Abstract
We aimed to scrutinize the risk factors for thrombosis in children with acute lymphoblastic leukemia treated with the Berlin-Frankfurt-Münster 95 protocol. The study population was 82 children younger than 16 years of age. The children were followed up for 10 years until January 2007. Thrombosis occurred in 10 (12%) of 82 patients during the treatment course, mainly after the M protocol. The most common risk factor was factor V Leiden (FVL; 15.6%). This was followed by methyleneterahydrofolate reductase (MTHFR; 9.3%), elevated lipoprotein (1.5%), and prothrombin (PT) 20210A (1.5%) in descending order. The risk of thrombosis was found to be significantly high in patients with FVL mutation (odds ratio = 7.1, 95% confidence interval = 1.6-30.5). The risk of thrombosis was not significant in patients with MTHFR and PT20210A mutation (P = .2). Age, catheter usage, FVL mutation, and prednisolone treatment are significant risk factors for thromboemboli occurrence.
Introduction
Thrombosis is a relatively rare but serious complication of acute lymphoblastic leukemia (ALL) in childhood. The incidence of thrombosis in ALL ranges between 1.1% and 36.7% and it develops due to genetic and acquired risk factors that operate collectively.1–4 Some of the essential risk factors are factor V Leiden (FVL) mutation, activated protein C resistance, prothrombin (PT)20210A mutation, methyleneterahydrofolate reductase (MTHFR) mutation, protein C deficiency, protein S deficiency, antithrombin III deficiency, central venous catheter, and some drugs.5,6
In this study, we aimed to scrutinize the risk factors for thrombosis in children with ALL treated in a tertiary center.
Materials and Methods
The study has been carried out at the Pediatric Hematology Unit of the Gazı University Faculty of Medicine on 82 children younger than 16 years who were diagnosed with ALL after January 1997 and treated with Berlin-Frankfurt-Münster (BFM) 95 protocol. The children were followed up for 10 years until January 2007. The ethical committee of the hospital accepted the study protocol.
The patients who died unrelated to thrombosis 15 days after the initiation of treatment protocol, the patients who had -catheter lumen blockage, and the patients who had thrombus at the tip of the catheter were not included in the study.
The diagnosis of thrombosis has been made with Doppler ultrasonography, magnetic resonance imaging, and echocardiogarphy.
The FVL, PT20210A, and MTHFR mutations have been determined using DNA polymerase chain reaction. Antiphospholipid antibodies, homocysteine, and lipoprotein a (Lp-a) levels have been measured with the use of standard laboratory techniques.
The collected data have been analyzed with SPSS 16.0 software package. The statistical comparisons of the collected data have been performed with Wilcoxon and Student t test, where appropriate. The correlation between the data has been done with the Pearson correlation analysis. The comparisons in terms of thrombosis risk have been realized through forward-Wald method. The statistical significance has been set at P < .05.
Results
The mean of age of the study population was 6.9 ± 4.2 years (minimum 1-maximum 15 years). The ratio of boys to girls was 1.3 (47 of 35).
None of the patients have had thrombosis at the index diagnosis. Thrombosis occurred in 10 (12%) of 82 patients during the treatment course.
There was no statistically significant difference in terms of gender between the groups (P = .4). However, 3 patients were younger than (3 of 10) and 7 patients were older than (7/10) 10 years. The thrombosis frequency was found to be statistically significant when comparison was done between the groups (P = .03).
Thrombosis most commonly occurred during the M protocol (Table 1).
The Thrombosis Formation According to the Treatment Protocol in Children With ALL.

Abbreviation: ALL, acute lymphoblastic leukemia.
The locations of thrombosis in order of frequency were right atrium (4 of 10), jugular vein (2 of 10), subclavian vein (1 of 10), right atrium plus brachiocephalic vein (1 of 10), right atrium plus tip of catheter (1 of 10), and stroke (1 of 10).
The total number of patients in whom we could investigate the hereditary risk factors was 64 (Table 2). The existence of risk factors was noticed in 19 (29.6%) of 64 patients. No risk factor was seen in 45 (70.4%) patients. There was only 1 risk factor in 16 (25%) patients whereas multiple risk factors were seen in 3 (4.6%) patients. The most common risk factor was FVL (15.6%). This was followed by MTHFR (9.3%), elevated Lp-a (1.5%), and PT20210A (1.5%) in descending order.
The Frequency of Thrombophilic Risk Factors in Children With ALL.

Abbreviations: ALL, acute lymphoblastic leukemia; AT, antithrombin; FVIII, factor VIII; FVL, factor V Leiden; Lp-a, lipoprotein a; MTHFR, methyleneterahydrofolate reductase; PC, protein C; PS, protein S; PT, prothrombin.
Thrombosis has been found in 8 (42.1%) of 19 patients who had thrombophilic risk factor, whereas thrombosis has been found in 2 (4.4%) of 45 patients who had no thrombophilic risk factor. When a comparison was done in terms of thrombosis development between the groups it has been noted that thrombosis occurrence was 10 times more likely in the thrombotic group (P = .002, odds ratio [OR] = 10.1, 95% confidence interval [CI] = 2.3-44.8).
The frequency of the thrombophilic risk factors that were determined in thrombotic patients has been given in Table 3. There was at least one risk factor in 8 (80%) of 10 patients with thrombosis whereas thrombophilic risk factor was found in 11 (20.3%) of 54 patients without thrombosis (P = .002).
The Frequency of Prothrombotic Risk Factors in 10 Patients With Thrombosis.
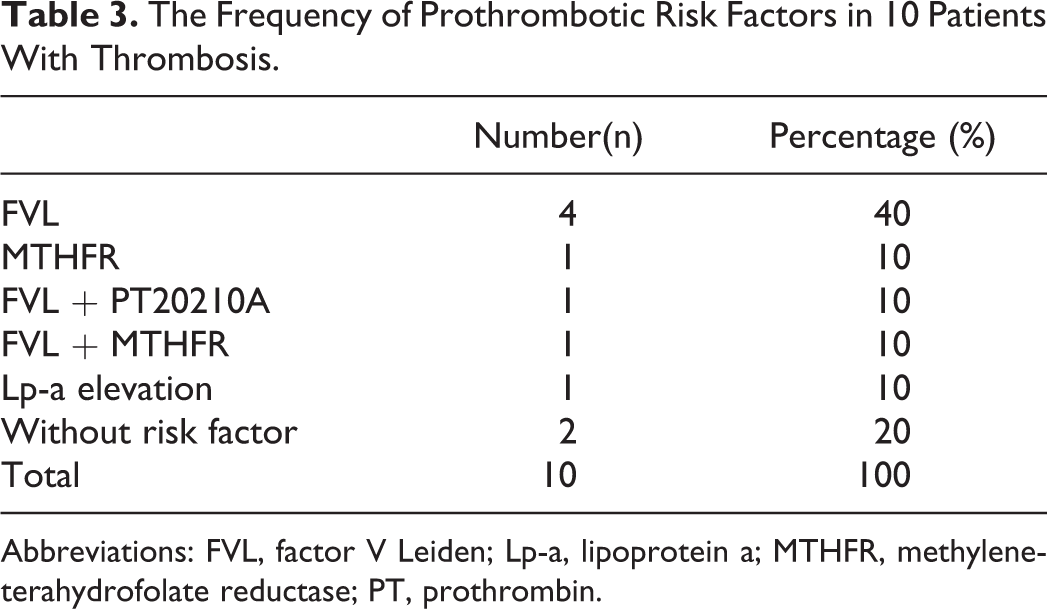
Abbreviations: FVL, factor V Leiden; Lp-a, lipoprotein a; MTHFR, methyleneterahydrofolate reductase; PT, prothrombin.
Thrombosis was present in 6 (46%) of 13 patients with FVL mutation. The frequency of FVL between the patients who had thrombosis was 60% (6 of 10). The FVL mutation has been noted in 7 (12.9%) of 54 patients without thrombosis (P = .002). The risk of thrombosis was found to be significantly high in patients with FVL mutation (OR = 7.1, 95% CI = 1.6-30.5). On the other hand, the risk of thrombosis was not significant in patients with MTHFR and PT20210A mutation (P = .2).
Discussion
The incidence of thrombosis in children with ALL varies between 1.1% and 36.7% and the actual mean is 3.2%.2,3 We found the thrombosis rate to be 12% in children who had had BFM-95 protocol for their treatments. This rate is in accordance with that reported by Nowak-Gottl et al. 3 Thrombosis is most commonly seen in the newborn and adolescence period during the childhood. A study reported that the incidence of thrombosis was higher in the children between 10 and 18 years compared to the children who were younger than 10 years. 7 Our study also showed that the thrombosis frequency in the children who were 10 years or older (70%) was significantly high compared to the children who were younger than 10 years (30%; P = .03). However, the children who are older than 10 years can be classified as high risk; hence, an intense chemotherapy treatment would increase the thrombosis risk.
The role of gender in thrombosis is not clear. Nowak-Gottl et al and Pui et al declared that the thrombosis risk was higher in girls and boys, respectively, 8 whereas our study did not find any significant difference between the genders (P > .05).
Nowak-Gottl et al compared the incidence of thrombosis in children with ALL who had prednisolone or dexamethasone as a steroid treatment in BFM-95 protocol and found that the incidence of thrombosis was 10.4% and 1.8% in prednisolone and dexamethasone groups, respectively. 3 Our study group was formed ofpatients who had taken only prednisolone and had thrombosis rate (12%) similar that similar to that of Nowak-Gottl et al’s ratio. Our finding supports that dexamethasone usage instead of prednisolone is more rationale due to higher thrombosis complication in BFM-95 protocol.
The thrombosis was commonly seen in the right atrium in our group (4 of 10). The right atrial thrombosis most commonly develops as a catheter complication without some rare exceptions. 9 Contrary to this, some studies have denoted that the thrombosis in children with ALL frequently localized in central nervous system (50%). 10 The localization of thrombosis in right atrium, subclavian vein, and jugular vein in the majority of cases show that the most important risk factor is cerebrovascular hemorrhage for the development of thromboemboli.
A hereditary thrombophilic factor is found at a rate of 58% in childhood thrombosis. Nowak-Gottl et al found the prothrombotic risk factor at a rate of 20% in children with ALL and they also declared that the incidence of thrombosis was 46.5% and 2.2% in risky and nonrisky groups, respectively. 8 In our study, we found the incidence of prothrombotic risk factor as 29.6%, the thrombosis frequency was 42.1% in risky group, whereas 4.4% in the nonrisky group.
We did not determine any homozygote FVL case, whereas the thrombosis risk was significantly high (7 times) in cases who had heterozygote FVL mutation. This finding shows that the screening for FVL mutation would be helpful in children with ALL during the diagnosis period. On the other hand, we did not find any significant correlation between thrombosis development and increased PT20210A and Lp-a levels and MTHPR mutations though MTHFR mutations are not important risk factors unless the patients have high homocysteine levels.
Conclusion
We conclude that age, catheter usage, FVL mutation, and prednisolone treatment are significant risk factors for thromboemboli occurrence.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
