Abstract
Objectives:
We hypothesized that the guideline-recommended peak anti-Xa levels for pregnant women with mechanical prosthetic heart valves (MPHVs) receiving adjusted dose low-molecular-weight heparin (LMWH) are associated with subtherapeutic trough levels and consequently with an inadequate level of anticoagulation.
Background:
Low-molecular-weight heparin is often used for anticoagulation in pregnant women including those with MPHV. American College of Cardiology/American Heart Association guidelines recommend monitoring of plasma anti-Xa factor peak levels and adjustment of the dose to achieve peak levels of 0.7 to 1.2 U/mL. In spite of these recommendations, cases of valve thrombosis during pregnancy continue to occur.
Methods and Results:
We studied 30 pregnant patients receiving anticoagulation for various indications with adjusted dose LMWH given subcutaneously twice a day which had both trough and peak anti-Xa levels throughout pregnancy for a total of 187 paired determinations. The recommended peak anti-Xa levels (0.7-1.2 U/mL) were obtained in 123 (66%) of the measurements but in 80% of them, the trough levels were found to be subtherapeutic (<0.6 U/mL). Subtherapeutic trough levels were found in 8 (73%) of the 11 measurements with peak levels of 0.7 to 0.79 U/mL, 17 (74%) of the 23 of 0.8 to 0.89 U/mL, 21 (72%) of the 29 of 0.9 to 0.99 U/mL, and 28 (44%) of the 63 of 1.0 to 1.2 U/mL. There were 42 measurements with peak anti-Xa levels >1.2 U/mL and even in these cases, 13 (31%) of the trough levels were found to be subtherapeutic.
Conclusions:
Anticoagulation with adjusted dose LMWH aimed to achieve guideline-recommended peak levels of anti-Xa for patients with MPHVs is commonly associated with subtherapeutic trough levels. Routine measurement of trough anti-Xa levels is therefore advisable in women with MPHV treated with LMWH during pregnancy to assure adequate level of anticoagulation.
Introduction
Pregnancy is associated with an increased risk of thrombosis in women with mechanical prosthetic heart valves (MPHVs). 1,2 Achieving a safe and effective anticoagulation for pregnant women with MPHV is challenging because all current anticoagulation regimens including oral anticoagulation (OAC) or heparins may be associated with important maternal and fetal complications. 1 –21 Substantial evidence exists for the efficacy and safety of low-molecular-weight heparin (LMWH) in the prevention and treatment of thromboembolism during pregnancy in patients with evidence of deep vein thrombosis and thrombophilia. 13 There has also been an increased experience with the use of this therapy in women with MPHVs. 2,14 –21 However, despite the use of guideline-recommended regimens, cases of valve thrombosis during pregnancy continue to occur. 1,2 Recent guidelines by the ACCP, 22 the American College of Cardiology/American Heart Association (AHA/ACC), 23 and the European society of cardiology 24 have recommended the use of LMWH in pregnant women with MPHVs with a dose adjusted to achieve a peak anti-Xa levels between 0.7 to 1.2 and 0.8 to 1.2 U/mL, respectively. In the absence of controlled clinical trials, these recommendations are not evidence based and need to be further investigated. In addition, preliminary data have suggested that therapeutic peak anti-Xa levels during pregnancy are often associated with subtherapeutic trough levels which may be responsible for thromboembolic complications. Indeed, the manufacturer of the LMWH enoxaparin recommends the measurement of peak and trough anti-Xa levels, 25 a recommendation adopted by some authors who advocated monitoring both trough and peak anti-Xa levels in pregnant women with MPHVs treated with LMWH. 21
The goal of our study was therefore to evaluate the relationship between guideline-recommended peak anti-Xa levels and trough levels in a relatively large number of women receiving anticoagulation with LMWH during pregnancy and examine the hypothesis that guideline-recommended peak anti-Xa levels are often associated with subtherapeutic trough levels.
Methods
We conducted a retrospective review of anti-Xa levels in pregnant women treated with weight-adjusted dose of LMWH (enoxaparin, Sanofi-Aventis, Sanofi Winthrop Industrie, France), administrated subcutaneously twice daily, starting at a dose of 1 mg/kg and up titrating it to a recommended peak anti-Xa level (obtained at 4 hours postinjection) of 0.8 to 1.2 U/mL. 21 Both trough and peak anti-Xa levels were monitored initially every 3 days until the target anti-Xa level was achieved and after that at least every 2 weeks. The induction of labor or cesarean section was typically planned at 38 weeks of gestation, and LMWH was withdrawn at least 36 hours before delivery and substituted with intravenous unfractioned heparin (UFH). At the onset of active labor, UFH was withheld so that epidural anesthesia could be administered. Depending on the degree of uterine bleeding, dose-adjusted UFH or LMWH was reinitiated 6 to 12 hours after delivery.
Anti-Xa levels were measured using a chromogenic antifactor Xa heparin assay, Berichrom Heparin reagent kit, Siemens (Kaplan Medical Center, Rehovot, Israel), or Rotachrom Heparin reagent kit, Diagnostica Stago (University of Southern California, Los Angeles, California), per manufacturer instructions with correlation coefficient of .92 and .97, respectively. In a comparison study of the 2 heparin assays, no significant difference in the heparin values was noted between assay kits (r = .93; slope = 0.98; P = .73). 26
Statistical Analysis
As this was an exploratory study not powered to examine causality, mostly descriptive statistics were used to present our data. Results for continuous variables were presented as mean ± standard deviation and for categorical variables as count (percentage). The trough and peak levels were compared by linear regression analysis. Correlation coefficients are expressed as r values, and P value of <.05 was considered statistically significant. This retrospective study was conducted with approval of the institutional review board.
Results
Patient Population
The data were obtained from 30 pregnant patients (mean age 31 ± 5 years) followed at the cardiology high-risk pregnancy service of the University of Southern California/Los Angeles County Hospital, Los Angeles, California (20 patients) and the Heart Institute, Kaplan Medical Center, Rehovot, Israel (10 patients) who received anticoagulation with LMWH given subcutaneously every 12 hours throughout their pregnancies, using the same protocol (number of determinations per patient ranged from 5 to 38). Indications for LMWH treatment were new-generation MPHV in 8 patients (3—aortic valve and 5—mitral valve); mitral stenosis with and without atrial fibrillation in 12 women (2 were postvalvuloplasty, 2 had left atrial thrombi, and 2 had a history of cerebrovascular accident); combined mitral stenosis and regurgitation in 4 women; dilated cardiomyopathy and pulmonary embolism in 1 woman; and venous thrombosis and pulmonary embolism in 5 women.
Complications
There were no fetal complications or maternal thrombembolic events. Maternal bleeding complications occurred in 2 women, the first had atrial fibrillation and developed intrauterine hematoma in the second trimester which did not require any intervention and the second had severe mitral stenosis and underwent balloon valvuloplasty, which was performed inadvertently less than 24 hours from discontinuation of the last LMWH dose and was complicated by the development of cardiac tamponade. This patient required pericardiocentesis and underwent urgent mitral valve replacement.
Anti-Xa Levels
There were overall 225 measurements of anti-Xa levels with 212 determinations of trough levels and 200 determinations of peak levels. Paired determinations of both peak and trough levels were obtained in 187 patients and were included in the present analysis. The mean peak anti-Xa level was 1.02 ± 0.28 U/mL and the trough was 0.55 ± 0.21 U/mL. Therapeutic peak levels of anti-Xa between 0.7 and 1.2 U/mL were measured in 123 (66%) of all 187 patients. Trough level was found to be subtherapeutic (anti-Xa level <0.6 U/mL) in 99 (80%) patients. Subtherapeutic trough levels were found in 8 (73%) of the 11 measurements with peak levels of 0.7 to 0.79 U/mL (n = 11), 17 (74%) of the 23 of 0.8 to 0.89 U/mL (n = 23), 21 (72%) of the 29 of 0.9 to 0.99 U/mL (n = 29), 28 (44%) of the 63 of 1.0 to 1.2 U/mL (n = 63), and 3 (7.1%) of the 42 of >1.2 U/mL (n = 42; Figure 1). There were 42 measurements of peak anti-Xa >1.2 U/mL and also 13 (31%) of them showed subtherapeutic trough levels. High peak levels >1.5 U/mL were found in only 6 (3%) of the measurements, 2 of these had trough levels > 0.8 U/mL, and the rest levels of 0.6 to 0.8 U/mL.

Percentage of subtherapeutic trough levels according to peak anti-Xa level categories.
A statistically significant correlation (r = .68) was observed between all measured peak and trough anti-Xa levels (Figure 2), which was preserved after adjustment to the gestational week (r = .70). In order to determine the individual patient relationship between the peak and the trough levels, we examined the peak to trough anti-Xa ratios stratified by pregnancy trimesters. Mean values of peak to trough ratio as measured in the different gestational trimesters are presented in Figure 3 (20% of both peak and trough anti-Xa levels were measured in the first trimester, 37% in the second, and 44% in the third). We found that this ratio becomes smaller and displays less variability with pregnancy progression (P < .001): the median (interquartile range) was 2.14 (1.0), 1.8 (0.63), and 1.71 (0.52) for the first, second, and third gestational trimesters, respectively.
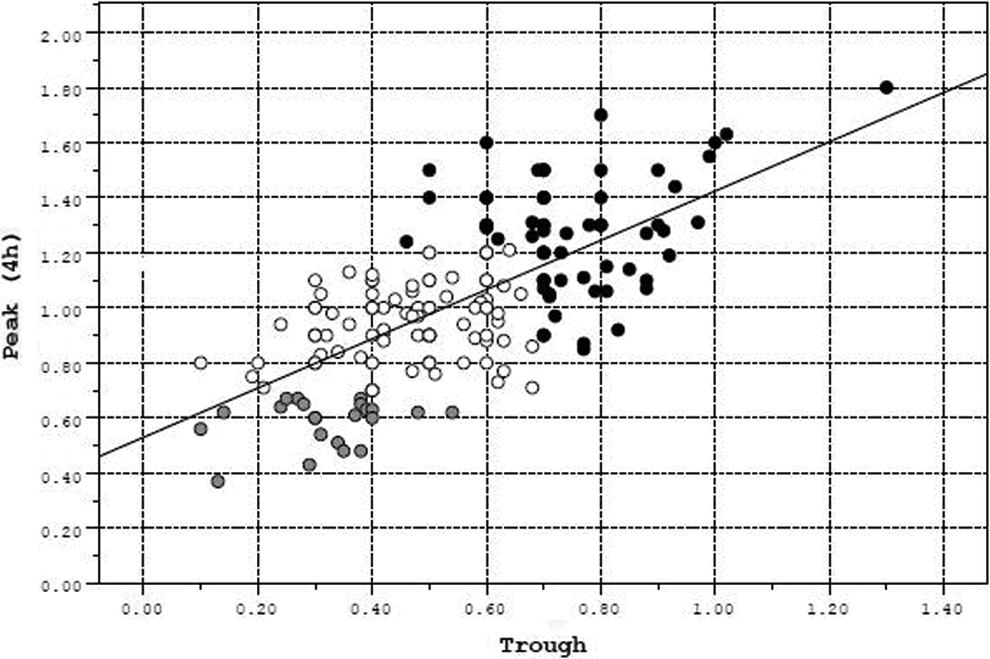
Correlation between peak and trough anti-Xa levels. Scatter plot of trough and peak anti-Xa level correlation. Therapeutic peak anti-Xa levels associated with subtherapeutic trough anti-Xa levels (constituting 80% of the therapeutic peak anti-Xa measurements) are marked in white circles; therapeutic peak anti-Xa levels associated with therapeutic trough anti-Xa levels are marked in black-filled circles (constituting 20% of the therapeutic peak anti-Xa measurements). In a minority (12%) of the patients, both peak and trough levels were subtherapeutic and they are marked in gray-filled circles.

Box plots of peak to trough anti-Xa ratios for each of the 3 gestational trimesters. The band at the middle of each box plot represents the median, the bottom and top of the boxes represent the 25th and 75th percentiles, and the whiskers at the ends of the boxes show the distance from the end of the box to the largest and smallest observed values that are less than 1.5 box lengths from either end of the box. Outliers displayed are cases with values between 1.5 and 3 box lengths.
Discussion
The main finding of the current study is that treatment with adjusted doses of LMWH, achieving guideline-recommended peak anti-Xa levels for women with MPHVs (between 0.7 and 1.2 U/mL), was associated with subtherapeutic trough anti-Xa levels in 80% of the patients.
Pregnancy is associated with increased risk of thrombosis in women with MPHVs, with a high mortality rate 1 –3,21 requiring effective anticoagulation. Although treatment with oral anticoagulants has been shown in general to be effective at reducing the risk of thromboembolic complications in pregnant women with MPHVs, isolated cases of thromboembolic events have been reported on this therapy. 3 The main concern with the use of OAC during pregnancy is related to a significant risk of fetal side effects as well as maternal bleeding. Although a number of recent studies have reported reduced fetal side effects with the use of low-dose OAC, 5 –9 other reports have clearly shown a high and unacceptable rate of fetal complications even with reduced OAC dose. 10 –12 Unfractionated heparin was previously the heparin of choice for the prevention of thromboembolic complications during pregnancy because it does not cross the placenta and is therefore associated with low direct risk to the fetus. 1,13,21 At the same time, however, the drug may be associated with important untoward effects including heparin-induced thrombocytopenia and osteopenia. 1-2,13 Increased volume of distribution due to higher blood volume during pregnancy and enhanced renal excretion due to increased glomerular filtration rate result in reduced half-life and a risk of inadequate level of anticoagulation, 13 which may be the cause for high reported rate (33%) of MPHVs thrombosis among women who received UFH throughout pregnancy. 3 Substitution of heparin for OAC in the first trimester was also associated with a high rate (9.5%) of maternal thromboembolic complications. 3
Therapy with LMWH in pregnancy seems an attractive and convenient alternative to anticoagulation of women with MPHV. Similar to UFH, LMWH also does not cross the placenta but has several advantages over UFH including longer half-life, higher bioavailability, more predictable dose response, and improved side effect profile. 27 Over the last 2 decades, there has been a growing experience with the use of LMWH treatment in pregnant women with MPHVs. Earlier published data on the use of LMWH in women with MPHV during pregnancy 1 were limited to small groups of patients or to isolated case reports with several of these cases complicated by valve thrombosis and even death. A careful review of the reported cases, however, suggested that most of them were associated with an inadequate dose, lack of monitoring, or subtherapeutic anti-Xa levels. 14 –22,28 A recent review by McLintock 2 has summarized the maternal and fetal complications in 92 women from 5 prospective cohort studies treated with dose-adjusted LMWH throughout pregnancy. 2 Nine episodes of valve thrombosis were reported and were attributed to poor compliance or suboptimal LMWH doses in the majority of the cases. 2 In addition, Yinon et al 17 has recently reported valve thrombosis and death in a women with a mechanical Medtronic Hall aortic valve who presented with a transient ischemic attack at 24-weeks’ gestation in spite of guideline-recommended levels of peak anti-Xa levels and a history of compliance with therapy, trough levels of anti-Xa however, were not measured.
Similar to UFH, the pharmacokinetics of LMWH have been shown to change during pregnancy with lower plasma concentrations, probably due to higher volume of distribution and enhanced renal clearance. 1,13 Therefore, dose adjustment of LMWH on the basis of only peak anti-Xa level may not achieve adequate level of anticoagulation around the clock. This suggestion was initially made by Barbour et al 29 who evaluated 138 peak and 112 troughs anti-Xa levels in 13 pregnancies in 12 patients who received LMWH for anticoagulation. These investigators demonstrated that with peak anti-Xa levels of 0.63, 0.70, and 0.69 U/mL measured at the first, second, and third trimesters, respectively, mean trough levels were only 0.21, 0.30, and 0.40 U/mL with only 9% of the measurements achieving a level of >0.5 U/mL. Even when peak levels were between 0.75 and 1.0 U/mL, only 15% of trough levels were >0.5 U/mL. Similarly, in a recent report in 15 pregnant women treated with LMWH at different gestational ages, a subtherapeutic peak anti-Xa level was demonstrated in 20% of the measurements and in 73% of trough levels despite “therapeutic” enoxaparin administration of 1 mg/kg twice a day. 30
The results of the present study also demonstrate a statistically significant correlation between all measured peak and trough anti-Xa levels, which remained preserved after adjustment to the gestational week. At the same time, however, individual prediction of trough levels based on peak levels seemed suboptimal in many cases. When using the peak to trough anti-Xa ratios stratified by pregnancy trimesters to assess the individual patient relationship between the peak and the trough levels, we found a significantly smaller variability in the second and third trimester compared to the first. The larger variability in the first trimester could be related to initiation of treatment and gradual up titration of LMWH to achieve the desirable peak and trough levels. Peak anti-Xa levels in second and third trimesters are more predictive of therapeutic trough levels such that peak levels of 1.28 U/mL in the second trimester and of 1.22 U/mL in the third trimester were associated with therapeutic trough levels in about 75% of patients. These findings may be of some clinical relevance in late pregnancy in cases where monitoring of anti-Xa trough levels is not possible. At the same time, however, our study provides further support to the importance of measuring trough anti-Xa levels to guide efficacy of anticoagulation with LMWH in pregnant women who are at high risk for the development of thromboembolic complications. Our findings based on a strict clinical protocol in a relatively large number of anti-Xa measurements demonstrate that monitoring peak anti-Xa level only is not a reliable guide for anticoagulation efficacy of LMWH during pregnancy. Adjusted dose LMWH achieving guideline-recommended peak anti-Xa levels between 0.7 and 1.2 U/mL was associated with subtherapeutic trough levels in as many as 80% of patients. In addition, although we found a statistically significant correlation between the group peak and the trough anti-Xa levels, there were large individual variations not allowing the prediction of trough levels based on peak levels. Equally important was the finding that therapeutic trough anti-Xa levels of ≥0.6 U/mL, as previously recommended by us for pregnant patients with MPHV, 21 were rarely associated with excessive peak anti-Xa levels and seem to be safe. Our findings, in addition to the continuing reports of valve thrombosis due to subtherapeutic anticoagulation with LMWH, suggest the importance of routine measurement and maintenance of trough levels at therapeutic range in order to assure adequate anticoagulation in pregnant women with MPHVs. To prevent excessive anticoagulation and possible bleeding complications, peak levels should also be monitored to maintain anti-Xa levels <1.5 U/mL. In case that therapeutic trough levels are associated with anti-Xa levels >1.5, a twice a day dosing has to be considered.
Limitations
Our study has a number of limitations. The patients included in our study received anticoagulation therapy during pregnancy for various reasons and not all patients had MPHVs. For this reason and although the incidence of bleeding complications could be evaluated in the entire study population, we were able to assess valve-related thromboembolic complications in only 8 patients with MPHV. At the same time, however, the primary goal of the study was to assess the relation between peak and trough anti-Xa levels that should not have been influenced by the indication for anticoagulation.
We defined subtherapeutic anticoagulation with LMWH as an anti-Xa level of ≤0.6 U/mL; this definition is arbitrary and was designed to provide a “safe” level of anticoagulation at any time. Freedom from thromboembolic events in all 8 patients with MPHV in this study and in 5 additional pregnancies managed with a similar protocol recently reported by Vijayan et al, 31 compared to a high incidence of valve thrombosis reported when trough anti-Xa levels were not monitored, 2 support the clinical rational of aiming at a trough anti-Xa level of ≥0.6 U/mL.
Summary and Recommendations
Anticoagulation with adjusted does LMWH titrated to achieve guideline-recommended peak anti-Xa levels for women with MPHVs is associated with subtherapeutic trough levels in over two-third of patients and is probably related to the enhanced clearance of LMWH during gestation. This finding provides a strong support for the need to change present guideline recommendations for anticoagulation in pregnant women with MPHVs treated with LMWH and include routine monitoring of trough anti-Xa levels to avoid subtherapeutic anticoagulation along with peak levels to prevent excessive anticoagulation that may be associated with increased incidence of bleeding.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
