Abstract
Objective
This study examines the clinical features and risk factors for postoperative venous thromboembolism in craniotomy patients with intracranial tumors.
Methods
In this study, we conducted an analysis using clinical data extracted from the Medical Information Mark for Intensive Care (MIMIC) database. Clinical data from the first day of ICU admission were utilized for our analysis. We employed univariate and multivariate logistic regression analyses to examine the factors influencing the outcome. A nomogram, constructed using the R software, was developed to predict the risk of secondary thromboembolism.
Results
A total of 728 eligible patients were included in this study, among whom the incidence of postoperative venous thromboembolism after craniotomy was found to be 19.2%. The age of the patients, along with their initial Glasgow Coma Scale score and prothrombin time, were identified as independent potential risk factors for the occurrence of venous thromboembolism in patients with intracranial tumors. These risk factors were incorporated into the predictive model, and a nomogram was constructed using the R software. By further conducting receiver operating characteristic curve analysis, the model exhibited an area under the curve of 0.7463 (95% CI 0.7056-0.7932). Simultaneously, we employed repeated sampling to construct a calibration curve, which demonstrated excellent model fit.
Conclusion
Patient age, Glasgow Coma Scale score, and prothrombin time have been identified as potential independent risk factors that may influence the occurrence of venous thromboembolism in patients with intracranial tumors.
Keywords
Introduction
Venous thromboembolism (VTE)—comprising deep-vein thrombosis (DVT) and pulmonary embolism (PE)—imposes a substantial global health burden. Its pathogenesis is driven by multiple, often co-existing, risk factors; indeed, most affected patients present with more than one. 1 Despite venous thromboembolism being a preventable cause of morbidity and mortality among critically ill patients, its awareness and recognition are often insufficient. 2 Venous thromboembolism is a frequent complication of intracranial tumors; even after adjusting for age and sex, brain tumors rank among the malignancies with the highest thrombosis risk. 3 Previous research findings indicate that the incidence rates of DVT and PE among patients undergoing craniotomy were reported as 31% and 5%, respectively. 4
Craniotomy for brain tumors sharply increases VTE risk; these patients develop DVT more often than those with other cancers or non-cancer surgeries. 5 A study focusing on post-craniotomy patients with brain tumors revealed an overall incidence rate of VTE at 3.5%. 6 Previous studies have reported a risk of VTE of 30% among patients undergoing craniotomy for meningiomas. 7 In a study encompassing 1002 adult patients with glioblastoma, the incidence rate of venous thromboembolism was found to be 16.1%. 8 Venous thromboembolism is recognized as a significant preventable factor contributing to morbidity and mortality among ICU patients, and is associated with adverse outcomes. 9 In a study encompassing 175,665 critically ill adult patients, the association between venous thromboembolism and increased risk of mortality in critically ill adults was confirmed. 10
The objective of this study is to investigate the incidence of VTE and associated risk factors among critically ill patients after craniotomy for intracranial tumors. The study aims to provide evidence-based insights to reduce the occurrence and mortality rate of VTE. We also attempted to establish a predictive model that incorporates potential independent risk factors to calculate an individual's risk of VTE and promptly identify latent conditions, aiming to improve the prognosis of patients with intracranial tumors.
Materials and Methods
Data Source
This study utilized a large-scale U.S. intensive care database called the Medical Information Mark for Intensive Care (MIMIC) as the primary data source. 11 The MIMIC database is a comprehensive and extensive clinical dataset.
Patient Information
We extracted patient data diagnosed with intracranial tumors from the MIMIC-IV database, and included patients who met the following criteria: those who underwent craniotomy due to intracranial neoplasms. Exclusion criteria included the following conditions: ① patients under the age of 18 or over the age of 80; ② hospitalization duration less than 24 h; ③ patients with multiple ICU admissions; ④ pregnant or postpartum patients; ⑤ patients with significant immune deficiencies, such as acquired immunodeficiency syndrome, tumors, or hematological disorders; ⑥ patients with a clinical data missing rate exceeding 10% in the database. Patients with >10% missing data were excluded. For variables with ≤10% missingness, five imputed datasets were generated via multivariate imputation by chained equations (MICE) using the mice package in R, with pooled estimates obtained according to Rubin's rules. The imputation model incorporated all analytic variables—including the outcome—and auxiliary covariates (eg, age and sex) deemed clinically relevant; baseline characteristics were compared before and after imputation to assess consistency.
Definition and Diagnostic Criteria
The diagnostic criteria for venous thromboembolism are based on the standards established by the American Academy of Family Physicians (AAFP) and the American College of Physicians (ACP). 12 Laboratory examinations and imaging studies play a crucial role in the diagnosis of venous thromboembolism. The diagnosis of DVT is confirmed through a comprehensive approach that includes assessment of clinical manifestations, laboratory investigations, and lower-extremity deep vein ultrasonography. The diagnosis of PE involves a comprehensive diagnostic approach, integrating the evaluation of clinical symptoms, laboratory parameters, and imaging examinations such as computerized tomography pulmonary angiography (CTPA) to confirm the diagnosis. 13
Data Collection
In this study, we extracted information related to demographic characteristics, clinical features, laboratory results, and clinically relevant diagnoses of ICU patients. Based on the exclusion criteria and research objectives, we collected relevant information about eligible study subjects, including age, gender, Glasgow Coma Scale (GCS) score, underlying comorbidities (such as cardiovascular diseases, chronic obstructive pulmonary disease, diabetes, chronic kidney disease, and chronic liver disease), white blood cell count (WBC), percentage of neutrophils (NEU), monocyte percentage (MONO), lymphocyte percentage (LY), red blood cell count (RBC), hemoglobin (HGB), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC), mean corpuscular volume (MCV), red blood cell distribution width (RDW), platelet count (PLT), prothrombin time (PT), activated partial thromboplastin time (APTT), international normalized ratio (INR), duration of hospitalization, duration of ICU stay, in-hospital mortality rate, and ICU mortality rate. The outcome measures were determined based on relevant definitions to assess the presence of venous thromboembolism in patients.
Construction, Validation, and Evaluation of the Predictive Model
We employed a random sampling approach to divide the data into training and validation sets in a ratio of 7:3. In the training set, we carried out the process of establishing the predictive model. Subsequently, we conducted model validation using the validation set to further assess the feasibility and performance of the model. For the analysis of baseline characteristics, we represented normally distributed continuous variables using mean and standard deviation, while skewed continuous variables were expressed using median and interquartile range. We employed appropriate statistical tests, such as the t-test, chi-square test, or Wilcoxon rank-sum test, to compare the characteristics between the two groups of patients based on specific circumstances.
To establish a well-calibrated Nomogram predictive model, we conducted univariate and multivariate logistic regression analyses to identify predictive factors. In the univariate analysis, only variables with a p-value less than 0.1 were selected for entry into the multivariable stepwise logistic regression. Prior to constructing the multivariable logistic regression model, we examined multicollinearity among all continuous and categorical predictors. Collinearity was quantified by the variance inflation factor (VIF), with a value >5 considered indicative of substantial multicollinearity. All calculated VIFs were <2.3 (range 1.1-2.3), indicating the absence of significant multicollinearity. Based on the results of this multifactorial analysis involving independent risk factors, we constructed the Nomogram predictive model using R software. Subsequently, we proceeded to validate and evaluate the model. Firstly, we assessed the accuracy of the model by calculating the area under the receiver operating characteristic (ROC) curve, also known as the AUC. The ROC curve and AUC are reported solely to illustrate the model's ability to rank-order risk, not to evaluate diagnostic accuracy. No threshold-based classification (eg, Youden's index) is proposed or interpreted. Next, we performed multiple iterations of bootstrapping validation and constructed a calibration curve to assess the consistency between the calibration curve and the reference curve. In statistical analysis, differences with a p-value less than 0.05 are considered statistically significant. The software used for analysis in this study included IBM SPSS Statistics 24.0 and R version 3.3.3.
Results
Patient Characteristics
Of the 1148 patients with intracranial tumors initially screened, 728 met all inclusion and exclusion criteria and were retained for analysis. The study flow is shown in Figure 1. Patients were divided into a training cohort (n = 510) and a validation cohort (n = 218). Baseline characteristics are summarized in Table 1. Overall, 728 patients with intracranial tumors were enrolled; their median age was 62 years (IQR 53-69), and 260 (35.7%) were male. Venous thromboembolism developed in 19.2% of the cohort.Patients were randomly assigned to training and validation cohorts. Baseline characteristics were compared between the two groups and found to be well balanced.

Research Flowchart.
Baseline Characteristics of Postoperative Patients with Intracranial Tumors.

Note: VTE, Venous Thromboembolism; GCS, Glasgow Coma Scale; CAD, Coronary Artery Disease; CHF, Congestive Heart Failure; COPD, Chronic Obstructive Pulmonary Disease; DM, Diabetes Mellitus; CKD, Chronic Kidney Disease; LF, Liver Failure; WBC, White Blood Cell Count; NEU, Percentage of neutrophils; MONO, Monocyte Percentage; LY, Lymphocyte Percentage; RBC, Red Blood Cell Count; HGB, Hemoglobin; MCH, Mean Corpuscular Hemoglobin; MCHC, Mean Corpuscular Hemoglobin Concentration; MCV, Mean Corpuscular Volume; PLT, Platelet RDW, Red Blood Cell Distribution Width; PT, Prothrombin Time; APTT, Activated Partial Thromboplastin Time; ICU, Intensive Care Unit. If all measurement data conform to a normal distribution, they are expressed as mean ± SD; otherwise, they are expressed as median (interquartile range, IQR).
Among the 510 patients in the training cohort (Table 2), median age was 62 years (IQR 55-70) and 172 (33.7%) were male; 98 (19.2%) developed venous thromboembolism (VTE). Stratified analysis showed that patients who remained VTE-free were younger [median 61 years (IQR 54-69)] than those who developed VTE [65 years (IQR 60-71); P < .001]. Patients who developed VTE presented with lower initial GCS scores [14 (12-15) versus 14 (14-15); p = 0.027] and prolonged coagulation times: PT was 13.2 s (12.2-14.5) versus 11.8 s (11.0-12.7), and APTT was 28.9 s (26.6-31.6) versus 28.5 s (26.3-32.3) (both P < .05). Full details are provided in Table 2. Multicollinearity among all predictors was assessed using VIFs. The largest VIF observed was 2.3, well below the conventional threshold of 5, so no variables were removed.
Characteristics of Patients and Diseases.

Note: VTE, Venous Thromboembolism; GCS, Glasgow Coma Scale; CAD, Coronary Artery Disease; CHF, Congestive Heart Failure; COPD, Chronic Obstructive Pulmonary Disease; DM, Diabetes Mellitus; CKD, Chronic Kidney Disease; LF, Liver Failure; WBC, White Blood Cell Count; NEU, Percentage of neutrophils; MONO, Monocyte Percentage; LY, Lymphocyte Percentage; RBC, Red Blood Cell Count; HGB, Hemoglobin; MCH, Mean Corpuscular Hemoglobin; MCHC, Mean Corpuscular Hemoglobin Concentration; MCV, Mean Corpuscular Volume; PLT, Platelet RDW, Red Blood Cell Distribution Width; PT, Prothrombin Time; APTT, Activated Partial Thromboplastin Time; ICU, Intensive Care Unit. If all measurement data conform to a normal distribution, they are expressed as mean ± SD; otherwise, they are expressed as median (interquartile range, IQR).
Predictive Factors for Postoperative Secondary Venous Thromboembolism in Craniotomy Patients
Subsequently, we conducted univariate and multivariable logistic regression analyses to identify predictive factors. Variables with a p-value of less than 0.1 in the univariate analysis were included in the univariate binary logistic regression. Significant variables from the univariate analysis were further incorporated into the multivariable logistic regression. Finally, these variables were introduced into the predictive model. The results of univariate and multivariable logistic regression analyses in the training cohort are presented in Table 3. We identified three variables – patient age, initial GCS score, and PT as potential risk factors for the development of secondary VTE in patients with intracranial tumors (Table 3). The three variables were further included in the multivariable logistic regression analysis. Age, initial GCS upon admission to the ICU, and PT were found to be independent risk factors, as shown in Table 3. We incorporated the aforementioned independent risk factors into the predictive model and constructed a Nomogram, as illustrated in Figure 2, using R software.
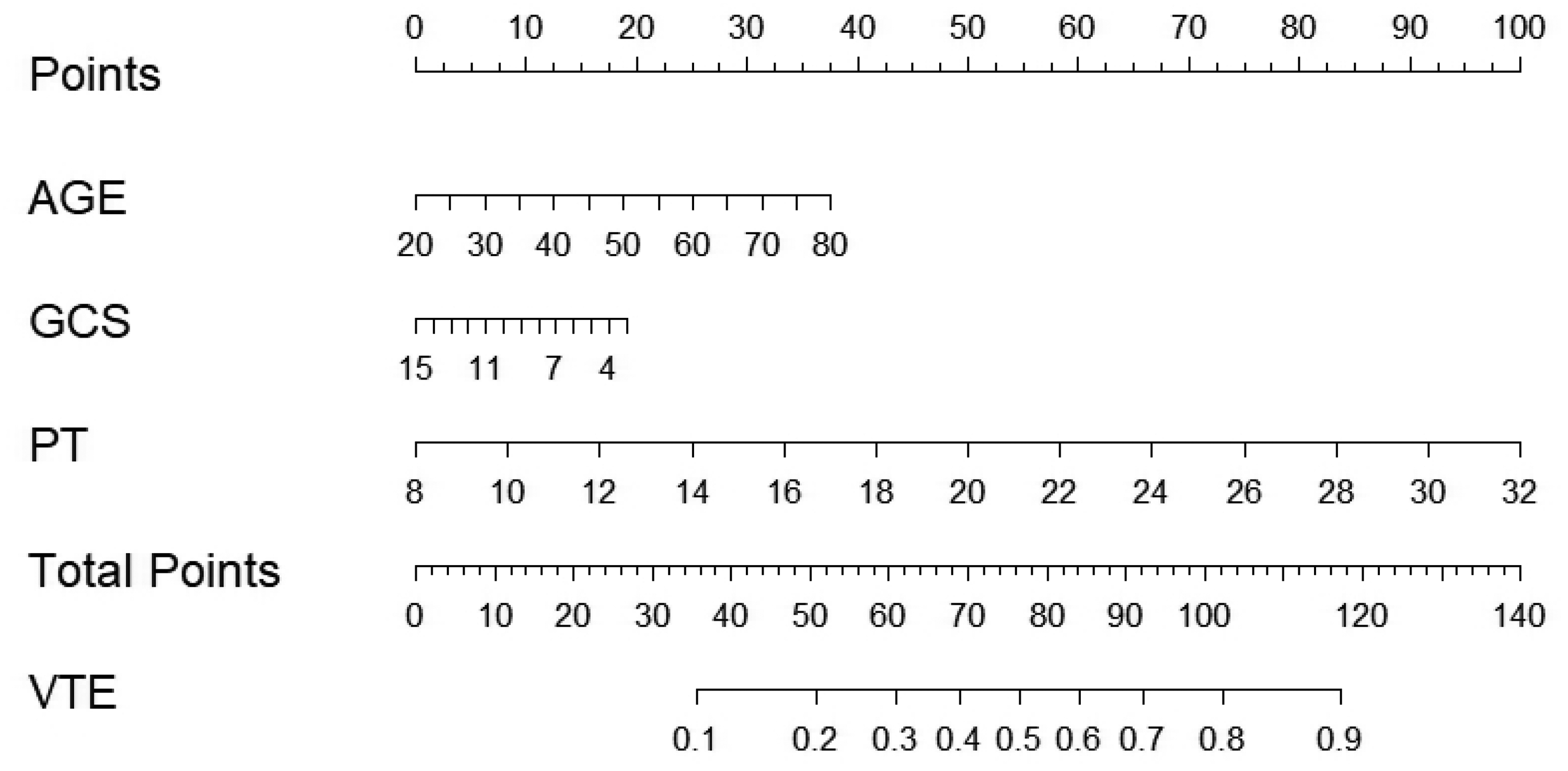
Nomogram for Predicting the Risk of Postoperative VTE in Patients with Intracranial Tumors.
Univariate and Multivariate Analysis of Postoperative VTE in Patients with Intracranial Tumors.

Note: VTE, Venous Thromboembolism; GCS, Glasgow Coma Scale; CAD, Coronary Artery Disease; CHF, Congestive Heart Failure; COPD, Chronic Obstructive Pulmonary Disease; CKD, Chronic Kidney Disease; RDW, Red Blood Cell Distribution Width; PT, Prothrombin Time; APTT, Activated Partial Thromboplastin Time.
Interpretation and Analysis of the Predictive Model Nomogram
The Nomogram (Figure 2) encompasses the following information: Firstly, it displays the variable names included in the predictive model, such as AGE, GCS, and PT. Each variable is associated with a labeled scale on the corresponding line segment, representing the range of possible values for that variable. The length of each line segment reflects the magnitude of contribution of that particular factor to the outcome event. Next are the scores, which include individual scores, referred to as “Points” in the graph. These represent the scores assigned to each variable at different values. Additionally, there is a total score, known as “Total Points,” which represents the cumulative sum of individual scores obtained by considering all variable values. Lastly, we have the predicted probability, denoted as “VTE” in the graph, which represents the likelihood of developing secondary VTE.
Determining the Threshold for Secondary Infection in the Predictive Model
After model establishment, we assessed the accuracy of the model using the area under the ROC curve (AUC). ROC analysis was performed to internally validate the Nomogram within the training cohort (Figure 3A) and externally validate the Nomogram within the validation cohort (Figure 3B). The internal validation of the ROC curve yielded an AUC value of 0.7463 (95% CI 0.7056-0.7932), indicating a favorable discriminatory power of the model within the training cohort. Similarly, the external validation of the ROC curve resulted in an AUC value of 0.7361 (95% CI 0.6498-0.8223), suggesting that the model possesses considerable predictive value. To further validate the effectiveness of the model, a calibration curve was constructed using R software. The calibration of the Nomogram was performed internally within the training cohort (Figure 4A) and externally within the validation cohort (Figure 4B). The calibration curves of the model displayed a reasonably acceptable fit to the standard ideal curve. Finally, to further assess its clinical validity, the decision curve (Figure 5) and clinical impact curve (Figure 6) were plotted using R. Most parts of the decision curve were above zero, indicating clinical validity. These results suggest that all models have certain predictive value.

The ROC Curve Verifies the Accuracy of the Training Dataset (3A) and Validation Dataset (3B) Models. Internal Validation in the Training Cohort Shows an AUC of 0.7463 (95% CI 0.7056-0.7932). in the Validation Cohort, the ROC Curve Yielded an AUC of 0.7361 (95% CI 0.6498-0.8223).This ROC Curve Reflects the Concordance (c-Statistic) Between Predicted Probabilities and Observed Outcomes, Without Implying Clinical Utility for Diagnosis.
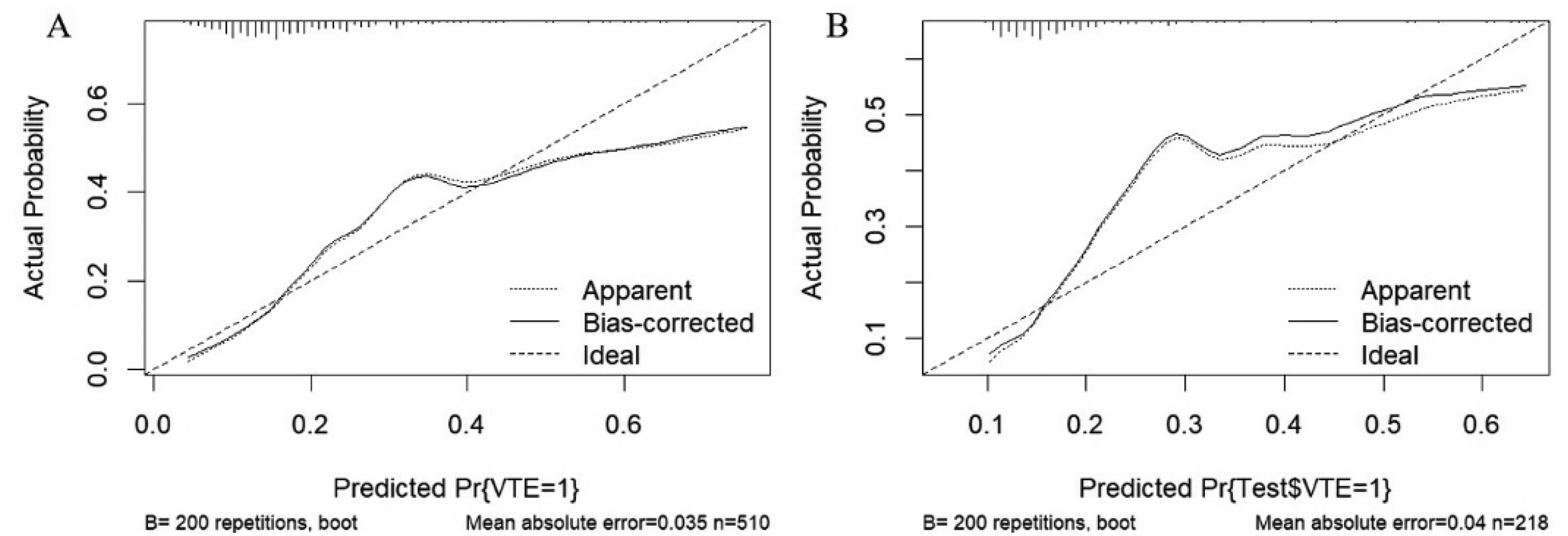
Calibration Curves of the Nomogram in the Training Dataset (4A) and Validation Dataset (4B). The Calibration Curves Show Calibration of the Nomogram in Terms of Agreement between the Predicted Risk of Secondary Infection.

Decision Curve Analysis of the Nomogram in the Training Dataset (5A) and Validation Dataset (5B).

Clinical Impact Curves of the Nomograms in the Training Dataset (6A) and Validation Dataset (6B).
Discussion
Preventing VTE poses a formidable challenge for critically ill patients, particularly those undergoing postoperative interventions for intracranial tumors. Similarly, predicting the risk of VTE holds paramount importance across all medical domains, particularly within the field of neurosurgery. Despite these challenges, there is a growing body of research focused on post-craniotomy patients, enabling clinicians to offer effective and safe thromboprophylaxis measures. The diagnosis of VTE holds crucial significance in the postoperative management of neurosurgical patients, as the use of anticoagulant therapy is associated with an increased risk of intracranial hemorrhagic complications.14,15 Research has demonstrated that in patients undergoing craniotomy for brain tumor resection, the risk of bleeding associated with anticoagulant therapy is estimated to be approximately 3%. 16 Malignant tumors are widely recognized as a significant risk factor for VTE due to their propensity to induce a hypercoagulable state. Research has revealed that the incidence of VTE in patients with malignancies can reach as high as 5.7%.17,18 Cases involving malignant tumors may also encompass additional factors that render patients susceptible to VTE, such as increased surgical complexity, elevated incidence of complications, and prolonged hospitalization. Moreover, the presence of VTE in tumor patients can significantly worsen their prognosis. 19 The heightened risk of VTE in patients with intracranial tumors has prompted us to develop a postoperative screening protocol aimed at detecting VTE occurrences. 20
In this study, a VTE incidence of 19.2% was observed among patients undergoing postoperative interventions for intracranial tumors. This finding is slightly higher than the incidence of VTE reported in a study investigating post-craniotomy patients, where the overall occurrence rate of VTE after craniotomy was found to be 7.7%. 21 Furthermore, our study revealed a hospital mortality rate of 66.3% among post-craniotomy patients who developed secondary VTE, which was significantly higher compared to patients without VTE. This finding aligns with previous research, which has demonstrated that VTE is a significant perioperative complication associated with increased length of hospital stay, healthcare costs, overall morbidity, and mortality rates among patients. 22 Furthermore, the incidence of cancer-associated thrombosis varies depending on the tumor site, but glioblastoma is recognized as one of the solid tumors with a remarkably high potential for thrombus formation. Patients with glioblastoma face a significantly elevated risk of developing VTE during the disease course, with an annual incidence rate as high as 30%. 23 In the field of neurosurgery, the majority of thrombotic events are directly associated with surgical procedures and typically occur in the operating room or intensive care unit. Therefore, we propose that lower extremity venous ultrasonography should be incorporated into the standard therapeutic protocol for VTE screening on the first postoperative day in high-risk patients. Moreover, this approach has been substantiated as highly effective in numerous studies. 7
We have developed a Nomogram model to predict the risk of secondary VTE in postoperative patients with intracranial tumors. Remarkably, this model has achieved acceptable predictive performance, with the AUC of 0.7463. Furthermore, the calibration curve also indicates that employing this model as an intervention strategy can yield improved outcomes. Previous studies have explored the risk factors for the occurrence of secondary VTE in post-craniotomy patients. The findings indicate an increased risk of VTE in both elderly individuals and those undergoing emergent craniotomies. 24 Furthermore, in order to prevent VTE in neurosurgical procedures, some researchers have identified the risk factors contributing to VTE and have developed predictive models for this complication.25,26 In patients undergoing craniotomy, it has been established that surgical duration exceeding 4 h is a significant factor associated with an increased incidence of DVT. 27 Moreover, studies have demonstrated that risk factors for VTE in the post-craniotomy setting include cumulative mechanical ventilation exceeding 48 h, unplanned reintubation within 30 days, postoperative infections, and postoperative coma lasting over 24 h. 28
Our study identified patient age, initial GCS upon ICU admission, and PT as potential risk factors for the development of secondary VTE in postoperative patients with intracranial tumors. Similar findings can be observed in other studies. Previous research has demonstrated that age is an independent predictive factor for postoperative VTE in patients with brain tumors. 29 Several studies have reported a notable increase in the risk of VTE among patients with brain tumors beyond the age of 55. 30 A study utilizing data from the American Surgical Quality Improvement Program revealed that patients aged 60 and above, who underwent craniotomy for brain tumors, had a 73% higher risk of developing VTE. 31 In the previous analysis of craniotomy cases from 2006 to 2010, factors associated with venous thromboembolism (VTE) encompassed variables such as age exceeding 60 years. 32 In a study involving adult patients with glioblastoma, it was confirmed that for each additional year of age, the likelihood of developing VTE increased by 3%. 8 In the elderly population, heightened thrombogenic activity, elevated plasma concentrations of coagulation factors, and diminished vascular function may be associated with an increased risk of thrombus formation. 33
The GCS is a tool utilized for assessing and quantifying the level of consciousness in patients. Over its 45 years of development, GCS has played a prominent role in the evaluation of the neurological system. The implementation of the GCS is straightforward and rapid, typically regarded as a statistically significant metric. 34 A lower total score on the GCS does not always signify more severe injury, nor does a higher score imply the opposite. Research suggests that although GCS has an impact on prognosis, it is insufficient to predict individual patient outcomes. 35 In the majority of neurosurgical institutions, patients scheduled for elective surgery are typically subjected to preoperative assessment of their coagulation parameters. PT is among the most commonly employed coagulation assays, utilized to assess tissue factor (TF) and the common coagulation pathway. PT is influenced by the activity of coagulation factors II (FII), V (FV), VII (FVII), X (FX), and fibrinogen. 36 Research has discovered that 12% of neurosurgical patients experiencing hemorrhagic or thrombotic complications may exhibit elevated PT. 37 An increasing body of evidence suggests that PT is a reliable tool for screening patients with coagulation dysfunction.38,39 Additionally, numerous studies have expanded upon our findings. A recent meta-analysis involving 49,620 patients with brain tumors revealed that congestive heart failure and a history of diabetes mellitus are risk factors for postoperative VTE in patients with brain tumors. 40 Although the Caprini Risk Assessment Model has been extensively validated in general surgical populations, it lacks neuro-specific variables such as the Glasgow Coma Scale and post-operative prolongation of prothrombin time. In our derivation cohort, 62% of the Caprini components were missing, precluding direct incorporation of this score into the predictive model. Nevertheless, our ongoing prospective validation phase has been designed to formally pit the Caprini RAM against the current nomogram, thereby quantifying any incremental predictive value afforded by the neuro-specific parameters.
Novelty in relation to previous work. Firstly, to the best of our knowledge, this is the first instance in which a nomogram has been utilized for predictive analysis of thromboembolism occurrence in patients with intracranial tumors. Secondly, Prior models typically pooled “all brain tumors” or “all neurosurgical procedures,” yielding marked heterogeneity and widely divergent VTE rates (3-31%). The present study, to our knowledge, is the first to restrict the cohort to intracranial-tumor patients on their first post-craniotomy ICU day, thereby enhancing clinical homogeneity and enabling results to be translated directly to bedside decision-making in the ICU.
Finally, Earlier models incorporated 7-12 variables, often including “operative duration” or “tumor histologic grade,” which are not immediately available after surgery. Our model achieves an AUC of 0.75 with only three routinely documented ICU variables—age, GCS, and PT—markedly enhancing clinical feasibility and ensuring broad applicability even in resource-limited settings.
However, this study does possess a few limitations. Firstly, as our analysis relies solely on the MIMIC-IV database—an observational cohort from a single tertiary-care hospital in the United States—the reported findings should be regarded as preliminary and require external validation. Second, critical pre-ICU clinical details are absent, including tumour histology, operative duration, and post-operative complications (eg, re-operation); their omission may introduce residual confounding. Third, the generalisability of our model remains untested; a prospective validation study is currently underway at Beijing Tiantan Hospital. Finally, the database lacked sufficient Caprini score components (62% missing), precluding incorporation of this well-validated instrument. Although the Caprini Risk Assessment Model is widely accepted for surgical patients, prospective collection of complete Caprini variables is needed to compare its predictive performance and additive value with the present nomogram.
Conclusion
The nomogram we constructed confirms that age, initial GCS upon ICU admission, and PT are potential risk factors for the occurrence of postoperative VTE in patients with intracranial tumors. Therefore, following the establishment of a personalized predictive model, individual assessment of post-craniotomy patients admitted to the ICU for the risk of developing secondary VTE) allows for targeted attention and proactive intervention in high-risk patients, aiming to improve patient outcomes at an early stage.
Footnotes
Acknowledgements
No assistance in the preparation of this article is to be declared.
Ethics Approval and Informed Consent Statements
Data from this study were all obtained from the public databases, all patient information was de-identified and patient consent was not required.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
