Abstract
Significant progress has been made in treating Coronavirus disease (COVID) – an infectious disease caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). An ominous turn in the pandemic is the evolving public health crisis emanating from persistent SARS-CoV-2 infection and its associated long-term impact. Long COVID or post–COVID syndrome describes protean symptoms that persist at least 3 months after the onset of acute illness and last for at least 2 months in individuals with a history of confirmed SARS-CoV-2 infection. Long COVID has become a public health concern. Millions of infected individuals are now facing chronic multi-organ failures, including neuropsychiatric, cardiovascular, pulmonary, and kidney complications. In general, the cause of long COVID syndrome is unclear but factors such as prolonged activation of immune responses, and viral persistence triggering transcription dysregulation of genes associated with normal thrombotic disease may play a role in cardiovascular complications. Although inflammatory biomarkers are reported in other disorders, it remains unclear whether similar biomarkers are associated with cardiovascular manifestations following COVID. Medications such as sulodexide directed at glycocalyx and coagulation have demonstrated benefits for long COVID in smaller studies. Here, we describe the outcomes of the symposium on the underlying cardiovascular mechanisms of the long COVID.
Keywords
Background
We organized the symposium on June 4, 2024, involving global experts. At the request of the Veterans Affairs (VA) long COVID team, we were tasked with developing special talks on the cardiovascular effects of long COVID. To comprehensively address this, we organized one-day international symposium to discuss long COVID impact on cardiovascular as well as other multi-organ systems (neurovascular system). The symposium centered around different international experts with a focus on epidemiology, economic impact, basic mechanisms, biomarkers and potential therapeutics.
Introduction
The global spread and persistence of coronavirus, SARS−CoV−2 continues to inflict a huge toll on the healthcare system, specifically immunocompromised and high−risk populations remain vulnerable to acute COVID. With increasing vaccination rates and other mitigation efforts, severe SARS-CoV-2 infection is declining. As of December 2024, there are 676+ million cases of COVID, with a total of 6.88+ million deaths, with 28 days death rate of 28 081 worldwide.1,2 A recent survey indicated that approximately 7% of adults and more than 1% of children (15 to 20 million Americans and more than 60 million globally) have had long COVID.3,4 An ominous development in the pandemic is the progression of many symptoms beyond 4 to 12 weeks− termed long COVID syndrome- an evolving public health crisis.5–7 Long COVID syndrome is a public health concern affecting millions of people worldwide with multisystem diseases like neurologic, psychiatric, cardiovascular, and pulmonary complications and impacting activities of daily living (ADLs), physical functioning, and professional and personal life. With the lack of information on the underlying molecular determinants and pathways that mediate these multisystem diseases, identifying circulating and other biomarkers can guide disease prognostication and generate a tool for classifying at−risk groups for developing novel and using existing therapeutics. The COVID pandemic has negatively affected the GDP by twice that of the Great Recession of 2007-2009 and 20 times that of the 9/11 terrorist attacks. 8
COVID is associated with multiple abnormalities in several organs months after infection. 9 Long COVID is broadly defined as signs, symptoms, and conditions that continue or develop after initial SARS−CoV−2 infection and can last for weeks, months, or years thereafter without completely resolving.5,10 Several studies have proposed potential contributions to long COVID not limited to viral persistence, including organ damage in targeted tissues, ongoing immune or inflammatory activation, downstream effects of Epstein-Barr virus (EBV) reactivation, endothelial activation or dysfunction to altered allergic profiles, impacts on platelet activation, clotting or micro clots, mast cell activation, autoimmunity microbiota dysbiosis, altered metabolic interactions with the immune system. 5 Local micro thrombosis resulting from hypercoagulability or mitochondrial failure can lead to structural alterations observed on brain Magnetic Resonance Imaging (MRI, decreased cortical thickness, decreased cerebral blood flow) compared to controls.11–13 Neurological symptoms, collectively, may be the most frequent manifestations of long COVID, while cardiovascular sequelae are less common, they remain challenging to diagnose, treat, and explain on a pathophysiological basis. Some cardiac pathology may reflect alterations in the autonomic system such as postural orthostatic hypotension (POTS). Additionally, although acute infection, as well as COVID-19 vaccination, may lead to myocardial inflammation, micro thrombosis, and alterations in systolic and diastolic dysfunction, much less is known about the underlying cardiovascular pathophysiology in long COVID aside from common phenotypic symptoms presentation such as chest pain or palpitations.
Definition of Long-COVID
According to the World Health Organization (WHO), long COVID is defined as a post-COVID condition that occurs in individuals with a history of probable or confirmed SARS-CoV-2 infection, usually 3 months from the onset of COVID, with symptoms that last for at least 2 months and cannot be explained by an alternative diagnosis. 14 Other names for long COVID include long-haul COVID, Post-COVID conditions (PCCs), Chronic COVID
Post-acute sequelae of COVID-19 (PASC). Common symptoms include fatigue, shortness of breath, cognitive dysfunction, and others that generally impact everyday functioning. Symptoms may be new onset following initial recovery from an acute COVID-19 episode or persist from the initial illness. Symptoms may also fluctuate or relapse over time. As per the Centers for Disease Control (CDC), long COVID is a chronic condition that occurs after SARS-CoV-2 infection and is present for at least 3 months. According to National Academies of Sciences (NAS), long COVID is an infection-associated chronic condition that occurs after SARS-CoV-2 infection and is present for at least 3-months as a continuous, relapsing and remitting, or progressive disease state that affects one or more organ systems. 3 Many limitations exist for the above definitions, for example, the absence of definitive biomarker profiles reflecting limitations in current scientific knowledge. 4
Long COVID and the Nervous System
Potentially, the greatest amount of data regarding long COVID comes from the neuropsychiatric fields. Neuropsychiatric symptoms associated with Long COVID are prominent, common, and may be severely debilitating in some patients. Frequently reported symptoms are chronic fatigue, post-exertional malaise, brain fog, cognitive impairment, memory deficits, inattentiveness, depression, anxiety, and alterations in smell or taste. Less commonly, patients may report new or worsening headaches, speech alterations, or abnormal movements. 15 Dysautonomia may also be a salient feature and some patients with long COVID will also manifest comorbid postural orthostatic tachycardia syndrome (POTS). While efforts to understand the pathophysiology of long COVID are ongoing, there is evidence that its etiology may involve viral persistence and reactivation, autoimmunity, immune dysfunction, dysbiosis of gut microbiota, endothelial dysfunction, microclots, and signaling alterations of the nervous system. 4 Many of these suspected mechanisms, and even several of the symptoms of long COVID, overlap with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), a post-viral syndrome typically associated with EBV.4,15 Our current understanding of long COVID and its neurological symptoms are being greatly aided by the decades of research already accomplished in ME/CFS. 16
There is a paucity of validated and reliable treatment options for long COVID, and more well-designed randomized clinical trials are needed. With the allocation of over $1.15 billion from Congress, 17 the NIH set up the researching COVID to enhance recovery (RECOVER) research network to further elucidate the mechanisms of long COVID and potential management options. Indeed, the initiative has produced several phase II clinical trials that are enrolling as of June 2024. Lastly, adequate knowledge of long COVID and related post-viral syndromes is lacking among clinicians, which results in misdiagnoses, delayed diagnoses, and adverse patient outcomes. Increased efforts among research and health organizations to educate frontline providers on these topics will be necessary to achieve progress with COVID. Our panel will discuss cardiovascular findings based on what we have learned from neuropsychiatric areas.
Epidemiology of Cardiovascular Manifestations of Long COVID
In an article published in 2023 by Swiss researchers and medical professionals, about 10%-35% of COVID patients may develop long COVID. 18 The repercussions from ongoing symptoms of COVID affect both the individual as well as their family and support system. An article in the Journal of Health Economics and Outcomes Research estimates 4 million people out of work due to extenuating COVID symptoms.19,20 Additionally, long COVID was recognized as a disability in America beginning in July 2021. A study of 15 308 participants aged 18-69 found that 12% were unemployed. 21
Economic Impact of Long-COVID
These circumstances lead to the identification of the 3 most prevalent types of economic impact found by David Cutler at Harvard University. In 2022, David Cutler published an updated article with the economic impact of long COVID in the United States. 19 In it, he describes the 3 main types of impact as loss of quality of life, lost earnings, and higher medical care. 19 The loss of quality-of-life stems from the uncertainty of the length of long COVID, resulting in a net loss of $2.2 trillion. The lost earning refers to shifts in workplace dynamics and wages. There are currently an estimated 8.4-9.6 million adults with at least 3 symptoms of long COVID. Additionally, 60% of the adult class is in the workplace. Because of long COVID symptoms, 70% of those with long COVID have faced reduction in hours worked or job loss resulting in a net earning loss of income of $1 trillion. Finally, the underlying issue with increasing cases of COVID is higher medical care. For example, he individual annual cost for myalgic encephalomyelitis and chronic fatigue syndrome ranges from $3000-$13 000. Additional annual medical spending is $528 billion for direct and indirect health services related to long COVID. Cutler found that the combined cost of loss of quality of life, lost earnings, and higher medical care amounts to $3.7 trillion. Taking this number as well as the individual lives affected by long COVID, we see a need for greater understanding and problem solving related to tackling long COVID through additional research and awareness.
Challenges in Cardiac Symptoms and Treatments in Long COVID
Cardiac symptoms are estimated to affect 10%-15% of those patients with long COVID. 22 The pathophysiology of long COVID cardiovascular manifestations is unlikely to be explained by a single process. Additionally, there is a subset of patients with persisting cardiovascular abnormalities which are sequelae of the damage from the initial COVID infection. A better understanding of the complexities surrounding the basic cellular mechanisms of long COVID will help to elucidate the pathophysiology, symptomatology, and potential therapeutic interventions.
No single etiology has been determined to be the culprit for long COVID cardiovascular symptoms and a single or multiple mechanisms may lead to similar “phenotypes” of the long COVID syndrome (ie, the “symptoms”). Inflammatory endothelial damage with microthrombi or myocyte damage and resultant fibrosis may be a final common pathway of multiple triggers leading to similar microscopic cellular damage. Importantly, the macro and microcellular relationship between different organ systems likely plays a critical role in the development of symptoms, particularly in long COVID neurocardiogenic manifestations and cardiopulmonary symptoms which are difficult, if not impossible, to isolate to a single organ system as the culprit for symptomatology. As an example, inflammatory and oxidative stress changes may lead to autonomic dysfunction with altered parasympathetic/sympathetic activity manifesting as POTS, inappropriate sinus tachycardia, or symptomatic palpitations.
Vaccination against COVID may impact the subsequent development of long COVID. There is an absence of randomized trials examining vaccination and long COVID related to the cardiovascular system, while prospective studies have demonstrated low, but measurable, adverse cardiovascular events from vaccination. 23 Significant study variation limits the ability to quantify the impact of vaccination on long COVID.
Studies focused on symptomatology commonly find that chest pain, heart failure, tachycardia, or palpitations are <10% of the symptoms in long COVID. Most studies are limited by their design but provide some insight into the frequency of cardiac symptoms in long COVID.24,25 Structural cardiac abnormalities have been found in patients that have recovered from the initial infection and may have a role in the Long COVID cardiovascular syndrome. Most studies evaluating structural cardiac abnormalities in long COVID are limited by the lack of a pre-COVID baseline. Thiele et al found that although the ejection fraction was normal in most patients six months after COVID infection, the global longitudinal strain (GLS) was abnormal in 68% (GLS is the fractional longitudinal shortening based upon speckle echocardiogram imaging). The right ventricular ejection fraction and the estimated pulmonary artery pressures were normal at six months. 26 Ramadan et al reviewed 35 studies involving over 50 000 patients at a median time of 48 days after the initial COVID infection. At six months, 30% had reduced global longitudinal strain, 10% had late gadolinium enhancement, 40% had diastolic dysfunction, and 18% had an elevated N-terminal proB-type natriuretic peptide. 22 Walter, et al evaluated patients with long COVID syndrome for cardiac abnormalities. 27 They found that left-sided heart failure in 1.14% of patients, secondary right-sided failure in 1.1%, and overall, a paucity of objective cardiac abnormalities identified principally by echocardiography. Overall, chest pain was reported in less than 1%-2% of patients. 27
Microvascular Alterations in Long COVID
Cardiovascular issues are not infrequent, and examples of medical conditions include myocardial injury, cardiac failure, arrhythmias, microvascular injury/thrombosis, and dysautonomia. The suggested molecular, pathobiological, and pathophysiological mechanisms associated with long COVID symptoms include myocardial hypertrophy, focal myocardial fibrosis, endothelial dysfunction and coagulation activation, neutrophil extracellular traps (NETs), reactive oxygen species (ROS), inflammation-coagulation (Factor XII), and direct complement activation (inflammation). 28 Diagnosis can be difficult and typically involves imaging tests: PET scans assess blood flow in the coronary arteries and can identify coronary microvascular disease. Additionally, cardiac MRI techniques offer comprehensive information about blood flow and heart function. Finally, invasive functional coronary angiography is a reliable procedure used to evaluate the coronary flow reserve (CFR) in micro-vessels 28 though limited due to its invasive nature (Figure 1).

Proposed pathomechanisms of long COVID.
Managing and recovering from a prolonged COVID can be difficult, but some measures may assist decrease symptoms associated with cardiovascular difficulties. We believe that, in addition to standard treatment strategies, a endothelial-“vasculoprotective” approach28,29 in conjunction with anti-inflammatory medications may assist treat these patients. However, there is currently no robust evidence-based data for long COVID therapy. It is impossible to predict which therapies will be beneficial until the exact cause of long COVID and its cardiovascular symptoms is determined (Figure 2).
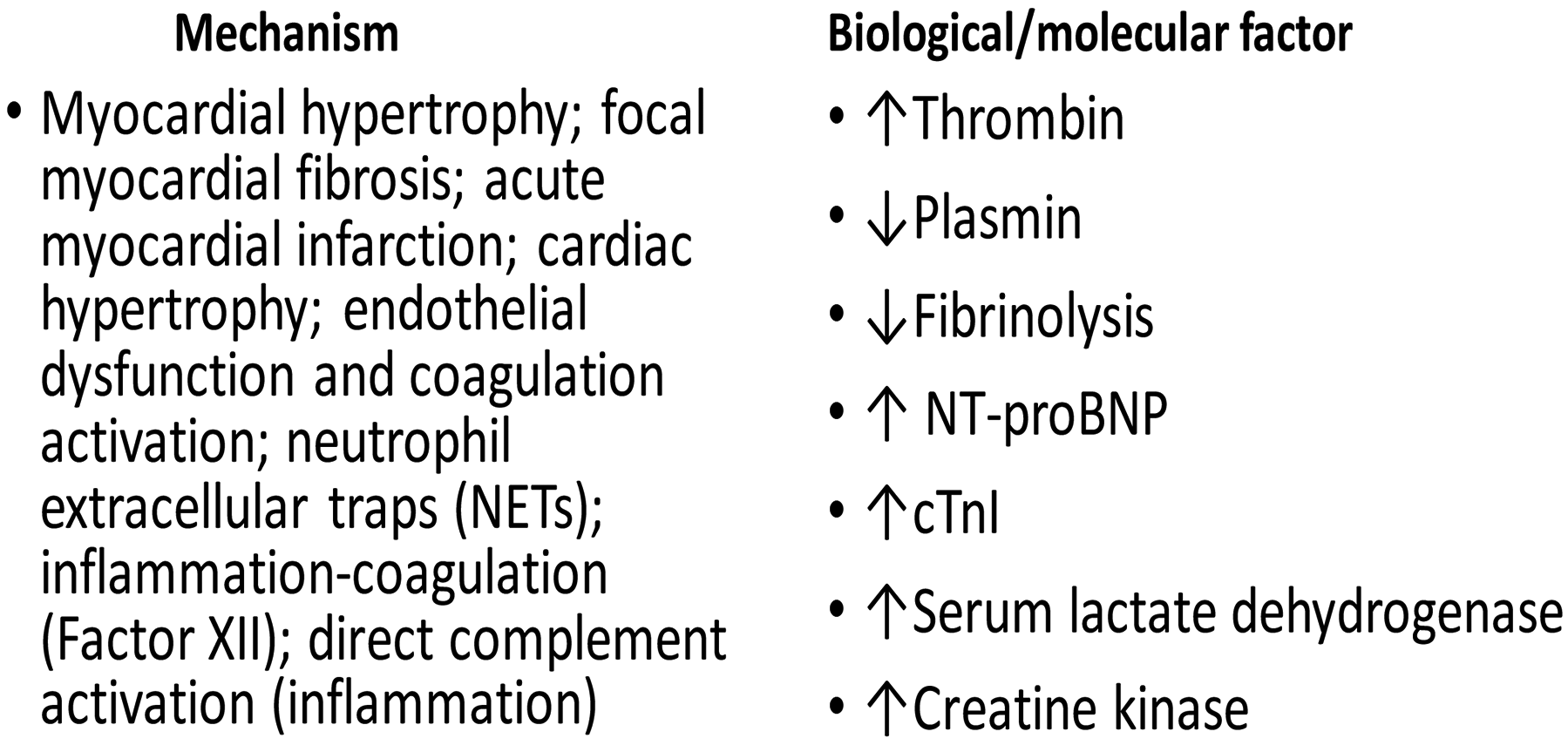
Proposed molecular, pathobiological, and pathophysiological mechanisms related to long COVID cardiovascular symptoms.
Mechanisms of Long COVID Syndrome and Modeling Approach for Identifying Disease Biomarkers
COVID- A Viral−Induced Immune Disorder
Identification of molecular determinants, such as inflammatory biomarkers, thrombotic factors, metabolomics, and downstream transcription regulators, would have significant translational potential, leading to improved outcomes. An unregulated hyper−inflammatory state can lead to cytokine storm, also called cytokine release syndrome (CRS), which was shown by our team and others to be the hallmark of acute COVID.
30
Given the complexity of pro− and anti−inflammatory cytokine/chemokine networks, methods must be developed to dissect the overall cellular and inflammatory profile to identify specific disease biomarkers. Inflammatory markers [leukocyte count, procalcitonin, C-reactive protein (CRP), interleukins (IL) 6 and 10] are elevated in intensive care unit (ICU) patients and those with severe COVID compared to persons with milder disease.31–33 The relationship between inflammatory cytokines, clinical symptoms as well as the clinical endothelial test result has been studied in a subset of 66 patient.
33
The majority of patients had increased levels of IP-10 (100%), IL-8 (95%), IFN-γ (95%), MCP-1 (80%), and TNF-α (70%). The serum concentration of IL-10 was below the threshold in 89% of subjects. Increased IL-6 and IL-27 levels were associated with fatigue and IL-8 was higher in patients with dyspnea. IL-1α level was significantly associated with increased risk of an EQI < 1, OR [95%CI] = 2.617 [1.17-5.855],
Role of the NLRP3 Inflammasome
Inflammasomes are large, micrometer−scale multiprotein cytosolic complexes activated in response to pathogen−associated molecular patterns (PAMPs) or damage−associated molecular patterns (DAMPs) and trigger proinflammatory responses as well as pyroptosis. 39 The NLRP3 inflammasome is known to be activated by highly pathogenic SARS−CoV, Middle East respiratory syndrome coronavirus (MERS−CoV), and mouse hepatitis virus.40–42 The activation of inflammasomes results in the release of caspase−1, a pathway that also includes secretion of IL−1β and IL−18 and a form of cell death called pyroptosis. 43 Patients with COVID have disrupted plasma membranes, indicating pyroptosis or other forms of necrosis. 44 Cytokines are markers of severe inflammation downstream of inflammasome activation, and can also be stimulated by nuclear factor (NF)−κB. 44 It is hypothesized that IL−1β acts as a pleiotropic proinflammatory cytokine. An exogenous IL−1 receptor antagonist (IL−1RA) completely abolishes IL−6 and tumor necrosis factor (TNF) secretion in SARS−CoV−2− infected primary monocytes. 45 Furthermore, bronchoalveolar lavage fluid (BALF) showed a significant elevation in IL−1β level in patients with moderate to severe COVID.46,47 Another strong indicator of inflammasome activation in COVID is IL−18, which is associated with severe COVID and has emerged as a highly predictive biomarker of death. 48
Role of Spike Protein and its Domains
Spike protein (S) is an essential structure in viruses that cause diseases both in humans and animals. SARS-CoV-2 spike protein is widely used in developing antiviral drugs and vaccines. The spike protein of SARS-CoV-2 consists of the S1-receptor binding (RBD) region composed of the N-terminal domain and the C-terminal domain. 49 It is the most important virulence factor and plays a key role in infection ranging from attachment, fusion, and spread of viral particles within the infected cells. It was recently reported that the spike proteins that are generated by COVID vaccines could be sustained in the body for several weeks, similar to other proteins made by the body, which has been reported in cases of myocarditis. 50 However, the immune system will quickly identify, attack, and destroy the spike proteins because it recognizes them as not part of the body. The majority of evidence for immune evasion is concentrated in a region centered on a conformational epitope consisting of residues at the N terminus (140-156 (N3 loop) and 246-260 (N5 loop). However, the role of spike protein in long COVID outcomes is yet to be established.
Role of Hepcidin, Spike Protein Peptide (COVIDIN), and Vaccines
While ferritin is shown to be highly expressed in COVID patients due to cytokine storm, IL-6 induction, contrastingly the serum iron and transferrin levels were both very low. 51 During hypoxic conditions, the signaling pathway involving hypoxia-inducible factors (HIFs) is also activated and is known to reduce hepcidin levels, thereby increasing extracellular iron levels. 52 Severe COVID is accompanied by hypoxia which strongly reduces hepcidin levels through erythropoietin (EPO) hormone, 52 and such iron dysregulation can be attributed to the products of SARS-CoV-2. In this context, the study showed part of the S1-RBD is proteolysis resistant against major human proteases and thus can present in very high concentrations once excess spike protein is degraded in dying/dead infected cells through phagocytosis. 50 In such a scenario, there is a hepcidin overdose which leads to an intracellular iron burden that overwhelms host cytoplasmic ferritin with high levels of iron-mediated free radicals leading to cell death via ferroptosis. Interestingly, our previous in-silico structural analysis of the spike protein sequence and host proteolysis processing suggests that the SARS-CoV-2 spike protein fragment mimics hepcidin.
This functional spike-derived peptide dubbed ‘Covidin’ thus may be intricately involved with host ferroportin binding and internalization leading to dysregulated host iron metabolism. In addition to increasing intracellular iron for viral replication, the presence of both host hepcidin and its molecular mimic Covidin at high levels will likely result in toxic levels of intracellular iron (ferroptosis). Interestingly, the study showed the Covidin peptide is resistant to human proteolytic machinery suggesting the persistence of this peptide post-spike degradation in infected individuals and upon autophagy/apoptosis or necrotic degradation of dead infected cells with surplus unassembled viral proteins.53,54 The Covidin peptide region was 100% conserved in all the major variants including the recent highly mutated Omicron strain. 55 The study strongly support a functional biological mimicry by Covidin of the natural host Hepcidin, an iron homeostatic hormone.53,54 There is another angle and controversial debate concerning the role of COVID vaccines on long COVID outcomes. Few studies have linked vaccine-induced immune thrombosis with thrombocytopenia (VITT), blood clots, and low blood platelet counts 56 in very rare cases, the causal mechanism is not known. However, multiple studies looking at the beneficial effect suggest that COVID vaccines have protective and therapeutic effects on long COVID. More robust comparative observational studies and trials are needed to clearly determine the effectiveness of vaccines in preventing and treating long COVID. 23 Therefore, it is strongly recommended that all people, even those with a history of COVID, receive vaccines to protect themselves against this fatal viral infection. 57
Application of Multi-Omics and Machine Learning Approach for Biomarkers Discovery
There is limited information on mechanistic modeling of biomarkers and identification of molecules affected during SARS−CoV−2 infection. Peripheral blood indicators are being used as biomarkers for early detection, diagnosis, risk stratification, and treatment of conditions such as infections, inflammatory disorders, degenerative diseases, and cancers.58,59 An extensive multi-omics analysis from plasma and PBMCs during the first week of SARS-CoV-2 infection provided an immunological profile that distinguished mild from moderate disease.60,61 A cross-omic interaction network demonstrated exaggerated transcription of pro-inflammatory cytokines accompanied by depleted cellular metabolites. 62 An ominous turn in the COVID pandemic is the evolving public health crisis of long COVID syndrome. With the lack of information on the underlying molecular determinants and pathways that mediate multi-organ sequelae, identification of circulating biomarkers can guide disease prognostication and generate a tool for classifying at−risk groups for novel and existing therapeutics. For example, targets such as the NOD-like receptor family, pyrin domain-containing 3 (NLRP3) inflammasome are currently under exploration for drug development.
Biomarkers in Long-COVID
There is limited information in this area, however, studies done so far have scantly reported the association of several circulating mediators. A comprehensive study should be carried out using in-vivo mouse models as well on large clinical cohorts to ascertain precise mechanisms underlying long-COVID. A systemic meta-analysis article using public articles reports that long COVID patients presents with elevated inflammatory biomarkers levels of IL-6, CRP, and TNF-a after SARS-CoV-2 infection for one or more months, possibly experiencing long COVID symptoms. 61 However, because of the heterogeneity of long COVID, no laboratory test could definitively distinguish long COVID from other diseases. This study recommended to include the following vascular markers: Endothelin-1 (ET-1), Hemoglobin (Hgb), Nitric oxide (NO), Platelet-derived growth factor-BB (PDGF-BB), Vascular endothelial growth factors (VEGF), Angiopoietin-2 (Ang-2), Col1A2, Col3A1, D-dimer, Erythrocyte sedimentation rate (ESR), Factor VIII:C, Matrix metalloproteinase-1 (MMP-1), MMP-9, Myeloperoxidase (MPO), Soluble intercellular adhesion molecule-1 (sICAM-1), soluble thrombomodulin (sTM), Soluble Vascular Cell Adhesion Molecule-1 (sVCAM-1), sVEGFR, Von Willebrand factor (VWF) antigen (VWF:Ag), VWF:pp, Artemin (ARTN), Alpha-smooth muscle actin (a-SMA), AXIN, Caspase-8 (CASP-8), Cystatin-C, Insulin-like growth factor binding protein-4 (IGFBP-4), miRNA21, Myeloid-related protein 8 (MRP8/14), Neutrophil gelatinase-associated lipocalin (NGAL), Osteoprotegerin (OPG), Oncostatin M (OSM), Sirtuin 2 (SIRT2), STAM-binding protein (STAMBP). 61 Conversely, it is recommended to use a holistic patient-centered approach to identify the associated long COVID biomarkers to use for disease prognosis and therapeutic targets.
Insights into Sulodexide and Long COVID
Sulodexide represents a drug in the glycosaminoglycan family composed by a mixture of 80% fast moving heparin and 20% dermatan sulfate with molecular weight of about 8-10 kDa and 27-32 kDa, respectively.
63
Notably, Sulodexide is the only orally active heparin-like drug.
64
It exhibits multiple actions interacting with antithrombin, heparin co-factor II and releasing tissue factor pathway inhibitor (TFPI), while also demonstrating profibrinolytic, antithrombotic and endothelial modulating properties without affecting hemostasis and or crossing the blood brain barrier. Sulodexide does not require monitoring and its effects on biomarkers of hemostatic modulation can be measured, reflecting its broad therapeutic index. Sulodexide exerts antithrombotic profibrinolytic and anti-inflammatory effects mediated by hemorheologic action, endothelial and other cellular modulations.
65
It also possesses anti-proliferative actions, improving the endothelial function by restoring the glycocalyx. Studies have confirmed that Sulodexide's bioavailability after oral administration and sustained actions, as indicated by thrombin generation inhibition and reduction in F1.2 biomarkers.
66
Furthermore, Sulodexide's validated properties include thrombin generation inhibition, lipolytic effect, anti-inflammatory effect, growth factor modulation, NETosis inhibition and complement modulation.
67
Sulodexide has shown efficacy in long COVID patients, with studies highlighting its efficacy in severe COVID syndrome68,69 after oral administration. Its potential benefits in managing long COVID syndrome include oral absorption, endothelial protection, anti-viral effects, cytoprotection and heparanase inhibition. As an antithrombotic drug, Sulodexide's uniqueness lies in its multiple effects including on

The endothelial glycocalyx is destructed by COVID. Endothelial glycocalyx (a) is naturally very dense (b) and has a regular net-shaped nanostructure (c-d-e). It is the main mechanical cell sensor of blood sheer stress that activates eNOS (endothelial NO synthase) (f) inducing endothelial nitric oxide (NO) release and therefore vascular media smooth muscle relaxation. Glycocalyx is dislocated by COVID and CV risk factors. We have chosen sulodexide, an oral mixture of two glycosaminoglycans with proven endothelial glycocalyx repair capabilities. By restoring the cell coat, sulodexide reinstates the eNOS activation mechanism and consequently the tissue microcirculation perfusion capabilities and its vasomotor adaptation to the local tissue metabolic needs.
Endothelial Function, Angina with Non-Obstructive Coronary Arteries (ANOCA), and the Tunisian Sulodexie Study in Long COVID
Tun-EndCOV is a multicenter study that included in its first phase, n = 798 COVID-19 survivors (>1 month) with a clinical screening for long-COVID symptoms and a fully automated non-invasive endothelial function test by a post-occlusive reactive hyperemia. A total of 397 (49.7%) patients had proven long lasting impaired endothelial that correlated independently and positively with non-respiratory long-COVID symptoms (Fatigue, chest pain, and neuro-cognitive difficulties).70,71
In the second phase of the Tun-EndCOV study we evaluated the potential benefits of sulodexide (AlfaSigma ®, Italy), a glycosaminoglycan mixture, in treating long-COVID symptoms and improving endothelial function. A multicenter quasi-experimental study included from the previous cohort, a subset of 290 patients with long-COVID symptoms and endothelial dysfunction. Patients were divided into a Sulodexide group and a control group. Sulodexide was administered for 21 days. Endothelial function and symptom severity were assessed at baseline and after treatment70,71 (Figure 4).

Endothelial function test to assess the mechanism of microcirculation and endothelial dysfunction. The test is a 15 min fully automated protocol done by a trained nurse or a fellow. In the first 5 min the Resting Blood flow from the index fingers is measured. A wrist cuff is then inflated to a suprasystolic BP during the 5 min of occlusion phase (stopping the blood flow). Then the cuff is deflated and a rapid rising blood flow is normally noted in the compensatory hyperemia phase. Many parameters are given and the Endothelium Quality Index (EQI) has been proven to be the best integrative index that correlates to the endothelium performance. The EQI is normal if greater than 2 and the lower it is the worse is endothelial performance.
Clinical characteristics were similar at inclusion in the two groups. The Sulodexide group improved significantly better in chest pain (83.7 vs 43.6%,
Endothelial Protection in Convalescent COVID-19 Patients. the Effect of Sulodexide on serum Levels of Biomarkers for Endothelial Dysfunction:
In a study of patients with cardiometabolic risk factors who were in a convalescent phase from moderate or severe COVID within 40 days of clinical presentation, there was elevated serum levels of biomarkers indicative of ongoing endothelial cell activation and dysfunction. Patients receiving sulodexide for eight weeks exhibited a reduction in TM, vWF, D-dimer, CRP, and IL-6 levels compared to placebo, suggesting a protective effect on the release of biomarkers associated with thromboinflammation and endothelial damage. There was no difference regarding sP-selectin (sP-sel), ICAM-1, VCAM-1 or fibrinogen at week 8 when compared to placebo. This suggest that early treatment with sulodexide may be beneficial in reducing endothelial injury in the convalescent phase of high-risk COVID patients, potentially leading to better clinical outcomes by mitigating post-COVID complications, including long-COVID syndrome. The complexity of post-COVID complications, including long COVID syndrome, and the insufficient knowledge about its etiology hinder the diagnosis and treatment of these individuals. Biomarkers can play a fundamental role in improving diagnosis, prognosis, and treatment stratification, potentially suggesting novel therapies. 72 In this study, we observed that patients in the convalescent phase from a moderate or severe COVID clinical presentation, who were treated with sulodexide for eight weeks within 40 days of initial symptoms, exhibited significantly lowered serum levels of TM, IL-6, CRP, D-dimer, and vWF compared to those who received a placebo.
Endothelial damage is a hallmark of cardiovascular post-COVID complications, particularly in patients with dysmetabolic disorders such as hypertension, diabetes, and obesity. These conditions share a chronic inflammatory state, characterized by elevated plasma levels of TM, vWF, ICAM-1, VCAM-1, CRP and P-selectin, which can persist during the convalescent phase and suggest ongoing endothelial cell activation and dysfunction.73–80 Chan et al identify that combined risk factors could have a relative risk (RR) of 6.98 (95% CI, 5.2-9.2) on outcomes. 81 Our studied population included patients with cardiometabolic disorders and severe inflammation from a recovering phase of COVID, reflected in the elevated baseline serum biomarkers of most patients.
As the primary endpoint in the study, TM, an endothelial cell surface receptor, serves as a biomarker for diagnosing endothelial damage and dysfunction, representing an independent prognostic biomarker.73,82,83 Fogarty et al observed that TM levels remained significantly elevated in convalescent COVID compared with controls (median 5.3 vs 4.1 ng/ml;

Thrombomodulin levels in serum of all subjects. The figure depicts thrombomodulin (TM) serum levels in all patients over an 8-week follow-up period. While there is a general decrease in TM levels from baseline to week 4, with further reduction by week 8, some patients exhibited spikes in TM levels of varying magnitudes, likely indicating episodic reactivation of the inflammatory process.
vWF, D-dimer, CRP, and IL-6 are frequently reported elevated in convalescent COVID patients and related to long COVID syndrome.61,77,78 vWF synthesized mainly in endothelial cells and generally involved in hypercoagulation, is also a well-known marker of endothelial injury.
84
Kruger et al reported the presence of microclots resistant to fibrinolysis, containing numerous inflammatory molecules, including vWF.
85
Giri et al noted that TM plays a key role in maintaining EC quiescence, with vWF expression and secretion markedly increased in TM-deficient ECs,
86
and also reported in COVID convalescent patients.
78
In the study, the combined effect on TM and vWF showed a marked correlation (
CRP, a pentameric protein synthesized primarily in the liver under the action of IL-6, and D-dimer, a fibrin degradation product present in the blood after a blood clot is degraded by fibrinolysis, widely studied in the acute phase of hospitalized patients, have seen their focus or research shifted of late to their role in long COVID.61,68,75,80,89–91 Our group, in a randomized placebo-controlled outpatient trial, showed that the patients in the sulodexide group had fewer hospitalizations, less oxygen requirement, and lower D-dimer and CRP levels. Additionally, treatment with sulodexide within three days of onset could improve the clinical outcomes of COVID patients. 68 The reduction in serum levels of D-dimer and CRP found in the acute setting was also present in the convalescent phase of this study.
Vascular Manifestations with Thrombo-Inflammatory Process
Long COVID is characterized by a structural, reservoir, and thrombo-inflammatory vascular changes. These changes indicate disease progression beyond the increased incidence of deep vein thrombosis and pulmonary embolism seen in the acute phase. 92 Clinically and subclinically, there are numerous vascular manifestations of long COVID. Notably, there is a higher frequency of PET scan evidence of medium and large vessel vasculitis in patients with long COVID compared to those who have recovered without developing long COVID. There is also evidence of perivascular infiltration in the dermal microvasculature, presenting as periodontitis-like disease, a reduction in vascular density, and alterations in the size and stiffness of blood cells. 11 While direct evidence of a viral reservoir in long COVID is limited, it is suspected to contribute to endothelial dysfunction.
The thrombo-inflammatory vascular pathobiology in long COVID is complex.92–94 In addition to the initial cytokine storm, there is evidence of elevated levels of vascular transformation blood biomarkers, 95 including P-selectin and angiopoietin-1 (ANG1). Initial endothelial damage mediates cytokines and activates platelets and neutrophils. Concomitantly, platelets release procoagulant microvesicles and prothrombotic substances such as von Willebrand factor (VWF), P-selectin, and platelet factor 4 (PF4). 23 Elevated VWF promotes platelet aggregation. The endothelium loses its glycocalyx and thrombomodulin, becoming thrombogenic. In the acute setting, endothelitis progresses to the activation of neutrophil extracellular traps (NETs), which play an integral role in long COVID pathobiology. NET activation is mediated by various factors, including pathogen-associated molecular patterns (PAMPs), damage-associated molecular patterns (DAMPs), activated platelets, complement proteins, autoantibodies, and immune complexes. These stimuli induce the activation of NADPH oxidase and the generation of reactive oxygen species through increased cytosolic calcium, leading to the activation of neutrophil elastase and myeloperoxidase, which subsequently decondense nuclear chromatin. Research is ongoing to define therapeutic targets for this process.
There is growing evidence of microclots detected in both acute COVID and long COVID. The presence of these amyloid aggregates is not pathognomonic of long COVID. Amyloid aggregates are formed by the misfolding of various proteins, with their polypeptide chains forming β-sheet structures that aggregate into long fibers. Research suggests that fibrinogen, one of the amyloid precursors, generates microclots resistant to fibrinolysis, which can obstruct microvessels.22,94 Currently, there is no definitive indication to anticoagulated patients with this finding unless a clinical event occurs. 96 Ongoing research aims to determine whether anticoagulant management would be beneficial in such cases.
Hematological Manifestations of Long COVID
Long COVID is emerging as a new dimension of the public health crisis triggered by the pandemic. Social inequalities and geographic disparities profoundly shaped the impact of COVID. Factors such as access to and quality of the public healthcare system, socioeconomic status of the citizens, and living conditions play crucial roles in determining individuals’ vulnerability to the SARS-CoV-2 infection. Predisposing risk factors for severe COVID are male gender, underlying cardiovascular disease or cardiovascular risk factors (including diabetes, high blood pressure, obesity, and advanced age). Venous thromboembolism (VTE) is also a risk factor for deterioration of COVID and is independently related to mortality. 97
A large epidemiological study conducted in Africa countries, enrolled 9.5 million confirmed COVID cases and showed that the prevalence of long COVID in African populations raises up to 41% (CI:26%-56%). Major clinical predictors for high risk of long COVID are cardiovascular risk factors (hypertension, obesity, hyperlipidemia, diabetes mellitus), cardiovascular disease (ischemic heart disease, arterial or venous thrombosis, HIV infection, chronic obstructive pulmonary disease/asthma, active cancer, tuberculosis, renal disease and psychiatric diseases. 98 Delayed arterial thrombosis or VTE post COVID is an emerging health problem and constitutes an additional problem associated with long COVID.99–101 European studies show that up to 90 days post-diagnosis of symptomatic COVID, the incidence of VTE (deep vein thrombosis or pulmonary embolism) and arterial thrombosis is 2-8/1000 and 1-8/1000 COVID patients respectively.102–104 Early since the recognition of the long COVID the European Society of Cardiology suggested that the evaluation of endothelial function, in addition to myocardial injury and respiratory function markers in COVID survivors may represent possible means for early detection of vascular sequelae post-COVID. 105 Data from prospective longitudinal observational studies show that persistent hypercoagulability and endothelial cell activation after the recovery of the acute phase of the disease are associated with long COVID symptoms.
The prospective observational study ROADMAP-post COVID, enrolled COVID survivors (n = 208) and 30 healthy individuals. Survivors showed significantly higher levels of D-dimer, fibrin monomers (FM), Tissue factor pathway inhibitor (TFPI), soluble sTM and heparanase as compared to the control group. Survivors had significantly shorter the procoagulant phospholipid-dependent clotting time (PPL-ct). Elevated sTM and FM levels were observed in about 9% of survivors. Elevated levels of D-dimer, heparinase, and TFPI was found in about 25% of the survivors. Procoag-PPL ct was shorter than the lower normal limit in 8%of the survivors and was associated with female gender. Elevated FM was also associated with female gender whereas the increase of heparanase was independently associated with male gender. 106 The persistent endothelial activation in patients with long COVID is further supported by the decrease levels of the ADAMTS-13. The implication of neutrophil activation in patients with long COVID is indicated by the CD10 low-density hyperreactive phenotype and increased PLA formation with consequent pulmonary dysfunction. 94
A prospective study on 50 COVID survivors, showed that two months following SARS-CoV-2 infection, ongoing endotheliopathy is a common finding, independent of the severity of the acute phase. Thrombin generation was increased and the levels of factors VIII, and von Willebrand and soluble thrombomodulin remain persistently elevated in a about 20% of patients following apparent resolution of acute COVID. Persistent endotheliopathy appeared to occur independently of ongoing acute phase response or NETosis and is associated with enhanced thrombin generation potential. 78 Nevertheless, there is no evidence establishing a link between critical COVID and the presence of antiphospholipid antibodies. 107 The large prospective epidemiological study conducted in African populations showed that patients with long COVID had higher levels of microclot and platelet-poor plasma viscosity, serum A-Amyloid, α-2 antiplasmin, platelet factor 4 von Willebrand Factor (VWF), endothelial-leukocyte adhesion molecule 1 (E-selectin), platelet endothelial cell adhesion molecule-1 (PECAM-1). 98 A longitudinal, prospective, single-center observational study on 60 patients with severe COVID admitted to the ICU and 25 patients with mild COVID treated at home, showed that after 4 weeks from the acute phase, extracellular vesicles -expressing platelet antigens (CD41A), endothelial antigens (CD31) and TF (CD142) were significantly increased in about 10% and 20% of patients with mild COVID or severe COVID respectively. 108
Current data suggest that cardiovascular events following COVID may be linked to persistent immune dysregulation, endothelial cell activation, and hypercoagulability. Patients experiencing prolonged symptoms often exhibit elevated levels of proinflammatory molecules, including tumor necrosis factor-α, interferon-γ, and interleukins 2 and 6. This immune dysregulation can trigger the activation of the coagulation pathway, leading to the formation of extensive microclots during both acute COVID and long COVID. These microclots are likely a significant mechanism underlying persistent symptoms and cardiovascular complications. 109
Discussion
Sulodexide has been useful in COVID management with validation the oral absorption concept 64 in Tunisian studies. Owing to the sustained anti-inflammatory properties and cytoprotection, sulodexide is ideal for long COVID syndrome. Sulodexide may be monitored by using sensitive biomarkers such as thrombomodulin, IL-6 and TNFa and CRP. Additional studies are needed for complement dosing and frequency of administration. Sulodexide is the only drug in its class for oral administration, this will be useful for long COVID syndrome. There are some remaining questions regarding the use of antithrombotics such as sulodexide for the efficacy in long COVID syndrome. Such as – what is the relevance of vaccine usage in terms of the adverse effects such as long COVID syndrome. Additionally, what is the relative benefit for the effectiveness of sulodexide in long COVID syndrome. The pathophysiology of long COVID syndrome is complex and multifactorial. This short synopsis underlining the effect of sulodexide underscores the importance of this drug in large clinical trials as it validates the effectiveness in long COVID syndrome. Furthermore, drugs such as sulodexide which are broadly known as GAGs (expand) based drugs can be protective in long term management of COVID. Sulodexide represents the first orally drug reduces the thrombo inflammatory biomarkers in long COVID.
Given the substantial public health impact of long COVID, there is an urgent need for a comprehensive strategy that focuses on (a) Profiling COVID patients at risk of developing long COVID using validated clinic-biological risk scores, (b) Prevention of long COVID-19 through targeted community interventions, (c) Treating long COVID, with particular emphasis on investigating the role of antithrombotic agents in selected groups of patients, such as those with cardiovascular disease, cardiovascular risk factors, cancer, or HIV infection. Additionally, it is crucial to intensify research on therapeutic agents that modulate endothelial cell activation to address these challenges effectively.
To identify the biomarkers associated with long COVID syndrome, studies should be designed to evaluate both retrospective and prospective clinical cohorts who have recovered from COVID. The biological samples need to be assessed for integrating thrombotic factors, primary metabolites, markers of inflammation, and host genetics for biomarkers identification and target specific drug syntheses and discovery pipeline.110–112 Far more research is needed to understand the mechanisms leading to common cardiac symptoms as well. Despite some patients with long COVID presenting with chest pain or palpitations, very little is known about the mechanisms contributing to these symptoms.
Conclusion and Future Directions
Long COVID is recognized as a significant and complex sequela in up to a third of patients who have recovered from acute COVID infection. The underlying mechanisms resulting in long COVID are not understood, but there are several potential mechanisms which include dysregulation of hemostasis and clotting, the reactivation of EBV, the interruption of the glycocalyx, or activation of different genes. Treatment is targeted at the underlying endothelial abnormalities and sulodexide is a promising agent from both a pathophysiological basis, but also on clinical improvement in symptoms. Long COVID requires intense investigation to better elucidate the underlying mechanisms. Although no specific mechanism seems to be responsible for clinical manifestations, our group recommends the following: (1)Broad-based research which aims to evaluate underlying genetic and proteomic alterations is necessary to advance the field. (2)Using machine learning and artificial intelligence approach to interrogate large proteomic and genetic data (omics), a better understanding and refinement of causative factors is possible.
Footnotes
Acknowledgements
We thank the speakers as noted in the authorship as well as the VA organizing team.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
