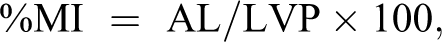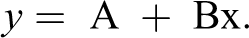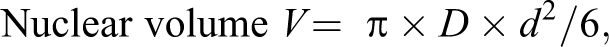Abstract
We previously showed that digitoxin prolongs the survival of rats with heart failure due to myocardial infarction (MI). In this study, we evaluated the effect of digitoxin on myocardial structure, ventricular function, and proteins involved in calcium kinetics. Seventy-two rats with MI >35% of the left ventricle were randomly assigned to 4 treatment groups: sham (n = 15), digitoxin (n = 11), infarction (n = 20), and infarction + digitoxin (n = 26). The rats were assessed 120 days after surgery by echocardiogram, hemodynamics, papillary muscle mechanics, collagen content, cardiomyocyte nuclear volume, and Western blot analysis of proteins involved in calcium kinetics. Digitoxin was administered via the rat chow. Two-way analysis of variance was used for comparisons. Myocardial infarction caused inotropic impairment, pulmonary congestion, increase of nuclear volume, myocardial collagen, and Na+/Ca2+ exchanger levels, and decreased SERCA2 and phosphorylated phospholamban levels. Treatment with digitoxin showed improvements in cardiac remodeling, inotropism, ventricular performance, pulmonary congestion, collagen accumulation, nuclear volume, and proteins involved in calcium kinetics. In rats with heart failure due to MI, long-term treatment with digitoxin attenuates congestive heart failure, mitigates myocardial remodeling and contractile impairment, and preserves myocardial levels of proteins involved in calcium kinetics.
Keywords
Introduction
After the results of the Digitalis Investigator Group (DIG) trial was published (1997), 1 the use of cardiac glycosides decreased dramatically. The DIG trial followed up 6800 patients with heart failure (HF) and reported that, although long-term administration of digoxin did not affect all-cause mortality, hospitalization due to HF was decreased and clinical symptoms were improved. However, post hoc analyses of the DIG trial data 2,3 suggested that digoxin can increase the survival rate. Specifically, serum concentrations of digitalis lower than 0.9 ng/mL were associated with lower mortality. Recent studies have suggested the usefulness of conducting another clinical trial to restudy the effect of cardiac glycosides on survival in patients with HF 2 -4 ; however, this would be difficult for practical reasons. As an alternative, in vivo studies in animals can help to define the role of cardiac glycosides in the treatment of HF. Experimental studies can help to define the role of cardiac glycosides in the treatment of HF. In our previous study, Helber et al 5 monitored infarcted rats for 280 days and showed that digitoxin increased the survival rate. Thereafter, we showed that the effect of digitoxin was dependent on the severity of cardiac impairment, improving survival in rats with major MI and severe HF. 6 In contrast, rats with lower MI and less impaired ventricular function had lower survival rates.
Traditionally, digitalis promotes a specific inhibition of the Na+/K+ pump, inducing a positive inotropic effect by increasing the intracellular Na+ concentration, which in turn induces an increase in the intracellular Ca2+ concentration by the Na+/Ca2+ exchange and an increase in the Ca2+ pool of the sarcoplasmic reticulum. That is to say: traditionally, therapeutic benefits of digitalis are considered to be based upon inotropic effect. In this study, we used the rat model of secondary congestive HF due to myocardial infarction (MI) to evaluate the effects of long-term treatment with cardiac glycosides, aiming to assess not only myocardial contractile function and congestive state but focusing myocardial structures and calcium kinetics proteins remodeling.
Materials and Methods
Animals
Virgin female Wistar rats (body weight, 180-210 g) underwent coronary artery ligation according to a previously described method. 7 -9 They were housed in clear polyethylene cages with up to 4 rats in each. The cages were kept in the same room with a 12-hour light–dark cycle and independent ventilation, temperature, and humidity controls.
Five days after the coronary occlusion or sham procedure, the rats were anesthetized (ketamine, 50 mg/kg and xylazine, 10 mg/kg; intraperitoneal injection) and examined by echocardiography to determine the MI size. Rats with MI involving <35% of the left ventricle (LV) perimeter were not used in this study. Rats with MI involving ≥35% of the LV (n = 72) were identified by an ID 100 implantable Trovan transponder (Trovan Electronic Identification System, Ulm, Germany) affixed to the nape of the neck and randomly assigned to one of the following 4 treatment groups: (1) sham operation (S: n = 15), (2) sham operation + digitoxin (SD: n = 11), (3) infarct produced by coronary occlusion (infarcted control, IC: n = 20), and (4) infarct + digitoxin (ID: n = 26). The animals received standard rat chow containing 0.6% sodium (Nuvilab CR1, Nuvital, Curitiba, PR, Brazil). Digitoxin was administered in the rat chow according to a technique previously described by our group 10 at a concentration that permitted the ingestion of approximately 0.1 mg/100 g/d, as recommended by other studies. 11 -13 The rats were observed for 120 days.
The study was conducted in accordance with the Guide for the Care and Use of Laboratory Animals (National Institutes of Health publication no. 96-23, revised, 1996). The protocol was approved by our institutional Research Ethics Committee of Federal University of São Paulo (number 1277/06).
Doppler Echocardiogram
Myocardial infarct size was measured by 2-dimensional echocardiogram (HP Sonos500; Philips Medical Systems, Andover, Massachusetts) based on LV basal, mid transverse, and apical transverse views. Three measures of the LV cavity perimeter and infarct arc length were obtained in diastole for each transverse view. The infarct size (%MI) for each segment, expressed as the proportion of the LV perimeter of each transverse view, was calculated by using the following equation:
where AL is the arc length of the MI scar, and VCP is ventricular cross-section perimeter. The total infarct size of each rat was calculated as the mean %MI of the 3 segments. In a previous study, we showed that this approach provides credible estimates of infarct size that are in agreement with size determined by tetrazolium staining. 14
At 120 days after the coronary occlusion procedure, another echocardiogram study was conducted to obtain 1- and 2-dimensional real-time images and to measure flow velocity by spectral Doppler. The images were obtained at a depth of 2.0 cm. The rats were placed in the left lateral decubitus position, and images were obtained in the parasternal longitudinal plane. The transducer was then rotated 90° clockwise to obtain transverse images of the heart in 3 planes of the LV (basal, middle, and apical). The images were recorded on videotape for further analysis.
The echocardiogram allowed the analysis of the LV diastolic area (LVdA) and LV systolic area (LVsA). Systolic function was assessed by the fractional area change (%FAC) of the transverse areas according to the following formula:
Diastolic function was assessed using indices derived from the velocity curve of mitral diastolic flow as follows: peaks of E and A waves and the E/A ratio.
Hemodynamics and Lung Water Content
After the echocardiographic study, the animals underwent left and right heart catheterizations to measure systemic and right ventricular (RV) pressures. Catheterization of the LV was performed via the right carotid artery by inserting a Millar micromanometer (Mikro Tip 2F; Millar Instruments Inc, Houston, Texas) and placing the tip of the catheter inside the LV chamber. The intraventricular pressures and first temporal derivatives (dP/dt) were evaluated. To determine RV pressure, right catheterization was performed via the right external jugular vein using an 8-cm polyethylene PE20 catheter (natural resonance frequency: 62 Hz). The hemodynamic measurements were assessed using software AcqKnowledge version 3.5.7 for Windows (Biopac Systems, Santa Barbara, California; http://www.biopac.com).
Lung water content (%H2O) was determined on the basis of the wet and dry lung weight, utilizing the formula:
Study of Myocardial Mechanics in the Papillary Muscle
Immediately after assessing the hemodynamics, the heart was quickly removed and put in an oxygenated Krebs-Henseleit solution. The posterior papillary muscle of LV was carefully dissected and vertically mounted in an organ bath containing the Krebs-Henseleit solution 15 warmed at 29°C and 100% oxygenated. Using the software AcqKnowledge version 3.7.3 for Windows (Biopac Systems; http://www.biopac.com), the following variables were determined: maximum developed tension (DTmax), rate of positive (+dT/dt) and negative (−dT/dt) developed tension (DT), and resting tension (RT).
The mechanics of the papillary muscle was assessed at muscle lengths of 92%, 94%, 96%, 98%, and 100% of the optimal length (Lmax), to define the length–active tension relations. The values found were adjusted using the linear equation:
Histomorphometric Analysis
After the animals were killed, the hearts were put in a 10% buffered formaldehyde solution for 24 hours and then dehydrated in increasing concentrations of ethanol, cleared in xylene, and embedded in paraffin. The embedded tissue was cut into 4-µm sections with a Minot microtome (Leica, Wetzlar, Germany, RM 2035), which were mounted on glass slides and dried at 37°C. The slides were analyzed under an Olympus BX40 microscope coupled to a Sony CMA-D2 digital video camera, and the images were digitalized using the program Q-Capture Pro 6.0 for further histomorphometric analysis using the software Image Tool 3.0 (UTHCSA).
To analyze the nuclear volume of cardiomyocytes, the slides were stained with hematoxylin–eosin. The mean nuclear volume of septal myocytes of the left ventricular myocardium, far from the infarcted area, was calculated as a parameter of myocyte hypertrophy. The cardiac muscle fibers were visualized at a magnification of ×400, and the volume of each nucleus was calculated according to the following equation 16 :
where D is the largest nucleus diameter and d the largest diameter perpendicular to D.
Collagen in LV myocardium was evaluated by picrosirius red staining 17 of the tissue sections, which were then analyzed under polarized light. Images were captured at a magnification of ×400.
Calcium-Binding Proteins
Frozen LV remnant myocardial was homogenized using ice-cold lysis buffer and proteinase inhibitor cocktail, and lysates (20 μg protein) were separated by 10% SDS-PAGE. The protein was then transferred to PVDF membranes (Amersham Biosciences, New Jersey, USA) and transfer efficiency was evaluated by staining with 0.5% Ponceau S. After blocking with 5% nonfat dry milk for 2 hours at room temperature, PVDF membranes were probed with the primary antibodies (Santa Cruz Biotechnology, Inc, Santa Cruz, California): rabbit anti-Serca 2 (1:1000), rabbit anti-phospholamban Ser 16 (1:2500), rabbit anti-Na+/Ca++ exchanger (1:100), rabbit anti-α1 Na+/K+-ATPase (1:1000), and rabbit anti-α2 Na+/K+-ATPase (1:1000). After night incubation, the membranes were washed 5 times with phosphate-buffered saline and incubated for 2 hours with horseradish peroxidase-conjugated goat antirabbit antibodies (1:20,000; Bio-Rad, Hercules, California), washed 5 times with blocking buffer, and then rinsed twice with phosphate-buffered saline. Antibody binding was detected by using the SuperSignal West Dura Extended Duration chemiluminescence substrate (Thermo Scientific, Rockford, Illinois), and images were obtained using an LAS-4000 system (Fujifilm Life Science, Stamford, USA). Quantification of target proteins was normalized for the corresponding mean of each group.
Statistical Analysis
Results were presented as mean ± standard error of the mean. The 4 treatment groups were compared by 2-way analysis of variance, followed by the post hoc Tukey test; P ≤ .05 was considered significant. The t test was used to compare 2 groups. Data analysis and figure preparation were carried out by using GraphPad Prism version 6.1 (GraphPad Software, San Diego, California).
Results
Infarction Size
Echocardiographic assessment showed that mean infarct size of rats that received digitoxin (ID group, 44.4% ± 6.1) did not differ significantly from that of the untreated rats (IC group, 44.2% ± 6.8; P = .87 by t test).
Doppler Echocardiogram
Results of Doppler echocardiogram (Table 1) indicated impaired ventricular function in the infarcted rats compared with rats that underwent the sham surgery. Compared with the IC group, LDdA and LVsA values were lower in the ID group and the FAC was higher. The E waves and the E/A ratio were higher in the IC group compared with the other groups, whereas these indicators of diastolic function did not differ significantly between the ID group and the sham operation groups. Taken together, these results show that digitoxin attenuated the left ventricular systolic and diastolic function impairment due to MI.
Values (x ± SEM) of ECHO, Hemodynamics, and Pulmonary Water Content.
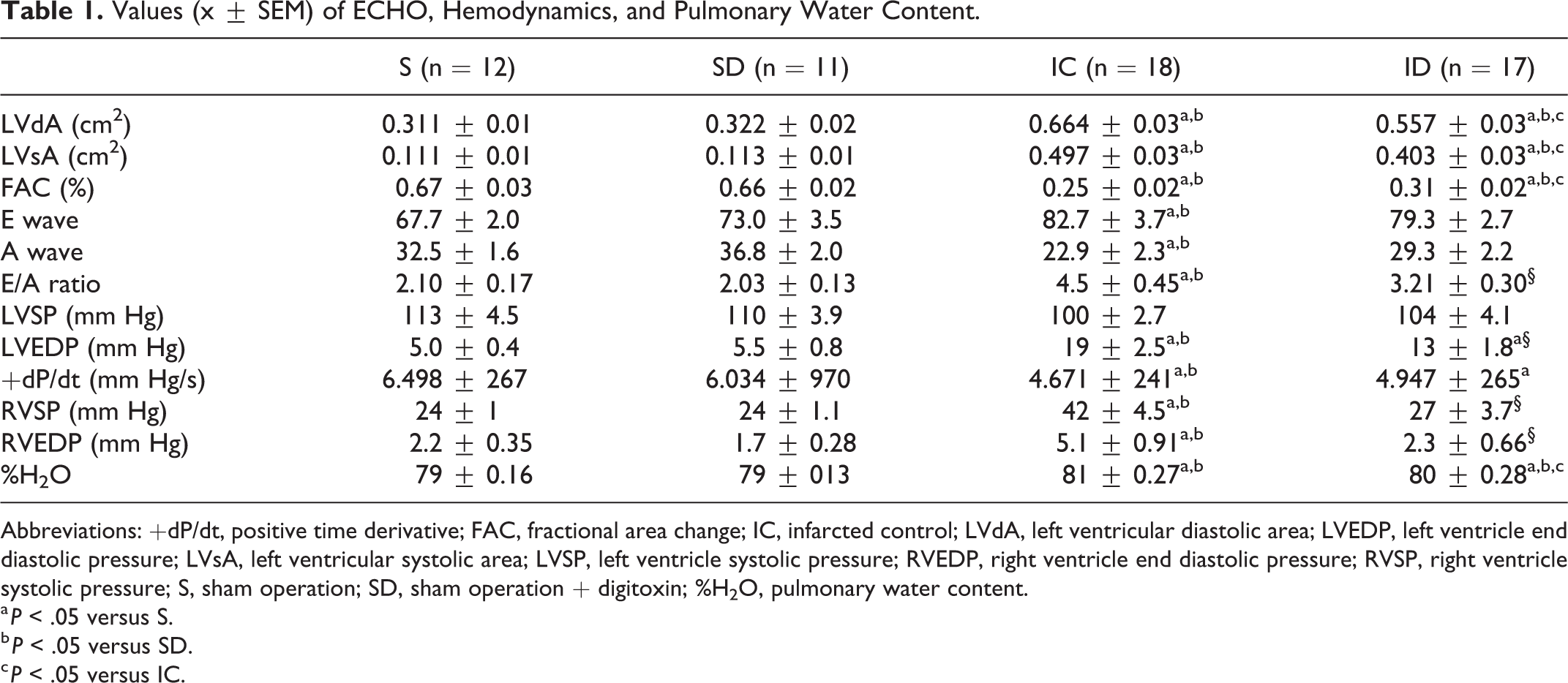
Abbreviations: +dP/dt, positive time derivative; FAC, fractional area change; IC, infarcted control; LVdA, left ventricular diastolic area; LVEDP, left ventricle end diastolic pressure; LVsA, left ventricular systolic area; LVSP, left ventricle systolic pressure; RVEDP, right ventricle end diastolic pressure; RVSP, right ventricle systolic pressure; S, sham operation; SD, sham operation + digitoxin; %H2O, pulmonary water content.
a P < .05 versus S.
b P < .05 versus SD.
c P < .05 versus IC.
Hemodynamics and Lung Water Content
No differences in LV systolic pressure were observed between treatment groups (Table 1). However, LV end-diastolic pressures were higher in the infarcted rats than in the sham operation groups. Interestingly, mean end-diastolic pressure in the ID group was lower than that of the IC group. The +dP/dt values of the infarcted rats were lower than those of the noninfarcted rats, but the IC and ID groups did not differ significantly. The RV systolic pressure of the IC group was higher than that of the other groups. The RV systolic pressure of ID did not differ significantly from that of noninfarcted rats, suggesting that digitoxin treatment prevented pulmonary hypertension in the infarcted rats. Similar results were observed when comparing RV end-diastolic pressure between treatment groups.
Lung water content was also higher in infarcted rats than in noninfarcted rats. However, lung water content was lower in the ID group compared with the IC group, indicating that digitoxin decreased pulmonary congestion in the infarcted rats.
Papillary Muscle Mechanics
Our results (Table 2) showed that developed tension in the infarcted rat was lower than that of noninfarcted rats. However, the ability of the papillary muscle to generate force was greater in the ID group than in the IC group. Similar results were found for +dT/dt. The animals in the IC group had reduced −dT/d values compared with those of S, SD, and ID groups. However, −/+dT/dt increased in ID group, showing that relaxation was increased after digitoxin.
Values (x ± SEM) of the Developed Tensions (DT), Resting Tension (RT), Positive (+dT/dt), and Negative (dT/dt) Derivatives and Ratio −/+dTdt.

Abbreviations: IC, infarcted control; S, sham operation; SD, sham operation + digitoxin.
a P < .05 versus S.
b P < .05 versus SD.
c P < .05 versus IC.
We found that tension correlated with resting length (Frank-Starling mechanism), with correlation coefficients >0.97 (Figure 1). The slope of the IC group (0.103 ± 0.02) was lower than that of the other treatment groups; slopes for the other 3 treatment groups did not differ significantly (S, 0.288 ± 0.04; SD, 0.275 ± 0.08; ID, 0.240 ± 0.02). This result indicates that digitoxin ameliorated the altered myofilament calcium sensitivity in the infarcted rats (Frank-Starling mechanism). Mean values for RT (g/mm2) at the optimal muscle length did not differ significantly between groups (Table 2).
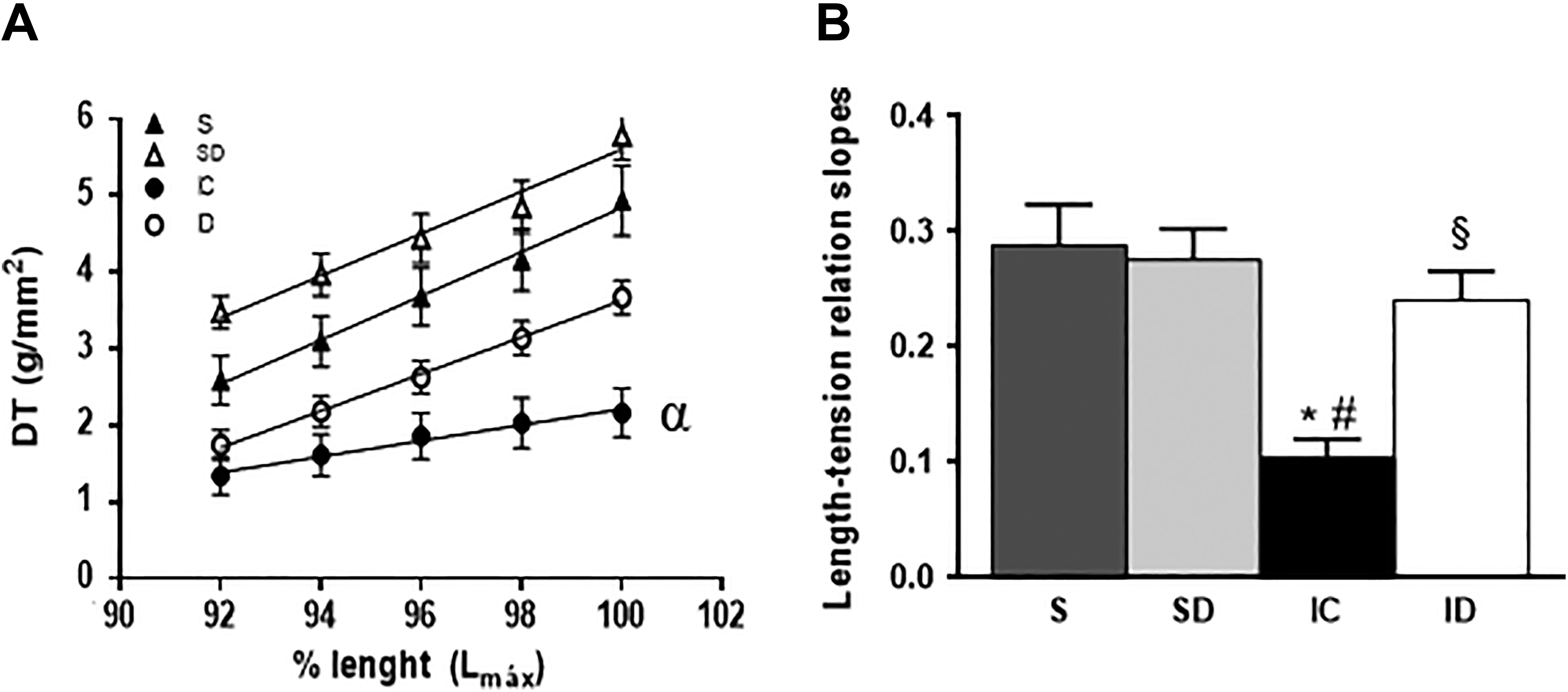
Values (x ± standard error of the mean) of developed tension (DT) at various levels of papillary muscle length. α: lower than the others (A) and the slopes of the tension/relations curves (B).
Cardiomyocyte Nuclear Volume
Nuclear volumes of cardiomyocytes (μm3), which were used to estimate cardiomyocyte hypertrophy, are shown in Figure 2A. Rats in the IC group (240 ± 31) had significantly higher nuclear volumes compared with the other treatment groups, whereas mean nuclear volume in the ID group (157 ± 11) did not differ significantly from that of noninfarcted groups (S: 154 ± 14; SD: 154 ± 20).
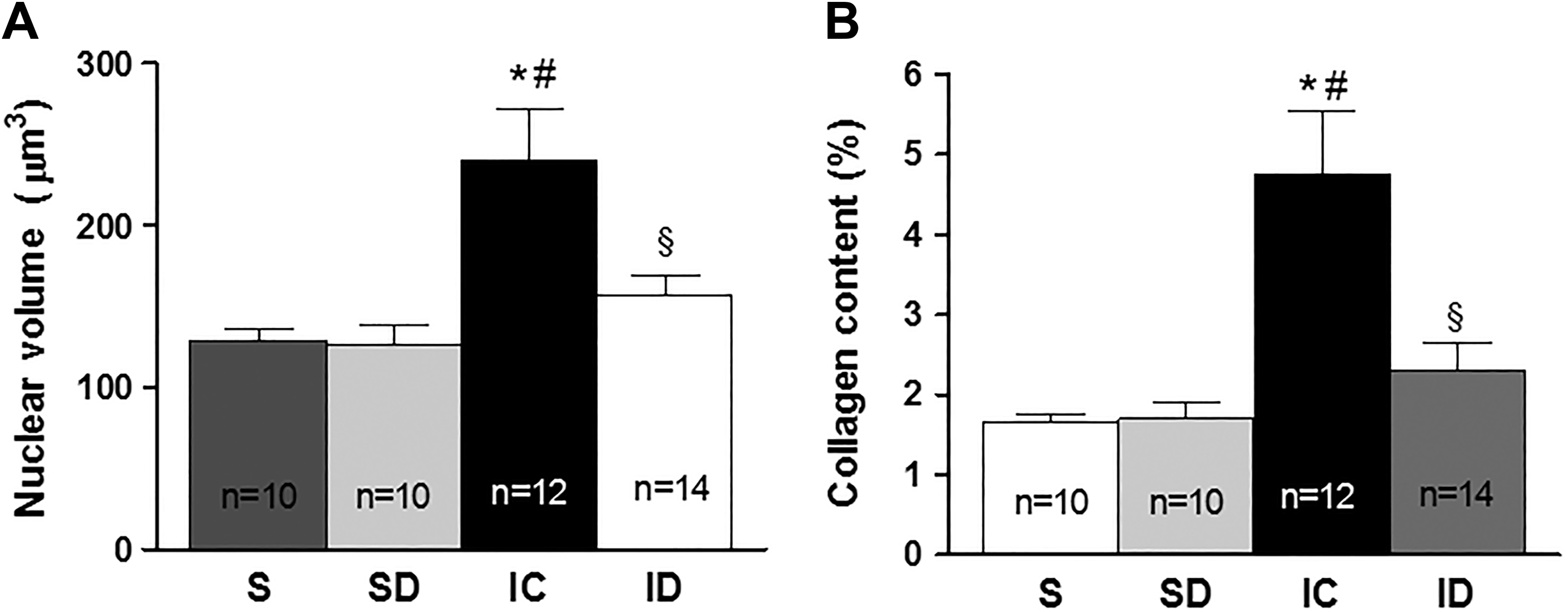
Values (x ± standard error of the mean) of (A) nuclear volume and (B) collagen content. *P < .05 versus S, #P < .05 versus SD, and §P < .05 versus IC. IC indicates infarcted control; S, sham operation; SD, sham operation + digitoxin.
Collagen Quantification
We found that myocardial collagen content (%) was significantly increased in the IC group (4.76 ± 0.8) compared (Figure 2B) with noninfarcted rats (S: 1.65 ± 0.1; SD: 1.69 ± 0.2). In contrast, myocardial collagen content in the ID group (2.30 ± 0.3) did not differ significantly from that of noninfarcted rats.
Calcium-Binding Proteins
Level of protein associated with calcium kinetics (Figure 3) in the LV remote tissue were determined by Western blotting. Our results show that levels of SERCA2 and serine 16–phosphorylated phospholamban were significantly lower in the IC group than in noninfarcted rats; levels did not differ significantly from those of the noninfarcted rats. Levels of the Na+/Ca2+ exchanger were significantly higher in the IC group than in the other 3 treatment groups. Interestingly, these proteins changed in the same sense than variations of dT/dt. The level of the Na+/Ca2+ exchanger (NCX) in the IC group was higher than that in the noninfarcted animals, while the data in the ID group did not differ compared to other groups. The protein content of the ∝2 subunit of Na+/K+-ATPAse in the ID group was higher than that in all other groups; however, the only statistically significant result was found when compared with the IC group.
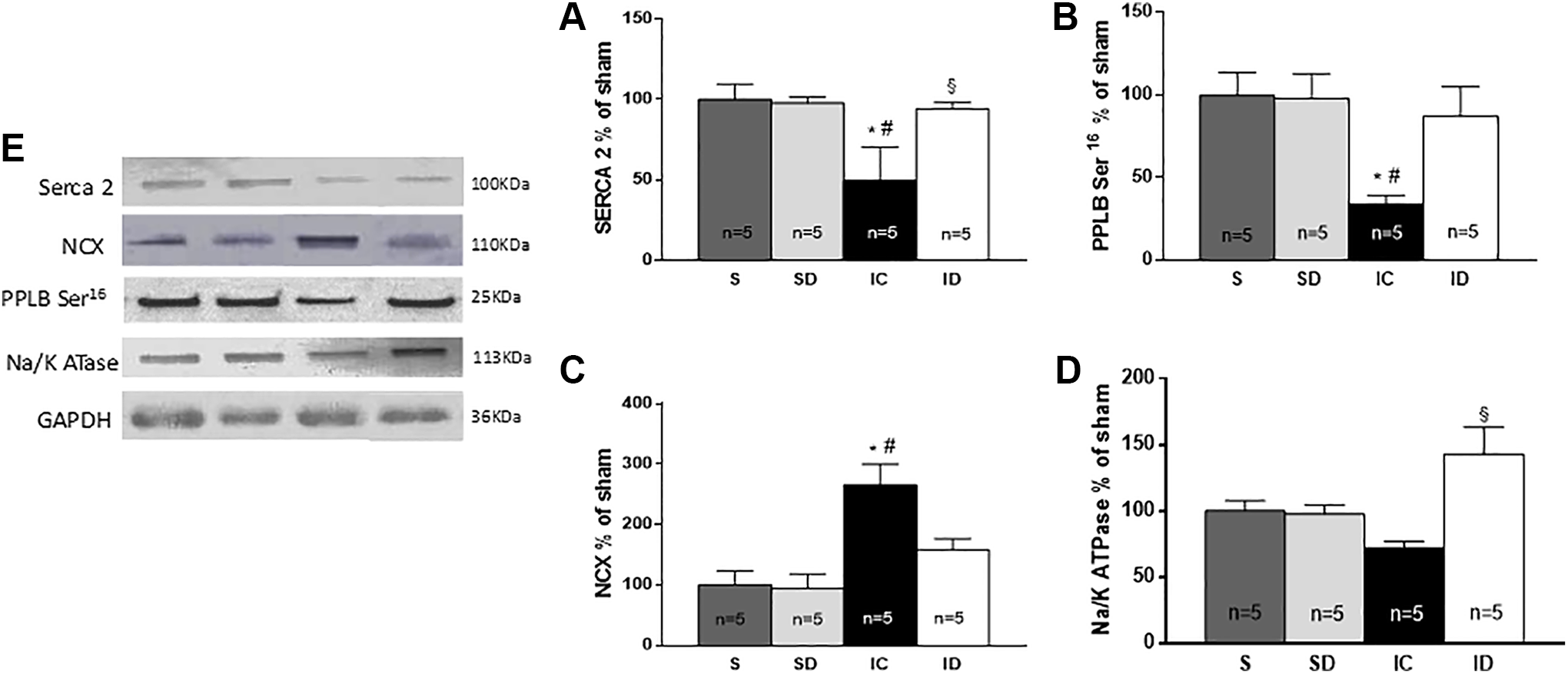
Quantification of proteins by Western blotting in percentage of sham in the myocardium, far from the infarction, in different groups. (A) SERCA 2, (B) phosphorylated phospholamban (Ser 16 ), (C) Na+/Ca2+ exchanger, and (D) Na+/K+-ATPase subunit α2, (E) Western blot of the proteins. *P < .05 versus S, #P < .05 versus SD, and §P < .05 versus IC. IC indicates infarcted control; S, sham operation; SD, sham operation + digitoxin.
Discussion
In recent years, the use of cardiac glycosides has declined, primarily because of the results of the DIG trial, which reported that digoxin treatment did not increase survival in patients with HF. 1 However, more recent analyses of the DIG trial data provided new interpretations of the results. Results of subgroup analysis showed that digoxin decreased the mortality rate in HF patients under certain conditions. Indeed, serum digoxin level was associated with survival. 18 -23 The mortality rate in patients with digoxin levels <0.9 ng/mL was significantly lower than that of untreated patients. 19,22 Conversely, increased mortality was observed in patients with serum digoxin levels >0.9 ng/mL. Our previous studies also showed increased survival in rats with post-MI congestive HF that were treated with digitoxin. 5,6
Our results suggested several mechanisms underlying the beneficial effects of digitoxin, which have not been previously described. We found that infarcted rats treated with digitoxin showed significant reductions in nuclear volume expansion and collagen accumulation, indicating attenuation of the myocardial remodeling. The functional effects of digitoxin included a reduction in LV end-diastolic pressure, RV systolic pressure, and lung water content, indicating an improvement in congestive HF. Particular interest for the left ventricular performance enhancement is the previously undescribed preservation of the length–tension relations in treated rats, that is to say, improvement of Frank-Starling mechanism, which were altered in the untreated infarcted rats.
These findings obtained from animal experiments help us understand the results of previous human clinical trials. They 22 -26 showed improvement in HF symptoms with administration of the cardiac glycoside digitoxin and a worsening of congestive symptoms without treatment. Taken together, our results indicate the beneficial effects of long-term cardiac glycoside use on myocardial remodeling and HF.
Our results showing the effects of MI on the levels of proteins involved in calcium kinetics are consistent with results of previous studies. 27 -32 The finding that digitoxin preserved myocardial levels of SERCA2, phosphorylated phospholamban, and the Na+/Ca2+ exchanger protein, and increased Na+/K+ ATPase levels appears to be novel and is consistent with the observed functional improvement, as assessed by echocardiography, hemodynamics, and myocardial mechanics analyses.
We must emphasize the importance of decreased myocardial remodeling in the infarcted rats treated with digitoxin. This beneficial effect of cardiac glycosides has already been described in other types of cardiac injury. 11 -13,32
In conclusion, our data indicate that long-term use of digitoxin in HF resulting from MI decreases myocardial hypertrophy in rats and significantly improves contractile function. These findings improve our understanding of the beneficial effects of glycosides in patients with congestive HF.
Clinical Implications
The post hoc analyses of the DIG trial results challenge the conclusion that cardiac glycosides do not increase survival rates of patients with HF. In previous studies, we found that digitoxin prolonged the survival of infarcted rats with HF (Helber et al, dos Santos et al). The findings of the present study clearly show that long-term use of digitoxin mitigates adverse myocardial remodeling in rats. Considering the difficulty of conducting a new clinical trial to test the effect of cardiac glycosides in humans, the results of animal studies may be useful to more accurately define the role of cardiac glycosides in the treatment of patients with HF. Since this work and our previous studies were conducted in female rats, it needs to be considered that a gender feature might contribute to our results.
Footnotes
Authors’ Note
The authors take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation.
Author Contributions
Camila T. Picollo made substantial contributions to the conception and design of the work; acquisition, analysis, and interpretation of data for the work; drafting the work; revising it critically for important intellectual content; final approval of the version to be published; and agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Alexandra A. dos Santos made substantial contributions to the acquisition, analysis, and interpretation of data for the work; revising it critically for important intellectual content; final approval of the version to be published; and agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Ednei L. Antonio made substantial contributions to the acquisition, analysis, and interpretation of data for the work; revising it critically for important intellectual content; final approval of the version to be published; and agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Jairo M. A. Silva made substantial contributions to the acquisition, analysis, and interpretation of data for the work; final approval of the version to be published; and agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Danilo S. Bocalini made substantial contributions to the acquisition, analysis, and interpretation of data for the work; final approval of the version to be published; and agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Andrey J. Serra made contribution to analysis and interpretation of data for the work; revising it critically for important intellectual content; final approval of the version to be published; and agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Silvia S. M. Ibara made substantial contributions to the acquisition, analysis, and interpretation of data for the work; revising it critically for important intellectual content; final approval of the version to be published; and agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Paulo J. F. Tucci made substantial contributions to the conception and design of the work; analysis and interpretation of data for the work; drafting the work; revising it critically for important intellectual content; final approval of the version to be published; and agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: FAPESP (09/54225-8), CNPq (478740/2011-5), Capes, Unifesp.