Abstract
Introduction
Thrombophilia (TP) has been proposed as a potential contributor to preeclampsia (PE). However, there is not a clear consensus on testing PE patients for TP due to contradictory evidences on this association. This study aims to identify what conditions of women with PE are associated with acquired and hereditary TP, and, additionally, to build a model of TP probability using these characteristics.
Material and Methods
Retrospective unicentric analysis of women diagnosed with PE referred for TP testing between May 2019 and May 2024.
Results
In 95 women, 19 (20%) tested positive for TP, of which nine (47%) had antiphospholipid syndrome (APS), six (32%) were heterozygous for PT G20210A, two (11%) had ADAMTS13 deficiency, one (5%) had PS deficiency and one (5%) had heterozygous FV Leiden. In multivariate analysis, intrauterine growth retardation (IUGR; odds ratio (OR) 0.08, 95% confidence interval (CI) 0.01-0.55) and history of previous abortions (OR 0.22, 95%CI 0.06-0.96) were negatively associated with TP. The group of women with both, one or none of those traits showed respectively a TP prevalence of 0%, 15% and 32%. The higher the prevalence of TP, the lower the incidence of placental insufficiency and prematurity (P < .05).
Conclusions
In pregnant women with PE, a history of previous abortions and IUGR were independently associated with the absence of TP. Women without these characteristics would probably benefit most from a Hematology consultation that includes a TP screening. A multinational standard TP screening framework for future studies is warranted to further our understanding of the role of TP in PE and to identify risk-groups for testing.
Keywords
Highlights
The prevalence of acquired and hereditary thrombophilia in preeclampsia was 20%
Previous abortions and intrauterine growth restriction were independently associated with the absence of positive results in the thrombophilia workup.
Introduction
Preeclampsia (PE) affects 3%-5% of all pregnancies and causes significant maternal and fetal morbimortality. 1 PE stems from dysfunctional placental arteriolar remodeling, with hypoperfusion that leads to proinflammatory and antiangiogenic stimuli into the maternal bloodstream, resulting in arterial hypertension and multiorgan failure in the mother, and intrauterine growth restriction (IUGR), prematurity and even fetal miscarriage.1-3
Thrombophilia (TP) is defined as a genetic and/or acquired hemostasis disorder that predisposes to the development of thromboembolic episodes. TP has been historically linked to PE 1 by increasing the hypercoagulable state of an already prothrombotic environment during pregnancy, which may impair placental perfusion and trigger/worsen PE and other gestational adverse events. 4 Evidence suggests that early and severe PE may be more frequently caused by TP, due to the cumulative effect from TP on pregnancy. 1 However, there is opposing data on this association, especially regarding hereditary TP.3-6 Consequently, there is no clear consensus between different guidelines on the indication of TP screening in women with PE,7,8 and more evidence is needed to aid the clinical decision of when to order TP testing in this setting.
This study aims to identify what conditions of women with PE are associated with the presence of acquired and hereditary TP, and, furthermore, to build a model of TP probability using these characteristics.
Methods
Study Population and Inclusion Criteria
This retrospective, sequential, single center, non-interventional analysis includes all consecutive women aged ≥18 years referred for TP investigation in the context of PE in a previous pregnancy, by the Gynecology and Obstetrics and the Internal Medicine departments, to the Hemostasis and Hemotherapy Unit, from The University and Polytechnic La Fe Hospital of Valencia, Spain, between May 2019 and May 2024. All included women had not previously been referred for TP screening and, therefore, had not been prescribed prophylaxis with acetylsalicylic acid nor low molecular weight heparin by a hematologist during pregnancy. All patients signed an informed consent for TP testing and genetic studies. This study was performed according to the Declaration of Helsinki, and procedures were approved by the institutional ethics committee.
Procedures and Definitions
A purposely designed TP panel included testing for antiphospholipid antibodies (lupus anticoagulant, anticardiolipin antibodies, anti-β2-glycoprotein-I antibodies), protein C (PC) deficiency, protein S (PS) deficiency, antithrombin (AT) deficiency, screening activated protein C resistance and confirmatory genetic factor V Leiden (FVL) testing, prothrombin (F2) mutation G20210A and ADAMTS13 levels measurement. Antiphospholipid syndrome (APS) was diagnosed according to consensus guidelines, 9 with laboratorial tests conducted on two separate samples collected at least three months apart. PC, PS and AT deficiency laboratorial diagnosis followed international recommendations.10-12 Accordingly, at our laboratory, antiphospholipid antibodies were considered positive when anticardiolipin (ACA) IgG and/or IgM antibodies were present in titers >20 U/mL, and antibeta2-glycoprotein I (anti-B2-GPI) IgG and/or IgM antibodies were present in titers >20 U/mL; PC deficiency was diagnosed when values were below 70%, while PS deficiency was considered at 65% and AT deficiency at 70%. Finally, ADAMTS13 deficiency was diagnosed when values were below 50%.
PE diagnosis and severity grading followed guideline recommendations. 13 Preterm birth was defined as natural or induced birth before 37 weeks of gestation. Fetal death was defined as any death occurring during gestation or labor. Placental insufficiency and IUGR were classified according to obstetric charts and ultrasound reports by the attending obstetrician team that followed the pregnancy. For history of abortion, we only considered involuntary pregnancy termination, up to 22 weeks of gestation, according to WHO definition 14 ; all other forms of abortion were not considered for patient and family history. Upon 22 weeks of gestation, the death of the fetus was considered fetal loss.
Endpoints and Statistical Analysis
Considering the limitations of the sample size and the absence of a control group, the primary objective was not to identify TP risks factors for developing PE, but instead we focused on identifying conditions of a selected sample of women diagnosed with PE that show a significant association (positive or negative) with the presence of TP. The secondary endpoint was to propose a pretest model to predict TP diagnosis probability in PE when these conditions are present.
Categorical variables were presented as percentages and compared using Pearson chi-square test (expected group size ≥5) or Fisher's exact test (expected group size <5), and continuous variables as median values with ranges and compared with Mann-Whitney U or Kruskal-Walis test as appropriate. Binomial logistic regression models were used for TP detection univariable and multivariable models. Variable selection in the multivariable model was performed using automatic selection by stepwise backward regression from a full model with all variables of interest. The level of significance was set at <.05 (2-sided) for all comparisons. Statistical analysis was conducted using R (The CRAN project).
Results
Patient, Pregnancy with PE and TP Characteristics
The main patient and PE characteristics of 95 women included in the study are summarized in Table 1. The median age at referral was 35 years (range, 20-55), 48% were nulliparous, 28% had a previous abortion and 6% a previous episode of thrombosis (2 cases of cerebral thrombosis and 4 cases of deep vein thrombosis [DVT], one of them in the context of systemic lupus erythematous). Median time to delivery in the PE pregnancy was 32 weeks (range 20 to 40 weeks), with 64% premature deliveries and 18% fetal deaths. Characteristics were comparable between patients with or without TP, except for patients with TP having more severe PE (P = .010) and less IUGR (P = .021). Out of the 19 women (20%) with identified TP, nine (47%) had APS, six (32%) were heterozygous for PT G20210A, two (11%) had ADAMTS13 deficiency (median levels of 43.3% in one case and 38.7% in the other), one (5%) had PS deficiency (confirmed by genetic testing) and one (5%) had heterozygous FVL. Regarding the nine APS patients, three were positive for lupus anticoagulant, six for ACA (median 120 U/mL, range 42-485; four high positive), and four for anti-B2-GPI (median 938 U/mL, range 76-2292; three high positive). One APS case was based on lupus anticoagulant alone, two were triple positive, two were ACA and anti-B2-GPI positive, while the remainder were either ACA or anti-B2-GPI positive. Within the whole sample, the prevalence of studied TP is: 9% of APS, 6% of PT G20210A mutation, 1% for PS deficiency, 1% for heterozygous FVL and 2% for ADAMTS13 deficiency. We did not find any cases of PC or AT deficiency.
Patient and pregnancy characteristics.

Note: P values <0.05 are in bold.
Legend: PE, preeclampsia; IUGR, intrauterine growth restriction.
Risk Factors and Risk Groups for Inherited and Acquired TP
Univariable analysis only identified IUGR as decreasing the odds ratio (OR) of having TP at referral (Table 2). Using a stepwise backward model with all clinical variables, the final multivariable model included previous abortions, family history of thrombosis, abortion or PE, severe PE, placental insufficiency, and IUGR. In this model, the presence of previous abortions (OR 0.22, 95% confidence interval [95% CI] 0.06-0.96) and IUGR (OR 0.08, 95% CI 0.01-0.55) were independently associated with the absence of inherited or acquired TP (Table 2).
Factors for Increased Risk of Inherited and Acquired TP.

Note: P values <0.05 are in bold.
Legend: TP, thrombophilia; PE, preeclampsia; IUGR, intrauterine growth restriction.
Using these two independent variables, we built a model with three risk groups for being diagnosed of TP: low-risk (presence of both previous abortions and IUGR), intermediate-risk (presence of one of these factors), high-risk (absence of both factors). The presence of TP was 0%, 15% and 32% for the low, intermediate, and high-risk groups, respectively. Patient and pregnancy characteristics for each risk strata are summarized in Table 3. Besides differences in abortions and IUGR, the higher the risk for TP, the lower the incidence of placental insufficiency and prematurity of the pregnancy (P < .05).
Patient, Pregnancy, and TP Diagnosis According to TP Risk Groups.
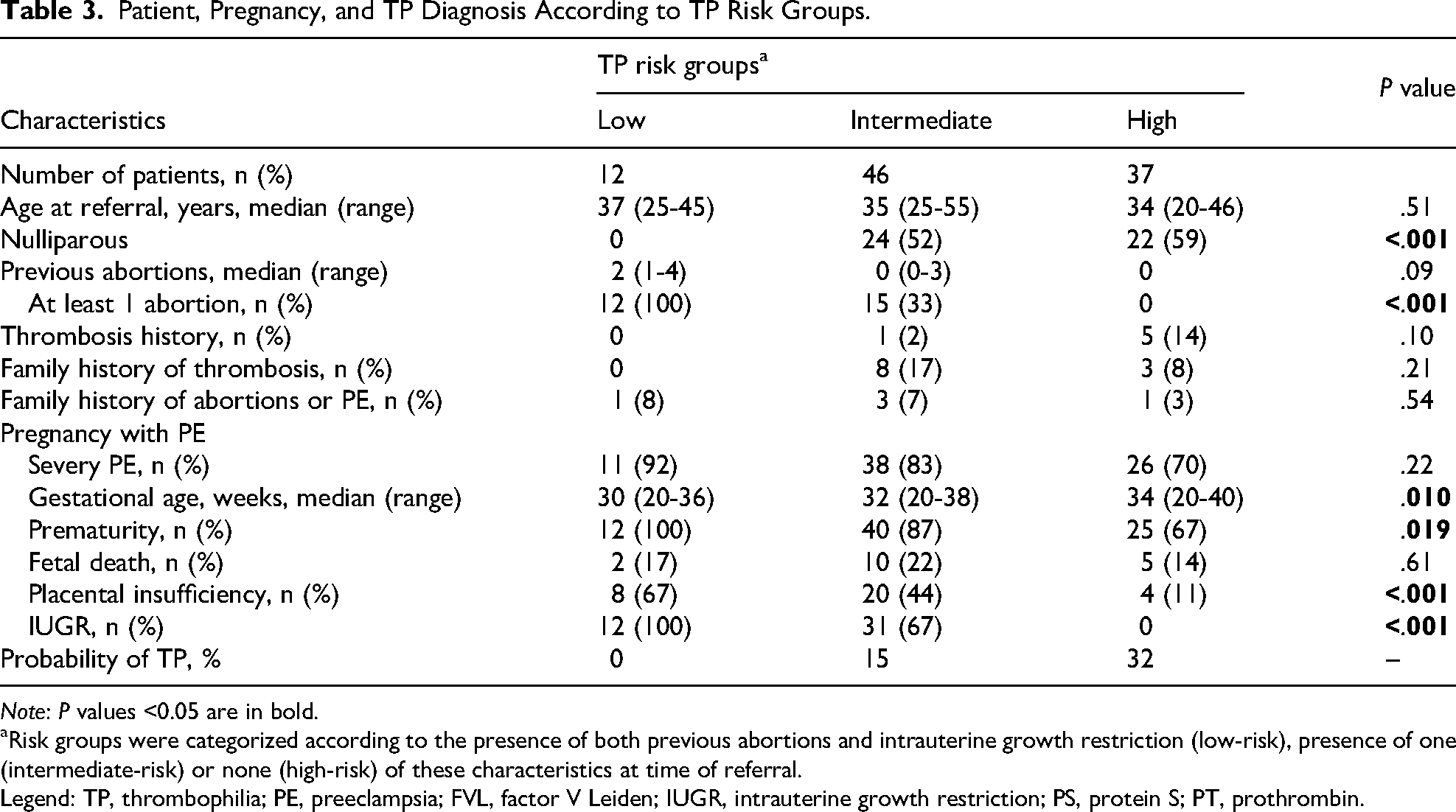
Note: P values <0.05 are in bold.
Risk groups were categorized according to the presence of both previous abortions and intrauterine growth restriction (low-risk), presence of one (intermediate-risk) or none (high-risk) of these characteristics at time of referral.
Legend: TP, thrombophilia; PE, preeclampsia; FVL, factor V Leiden; IUGR, intrauterine growth restriction; PS, protein S; PT, prothrombin.
Separately analyzing acquired (n = 9) and inherited (n = 10) TP, in multivariate analysis only IUGR was independently associated with the absence of acquired TP (OR 0.10, 95% CI 0.01-0.87), while inherited TP was not associated in any way with any factor.
Discussion
This study of selected women with history of PE shows a prevalence of acquired and/or inherited TP of 20%. We found that any previous abortions and IUGR were independently associated with the absence of TP, and that none of the analyzed factors were positively associated with TP. Additionally, we developed a model that categorizes women with background of PE into three groups of varying probabilities of having TP. While these findings should be interpreted with caution and warrant confirmatory studies, they can nonetheless inform of pretest probability in clinical practice.
One of the most challenging aspects of TP assessment in PE is building a consensus on which TP factors should be studied. On the one hand, we included ADAMTS13, which has been associated with PE in some studies, 15 but there is also contradictory evidence on its role. 16 On the other hand, we did not study other factors that have also been inconsistently reported to worsen pregnancy outcomes, such as plasminogen activator inhibitor type 1 (PAI-1) polymorphisms17-19 and methylenetetrahydrofolate reductase (MTHFR) mutations.20,21 Yet, even for classic TP factors, there is conflicting evidence regarding the development of PE during pregnancy.3,5 Efforts should be made to harmonize an international TP panel in PE, which will facilitate study comparison across different populations.
Owing to the difficulty to provide consistent evidence of the association between TP and PE, another challenging facet is whether to study TP at all. Sample size, ethnic variations and inclusion criteria may contribute to interstudy discrepancies, and it is therefore important to contextualize our sample within the national and worldwide panorama, which is represented in Table 4. Overall, the prevalence of inherited TP in our sample seems to be similar and below to that of the general population and patients with a history of DVT, respectively. The prevalence of APS seems to be closer to populations with thrombosis and higher than in overall population. Valencia is an area where 16.5% of the population is of foreign nationality, 22 with substantial immigrant communities from other European countries, Northwest Africa, Latin-America and Eastern Asia, which adds another layer of complexity, so comparisons should be done cautiously. There is no available data for mild and moderate ADAMTS13 deficiency, perhaps because, so far, all studies have been focused on the prevalence of severe deficit as the cause of thrombotic thrombocytopenic purpura. 23
Prevalence of Main TP in Both General Population and our Sample.

Legend: *Prevalence for antiphospholipid antibodies. APS, antiphospholipid syndrome; AT, antithrombin; DVT, deep vein thrombosis; ; FVL, factor V; PC, protein C Leiden; PS, protein S; PT, prothrombin.
When comparing the characteristics of our sample based on TP testing findings, we observe a significant difference between the prevalence of severe PE and IUGR (Table 1). For the first, all patients who tested positive for TP suffered from severe PE, which is consistent with findings in previous studies.5,37 As pointed out by Bohiltea R et al, 37 and in line with our findings, there may be a need to consider a clinical and physio-pathological spectrum within PE, where TP only plays a significant role in severe PE. Regarding IUGR, it was more frequently reported in patients who tested negative for TP. Although there is contradicting evidence, and considering the TP test panel used in our study, the relation between TP and IUGR is uncertain.20,38-41 Interestingly, and also in line with our findings, one study found that the association between IUGR and fetal TP was stronger in the absence of PE and pregnancy-induced hypertension, 42 which suggests that in PE with IUGR, other maternal-fetal factors may be at play.
Using a multivariable regression model, we identified two variables (previous abortions and IUGR) that allowed for the division of PE into three risk groups (Table 3), where the “low-risk” group (both variables present) is characterized by TP as not having a major role in the genesis of PE. Placental insufficiency is the most frequent cause of IUGR and is known to be associated with prematurity, 43 which in turn may jeopardize fetal viability. Additionally, placental histology in cases of early-onset PE and IUGR is not different between women with or without TP, 44 and studies on pre-term birth did not find an association between PE and the presence of TP.4,45 Therefore, our “low-risk” group is composed of women with vascular factors beyond TP, which have resulted in previous abortions and have complicated the current pregnancy with IUGR, placental insufficiency, PE, and most likely ended with a premature labor. As we remove IUGR and previous abortions in the “intermediate-risk” (only one factor present) and “high-risk” (none present) groups, the probability of TP being detected in a PE episode increases to 15% and to 32%, respectively. Nevertheless, it should be kept in mind that this analysis was primarily designed to identify patient characteristics that may benefit more from TP screening. Furthermore, we did not use recurrent pregnancy loss 46 as a criterion for this analysis. Further research is needed to confirm whether these risk-group features are based on distinct pathophysiological pathways.
This study has some limitations that should be considered, starting with a selection bias. Our study sample consists of women with previous PE that were referred from the Obstetrics and Internal Medicine clinics for TP testing, but, presumably, some women may have not been correctly included for several reasons, such as not having been diagnosed or followed in the past, or differences in criteria when considering whom to refer. This places the onus on the referring physician to subjectively classify PE episodes as significant enough to warrant a TP study. Nevertheless, it must be said that, up to now, the internal consensus at our institution favors referring every single patient affected by PE to be screened, hence the interest of this investigation to focus in defining the pretest probability of having a positive TP study to optimize resources. Another constraint of our study is data collection on variables that may be related to the presence of both PE and TP, which was not exhaustive. Furthermore, the limited sample size may have led to the omission of clinically significant associations, reducing result generalizability and limiting the strength of our conclusions. It may also have affected the analysis when considering acquired and inherited TP separately, while differences in comorbidities and interindividual factors could further account for the observed contrasts. Finally, the TP panel we report may not be universally applied in all centers, which may hinder study comparison and results generalizability.
On the other hand, our study has notable strengths. Firstly, we report on an extensive multiagent panel for TP testing, which exceeds common clinical practice, while most studies focus on individual factors. Additionally, we developed a prediction model with easily accessible clinical variables. If validated in future studies, this model could be integrated into routine clinical counselling. Finally, while most studies typically report on the risk of PE in the context of TP, we present what we believe to be, to the best of our knowledge, an innovative and practical approach by investigating TP in women referred to Hematology after experiencing an episode of PE.
In conclusion, in pregnant women with PE, a history of previous abortions and IUGR are independently associated with the absence of TP. Women without these characteristics would probably benefit most from a Hematology consultation that includes a TP screening. A multinational standard TP screening framework for future studies is warranted to further our understanding of the role of TP in PE and identify risk-groups for TP testing during pregnancy to improve maternal-fetal outcomes.
Footnotes
Acknowledgements
We want to thank patients, nurses, and all colleagues that collaborate with our department.
Authorship Statement
A.G., P.C., M.C.G. and E.T contributed to data retrieval. A.G. and P.C. performed the statistical analysis. A.G., P.C., E.T. and S.B. drafted the manuscript and integrated changes and suggestions made by M.C.G., A.B., F.F., A.C., AM and S.H. All authors reviewed and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
