Abstract
Background
Prosthetic heart valve thrombosis (PHVT) is a serious and potentially life-threatening complication that affects patients with mechanical heart valves. Timely and precise prediction of PHVT is essential for prompt intervention. This study aims to assess the association between the ratio of red blood cell distribution width (RDW) and platelet (PLT) count with PHVT.
Methods
We conducted a retrospective analysis of 297 transesophageal echocardiography examinations performed between January 2007 and October 2022 on patients with mechanical mitral prosthetic valves. This cohort included 161 patients diagnosed with PHVT and 136 patients with functional prosthetic valves.
Results
Patients with PHVT were, on average, older than those with normofunctional valves (56 vs 53 years, p = .046). Univariable analysis indicated that advanced age, heart failure (HF), chronic renal failure, COPD, reduced LVEF, ineffective anticoagulation, elevated D-dimer levels, and an elevated RDW-to-PLT ratio were associated with PHVT. The multivariable logistic regression analysis identified elevated RDW-to-PLT ratio (OR: 1.278, 95% CI: 1.142-1.327, p = .001), ineffective anticoagulation, HF, and D-dimer were independently associated with PHVT. The ROC curve analysis demonstrated that the RDW-to-PLT ratio exhibited moderate diagnostic performance, with a cut-off value of 0.065, sensitivity of 65%, and specificity of 66%.
Conclusion
This is the first study demonstrating that the higher RDW-to-PLT ratio is associated with PHVT. Further studies are necessary to validate these findings in broader clinical settings.
Introduction
Prosthetic heart valve thrombosis (PHVT) refers to the obstruction of a prosthetic valve caused by non-infective thrombotic material. This condition is a significant and potentially life-threatening complication faced by patients with either mechanical or bioprosthetic heart valves. PHVT is characterized by the formation of a thrombus on the surface of the prosthetic valve, which can lead to valve obstruction, systemic embolization, and impaired valve function. The incidence of PHVT can vary widely and is influenced by factors such as valve type, valve position, and individual patient characteristics. It is noted that the risk of thrombosis is higher in mechanical valves compared to bioprosthetic valves, with the incidence ranging from 0.6% to 6% in left-sided valves and up to 20% in tricuspid valves. 1 The etiopathogenesis of PHVT is multifactorial, involving several complex mechanisms. The primary mechanism relates to the interactions between corpuscular blood components, plasma, and the surfaces of the prosthesis. This process begins with the adsorption of plasma proteins onto the prosthetic material, which is typically followed by platelet (PLT) adhesion. The secondary mechanism pertains to how transprosthetic blood flow influences local thrombus formation; turbulent flow can elevate shear stress within the bloodstream, thereby promoting thrombosis. The tertiary mechanism involves insufficient anticoagulation, which is indicated by the rates of valve thrombosis for specific prostheses in relation to particular international normalized ratio (INR) levels.2,3 Diagnosing PHVT necessitates a comprehensive approach that includes clinical evaluation, imaging studies, and laboratory tests. Transesophageal echocardiography (TEE) is the gold standard for diagnosing PHVT due to its high sensitivity and specificity. 2 Management of PHVT involves a multifaceted strategy that encompasses medical therapy with anticoagulants or fibrinolytic agents and, in severe cases, surgical intervention. The choice of treatment is determined by the patient's clinical status, the size and location of the thrombus, and the associated risk of bleeding complications. 4 Despite advancements in both diagnostic and therapeutic strategies, PHVT continues to pose a significant clinical challenge, underscoring the need for ongoing research and improved methods for prevention, early detection, and effective management.
A complete blood count (CBC) is one of the most common and cost-effective laboratory tests conducted in hospitals. Numerous studies have assessed the efficacy of various hematological parameters derived from CBC in predicting disease severity and mortality risk. Among these parameters, red blood cell distribution width (RDW) holds particular significance. RDW measures the variability in red blood cell volume and is instrumental in classifying different types of anemia. 5 Elevated RDW is recognized as an inflammatory marker and has been correlated with poor outcomes in a range of conditions, including heart failure (HF), cancers, acute kidney injury, and sepsis.6–9 Additionally, PLT play a critical role in modulating inflammation and innate immunity; they adhere to the endothelium and promote neutrophil chemotaxis, infiltration, and the secretion of pro-inflammatory chemokines during acute inflammatory responses. 10 Abnormal PLT counts—either elevated or reduced—in severe diseases are indicative of mortality risk.11–13 The RDW-to-PLT ratio emerges as a novel and straightforward indicator of inflammation and thrombosis. Recent research has highlighted the clinical importance of this ratio as both a diagnostic and prognostic biomarker. Notably, the RDW-to-PLT ratio has demonstrated utility in identifying certain non-cardiac conditions and their prognoses.14–16 Moreover, it serves as a valuable predictor of adverse outcomes in patients with acute ST-elevation myocardial infarction (STEMI). 17 However, to our knowledge, no studies have explored the relationship between the RDW-to-PLT ratio and PHVT in the existing literature. Accordingly, this study aims to assess the predictive value of the RDW-to-PLT ratio in the context of PHVT.
Methods
A total of four hundred and twelve consecutive TEE examinations conducted between January 2007 and October 2022 were reviewed at our institution. These examinations involved patients with mechanical prosthetic valves in the mitral position, addressing various clinical indications. Patients with fever or signs of infection (suspected infective endocarditis) (49 patients), prosthetic valve obstruction without thrombus (56 patients), and moderate-to-severe paravalvular regurgitation without thrombus (10 patients) were excluded from the study. Moreover, patients with severe anemia (hemoglobin <7 g/dL) were also excluded from the study. Additionally, given that bioprosthetic heart valve thrombosis (BPHVT) is a less common condition compared to mechanical prostheses, patients diagnosed with BPHVT were also excluded. Ultimately, after applying the exclusion criteria, 297 patients with a mitral prosthetic valve were included in the study. This population comprised 161 patients diagnosed with PHVT via TEE and 136 patients with functional prosthetic valves. TEE was utilized for the definitive diagnosis of PHVT.
The local ethics committee approved the study (Ankara Etlik City Hospital, 26/06/2024, AEŞH-BADEK-2024-614), which was conducted in accordance with the recommendations outlined in the Declaration of Helsinki regarding biomedical research involving human subjects. Due to the retrospective nature of the study, the requirement for a written informed consent form from each participant was waived.
TEE Examination
Following an overnight fast, TEE examinations were standardized, with all subjects positioned in the lateral decubitus position. The Vivid-Med System Seven, equipped with a multiplane TEE probe (GE Medical Systems, Hortan, Norway), was employed for these examinations. Informed consent was explicitly obtained for the TEE procedure, which was performed under conscious sedation with the hypopharynx anesthetized. Two cardiologists, blinded to the patients’ clinical information, acquired and analyzed multiple views. Thrombus was identified as a homogeneous, mobile, or fixed mass exhibiting a lower density than the pannus at the valve occluder or struts. This finding was observed in all patients diagnosed with PHVT. The differentiation between thrombus and pannus overgrowth was primarily based on TEE findings, as previously established in the literature. 18
Laboratory Data Analysis
Venous blood samples were collected from all patients prior to the TEE examination for hematological and biochemical assessments. Hematologic parameters were analyzed using an automated Coulter LH 780 Hematology Analyzer (Beckman Coulter Ireland Inc., Mervue, Galway, Ireland). This analysis provided essential data for evaluating the patients’ hematologic status and biochemical profile, aiding in the comprehensive assessment of PHVT. The RDW-to-PLT ratio was calculated by dividing the RDW value by the PLT count (×103), both of which were obtained from the same automated blood sample taken at the time of admission to the study. Furthermore, the following formula is used to calculate the systemic immune-inflammation index (SII): (PLT count × neutrophil count)/lymphocyte count. 19
Definitions
Hypertension is defined as the administration of antihypertensive therapy or a systolic blood pressure exceeding 140 mm Hg and/or a diastolic blood pressure exceeding 90 mm Hg on at least two separate measurements during hospitalization. Diabetes mellitus (DM) is characterized by the use of oral hypoglycemic agents or insulin or by fasting blood glucose levels that meet the criteria established by the European Society of Cardiology. 20 A stroke is classified as a neurological deficit lasting less than 24 h, identified as a transient ischemic attack, or as a stroke if the deficit persists beyond 24 h. Preexisting chronic renal failure is defined as an estimated glomerular filtration rate of less than 60 mL/min/1.73 m2 for a duration exceeding three months, regardless of any kidney damage. Ineffective anticoagulation is identified by having one or more measurements of the INR below 2.5 within the preceding three months.
Statistical Analysis
Categorical variables were presented as counts and percentages, with analyses conducted using the Chi-square test. Continuous variables were expressed as mean ± SD for normally distributed data and as median (minimum-maximum) for non-normally distributed data. The normality of the data across patient groups was assessed using the Kolmogorov-Smirnov test. Normally distributed data were analyzed with the Student's t-test, while the Mann-Whitney U test was applied to non-normally distributed data. Logistic regression analysis was performed to establish independently associated factors with PHVT. After conducting the multivariable analysis, the optimal RDW-to-PLT ratio was determined through receiver operating characteristic (ROC) curve analysis. A p-value of less than .05 was considered statistically significant for all tests. Data analysis was carried out using SPSS version 22 (IBM Corp.; Armonk, NY, USA).
Results
Table 1 outlines the baseline characteristics, echocardiographic, and laboratory findings of patients with and without PHVT.
Baseline Characteristics, Echocardiographic and Laboratory Findings of Patients with and Without Prosthetic Heart Valve Thrombosis (PHVT).

Abbreviations: CHA2DS2-VASc, congestive heart failure, hypertension, age ≥75, diabetes, stroke, vascular disease, age 65–74, sex category; MCV, mean corpuscular volume; MPV, mean platelet volume; PLT, platelet; RDW, red blood cell distribution width; ACE-i, angiotensin converting enzyme inhibitors; ARB, angiotensin-II receptor blockers.
There was no significant difference in gender between the two groups. However, patients with PHVT were older than those with a normofunctional valve, averaging 56 years compared to 53 years (p = .046). The prevalence of hypertension, coronary artery disease (CAD), atrial fibrillation (AF), prior myocardial infarction, and DM was comparable in both groups (p > .05). Chronic renal failure was notably more common in patients with PHVT, occurring in 11.2% of this group versus 4.4% in the other (p = .033). Similarly, chronic obstructive pulmonary disease (COPD) was more prevalent in the PHVT group (16.1% vs 8.1%, p = .036). Moreover, patients with PHVT exhibited a higher rate of congestive heart failure (27.3% vs 13.2%, p = .003), as well as an increased incidence of cerebrovascular accidents (16.1% vs 5.9%, p = .006). As anticipated, instances of ineffective anticoagulation were significantly more frequent among those with PHVT (57.1% vs 32.4%, p < .001). When comparing the CHA2DS2-VASc scores, it was found that patients with PHVT had a significantly higher score than those without (2.0 vs 1.6, p = .005). The prevalence of anemia and malignancy was similar in both groups. According to echocardiographic assessments, the left ventricular ejection fraction (LVEF) was lower in patients with PHVT (48% vs 53%, p < .001). In contrast, the left atrium anteroposterior diameter did not show any significant difference between the groups.
Regarding laboratory findings, the PLT count was significantly lower in the PHVT group compared to the other group, with values of 229 versus 255 × 103/µL (p < .001). Additionally, the RDW was elevated in patients with PHVT, measuring 16.3% compared to 14% in the other group (p < .001). Furthermore, creatinine levels were higher in the PHVT group, recorded at 1.00 mg/dL versus 0.8 mg/dL (p = .005). The RDW-to-PLT ratio was also notably higher in the PHVT group, at 0.082, compared to 0.062 in the other group (p < .001). Similarly, serum D-dimer levels were significantly higher in patients with PHVT (p < .001). On the other hand, no significant differences were observed between the two groups regarding C-reactive protein, serum albumin, and the SII.
When evaluating medication use between the two groups, patients with and without PHVT exhibited similar characteristics in terms of antiplatelet, warfarin, statin, beta-blocker, angiotensin-converting enzyme inhibitor or angiotensin receptor blocker, and antidiabetic drug usage.
Independently Associated Factors with PHVT
Table 2 summarizes the univariable and multivariable model identifying independent risk factors for PHVT. The univariable analysis revealed several variables significantly associated with PHVT, including advanced age, chronic renal failure, COPD, congestive heart failure, CHA2DS2-VASc score, decreased LVEF, ineffective anticoagulation, elevated serum creatinine levels, decreased PLT count, increased RDW, elevated D-dimer levels, and a higher RDW-to-PLT ratio.
Univariable and Multivariable Logistic Regression Models for Predicting Prosthetic Heart Valve Thrombosis.

Abbreviations: COPD, chronic obstructive pulmonary disease; CHA2DS2-VASc, congestive heart failure, hypertension, age ≥75, diabetes, stroke, vascular disease, age 65–74, sex category; LVEF, left ventricular ejecton fraction; RDW, red blood cell distribution width; PLT, platelet; OR, odds ratio; CI, confidence interval.
The multivariable logistic regression analysis indicated that congestive heart failure [odds ratio (OR): 2.871, 95% confidence interval (CI): 1.412-5.103, p = .006], decreased LVEF (OR: 1.165, 95% CI: 1.085-1.314, p = .005), ineffective anticoagulation (OR: 2.703, 95% CI: 1.621-4.791, p < .001), a higher RDW-to-PLT ratio (OR: 1.278, 95% CI: 1.142-1.327, p = .001), and elevated D-dimer levels (OR: 1.451, 95% CI: 1.103-1.908, p = .002) emerged as independently associated with PHVT. The ROC curve analysis for the RDW-to-PLT ratio demonstrated an area under the curve of 0.693 (95% CI: 0.633-0.753), with a cut-off value of 0.065, sensitivity of 65%, and specificity of 66% (p = .001) (Figure 1).

Diagnostic performance of the RDW-to-PLT ratio for predicting prosthetic heart valve thrombosis. Abbreviations: RDW, red blood cell distribution width; PLT, platelet; AUC, area under curve; CI, confidence interval.
In the analysis, patients were categorized into two groups based on the RDW-to-PLT ratio using a cut-off value of 0.065 (with 166 patients having a ratio ≥ 0.065 and 131 patients having a ratio < 0.065). No significant differences were noted between the two groups regarding gender, hypertension, DM, CAD, myocardial infarction, HF, cerebrovascular events, and chronic renal failure (p > .05 for all parameters individually). However, patients with an RDW-to-PLT ratio of 0.065 or higher demonstrated a significantly increased rate of PHVT compared to their counterparts (62% vs 43%, p = .001) (Figure 2).
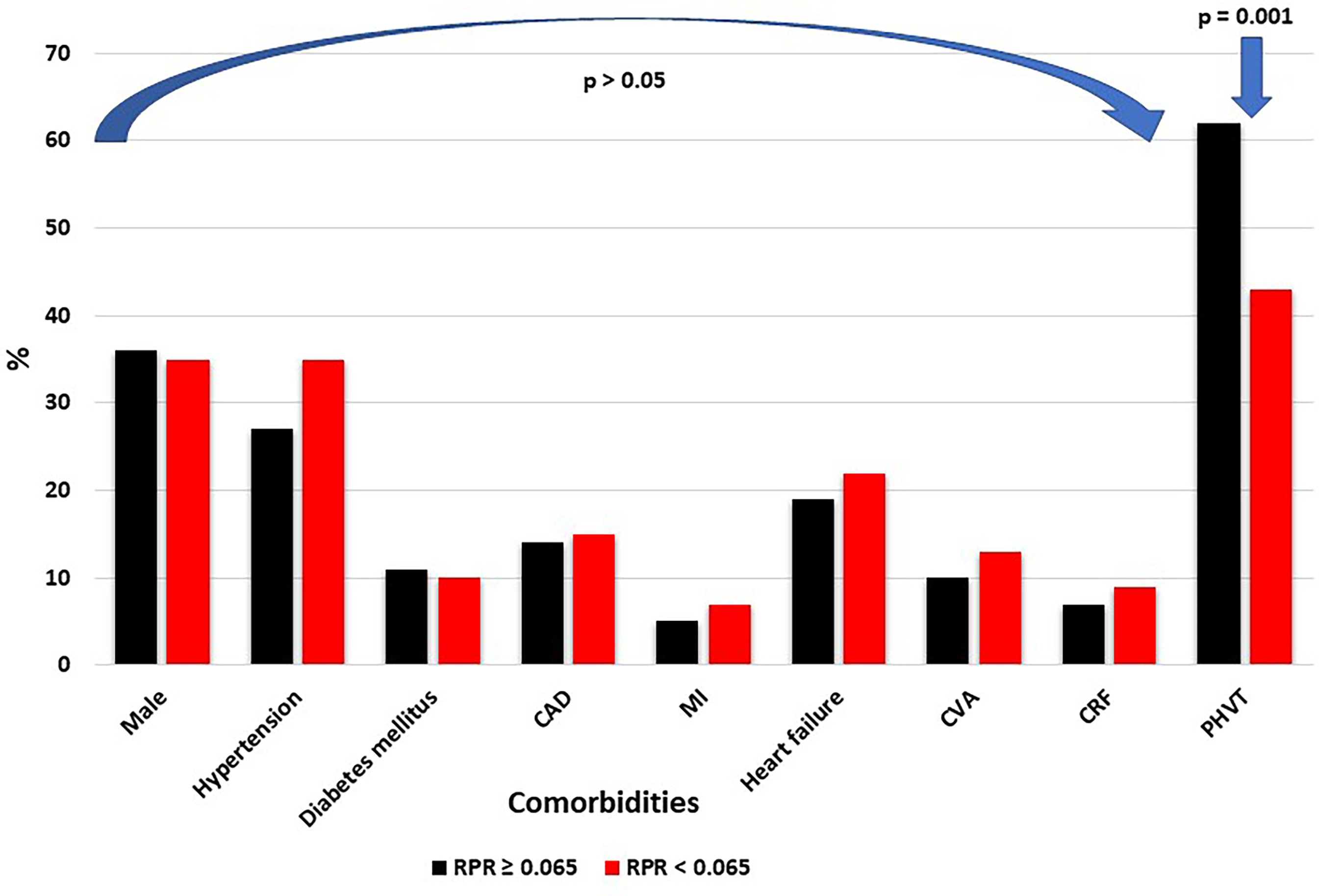
Comparison of comorbid conditions in patients based on the RDW-to-PLT ratio. Abbreviations: RDW, red blood cell distribution width; PLT, platelet; RPR, RDW to PLT ratio; CAD, coronary artery disease; MI, myocardial infarction; CVA, cerebrovascular accident; CRF, chronic renal failure; PHVT, prosthetic heart valve thrombosis.
Discussion
The current study presents compelling findings that could be significant in clinical practice for predicting PHVT in patients with mechanical mitral valves. A key takeaway from our research is that higher ratios of RDW-to-PLT were independently associated with PHVT. To our knowledge, this relationship has not been documented in prior research. Furthermore, our analysis identified ineffective anticoagulation, reduced LVEF, congestive heart failure, and elevated D-dimer levels as independent risk factors for PHVT.
The traditional risk factors for PHVT encompass insufficient endothelialization, AF, left atrial enlargement, multiple valve replacements, ventricular dysfunction, pannus formation, pharmacological treatments, malignancies, systemic diseases, pregnancy, genetic predispositions, specific autoantibodies, and heparin-induced thrombocytopenia.3,21 Moreover, inflammation and thrombosis are closely linked, with each process reinforcing the other. Inflammatory cytokines such as interleukin-1 and TNF-alpha stimulate endothelial cells to express pro-coagulant molecules, including tissue factor and von Willebrand factor, facilitating PLT adhesion and aggregation that ultimately leads to thrombosis.22,23 Additionally, mediators like thromboxane A2 and PLT-activating factor further activate PLTs, causing the release of more cytokines and establishing a feedback loop that exacerbates both inflammation and thrombosis. 24 Inflammation also triggers the coagulation cascade by releasing tissue factor, which promotes thrombin formation and the development of fibrin clots. Chronic inflammation heightens the risk of clotting by sustaining a hypercoagulable state. 24
The measurement of RDW is routinely conducted in the assessment of hematological disorders. However, recent advancements and studies have indicated that RDW may also serve as a marker of inflammation and possess prognostic significance in certain cardiovascular diseases. Elevated RDW levels have been linked to negative outcomes in patients with HF and have been associated with increased mortality and cardiovascular events independent of anemia status.25,26 Furthermore, thrombosis is another condition correlated with RDW. Past research has demonstrated that higher RDW levels can anticipate venous thromboembolism, stent thrombosis, catheter-related thrombosis, and cerebral vein thrombosis.27–30 Although specific analyses exploring the relationship between RDW and PHVT are lacking in the literature, our study's findings—showing significantly higher RDW levels in the PHVT group—support the established relationship between RDW and thrombosis in previous research. While RDW may not be as critical as PLT, which plays a central role in primary hemostasis, it appears to hold significant relevance in thrombotic events through mechanisms that remain to be fully elucidated. The notable relationship between RDW and PLT has been examined in various pathologies. Indeed, research has demonstrated that elevated RDW-to-PLT ratios possess both diagnostic and prognostic importance in conditions such as neonatal sepsis, hepatic fibrosis, burns, acute kidney injury, and acute pancreatitis.15,31–34 However, studies focusing on this biomarker within the realm of cardiology are limited. A recent comprehensive investigation revealed that a higher RDW-to-PLT ratio in the adult population of the United States correlates with the prevalence of cardiovascular disease. 35 Furthermore, a study by Yao et al indicated that patients with acute myocardial infarction and elevated RDW-to-PLT ratios experienced significantly higher in-hospital mortality rates. 36 Similar findings were observed in patients with acute STEMI. 17 Given that acute coronary syndrome is intrinsically a thrombotic process, the outcomes from these two studies underscore the value of the RDW-to-PLT ratio. Notably, our research marks the first identification of an increased RDW-to-PLT ratio as an independent risk factor for PHVT, thereby highlighting the significance of this straightforward hematologic marker.
Another prominent finding of our study is the independent association between elevated serum D-dimer levels and PHVT. As a fibrin degradation product, D-dimer levels are expected to increase with a higher thrombus burden; however, there is a limited number of studies in the literature explicitly investigating the relationship between D-dimer and PHVT. Previously, Nazlı et al conducted a prospective study involving 97 PHVT patients and demonstrated that elevated plasma D-dimer levels could predict PHVT. 37 Similarly, another retrospective analysis including 57 PHVT patients also identified an association between elevated D-dimer levels and PHVT. 38 Considering that the number of PHVT patients in our study exceeds the total number of patients in these two previous studies, our findings significantly reinforce the relationship between D-dimer levels and PHVT.
The effectiveness of anticoagulation therapy is significantly influenced by the achievement and maintenance of optimal therapeutic levels. Ineffective anticoagulation—whether resulting from subtherapeutic dosing, patient non-compliance, or pharmacodynamic variability—substantially heightens the risk of thrombotic events. 39 Our study found that ineffective anticoagulation was the most significant predictor of PHVT, associated with an almost 2.7-fold increase in risk.
HF and thrombosis are interconnected clinical conditions that significantly impact patient morbidity and mortality, involving intricate pathophysiological mechanisms. HF often results in reduced cardiac output and blood stasis, particularly within the venous system, which increases the likelihood of thrombus formation. The elevated venous pressure associated with heart failure can lead to endothelial dysfunction, heightening the risk of thrombosis. 40 Previous clinical studies have consistently indicated that left ventricular dysfunction may serve as a risk factor for PHVT. 41 The finding from our research that congestive heart failure is the strongest independent predictor of PHVT, associated with a threefold increase in risk, further supports these observations.
Study Limitations
This study is a single-center, retrospective, observational investigation with a limited patient cohort, which inherently reflects the limitations associated with a retrospective design. While there is potential for selection bias, efforts were made to include consecutive patients. Additionally, the absence of histopathological confirmation, which is essential for a definitive diagnosis of PHVT, should be noted. Nevertheless, TEE, regarded as the gold standard for diagnosing PHVT, was employed in this study. To enhance the reliability of our findings, we excluded patients whose results were difficult to interpret and those with prosthetic valve occlusion without evidence of thrombus. Furthermore, since the study focused exclusively on patients with PHVT in the mitral position, we were unable to assess the predictive value of the RDW-to-PLT ratio in those with PHVT in the aortic or right heart valves position. In addition, although it was not the primary focus of our study, we were unable to assess the impact of the RDW-to-PLT ratio on clinical outcomes in this patient population due to the lack of follow-up and outcome data. Another limitation that should be noted is that, although our analysis did not include patients with severe anemia, the proportion of anemic patients was very low, and no significant difference in anemia prevalence was observed between the two groups. However, the presence of anemia as a comorbidity may still have influenced the RDW results.
Conclusions
This study represents the first analysis to document an independent association between elevated the RDW-to-PLT ratios and the development of PHVT in patients with mechanical prosthetic mitral valves. Our findings highlight the RDW-to-PLT ratio as a valuable, non-invasive biomarker for predicting PHVT development and emphasize its potential for integration into routine clinical practice to enhance the early detection and management of PHVT in patients with left-sided mechanical prosthetic valves. By facilitating the early diagnosis and management of PHVT, this biomarker could serve as a promising addition to the current diagnostic toolkit, ultimately leading to improved outcomes in this high-risk population. However, further research is warranted to validate these findings and to establish whether the RDW-to-PLT ratio serves as a definitive risk factor for PHVT development across broader clinical settings.
Footnotes
Ethics Considerations and Consent to Participate
The Ethics Committee of Ankara Etlik City Hospital approved this retrospective study and waived the requirement for informed consent.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
