Abstract
Background
Coronary atherosclerosis (CAS) is a chronic inflammatory condition marked by damage to the coronary artery endothelium, lipid accumulation, and fibrosis. It stands as the principal etiology of coronary heart disease (CHD).
Aims
The rationale of this study was to investigate the clinical value and potential mechanism of miR-373-3p in carotid CAS.
Methods
A total of 95 patients with CAS and 35 controls were enrolled in the study. RT-qPCR was used to evaluate the relative expression of miR-373-3p. ROC curve was used to analyze the diagnostic value of miR-373-3p in CAS. Logistic regression analysis was utilized to evaluate whether miR-373-3p serves as a risk factor for CAS. In addition, miR-373-3p overexpression and knockdown models of endothelial progenitor (EPCs) were established to investigate the mechanism of miR-373-3p in the regulation of EPCs.
Results
The level of miR-373-3p in CAS patients was significantly increased. MiR-373-3p can well distinguish patients with CAS and is a risk factor for CAS. The over-expression of miR-373-3p can substantially inhibit the proliferation, migration and invasion of EPCs, and stimulate the apoptosis of EPCs. MiR-373-3p is involved in the progression of CAS by targeting VEGFA.
Conclusions
As a highly sensitive potential biomarker, miR-373-3p can predict the occurrence and progression of CAS. Additionally, miR-373-3p is involved in the progression of CAS by targeting VEGFA, which may play an essential role in the pathogenesis of CAS.
Introduction
Coronary atherosclerosis (CAS) is a form of atherosclerosis (AS) affecting the coronary arteries. 1 This prevalent cardiovascular condition is characterized by the accumulation of lipids and complex sugars in the damaged endothelial cells (ECs) of these arteries, leading to atherosclerotic changes within the intima and media. 2 The progression of CAS is marked by an increasing buildup of both calcified and non-calcified plaques, which progressively narrows or can even occlude the coronary arteries. 3 Given that these arteries are crucial for supplying oxygenated blood to the heart, any stenosis or obstruction can profoundly impact both physical and mental health, elevating the risk of coronary heart disease (CHD), myocardial infarction, arrhythmias, and other severe conditions. 4 Consequently, CAS is a leading cause of mortality and long-term morbidity worldwide, with its prevalence rising with age. 5 The pathogenesis of CAS is intricate, involving a range of cell types, growth factors, cytokines, and regulatory molecules in its development and progression. 6 There is an urgent need to delve into potential targets at various levels to enhance the diagnosis and prognosis of CAS.
Coronary angiography, while considered the gold standard for diagnosing CAS, is not the preferred initial diagnostic tool due to its invasive nature and complex procedure. 7 Thus, identifying effective indicators for screening and evaluating CAS is crucial for clinical management. MicroRNAs (miRNAs), which are involved in various physiological and pathological processes, exhibit pronounced tissue and cell specificity. 8 Their detection in blood specimens is both straightforward and reliable, making them promising candidates for sensitive biomarkers in disease diagnosis. 9 Prior research has established a significant association between miRNAs and the development of AS and CHD. 10 Research has shown that miR-373 is associated with lipid metabolism. 11 Furthermore, studies have demonstrated the abnormality of miR-373-3p and its diagnostic value in patients with myocardial infarction (MI) and obstructive coronary artery disease (CAD). 12 However, studies focusing on miR-373-3p in the context of CAS are limited. Therefore, this study aims to investigate the expression and role of miR-373-3p in CAS, evaluating its potential as a diagnostic and risk assessment tool for this condition, and providing a new theoretical foundation for its clinical application.
Materials and Methods
General Information
During the period from July 2022 to March 2023, patients who underwent coronary angiography at the Department of Cardiology of The Sixth People's Hospital of Zibo, as well as control individuals who received physical examinations at the same institution, were selected as participants for this study. All subjects and their families consented to this study and signed an informed consent form. The study was meticulously reviewed and approved by the Medical Ethics Committee of The Sixth People's Hospital of Zibo.
A total of 95 patients with CAS were screened. Eligibility for the study required patients to have more than 50% stenosis in at least one of the three major coronary vessels-the left anterior descending artery, the left circumflex artery, or the right coronary artery-or more than 50% stenosis in the left main coronary artery, as confirmed by coronary angiography. A control group of 35 control individuals was selected, with inclusion criteria stipulating no stenosis or less than 30% stenosis in any of the aforementioned coronary arteries, including the left main coronary artery. Exclusion criteria encompassed severe heart valve disease, malignancies or severe hematological conditions, and diabetes mellitus.
Cell Culture and Processing
Endothelial progenitor cells (EPCs) were purchased from Shanghai Yubo Biotechnology Co., LD The EPCs were cultivated in fresh endothelial basal medium enriched with 10% FBS, maintained in a 5% CO2 incubator at a temperature of 37 °C. EPCs in the logarithmic growth phase were selected, and transfection was conducted utilizing Lipofectamine 3000 (Invitrogen, USA). Subsequent experiments were carried out 48 h post-transfection. The cell transfection experiments were categorized as follows: EPCs were transfected with a mimic negative control (mimic NC), miR-373-3p mimic, inhibitor NC, or miR-373-3p inhibitors (MCE, USA).
Real-Time Quantitative PCR (RT-qPCR) Analysis
Fasting venous blood was collected from the subjects in a centrifuge tube, and then centrifuged to obtain plasma, which was stored in a cryogenic refrigerator at −80 °C until testing. RNA extraction was conducted with the miRNeasy plasma kit (Zhijie Fangyuan Technology, Beijing) to isolate miRNA. Cells from each group were collected and the supernatants were taken for testing after centrifugation. Total RNA from each cell group was extracted using TRIzol reagent (Invitrogen, USA). RNA concentration was determined with a Nanodrop2000c spectrophotometer (Thermo Scientific, USA) with the sample OD260 / OD280 ratio between 1.8 and 2.1 for the next experiment. The cDNA synthesis was performed using a reverse transcription kit (Takara, Japan). The BestarTM Sybr Green qPCR Master Mix Kit (Xinghan Sci&Tech, Shanghai) is used for real-time fluorescence quantitative PCR. The specific operating steps should be performed according to the manual. The PCR reaction system consists of 10 µL Bestar TM Sybr Green qPCR Master Mix, 0.4 µL each of the forward and reverse primers (concentration of 10 µmol/L), 1 µL cDNA sample (50 ng), and sterile water added to a total volume of 20 µL. The ABI 7500 real-time fluorescence quantitative PCR instrument for gene amplification is used for the experiment. The primers used were specific for miR-373-3p and reference U6, respectively. Amplification conditions were one cycle pre-denaturation at 95 °C for 10 min, followed by denaturation at 95 °C for 15 s and renaturation at 60 °C for 60 s, 45 cycles. There were three repetitions of the experiment. Reaction cycle curve and cycle threshold (Ct) were generated, and miR-373-3p expression level calculated using U6 as internal reference. miR-373-3p primer sequences were 5′-GAAGTGCTTCGATTTTG-3′ (forward) and 5′- CCAGTGCAGGGTCCGAGGT-3′ (reward). U6 primer sequences were 5′- GAAGTGCTTCGATTTTG −3′ (forward) and 5′- GGAACGCTTCACGAATTTGC-3′ (reward).
Cell Counting kit-8 (CCK- 8) Assay
The cells were removed from the incubator after 48 h of transfection. The cells were rinsed with an appropriate amount of PBS, the PBS was discarded and trypsin was added and the cells were incubated for 3 min. Cells were harvested, 2 mL DMEM complete medium was added to the 6-well plate, adherent cells were gently blown, collected in a 15 ml centrifuge tube, centrifuged (1000 rpm for 5 min) and supernatant discarded after centrifugation. Cells were resuspended in DMEM and seeded in 96-well plates at a density of approximately 3000 cells per well. Cells were incubated for 1 h in an incubator. After removal, 10 µL of CCK-8 solution (Rebo, Shanghai) was added to each well in the incubator. The OD values at different time points of 0, 24, 48 and 72 h were recorded and analyzed. The absorbance at 450 nm was measured using a microplate reader. Three composite wells were established for each sample set and the experiment repeated three times.
Transwell Assay
The cell culture reagent and 8 μm Transwell chambers (Corning, Shanghai) were incubated at 37 °C. Cells, in optimal growth conditions for each group, were collected and subjected to a 24-h starvation period. The cells were then digested, resuspended, and the concentration adjusted to 2 × 105/ml using serum-free medium. For migration assays, appropriate gelatin was applied to the Transwell device, while collagen membranes were used for invasion assays. DMEM medium containing 10% PBS was added to the lower chamber, and the cell suspension was introduced into the upper chamber of the Transwell. The chambers were incubated for 24 h, after which the number of migrating and invading cells was quantified. The experiment was repeated three times for each group of samples with three compound wells.
Cell Apoptosis Assay
EPCs in each group were cultured for 48 h after transfection and harvested. The cells were digested with 0.25% EDTA-free trypsin. The supernatant was removed by centrifugation after termination and the cells were collected. The cells were then transferred to a centrifuge tube, resuspended twice with pre-cooled PBS, washed and prepared as a cell suspension by adding 300 μL binding buffer. Subsequently, 5 µL Annexin V-FITC was added to the cell suspension, which was mixed evenly and incubated for 15 min under shaded conditions, followed by 10 µL PI, which was mixed gently and incubated for 10 min under shaded conditions. The cells were then subjected to flow cytometric analysis. The flow cytometry data were used for analysis and calculation of the apoptosis rate. Each sample set was run in triplicate and the experiment repeated three times.
Luciferase Reporter Assay
The bioinformatics database Starbase (https://rnasysu.com/encori/ago) predicted the target gene of miR-373-3p. VEGFA 3′ UTR was amplified and cloned into the pGL3 reporter vector to construct a wild-type VEGFA vector (VEGFA-WT). A mutant VEGFA vector (VEGFA-MUT) was constructed by combining a mutation in the VEGFA gene. VEGFA-WT or VEGFA-MUT were transfected into EPCs with miR-373-3p mimics or miR-373-3p inhibitors. A dual luciferase reporter system (Promega, USA) was used to determine relative luciferase activity. The results were normalized to the Renilla luciferase activity. Three replicates were made of each sample and the experiment repeated three times.
Statistical Analysis
SPSS 26.0 and GraphPad 9.0 were employed for data analysis. Measurement data are presented as mean ± SD. Independent sample t-tests were utilized to compare parameters between two groups, while one-way analysis of variance was applied for comparisons among multiple groups. The Area Under the Curve (AUC) and 95% Confidence Interval (CI) for miR-373-3p in predicting CAS were determined using Receiver Operating Characteristic (ROC) curves. Logistic regression was used to analyze risk factors for CAS. Statistical significance was set at P < .05.
Results
Clinical Data of Subjects
The comparison results of general data and routine index information of the two groups of subjects are shown in Table 1. There was no significant difference in general data (age, gender, BMI, smoking, drinking, hypertension and hyperlipidemia) between the two groups (P > .05). Additionally, total cholesterol (TC) and low-density lipoprotein (LDL) were significantly different between the two groups (P < .001), while triglycerides (TG) and high-density lipoprotein (HDL) were not significantly different and had no statistical significance (P > .05).
Comparison of the Clinical Data of Subjects.

CAS, coronary atherosclerosis; BMI, Body mass index; TC, Serum total cholesterol; TG, Triglycerides; LDL, Low-density lipoprotein; HDL, High- density lipoprotein. ***P < .001.
Expression and Diagnostic Value of miR-373-3p
To investigate and analyze the relationship between miR-373-3p and CAS, we evaluated the expression of miR-373-3p in both the CAS and control groups using RT-qPCR. Our findings revealed that the plasma levels of miR-373-3p in CAS patients were markedly elevated compared to those in the control group (P < .001) (Figure 1A). To assess the diagnostic potential of miR-373-3p for CAS, we employed an ROC curve analysis, which yielded an AUC of 0.915 (P < .001). The 95% confidence interval was 0.868-0.966, with sensitivity and specificity values of 81.1% and 91.4%, respectively (Figure 1B).
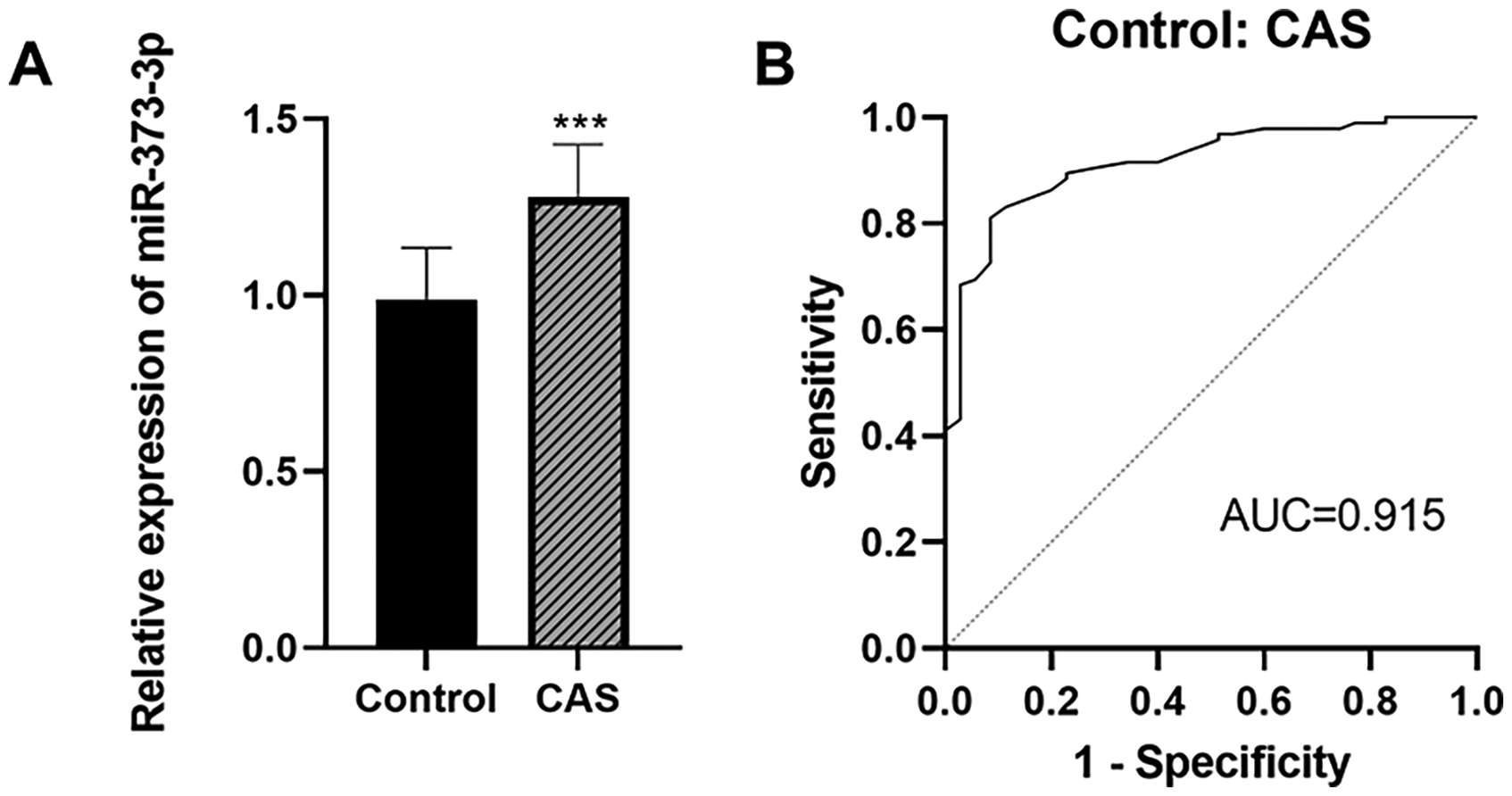
Expression and diagnostic value of miR-373-3p. The relative expression of miR-373-3p in plasma of CAS patients was significantly increased (A). The AUC for miR-373-3p in differentiating between controls and CAS patients was 0.915 (95%CI = 0.868-0.966), with sensitivity and specificity values of 81.1% and 91.4%, respectively (B). ***P < .001.
Analysis of Risk Factors for CAS
To explore the association between miR-373-3p and CAS, a logistic regression model was employed for multivariate analysis. The findings indicated that hypertension, hyperlipidemia, and miR-373-3p were significant risk factors for CAS (P < .05). In addition, smoking, TG and LDL may be risk factors for CAS, but the differences did not reach statistical significance (P > .05) (Table 2).
Logistic Regression Analyzed Risk Factors for CAS.

CAS, Coronary atherosclerosis; HR, Hazard Ratio; CI; Confidence Interval; BMI, Body mass index; TC, Serum total cholesterol; TG, Triglycerides; LDL, Low-density lipoprotein; HDL, High -density lipoprotein. *P < .05.
Effect of miR-373-3p on the Growth of EPCs
To explore the impact of miR-373-3p overexpression or inhibition on EPC proliferation, EPCs were transfected with miR-373-3p mimics, resulting in a significant increase in the relative expression levels of miR-373-3p compared to controls (P < .001). Conversely, transfection with miR-373-3p inhibitors led to a marked reduction in miR-373-3p expression (P < .01), demonstrating the effectiveness of the transfections (Figure 2A). Subsequent cell proliferation analysis showed that overexpression of miR-373-3p significantly inhibited EPCs proliferation, while inhibition of miR-373-3p had the opposite effect (Figure 2B). Flowcytometric analysis showed that the apoptosis rate of EPC was substantially higher after overexpression of miR-373-3p than in the control groups, while the opposite effect was observed after knockdown of miR-373-3p (Figure 2C).
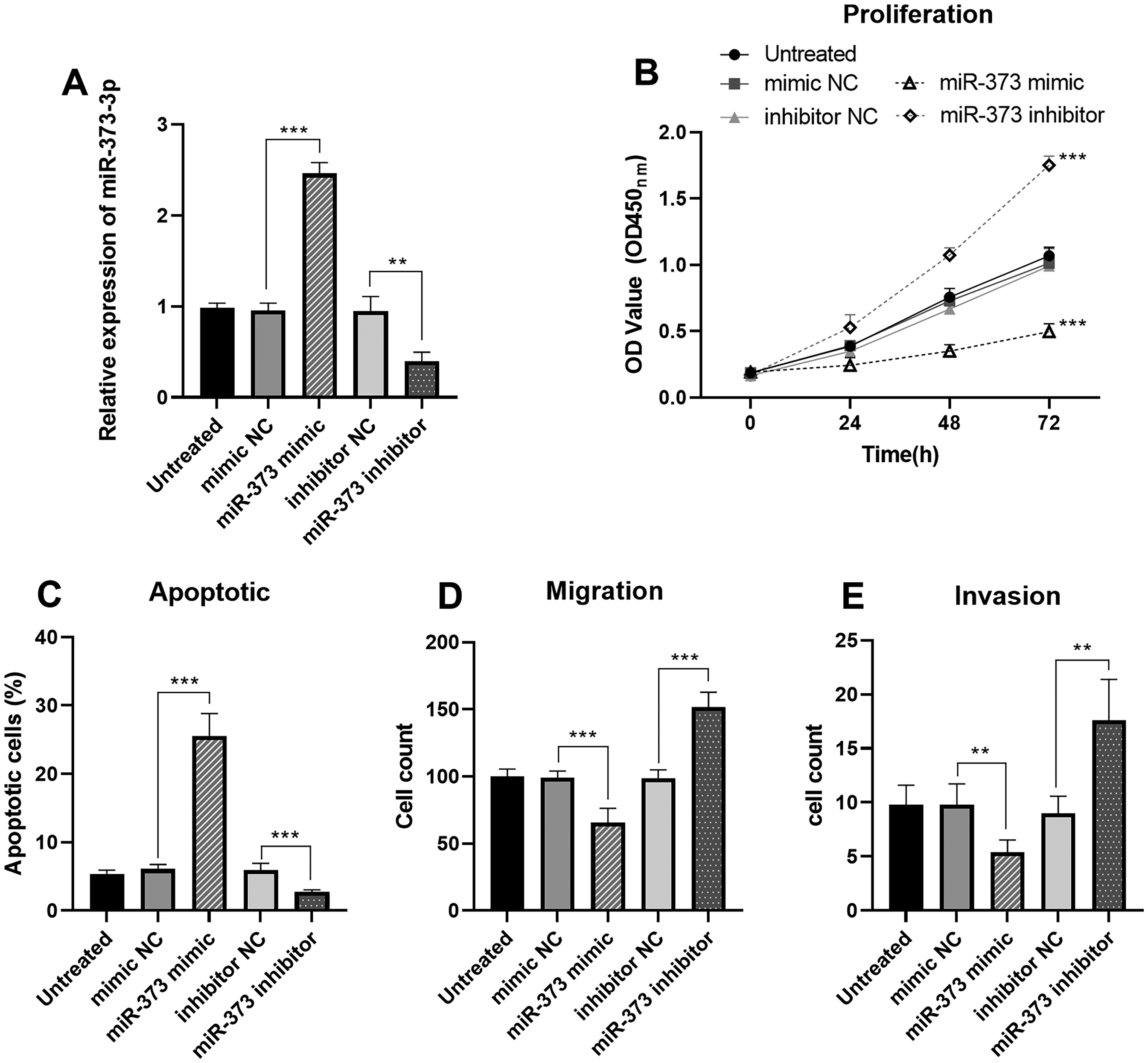
Effects of miR-373-3p on the function of EPCs. The transfection effect of EPCs was favorable (A). Overexpression of miR-373-3p inhibited the proliferation of EPCs, but promoted the apoptosis of EPCs (B-C). Overexpression of miR-373-3p inhibited the migration and invasion of EPCs (D-E). ** P < .001, *** P < .001.
Effect of miR-373-3p on EPCs Activity
The effect of miR-373-3p overexpression or inhibition on EPCs viability was further investigated. Transwell assay showed that overexpression of miR-373-3p significantly inhibited cell migration and invasion, while inhibition of miR-373-3p had the opposite effect (Figure 2D-E).
Relating miR-373-3p to VEGFA
The mechanism of miR-373-3p in CAS was further analyzed. We found that VEGFA was the target gene of miR-373-3p. Their binding sites are shown in Figure 3A. Dual-luciferase reporter assay confirmed that miR-373-3p directly targets VEGFA in EPCs (Figure 3B). The upregulation of miR-373-3p significantly inhibited the expression of VEGFA in EPCs, whereas the knockdown of miR-373-3p had the opposite effect (Figure 3C). In addition, the expression level of VEGFA was significantly lower in CAS patients than in controls (P < .001) (Figure 3D). Pearson correlation analysis showed that miR-373-3p was negatively correlated with VEGFA in CAS patients (r = −0.704, P < .001) (Figure 3E).

Relating miR-373-3p to VEGFA. Binding site of miR-373-3p to VEGFA (A). Dual-luciferase reporter assay confirmed that miR-373-3p directly targets VEGFA in EPCs (B). Overexpression of miR-373-3p markedly inhibited VEGFA levels in EPCs (C). The expression level of VEGFA was significantly lower in CAS patients than in controls (D). MiR-373-3p was negatively related to VEGFA in CAS patients (r = −0.704, P < .001) (E). ** P < .001, *** P < .001.
Discussion
CAS is a persistent inflammatory disorder marked by endothelial damage in the coronary arteries, lipid accumulation, and fibrosis. 13 Current research underscores CAS as a principal contributor to CHD, which presents a significant global public health challenge due to its high rates of disability and mortality. 14 Consequently, identifying specific biomarkers associated with CAS could facilitate the early diagnosis and intervention of CHD, and offer a foundation for future gene-targeted therapies. MiRNAs were discovered and demonstrated to play key roles in various diseases including cancer. 15 Related studies have reported that some miRNAs are associated with CAS. 16 Wang et al identified miR-182-5p and miR-9-5p as upregulated in CAS. 17 MiR-381 may regulate the occurrence of CAS. 18 Previous studies have shown that miR-373-3p is upregulated in MI and CAD.12,19 Our research findings indicate that miR-373-3p is significantly elevated in the plasma of patients with CAS. This increase suggests that high plasma levels of miR-373-3p are pivotal in CAS pathology. Furthermore, miR-373-3p exhibits high accuracy in differentiating CAS patients, indicating its potential as a predictive marker for CAS. Notably, multivariate analysis results further supported plasma miR-373-3p as a risk factor for CAS lesions. Our results further suggest that miR-373-3p upregulation is associated with CAS development and progression. Therefore, we further established miR-373-3p overexpressing and knockdown EPCs models in vitro to further explore its potential mechanism in CAS.
EPCs play an integral role in several pathophysiological processes, including wound repair, tumor growth and angiogenesis. 20 Endothelial dysfunction and inflammation are the initial links of AS. 21 Studies have shown that miR-221/222 plays an important role in endothelial physiology. 22 Existing research highlights the relationship between endothelial cell dysfunction and CAS, underscoring the significance of studying the functional mechanisms of these cells to better understand CAS pathogenesis. 23 Our results showed that the expression of miR-373-3p could significantly affect the function of EPCs. This indicates that miR-373-3p may be involved in the onset and development of CAS by affecting the physiological role of EPCs. In addition, VEGF is a high-specific pro-vascular ECs growth factor. It can promote the increase of vascular permeability and angiogenesis.24,25 It has been shown that miR-206 inhibits CAD progression by regulating VEGF expression. 26 Jin et al showed that miR-214 affects CAD pathogenesis by targeting VEGF. 27 VEGFA is a member of the VEGF family. 28 It is the first identified factor to regulate vascular permeability, which helps to induce the proliferation and migration of vascular ECs and plays an essential role in the process of deriving new blood vessels from the existing vascular system.29,30 Our results indicated that VEGFA was a target gene of miR-373-3p. miR-373-3p is involved in the progression of CAS by targeting VEGFA. The results elucidate the mechanism underlying miR-373-3p in CAS.
Research has evidence that miR-373-3p targeted DKK1 and promoted EMT-induced metastasis in tongue squamous cell carcinoma through the Wnt /β-Catenin pathway. 31 Qu et al showed that mir-373-3p mediated TGF-β/Smad4 signaling pathway to alleviate the malignant characteristics of glioma cells. 32 Our study demonstrated that miR-373-3p targeted VEGFA to regulate the function of EPCs and participate in the progression of CAS. However, further studies are needed to fully elucidate the specific mechanism of miR-373-3p in CAS.
The current experiment has notable limitations, including selection bias in the recruitment of research subjects and a lack of long-term follow-up. To address these issues, we will enhance the experimental design and expand the sample size. Further investigation into the specific mechanisms by which miR-373-3p regulates the onset and progression of CAS will refine the experimental outcomes and provide additional data to support the future development of precision treatments for CAS.
Taken together, miR-373-3p emerges as a promising and sensitive biomarker for forecasting both the onset and progression of CAS. Additionally, miR-373-3p is involved in the progression of CAS by targeting VEGFA, which may have an essential role in the pathophysiological process of CAS.
Footnotes
Consent for Publication
All patients provided written informed consent.
Data Availability Statement
Corresponding authors may provide data and materials.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Considerations
The study protocol was approved by The Sixth People's Hospital of Zibo. All procedures performed in studies involving human participants were in accordance with the 1964 Helsinki Declaration and later versions.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
