) in the ICU.4,17 Platelet count >250 × 103/µL upon ICU admission and the lowest haemoglobin level ≥90 g/L during hospitalization in the ICU were defined as risk factors based on the study by Viarasilpa et al 4 CVC was defined as single-, double-, or triple- lumen dialysis or tunnel catheters inserted into the internal jugular, subclavian, or femoral vein and peripheral insertion of a central catheter (PICC). 17 Anticoagulant prophylaxis was defined as the administration of at least one prophylactic dose of low molecular weight heparin, or direct acting oral anticoagulant before VTE diagnosis. Patients requiring full dose therapeutic anticoagulation were not included into the analysis. Data were collected by two researchers to ensure accuracy and completeness. In the case of any dispute, the study designer would discuss and make the final decision. Ethical approval was obtained from the Ethics Committee of the First Affiliated Hospital of Chongqing Medical University (Document No. 2021-735). This was a retrospective corhort study and we did not provide informed consent to the patients included in this study. All data were anonymize, and there was no significant risk.
Statistical Analysis
All data were analyzed using SPSS (version 22 (IBM Corp, Armonk, NY). Descriptive analysis is reported as percentage, mean ± standard deviation, or median (interquartile spacing). The influence factors in patients with and without VTE were compared using the t-test, Wilcoxon rank-sum tests, and chi-square tests. Multivariable logistic regression analysis (variable screening method: backward: Wald, variable inclusion criterion α = 0.05, and elimination criterion 0.1) was used to evaluate the independent factors of VTE. The ICU-VTE prediction model was developed from the statistically significant predictors obtained using multivariate analysis (P < 0.05). We assigned a point score for each variable according to the value of the regression coefficient (β) of the model. After the final prediction model was generated using the entire study sample as a derivation cohort, we performed internal validation to assess stability by applying the score to a randomly selected subset of the derivation cohort (30%). Receiver operating characteristic (ROC) curves were used to assess internal validation and compare the ICU-VTE prediction model with the ICU-VTE scores, Caprini RAM, and PPS. We divided each of the ICU-VTE prediction model scores into three groups (low, intermediate, and high-risk), and the chi-square tests were used to calculate the odds ratio (OR) with 95% confidence interval (CI) using the low-risk group as the control.
Results
Demographics
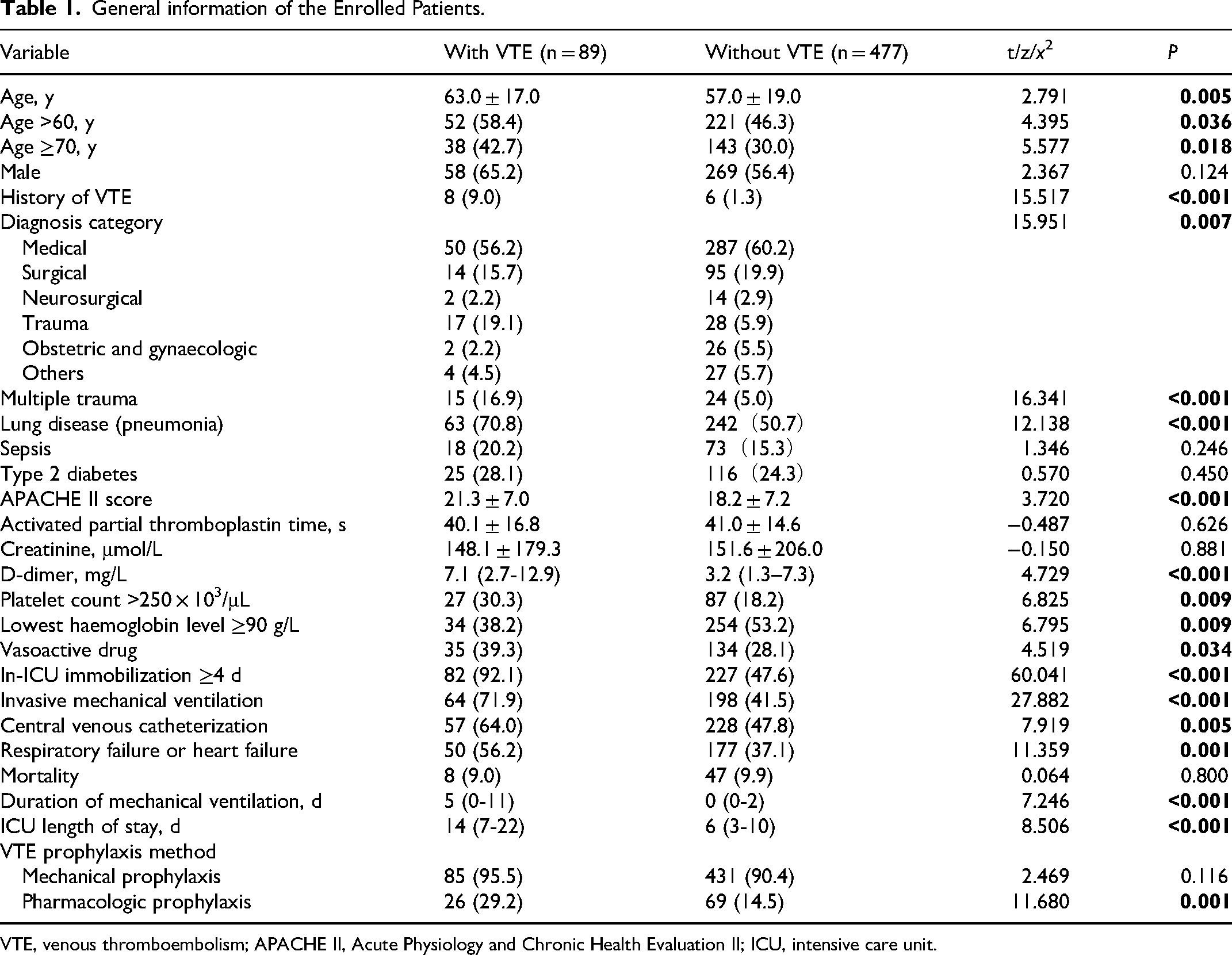
Of the 878 ICU patients screened between June 2018 and October 2022, 312 patients who met the exclusion criteria were excluded, leaving 566 patients in the final analysis (Fig. 1). The age distribution of the 566 patients ranged from 18 to 97 years, with an average of 57.9 ± 18.8 years, and 327 patients (57.8%) were male. Of the included patients, 89 (15.7%) were diagnosed with VTE, including 82 (92.1%) cases of DVT in the lower limbs, 2 (2.3%) cases of DVT in the upper limbs, 2 (2.3%) cases of DVT in the upper and lower limbs, and 3 (3.3%) cases of PE. Of the 82 patients with DVT in the lower limbs, 70 (85.4%) had distal DVT. Mechanical prophylaxis for VTE was given to 91.2% of the overall study population, while the proportion of pharmacologic prophylaxis was 16.8%. The days from ICU admission to VTE diagnosis ranged from 2 to 51 days, with an average of 7.9 days. The proportion of asymptomatic and symptomatic VTE patients was 62.9% and 37.1%, respectively. (data not shown). The maximum number of patients diagnosed with VTE was 57 within 2–7 days of ICU stay, and 82 within 14 days, accounting for 92.1%. (Fig. 2). Patients who developed VTE had longer ICU length of stay and duration of mechanical ventilation than those who did not but had similar mortality rates (Table 1). The patient characteristics and outcomes of the validation cohort were similar to those of the overall cohort (Supplemental Table 1). Missing data were provided in Supplemental Table 2.
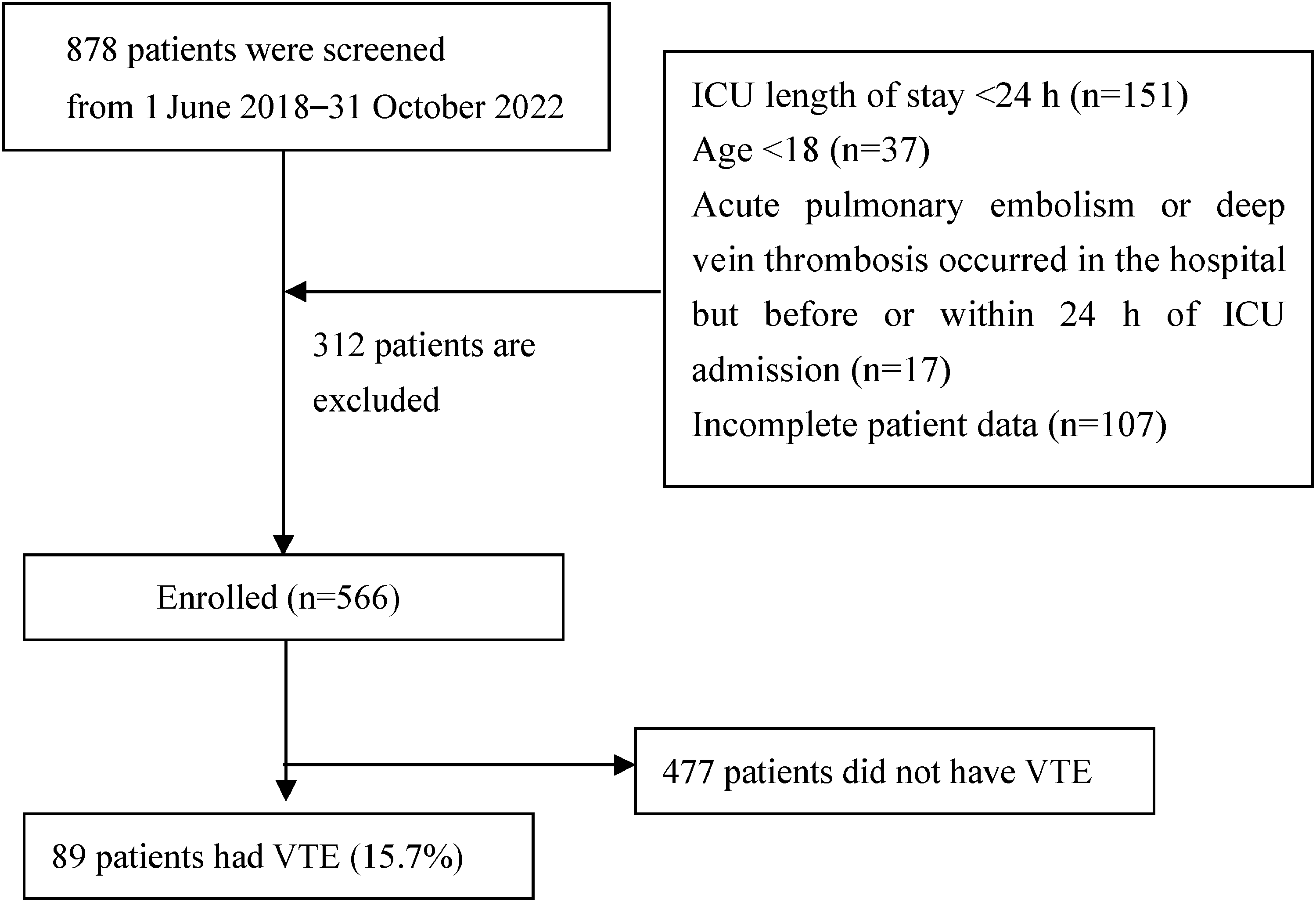
Consort flow diagram of the study procedure.
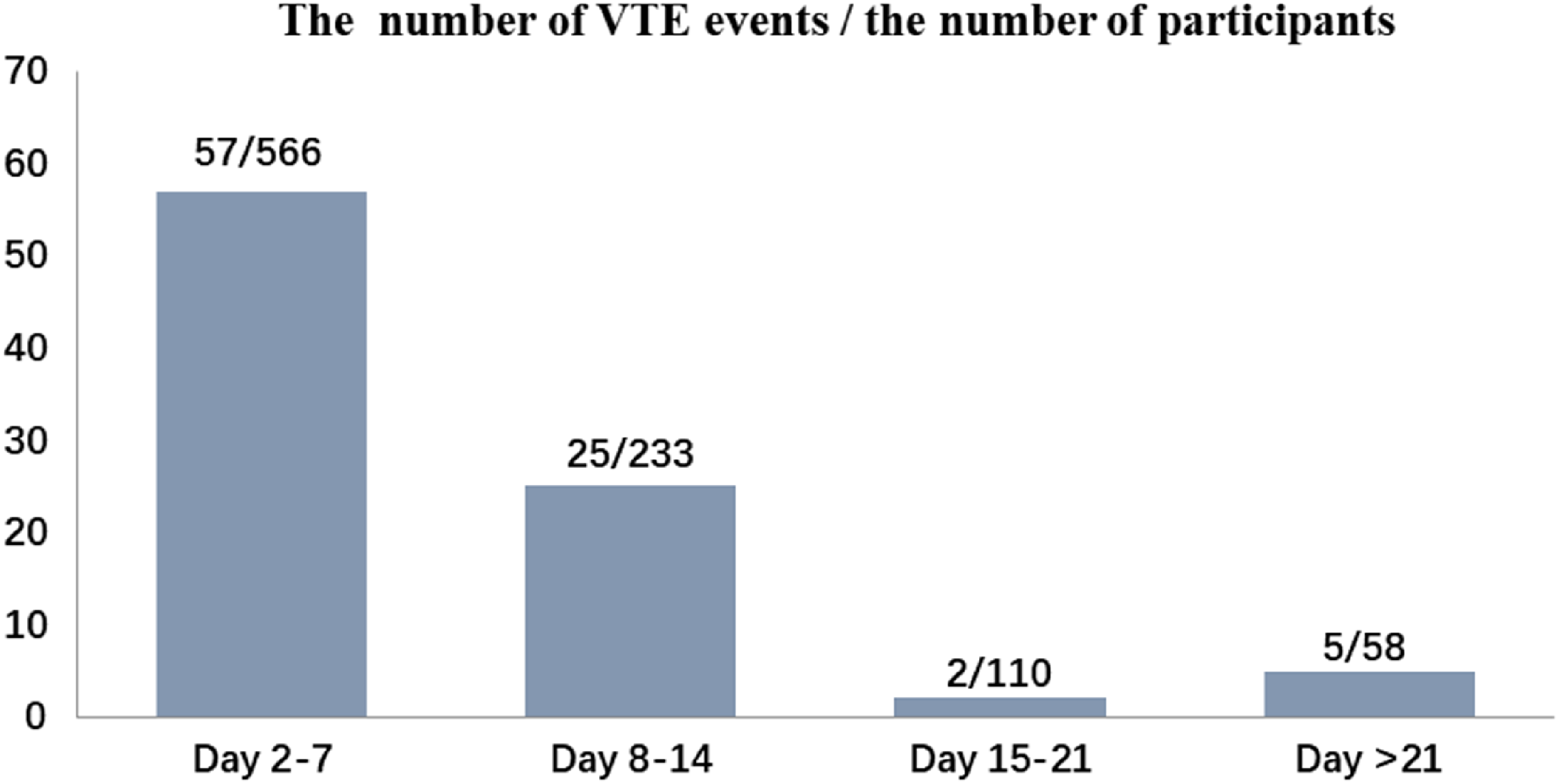
The actual number of VTE events occurring during each time-interval.
General information of the Enrolled Patients.
VTE, venous thromboembolism; APACHE II, Acute Physiology and Chronic Health Evaluation II; ICU, intensive care unit.
Predictors of VTE in ICU Patients
In the univariate analysis, factors including age (>60 years and ≥70 years), history of VTE, diagnosis category, multiple trauma, lung disease (pneumonia), APACHE II score, D-dimer, platelet count >250 × 103/µL, the lowest haemoglobin level ≥90 g/L, vasoactive drug use, immobilization ≥4 days, invasive mechanical ventilation, respiratory failure or heart failure, CVC, and ICU length of stay were found to be statistically different and might be associated with VTE (Table 1).
Table 2 shows the results of the multi-variable analysis. Fifteen potential risk factors of VTE from factors that were statistically different in uni-variate analysis were analyzed using a multi-variable logistic regression model. ICU length of stay was not analyzed as a potential risk factor, as over 90% of patients were diagnosed with VTE within 14 days of ICU hospitalization. In the final model, eight predictors of VTE were identified: immobilization ≥4 days (OR 7.46; 95% CI, 3.21-17.31), history of VTE (OR 7.39; 95% CI, 2.16-25.28), multiple trauma (OR 4.41; 95% CI, 1.93-10.07), age ≥70 years (OR 2.42; 95% CI, 1.38-4.23), platelet count >250 × 103/µL (OR 2.38; 95% CI, 1.32-4.29), invasive mechanical ventilation (OR 2.31; 95% CI, 1.32-4.01), CVC (OR 1.92; 95% CI, 1.10-3.35), and respiratory failure or heart failure (OR 1.78; 95% CI, 1.03-3.07).
Multivariate Analysis of Risk Factors in ICU Patients.

S.E, standard error; CI, confidence interval; VTE, venous thromboembolism; ICU, intensive care unit.
Development of the ICU-VTE Prediction Model
Based on the results of the multi-variable analysis, we developed an ICU-VTE prediction model consisting of eight predictors. The score of each factor in this model was 0–3 points, and the total score was 0–16 points. Based on the incidence of VTE in different scoring categories, we divided 0–4 points into low risk, 5–6 points into intermediate risk, and ≥7 points into high risk categories, in which the incidence of VTE was 1.8%, 17.1%, and 44.1%, respectively.The rates of drug prophylaxis in the low risk, intermediate risk, and high-risk were 8.7%, 20.1%, and 33.1%, respectively(data not shown). In the present study, approximately half of all ICU patients were at low risk, and with low risk as the control, the OR values of patients at moderate and high-risk for VTE were 11.0 and 42.6 times higher, respectively (Table 3, Supplemental Table 3). The risk of VTE for each risk category for validation cohort was shown in Supplemental Table 4.
ICU-VTE Prediction Model.
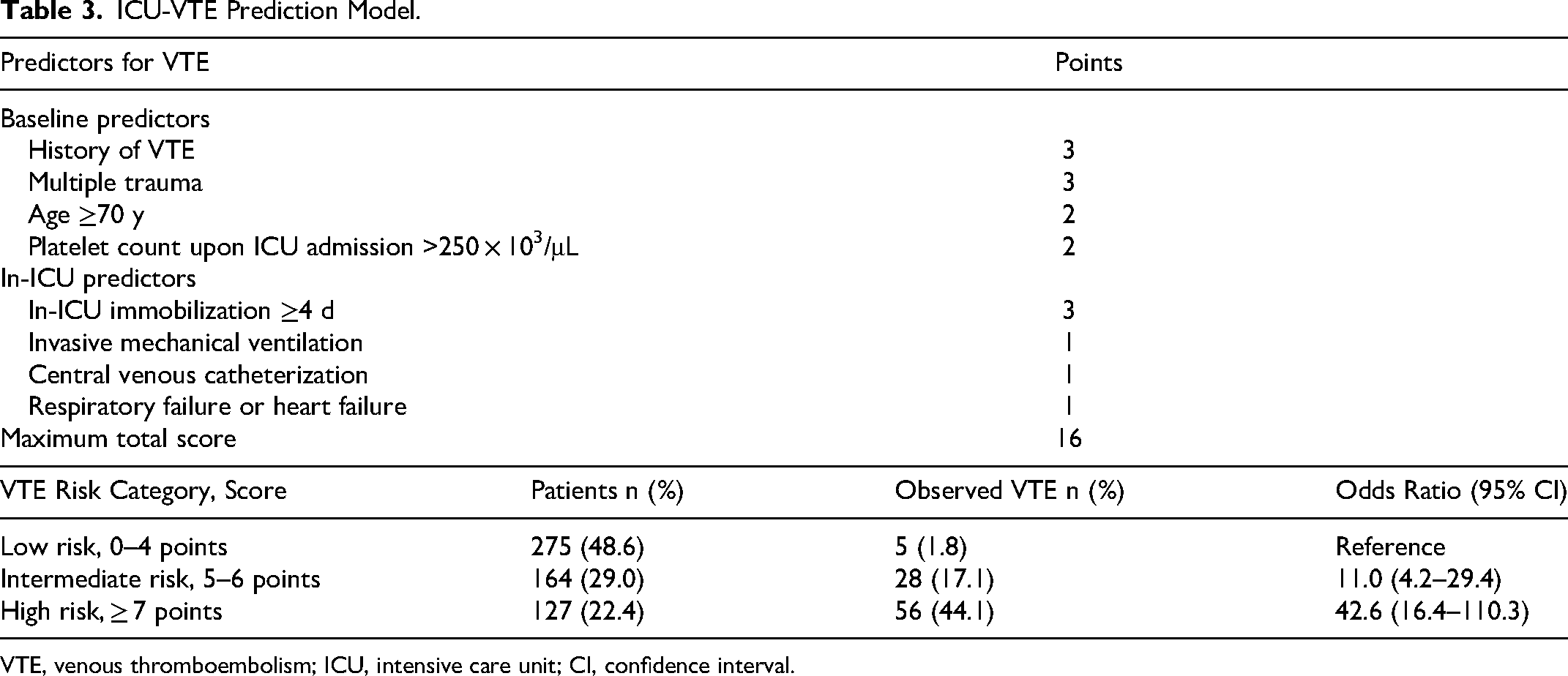
VTE, venous thromboembolism; ICU, intensive care unit; CI, confidence interval.
Validation of the ICU-VTE Prediction Model
A total of 161 patients were randomly selected for the model verification. The ROC curve showed that the AUC of the ICU-VTE prediction model in the validation cohort was 0.87 (95% CI, 0.808-0.918), indicating a high predictive value with a slightly higher AUC than that for the entire cohort (Fig. 3).
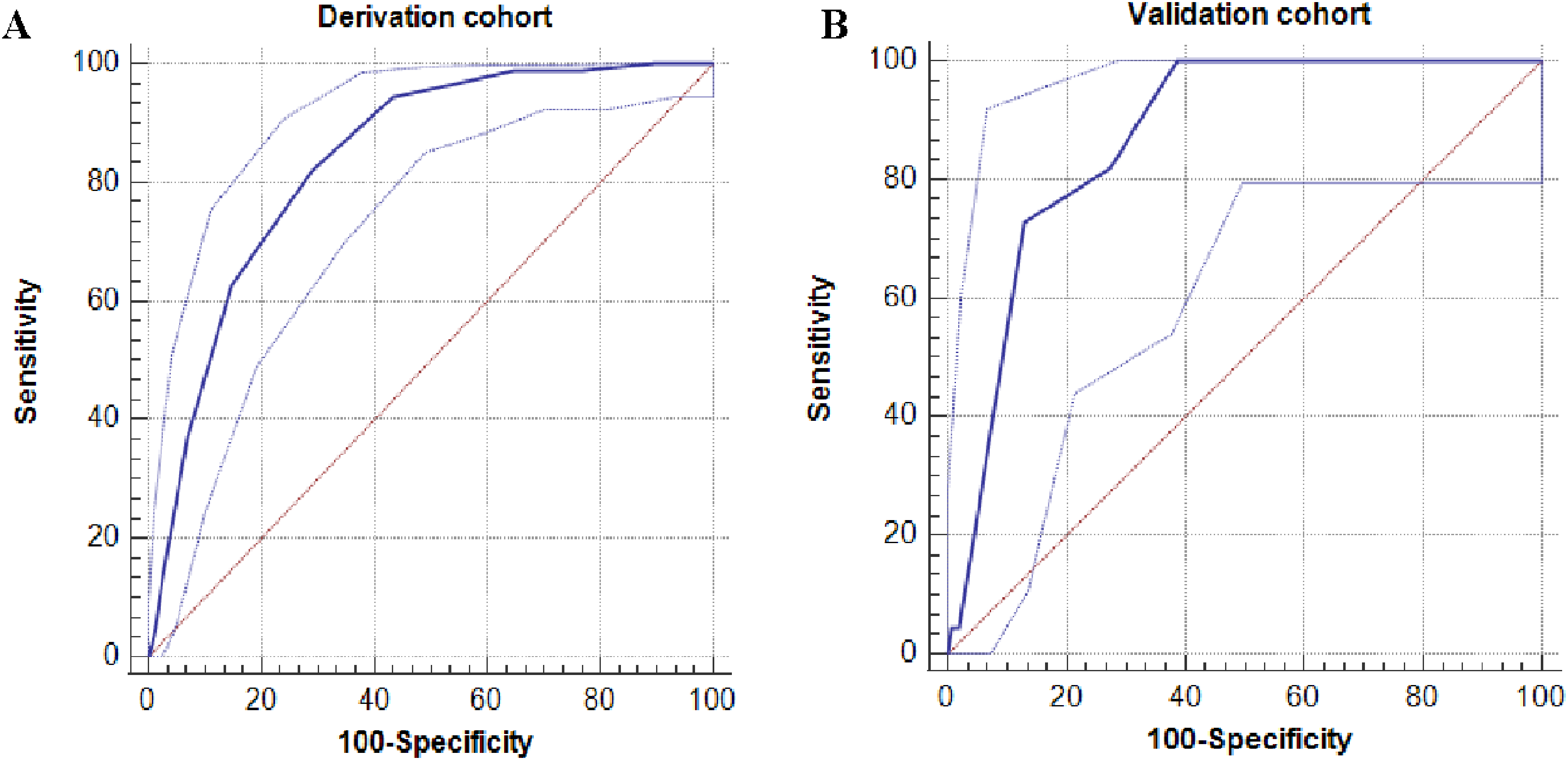
Area under the receiver operating characteristic curve for the derivation cohort is 0.83 (95% ci, 0.799-0.877) (A) and for the validation cohort is 0.87 (95% CI, 0.808–0.918) (B). CI, confidence interval.
We also conducted a comparative analysis of the four tools (ICU-VTE prediction model, ICU-VTE score, Caprini RAM, and PPS) and found that the scores of patients with VTE were higher than those of patients without VTE in all tools. On comparing the area under the curve (AUC) of the four tools, the new ICU-VTE prediction model had the maximum AUC of 0.838, while the PPS had the minimum AUC of 0.626; the differences were statistically significant between the ICU-VTE prediction model and the other three tools (ICU-VTE score, Z = 3.723, P < 0.001; Caprini RAM, Z = 6.212, P < 0.001; PPS, Z = 7.120, P < 0.001). This means that the ICU-VTE prediction model had a better predictive accuracy (Fig. 4, Table 4). The AUC for the model associated with symptomatic VTE was 0.824 (0.769, 0.878) (Supplemental Fig. 1).
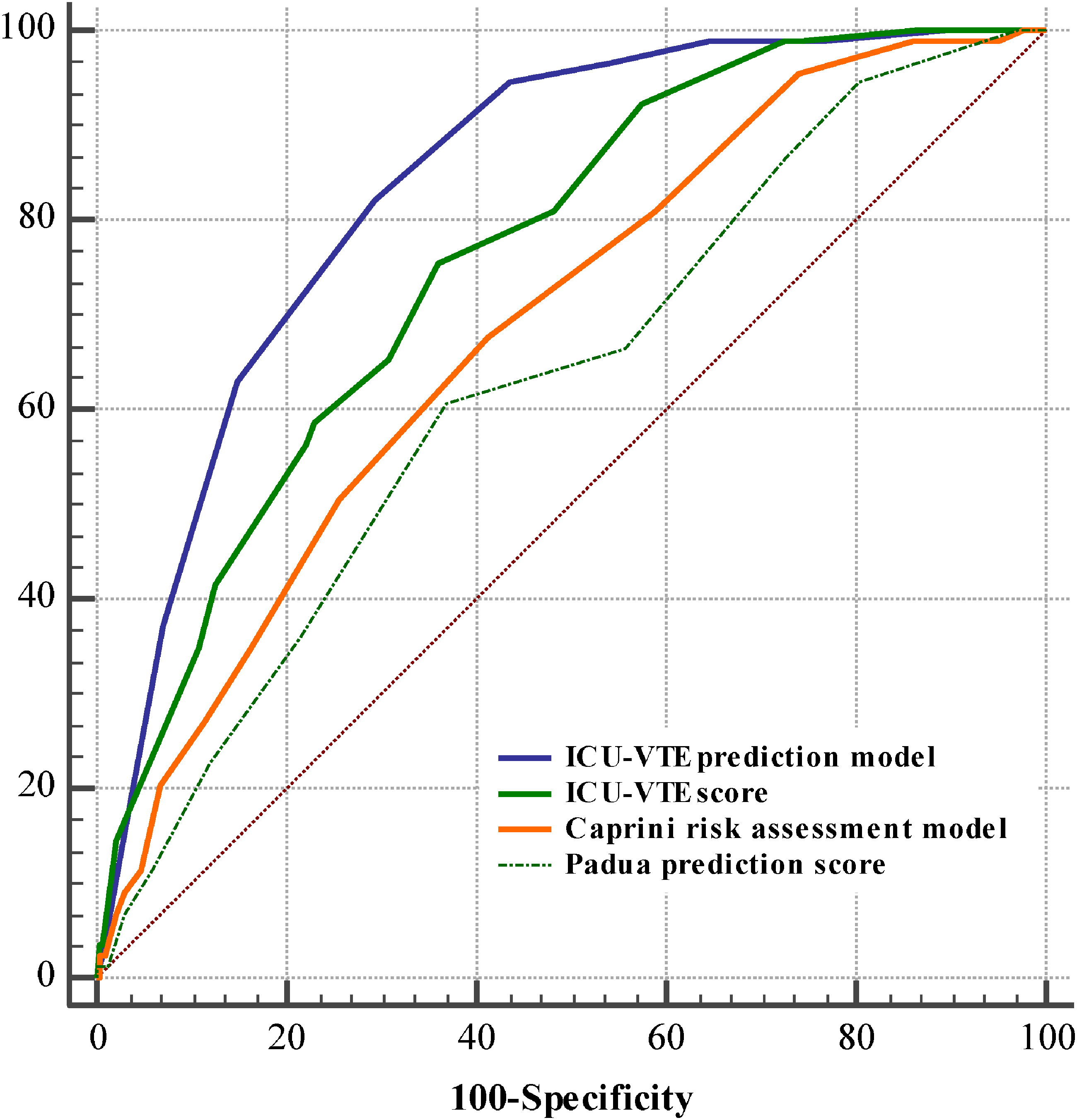
Area under the receiver operating characteristic curves for four VTE assessment tools VTE, venous thromboembolism; ICU, intensive care unit.
Comparison of the AUC Values for Different Prediction Tools.
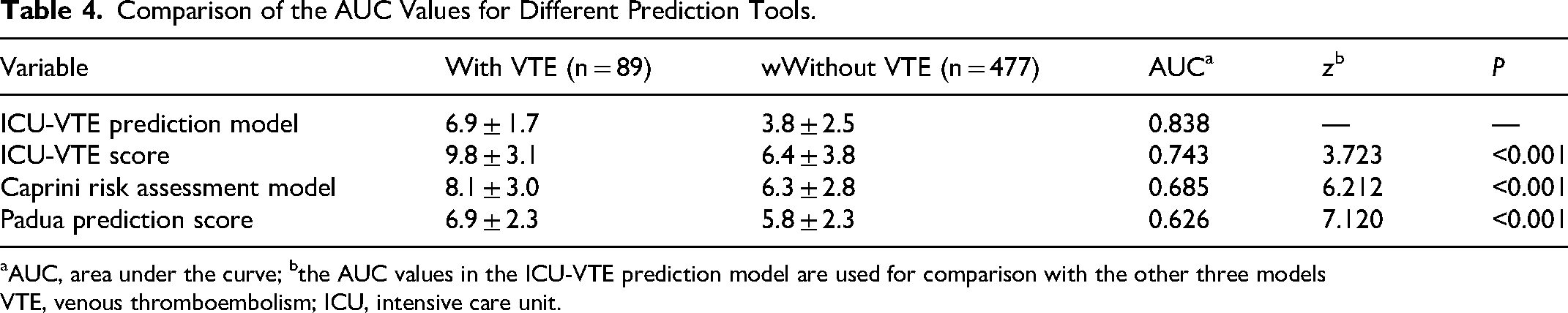
AUC, area under the curve; bthe AUC values in the ICU-VTE prediction model are used for comparison with the other three models
VTE, venous thromboembolism; ICU, intensive care unit.
Discussion
This retrospective corhort study found that the incidence of VTE was 15.7%, with lower extremity DVT accounting for 90%. More than half of the patients were diagnosed with VTE within 2–7 days of ICU admission and more than 90% of patients were diagnosed within 14 days of ICU admission, which was similar to the findings of previous studies.4,18 According to the analysis of influencing factors, eight predictors were identified, and the ICU-VTE prediction model was developed on this basis. The ICU-VTE prediction model has high predictive accuracy based on internal verification and comparison with other tools.
The ICU-VTE prediction model in the present study included eight independent risk factors at least four of which were similar to ICU-VTE score, the PPS, or Caprini RAM. Invasive mechanical ventilation as an independent risk factor is a common treatment for ICU patients. In the present study, the percentage of patients undergoing invasive mechanical ventilation among patients with and without VTE was 71.9% and 41.5%, respectively, which was consistent with a previous study. 4 A systematic review and meta-analysis of a large sample of 729,477 patients showed that invasive mechanical ventilation was a specific risk factor for VTE in ICU patients with an OR of 1.74. 16 Gupta et al 19 found that the incidence of DVT in mechanically ventilated patients was higher than that in the group without mechanical ventilation, even though anticoagulant drugs were administered. When mechanical ventilation is used in critically ill patients, the invasive operation destroys the integrity of the vascular wall; thus, the negative thoracic pressure under the normal physiological state is transformed into positive thoracic pressure, which affects the return blood volume and increases the risk of VTE. 20 Another explanation is that mechanical ventilation may lead to pulmonary and systemic coagulation dysfunction. 21 In addition, invasive mechanical ventilation may lead to immobility; therefore, early mobilization, including in-bed and ambulation activities, should be performed in these patients. 22 Patency considers that low platelet counts are a protective factor, while high platelet counts can easily increase the risk of VTE. Few studies have reported elevated platelet count as a risk factor for VTE in ICU patients, but more often in patients with tumors.23,24 Three studies reported elevated platelet count as a risk factor for VTE in ICU patients, with results consistent with those in the two studies of patients. Two of these studies defined a platelet count >250 × 103/µL4,25 as a risk factor, while the other defined a platelet count >166 × 103/µL as a risk factor. 26 The latter study may have inadvertently reported the average platelet count, since the mean platelet count of 170 × 103/µL is greater than 166 × 103/µL, even in patients without VTE in the present study.
Based on the distribution of the ICU-VTE prediction model scores and incidence of VTE, we found that these scores were clearly divided into three categories. Low-risk patients with scores of 0–4 points had an overall 1.81% rate of VTE. Patients were at intermediate risk, scoring 5–6 points, and the overall incidence of VTE was 17.1%, which was 11.1 times the risk of VTE among low-risk patients. In clinical nursing evaluation, patients aged ≥70 years who have multiple injuries, immobilized for ≥4 days, or a history of VTE are automatically assigned to the intermediate-risk group. The high-risk patients with a score of 7 or above, and the overall incidence of VTE was up to 44.1%. Compared with other common VTE assessment tools, the new VTE prediction model is an ICU-specific and accurate risk assessment model. The ICU-VTE score was designed to evaluate symptomatic VTE in ICU patients.The score consists of the following six risk factors: CVC, immobilization ≥4 days, prior history of VTE, invasive mechanical ventilation, the lowest haemoglobin level ≥90 g/L, and platelet count >250 × 103/µL. The AUC of the model was 0.87 for symptomatic VTE is higher than that in our model. 4 However, when we used it to predict symptomatic or asymptomatic VTE, the AUC was 0.743 for the ICU-VTE score, compared with 0.838 for the new ICU-VTE prediction model. The other two tools used to assess the risk factors of VTE in ICU patients are the PPS and Caprini RAM.14,27 The PPS includes 11 risk factors, with a score of ≥4 points classified as high-risk, and is used primarily in medical patients. 12 The Caprini RAM contains 40 risk factors, and high risk is defined as a score ≥5 points, which is used in surgical patients. 11 In recent years researchers have also used Caprini RAM for medical patients.28–30 Therefore, we also used the Caprini score to evaluate medical patients and compare them with the new model. The AUC of the new ICU-VTE prediction model was higher than that of the Caprini RAM (AUC = 0.685) and PPS (AUC = 0.626). In a similar study, the AUC of Caprini RAM was 0.709 and 0.616 for PPS in ICU non-surgical patients. 31
It is worth noting that the low proportion of patients on pharmacological prophylaxis in the present study may lowers the external validity of the study and this needs to be further validated in future studies. According to the guidelines of the American College of Chest Physicians and the American Society of Haematology, thromboprophylaxis should be undertaken for ICU patients. Physical prophylaxis is preferred in patients with a higher risk of bleeding, while pharmacological prophylaxis should be performed when the risk of bleeding is reduced.32,33 The proportion of patients in this study who received drug prophylaxis was 16.8%, much lower than the 94% of patients with VTE and 79% of patients without VTE reported by Viarasilpa et al 4 This may be related to the fact that the doctor neglects the drug anticoagulation to prevent VTE for fear of causing bleeding. Proportion of drug prophylaxis in this study is similar to a multi-center, observational study in which only 199 (21.2%) of 940 ICU patients from 54 hospitals receive drug prophylaxis in China. 34 Other studies show that pharmacological prophylaxis may be delayed, interrupted, or withheld because of concerns about bleeding and the need for invasive procedures.35,36 In addition, the definition of patients received pharmacological prophylaxis was during ICU stay or before VTE diagnosis, rather than during the entire hospital stay in the present study.
Moreover, the majority of VTE cases were screening-detected asymptomatic VTE in the present study. Ultrasound screening of high-risk or traumatized ICU patients may be a cost-effective method for early detection of DVT and prevention of fatal PE.37,38 The cost-effectiveness of full-dose anticoagulant therapy for patients with asymptomatic distal DVT was unknown. In a study of non-ICU patients, patients with cancer-associated distal DVT were found to have no differences in mortality and VTE recurrence rates with respect to cancer-associated proximal DVT, except for the incidence of fatal pulmonary embolism. 39 In addition, a meta-analysis showed that patients with distal DVT who received anticoagulant therapy with VKA benefited with a lower risk of thrombosis recurrence and little difference in major bleeding events, although there was an increase in clinically relevant non-major bleeding events and no difference in the incidence of pulmonary embolism compared with no intervention or placebo. 40 Therefore, the future study needs to explore whether there is a net-benefit to treating asymptomatic distal DVT screened by Ultrasound among the reduction in mortality, thrombotic recurrence, and symptomatic pulmonary embolism offsets the bleeding risk associated with full-dose anticoagulant therapy.
Our findings have several implications for clinical practice. ICU patients have a high incidence of VTE, and ICU-specific risk factors should be considered in risk assessment tools. Therefore, an ICU-specific VTE assessment tool is necessary. Our model meets this need and can assist ICU physicians in clinical decision-making and monitor observed and expected VTE rates as an indicator of department or hospital quality improvement. In the ICU-VTE prediction model, all eight risk factors can be automatically captured from the nursing system or electronic medical record system to generate real-time assessment scores, and the risk level of the current patient can be displayed. In the future, the risk level can be correlated with corresponding measures to provide doctors with direct reminders and assessments. Early active mobilization should be performed in patients with invasive mechanical ventilation without activity contraindications, CVC should be minimized or early extraction promoted, and physical or chemical preventive measures may be considered for patients with multiple injuries. Finally, in high-risk patients, ultrasound screening should be performed to detect DVT in time and prevent fatal PE.
The strengths of this study include the fact that the ICU-VTE prediction model can reflect the risk factors of VTE in ICU patients well. The risk factors in the current VTE assessment tools, such as the Caprini RAM, PPS, and ICU-VTE score were incorporated if they were found to be potential factors in ICU patients. The ICU-specific risk factors in the meta-analysis of large samples were also considered. In addition, internal verification and comparisons with the other three tools were used to verify the predictive accuracy of our model. This study had several limitations. Our sample size is small compared to that used to create the other VTE risk assessment tools (ICU-VTE score or Caprini RAM), and the data from one hospital are not representative enough. Moreover, this study is a retrospective study, and there may be information omissions; thus, it cannot be ruled out that the information does not fully reflect the patient's situation at that time. Furthermore, we only obtained data on VTE events during ICU admission, not after discharge from the ICU or hospital; hence, our model is limited to patients during ICU admission. Finally, although our model has high predictive accuracy in predicting symptomatic or asymptomatic VTE compared with the other three tools, we only conducted internal verification, and external verification was not performed. Therefore, in future studies, data from different hospitals will be selected for external verification.
In summary, ICU patients are still at high risk of VTE, despite taking physical or chemical preventive measures according to the guidelines. The ICU-VTE prediction model can provide accurate VTE risk stratification among ICU patients. Further studies will conduct prospective external verification in different ICUs in other hospitals. The effect of the proportion of enrolled patients receiving drug prophylaxis on the predicted value and whether there is a net-benefit to treating asymptomatic distal DVT. Moreover, software will be developed to embed the model into electronic systems and dynamically evaluate patients by automatically capturing data from electronic medical records and nursing systems.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241280624 - Supplemental material for Development of a Risk Assessment Tool for Venous Thromboembolism among Hospitalized Patients in the ICU
Supplemental material, sj-docx-1-cat-10.1177_10760296241280624 for Development of a Risk Assessment Tool for Venous Thromboembolism among Hospitalized Patients in the ICU by Chuanlin Zhang, Jie Mi, Xueqin Wang, Ruiying Gan, Xinyi Luo, Zhi Nie, Xiaoya Chen and Zeju Zhang in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Author Contributions
Zeju, Zhang analyzed the data. Chuanlin, Zhang and Jie, Mi designed the study and wrote the manuscript. Xueqin, Wang revised the manuscript. Ruiying, Gan; Xinyi, Luo; Zhi, Nie; and Xiaoya Chen collected the data. All the authors read and approved the final manuscript. Jie, Mi and Zeju, Zhang contributed equally to this article.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
Ethical approval was obtained from the Ethics Committee of the First Affiliated Hospital of Chongqing Medical University (Document No. 2021-735). Written informed consent was not required because all data were anonymised, and there was no significant risk.
Funding
This work was supported by The First Affiliated Hospital of Chongqing Medical University, Grant/Award Number: (PYJJ2019-228, and HLJJ2022-08) and School of Nursing, Chongqing Medical University, Grant/Award Number:(20230404), and Nursing Research Innovation Project of The First Affiliated Hospital of Chongqing Medical University, Grant/Award Number: (HLYB2024-07).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
