Abstract
Introduction
Persistent elevation of biomarkers associated with endothelial dysfunction in convalescent COVID-19 patients has been linked to an increased risk of long-term cardiovascular complications, including long COVID syndrome. Sulodexide, known for its vascular endothelial affinity, has demonstrated pleiotropic protective properties. This study aims to evaluate the impact of sulodexide on serum levels of endothelial dysfunction biomarkers in patients during the convalescent phase of COVID-19.
Methods
We conducted a double-blind, single-center, randomized, placebo-controlled trial in Mexico, comparing sulodexide (250 LRU orally, twice daily) with placebo over 8 weeks in adult patients during early COVID-19 convalescence. Differences in serum biomarkers between the groups were analyzed using repeated measures and post hoc tests, with Thrombomodulin (TM) as the primary endpoint.
Results
Among 206 analyzed patients (103 in each group), at week 8, the sulodexide group exhibited significantly lower mean levels of Thrombomodulin (TM) (25.2 ± 7.9 ng/mL vs 29.9 ± 14.7 ng/mL, P = .03), von Willebrand Factor (vWF) (232 ± 131 U/dL vs 266 ± 122 U/dL, P = .02) and Interleukin-6 (IL-6) (12.5 ± 13.2 pg/mL vs 16.2 ± 16.5 pg/mL, P = .03) compared to the placebo group. D-dimer and C reactive protein (CRP) in the sulodexide group were also lowered. No significant differences were observed for P-selectin, fibrinogen, VCAM-1, or ICAM-1 levels.
Conclusions
Patients in the convalescent phase of COVID-19 who received sulodexide for eight weeks showed a reduction in TM, vWF, D-dimer, CRP, and IL-6 serum levels compared to placebo. These findings suggest a potential protective effect of sulodexide against thromboinflammation and endothelial damage.
Keywords
Introduction
The coronavirus disease of 2019 (COVID-19) typically begins by affecting the respiratory tract and can range in clinical presentation from asymptomatic to mild or severe. 1 Since 2020, the virus has affected over 750 million people globally. 2 Benefiting from widespread vaccination efforts and the emergence of less lethal SARS-CoV-2 variants, the burden of severe COVID-19 has declined. Unfortunately, a new public health issue has emerged, and attention has shifted toward survivors who exhibit long-lasting symptoms following apparent clinical recovery. 3 A significant proportion of patients develop debilitating symptoms after acute infection, a postinfectious state termed long COVID-19 syndrome. 4
Increasing evidence suggests that COVID-19 can impact the vascular system directly, through viral infection, or indirectly, via a cytokine storm, leading to endothelial dysfunction, including endotheliitis and endotheliopathy. Consequently, COVID-19 is now recognized as both a microvascular and endothelial disease.5–10 Endothelial dysfunction in convalescent individuals has been associated with an increased risk of long-term cardiovascular complications, including thrombotic events, post-intensive care syndrome, and permanent organ damage.11,12 It has been reported that patients with chronic degenerative diseases, such as metabolic syndrome, obesity, and/or diabetes mellitus (DM), when affected with COVID-19, tend to be associated with a poorer prognosis. 13 These diseases share a chronic inflammatory state that, coupled with the severe inflammation of COVID-19, may promote an immunothrombotic status. 14
Given the wide-ranging impacts of long COVID-19 syndrome, significant research efforts have aimed to elucidate its etiology and pathophysiology to enhance patient outcomes.15,16 Current research has highlighted the potential role of various biomarkers that could monitor disease activity and therapeutic responses, differentiate clinical profiles to tailor interventions and confirm diagnoses in the context of other clinical conditions.17,18 However, consensus on which biomarkers are essential for tracking the transient nature of long COVID-19 syndrome is still lacking. 17
Studies involving patients recovering from COVID-19 have persistently identified increased levels of biomarkers for endothelial dysfunction like Thrombomodulin (TM) and von Willebrand factor (vWF). 4 Persistent elevation of these biomarkers in the post-acute phase may signify ongoing endothelial activation and a prothrombotic state. Moreover, the association of inflammatory biomarkers in COVID-19 infection, and inflammation-induced degradation of the glycocalyx layer in endothelial cells has been demonstrated,15,19 warranting close monitoring and potential intervention to mitigate cardiovascular complications.
Sulodexide is a mixture of two glycosaminoglycans (GAGs), one a fast-moving heparin fraction (80%), the other dermatan sulfate (20%). As a precursor for the synthesis of GAGs, sulodexide can help restore a shredded endothelial glycocalyx and prevent further degradation. 19 This improvement restores endothelial barrier function and allows the endothelium to better modulate the generation of key inflammatory molecules, while at the same time downregulating its response to them. 19 Recent studies have explored the potential benefits of sulodexide in mitigating endothelial dysfunction in COVID-19 patients, highlighting its role in improving vascular health and reducing disease severity.20,21 Sulodexide's antithrombotic, profibrinolytic, and anti-inflammatory effects may be significant against the thromboinflamatory state seen in COVID-19 patients. Finally, sulodexide is associated with a lower bleeding risk than oral anticoagulants. 19 therefore, studying sulodexide's effect on COVID-19 patients is reasonable.
Developing targeted therapies to preserve a healthy glycocalyx and endothelial function to improve long-term prognosis in convalescent COVID-19 patients has been the focus of ongoing research. 22 Despite these efforts, there remains a lack of reliable and effective treatments. 23 We aimed to assess whether sulodexide's pleiotropic effects influence serum levels of biomarkers for endothelial dysfunction in patients who are in a convalescent phase from a moderate or more severe clinical presentation of COVID-19.
Methods
Study Design
We carried out a prospective, single-center, double blinded, randomized placebo-controlled trial with a parallel-group design to assess the effect of sulodexide on biomarker levels of convalescent COVID-19 patients, as defined in Table 1. We conducted this trial in compliance with the Declaration of Helsinki. The trial was board-reviewed by the Comité de Bioseguridad de Investigación Biomédica para el Desarrollo de Fármacos S.A. de C.V. file CB 000001, and by RM Pharma Specialists S.A. de C.V. Research and Ethic Committee file CEI 000002. It was registered at clinicaltrials.gov (NCT05371925) and in Mexico at the Comisión Federal para la Protección contra Riesgos Sanitarios (COFEPRIS) with registry number 223300410A0037/2022. Patients approved the use of their laboratory test results and blood samples for research purposes after ensuring their confidentiality.
Baseline Demographic and Laboratory Data of the Studied Population.

vWF, von Willebrand Factor; ICAM, intercellular adhesion molecule; VCAM, vascular cell adhesion molecule; IL, interleukin; CRP, C reactive protein; n, number of patients; CI, confidence interval; SD, standard deviation; N/L, neutrophil to lymphocyte ratio; P/L, platelet to lymphocyte ratio.
Patients
Eligible patients were recruited through social media outreach and by engaging primary care physicians from both public and private clinics for early referrals. We also collaborated with social healthcare workers at hospitals treating COVID-19 patients. The majority of participants screened at our site were referred from these hospitals, which were generally well-informed about our inclusion criteria. Consequently, the rejection rate among screened patients at our site was low.
Patient inclusion and exclusion criteria are summarized in Supp. Table 1. Key inclusion criteria were: i. confirmed SARS-CoV-2 infection as determined by PCR in naso/oropharyngeal swabs or any other relevant specimen obtained during the disease, and rapid antigenic tests were also acceptable; ii. status on a convalescent COVID-19 phase, defined as at least 10 days after the onset of symptoms, no fever for at least 24 h without the use of antipyretics, and improvement of respiratory symptoms according to the quick COVID-19 Severity Index; and iii. status as suffering from chronic comorbidities deemed of high risk (>50%) for endothelial dysfunction. The percentage risk was calculated using the COVID-19 Health Complication (C19HC) calculator (IMSS, Gobierno de Mexico) that considers the importance of various chronic comorbidities. 24 Important exclusion criteria included: i. more than 40 days since the first COVID-19 clinical symptom; and ii. current anticoagulation treatment. Once the trial was underway, the initiation of anticoagulant medication at a prophylactic dose was not considered a criterion for exclusion. However, due to the possible risk of bleeding complications, these patients underwent stricter follow-ups.
Study subjects who qualified were immediately randomized after the informed consent was signed and scheduled for a blood sample test at the earliest convenience. Oral medication started after the blood sample had been taken.
If allocated to the study group, the patient received a sulodexide (Vessel Due F, Alfasigma SpA) oral dose of 250 LRU twice daily for 8 weeks. Sulodexide 250 LRU twice daily has been observed to produce effective plasma concentrations in vitro and is regularly prescribed in clinical practice. 19 If assigned to the control group, the patient received an oral dose of mask placebo (similar to sulodexide) twice daily for 8 weeks.
Study Protocol
Group allocation was performed at the research site with a four-block randomization sequence generated at a 1:1 ratio for IMP/placebo with no strata. The medical team in charge of the patients and treatment regimens was blinded to group allocation.
Apart from being used to provide study medication and perform follow-ups on study endpoints, the research site was not involved in the disease's primary treatment. Patients were encouraged to continue with the standard care recommended by their healthcare providers. Patients were allowed to receive concomitant therapies and full supportive care during the study, as appropriate. Relevant concomitant treatments received during the study, including those for COVID-19, were recorded in the CRF.
For follow-up at weeks four and eight, blood tests were scheduled, and adherence and adverse events were evaluated. Remote communication with participants or household members was available as necessary during the eight-week participation period.
If the patient's symptoms worsened, an emergency department examination was recommended with their healthcare provider. If the patient required hospital care, the oral dose of sulodexide or placebo was suspended but the gathering of data on the patient's progression continued, including data in the final analysis. The research team was not involved in any of the treatment decisions made during hospital care but, in such cases, follow-up was extended beyond eight weeks until an outcome could be defined or until the trial ended. If possible, sulodexide was resumed after discharge from the hospital.
Data Sources
Data was captured using Castor EDC software (Amsterdam, The Netherlands) for validation and monitoring and a hard copy was saved on file at the research site.
Study Endpoints
The primary endpoint was evaluated by comparing TM serum levels in the study group with those of the control group after four and eight weeks of treatment. Secondary endpoints (vWF, sP-sel, IL-6, ICAM-1, VCAM-1, D-dimer, CRP, Ti, and fibrinogen) were also assessed at weeks four and eight.
Baseline blood samples (∼10 mL) in a fasting state from all patients were obtained upon admission to the trial via clean venipuncture after signing the informed consent form. Participants were scheduled in the early morning and blood samples were collected in vacutainer tubes containing 3.2% sodium citrate (Becton Dickinson, Franklin Lakes, NJ, USA) and processed within 2 h of collection. Serum and plasma were separated by centrifugation at 3500 rpm for 10 min and then preserved at −70 °C until use for biomarker assessment. Complete blood cell count, glucose, and other routine chemical tests were also performed. The same process was performed for the follow-up visit assessment.
Outcomes were assessed as follows, serum levels of D-dimer and troponin I (TnI) (using a chemiluminescence assay), C-reactive protein (CRP) (using a turbidimetric assay) with an automatic 3H Hematology Analyzer (KONTROLab Instruments Inc., Hamburg, Germany) and a Cobas b 121 multiparametric analyzer (Roche Diagnostics, Rotkreuz, Switzerland). The Enzyme-Linked Immunosorbent Assay (ELISA) method, using commercially available assay kits from the same distributor (MyBioSource, Inc., San Diego, USA) and production lot, was employed to measure serum levels of TM (lot no. MBS2701918), P-selectin (sP-sel) (lot no. MBS355417), vWF (lot no. MBS2880826), interleukin 6 (IL-6) (lot no. MBS2701078), intercellular adhesion molecule (ICAM-1) (lot no. MBS265404), and vascular cell adhesion molecule (VCAM-1) (lot no. MBS2701976). These measurements were performed according to the manufacturer's instructions using an ELISA plate reader (BMG LABTECH, Offenburg, Germany). Each measurement was duplicated, and biomarkers serum levels were determined by extrapolation from a standard curve generated for each set assayed. The intra-assay precision (precision within an assay) was CV <8%. ELISA assays were conducted periodically as sufficient samples were collected to complete a 96-well plate.
All laboratory outcome tests were conducted in the same facility, adhering to strict safety protocols. The reference ranges used were specific to the local laboratory at the trial site. Adverse events were assessed at weeks four and eight, along with self-reported treatment adherence.
Statistical Analysis
Statistical analyses were conducted using SAS version 9.4 (Statistical Analysis Software, Cary, NC, USA). Continuous variables were expressed as mean ± standard deviation and compared using the Student's t-test, while categorical variables were analyzed using the Chi-square test or Fisher's exact test. A P-value of less than .05 was considered statistically significant.
Primary and secondary outcome measures between treatment groups were compared using Repeated Measures ANOVA when normality was assumed, or the Friedman test with post hoc pairwise comparisons via the Wilcoxon Signed-Rank test when normality was not met. Bonferroni correction was applied to control for type I error in multiple comparisons.
Proportions between the two intervention arms were compared using Fisher's exact test and presented as relative risk (RR) or reduction of RR (1− RR *100%) when RR was less than 1. Adjustments for covariates and potential confounders were made using Poisson regression with a log link and robust covariance estimates (Huber method). Continuous variables were compared between groups using the Wilcoxon rank-sum test, with the effect presented as the mean difference.
Trial populations were analyzed by intention-to-treat (ITT) for all endpoints and per-protocol for the primary endpoint. If less than 5% of data were missing for any primary or secondary outcome, a complete case analysis was performed without imputing missing values, as no data imputation was necessary.
The sample size was calculated using a repeated measures ANOVA F-test for the difference between two groups across three time points. With an effect size of 0.2, a power of 85%, an alpha error of 0.05, and a 1:1 group distribution, the final sample size was 200 patients (100 per group), including a 10% allowance for attrition.
Results
Between June 2023 and February 2024, a total of 218 patients were assessed for eligibility, of which 206 were included in the final analysis (Figure 1). The patients were randomized into either a study group or a control group, with 103 patients in each group. Among the participants, 130 (63%) were female. The mean age was 61 ± 13.3 years, and the mean body mass index (BMI) was 28.6 ± 4.4 kg/m². There were no significant demographic differences between the groups (Table 1).
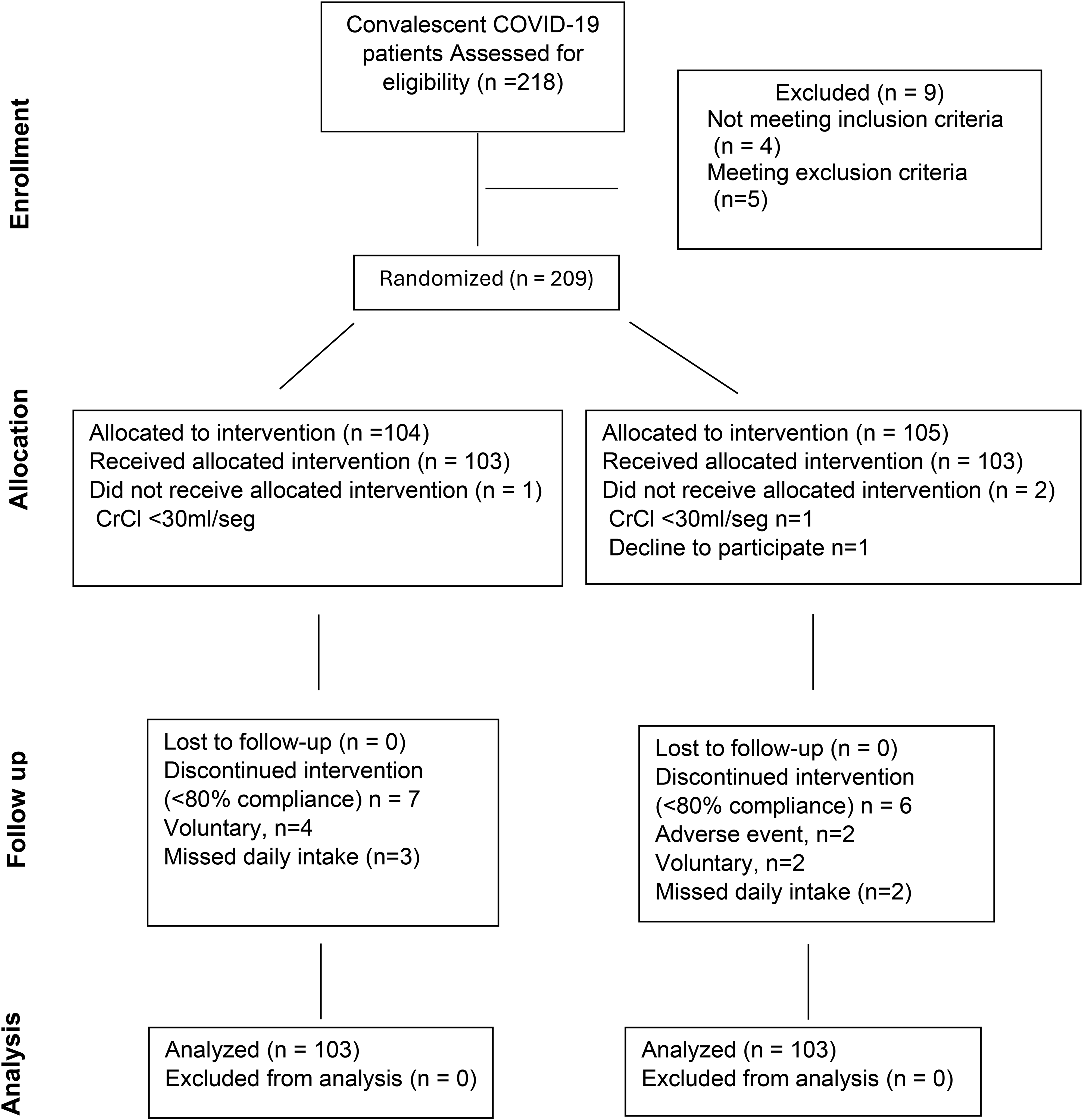
Consort table.
Hypertension was the most frequent chronic comorbidity, reported in 122 patients (59.2%). The study population's average time from initial symptoms to study recruitment was 22 ± 6.2 days. Of the patients, 192 (93%) received at least one COVID-19 vaccine (although the average time since the last vaccine dose before recruitment was 14.3 ± 7 months), and 68 (33%) received a mixed brand of vaccine for the second or booster dose. Aside from the current COVID-19 infection, no previous symptomatic COVID-19 presentation was reported by 73 (35.4%) patients. Of the recruited patients, 101 (49%) were in a convalescent phase of a severe COVID-19 clinical presentation (Table 2). Other than patients with DM being in higher proportion in the sulodexide group, no significant differences were found between the groups.
COVID-19 Characteristics. Vaccination status, Number of Applied Doses, Clinical Severity, Previous Cases, and Time from Initial Symptoms in the Studied Population.

COVID-19 clinical presentation besides the current trial inclusion.
SD, standard deviation; n, number of patients; %, percentage.
General laboratory and biomarker mean baseline values are included in Table 1. Of interest, the neutrophil/lymphocyte ratio, CRP, D-dimer, and fibrinogen were all above the normal range, as were the mean levels of TM, P-selectin, vWF, IL-6, ICAM-1, VCAM-1. Troponin I was elevated only in two patients from baseline, one in each group. While most patients showed a general decline in biomarker serum levels over time, several experienced a spike at either week four or week eight.
Primary Endpoint
The differences between groups of TM serum levels at three-time points and post hoc analyses are displayed in Figure 2. Pairwise comparison of TM serum levels did not show a significant difference in the sulodexide group compared to the placebo group at week four (37.4 ± 10.6 vs 39.2 ± 14.3 ng/mL, P = .154) in the ITT or per-protocol population analysis. At week eight, TM serum levels in the sulodexide group showed significant improvement compared to the placebo group in the ITT (25.2 ± 7.9 vs 29.9 ± 14.7 ng/mL, P = .032) and per-protocol population analyses (25.1 ± 10 vs 30.3 ± 14.9 ng/mL, P = .036). Although more patients in the sulodexide group reached a normal TM value at week eight when compared to the placebo group (15 [14.3%] vs 8 [7.7%], OR 0.49 [CI 0.19-1.22], P = .12), it did not reach statistically significant.

Thrombomodulin and von Willebrand Factor change between the sulodexide and placebo groups from baseline throughout week 4 and week 8. The figure shows the change in serum levels of TM (A) and vWF (B) over time. Comparisons were made using the Friedman test for multiple measurements and post hoc pairwise comparisons using the Wilcoxon Signed-Rank test for the measurements at different time points, adjusted for multiple comparisons with Bonferroni correction. Significant differences were observed over time and between groups from baseline to week 8. vWF, von Willebrand Factor; TM, thrombomodulin; npairs, number of patients in each group; df, degrees of freedom. Mean value, ----- median value.
Correlations were calculated between TM and secondary endpoints, and only positive and statistically significant correlations were observed between TM and vWF (r = 0.331, P ≤ .001).
Secondary Endpoints
At week four, when comparing the sulodexide group to the placebo group, mean levels of CRP (12.3 ± 15 vs 16.8 ± 19.2 mg/L, P = .032) and D-dimer (621.8 ± 578.7 vs 891.2 ± 1028.3 ng FEU/mL, P = .01) showed significant improvement. At week eight, the pairwise comparison showed improvement in serum levels of vWF (232.9 ± 131.2 vs 266 ± 122.3 U/dL, P = .028), IL-6 (12.5 ± 13.2 vs 16.2 ± 16.5 pg/mL, P = .03), D-dimer (511.3 ± 407.9 vs 781.4 ± 998.3 ng FEU/mL, P = .006), and CRP (8.2 ± 8.8 vs 15.8 ± 19.7 mg/L, P < .001) in the sulodexide group when compared to the placebo group (Figure 3). No difference between the groups in mean serum levels of P-selectin, fibrinogen, VCAM-1, or ICAM-1 were found. Mean serum biomarker levels are detailed in Table 3.
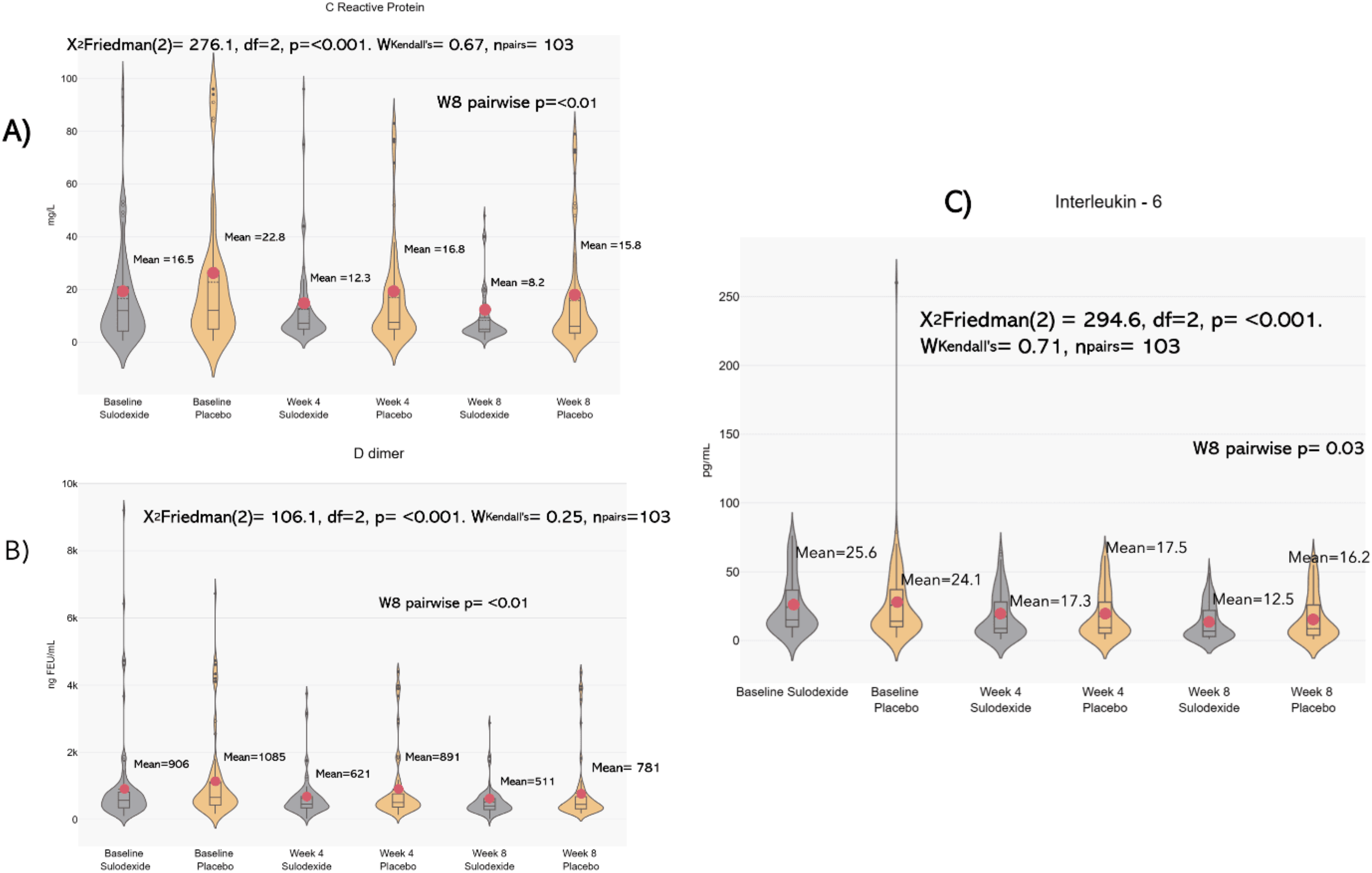
Biomarker change between the sulodexide and placebo groups from baseline throughout week 4 and week 8. The figure shows the change in serum levels of different biomarkers over time. Comparisons were made using the Friedman test for multiple measurements and post hoc pairwise comparisons using the Wilcoxon Signed-Rank test for the measurements at different time points, adjusted for multiple comparisons with Bonferroni correction. A (CRP), B (D-dimer) and C (IL-6) show significant differences through time and between groups from baseline to week 8. vWF, von Willebrand Factor; IL, interleukin; CRP, C reactive protein; npairs, number of patients in each group; df, degrees of freedom. Mean value, ----- median value.
Laboratory Outcomes and Between Groups Comparison at Week 4 and Week 8.

TM, thrombomodulin; vWF, von Willebrand Factor; ICAM, intercellular adhesion molecule; VCAM, vascular cell adhesion molecule; IL, interleukin; CRP, C reactive protein; n, number of patients; CI, confidence interval.
P values were determined by The Wilcoxon signed-rank post hoc tests with Bonferroni correction for multiple comparisons.
Adverse Events and Concomitant Medication
Sulodexide was well tolerated in the study population considering that only 8 (8.2%) patients reported an adverse event, all of which were non-serious. Furthermore, the number of patients reporting adverse events in the placebo group was equal in number and proportion to those in the treatment group, confirming sulodexide's safety (Supp. Table 2).
Concomitant medications were evaluated as potential confounding factors in this study. The distribution of concomitant medications between the treatment groups was not balanced, and statistical adjustments were made to account for any potential differences. As a result, no significant confounding from concomitant medications was identified.
Discussion
The complexity of post-COVID-19 complications, including long COVID-19 syndrome, and insufficient knowledge about their etiology, hinder the diagnosis and treatment of these individuals. Biomarkers play a fundamental role in improving diagnosis, prognosis, and treatment stratification, potentially suggesting novel therapies. 15 In this study, we observed that patients in the convalescent phase from a moderate or severe COVID-19 clinical presentation, who were treated with sulodexide for eight weeks within 40 days of initial symptoms, exhibited significantly lowered serum levels of TM, IL-6, CRP, D-dimer, and vWF compared to those who received a placebo.
Amounting evidence suggests that endothelial damage plays an important role in cardiovascular post-COVID-19 complications, particularly in patients with dysmetabolic disorders such as hypertension, diabetes, and obesity. These conditions share a chronic inflammatory state characterized by elevated plasma levels of TM, vWF, ICAM-1, VCAM-1, CRP, and P-selectin, which can persist during the convalescent phase and suggest ongoing endothelial cell activation and dysfunction.14,25–31 Chan et al report that combined risk factors could have a relative risk (RR) of 6.98 (95% CI, 5.2−9.2) on outcomes. 32 Our studied population included patients with cardiometabolic disorders and severe inflammation from a recovering phase of COVID-19, reflected in the elevated baseline serum biomarkers of most patients.
As the primary endpoint in our study, TM, an endothelial cell (EC) surface receptor, serves as a biomarker for diagnosing endothelial damage and dysfunction, representing an independent prognostic biomarker.25,33,34 Fogarty et al observed that TM levels remained significantly elevated in convalescent COVID-19 patients compared with controls (median 5.3 vs 4.1 ng/mL; P = .02, 95% CI 0.01-2.7). 30 In our study, TM serum levels were significantly lower in patients treated with sulodexide compared to placebo after eight weeks. However, the mean TM serum value remained above the normal range at eight weeks in both groups, possibly influenced by participants’ pre-existing chronic comorbidities.
vWF, D-dimer, CRP, and IL-6 are frequently reported to be elevated in convalescent COVID-19 patients and are related to long COVID-19 syndrome.29,30,35 vWF is also a well-known marker of endothelial injury. 36 Kruger et al reported the presence of microclots resistant to fibrinolysis, containing numerous inflammatory molecules, including vWF. 37 Giri et al noted that TM plays a key role in maintaining EC quiescence, with vWF expression and secretion markedly increased in TM-deficient ECs, 38 and also reported in COVID-19 convalescent patients. 30 In our study, the combined effect on TM and vWF showed a marked correlation (r = 0.3824) which could result from sulodexide's endothelial cell protection or restoration properties.
IL-6 is an important cytokine relevant to the outcome of COVID-19, including disease duration and severity. 22 Rajewska-Tabor et al reported that in vitro supplementation of sulodexide to serum collected from patients four months post-COVID-19 reduced the synthesis of IL-6 and vWF from coronary artery endothelial cells. 39 This correlated with our findings, as vWF and IL-6 serum levels were also significantly lower in the sulodexide group compared to placebo after eight weeks of treatment.
C-reactive protein (CRP), IL-6, and D-dimer were widely studied in the acute phase of COVID-19 hospitalized patients, with a recent focus on their role in long COVID-19.27,31,35,40–43 In a previous randomized placebo-controlled outpatient trial, our group showed that patients receiving 1000 LRU of sulodexide daily had fewer hospitalizations, less oxygen requirement, and lower D-dimer and CRP levels. Additionally, initiating sulodexide within three days of onset could improve the clinical outcomes of COVID-19 patients. 20 In the current study, a reduction in serum levels of D-dimer and CRP observed during the acute phase was also noted during the convalescent phase, despite the lower daily dose of 500 LRU. A higher dose may enhance sulodexide's antithrombotic effects, which could be beneficial in certain clinical scenarios.
sP-sel, ICAM-1, and VCAM-1 are other biomarkers related to post-COVID-19 complications.26,44–46 Even though sulodexide has reported improved serum levels in non-COVID-19-related diseases, 47 no significant difference was observed when compared to placebo at week four or week eight in this trial, in which the role of patients’ chronic comorbidities is still to be determined. 3
Sulodexide has been studied in the clinical setting with long COVID-19 patients. Charfeddine et al, in a multicenter, prospective trial of 290 patients with long-COVID-19 symptoms, found that sulodexide significantly improved clinically evaluated endothelial function (median delta-EQI 0.66 [0.6] vs 0.18 [0.3], p < 10−3) correlating with improved symptoms of chest pain (83.7 vs 43.6%, p < 10−3) and palpitations (85.2 vs 52.9%, p = .009). 21 Our findings may help explain these results through the observed reduction in endothelial and inflammatory serum biomarkers.
The present trial was subject to certain limitations. We did not conduct a dose-dependent analysis with 500 LRU bid, which has been reported as useful in other clinical settings while maintaining a favorable safety profile. Our study population did not include low-risk patients, even though biomarkers are also elevated in non-severe COVID-19 patients who can develop long COVID-19 syndrome. Therefore, the generalizability of our findings to those populations remains unknown. Our biomarker panel was limited. The trial does not indicate whether sulodexide would prevent long COVID if started during an emergency department visit or hospitalization for COVID-19, or whether it would be effective as a treatment in individuals who already have long COVID. The study group had a higher proportion of participants with DM. Furthermore, certain biomarkers may have been affected by participants’ pre-existing medical conditions or concurrent medication use.
Diagnosing and managing long COVID-19 is complex, and a ‘one size fits all’ approach is unlikely to be appropriate or effective. However, utilizing a core panel of biomarkers may aid in the early detection of post-COVID-19 complications.3,35,48 A therapeutic option capable of modulating a broad range of these biomarkers could be beneficial until more targeted treatments are identified. The findings from this study contribute to the growing body of research on sulodexide across different stages of COVID-19. With additional advantages such as low cost, favorable intestinal absorption, minimal adverse effects, and a reduced risk of major bleeding, 49 sulodexide presents a promising option in the limited therapeutic arsenal for post-COVID-19 complications, including long COVID-19.
Conclusions
In this study, patients with cardiometabolic risk factors who were in a convalescent phase from moderate or severe COVID-19 within 40 days of clinical presentation showed elevated serum levels of biomarkers indicative of ongoing endothelial cell activation and dysfunction. Patients receiving sulodexide for eight weeks exhibited a reduction in TM, vWF, D-dimer, CRP, and IL-6 levels compared to placebo, suggesting a protective effect on the release of biomarkers associated with thromboinflammation and endothelial damage.
Treatment with sulodexide may be beneficial in reducing endothelial injury in the convalescent phase of high-risk COVID-19 patients, potentially leading to better clinical outcomes.
Given the limited therapeutic options available to address endothelial dysfunction in high-risk convalescent COVID-19 patients, sulodexide should be considered, as it targets major components of post-COVID-19 known pathophysiology. However, more robust clinical investigations are warranted to confirm these findings.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241297647 - Supplemental material for Treatment with Sulodexide Downregulates Biomarkers for Endothelial Dysfunction in Convalescent COVID-19 Patients
Supplemental material, sj-docx-1-cat-10.1177_10760296241297647 for Treatment with Sulodexide Downregulates Biomarkers for Endothelial Dysfunction in Convalescent COVID-19 Patients by Alejandro J Gonzalez-Ochoa, Gyozo Szolnoky, Ana G Hernandez-Ibarra and Jawed Fareed in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-2-cat-10.1177_10760296241297647 - Supplemental material for Treatment with Sulodexide Downregulates Biomarkers for Endothelial Dysfunction in Convalescent COVID-19 Patients
Supplemental material, sj-docx-2-cat-10.1177_10760296241297647 for Treatment with Sulodexide Downregulates Biomarkers for Endothelial Dysfunction in Convalescent COVID-19 Patients by Alejandro J Gonzalez-Ochoa, Gyozo Szolnoky, Ana G Hernandez-Ibarra and Jawed Fareed in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Author Disclosures
Alejandro J Gonzalez Ochoa currently serves as a consultant for Alfasigma SpA, and has received travel and registration support for meetings from Alfasigma SpA.
Consent to Participate
All participants signed a physical consent form to participate in the trial and for the use of biological samples.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AJGO currently serves as a consultant for Alfasigma Mexico and has received support for travel and registration for medical meetings. Rest of Co-authors have no conflict of interest to declare
Ethical Considerations
This research was board-reviewed by the Comité de Bioseguridad de Investigación Biomédica para el Desarrollo de Fármacos S.A. de C.V. file CB 000001, and by RM Pharma Specialists S.A. de C.V. Research and Ethic Committee file CEI 000002.
Funding
This study was independently initiated by the lead researcher and sponsored by Centro Medico del Noroeste with limited financial support from Alfasigma SpA, with the latter providing the sulodexide and placebo capsules for the duration of the trial. Alfasigma SpA did not take part in enrolling trial participants; collecting, managing, analyzing, and interpreting the data; or the decision to submit the report for publication.
Alfasigma SpA,
Statements and Declarations
The research in this paper was presented as a poster at the 32nd Congress of the International Society on Thrombosis and Haemostasis (ISTH), held from June 22 to 26, 2024, in Bangkok, Thailand.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
