Abstract
Coronavirus disease 2019 (COVID-19) is associated with a high incidence of thromboembolic events, both venous and arterial. There are currently no specific clinical or laboratory markers to guide antithrombotic therapy for COVID-19 patients. Immature platelets represent a population of hyper-reactive platelets associated with arterial thrombotic events. This prospective study compared consecutive severe COVID-19 patients (n = 53, median age = 73 years) versus patients with sepsis from another origin (n = 41, median age = 69 years). Total platelet counts, immature platelet fraction (IPF) and immature platelet count (IPC) were determined by the Sysmex XN-3000 auto-analyzer on admission and at subsequent time-points. IPC levels three days after admission were significantly higher in the COVID-19 group compared to the sepsis group (13.4 × 109/ L [IQR 9.1–18.5] in the COVID-19 group vs 9 × 109/ L [5.5–14.7] in the sepsis group, P = 0.007). COVID-19 patients with respiratory disease show increased platelet turnover and reactivity, as seen in higher levels of immature platelet indices, especially IPC, compared to the sepsis control group. While these platelet indices remained high, CRP levels decreased, particularly in patients treated with tocilizumab. This reduction in CRP was not accompanied by any apparent clinical improvement. These findings suggest that immature platelets may serve as a biomarker for disease severity in COVID-19 patients and their CRP may not be a reliable marker for disease severity.
Keywords
Introduction
Coronavirus disease 2019 (COVID-19) became a pandemic with global effects on morbidity and mortality. 1 It can range from mild respiratory failure to multi-organ dysfunction, and can have a fatal outcome, like sepsis syndrome. 2 Severe COVID-19 is associated with increased risk of thromboembolic events, both venous and arterial. 3 Some of the thrombotic complications include venous thromboembolic events, stroke, acute limb ischemia and acute coronary syndromes.3,4 Platelets play a pivotal role in these processes, functioning not only as mediators of thrombosis but also as active participants in the immune response.5,6
Immature platelet fraction (IPF) represents the youngest platelets recently released from the bone marrow and emerges as a critical biomarker for evaluating real-time thrombopoiesis. 7 These platelets are larger, richer in RNA content, and more biochemically active than their mature counterparts. 8 The diagnostic value of IPF extends beyond COVID-19, demonstrating utility in conditions such as immune thrombocytopenic purpura (ITP), thrombotic thrombocytopenic purpura (TTP), sepsis, and bone marrow recovery after chemotherapy.8,9 Elevated IPF levels reflect compensatory bone marrow responses to peripheral platelet destruction or consumption, aiding in the differentiation of thrombocytopenic states caused by central versus peripheral mechanisms. 10
Recent studies have highlighted the prognostic value of immature platelet indices, such as IPF% and IPC, in predicting severe disease outcomes and increased mortality risks in both adult and pediatric COVID-19 patients.11,12 These indices have been associated with ICU admission, prolonged hospitalization, and mortality in adults, as well as disease severity in pediatric patients. This highlights the potential utility of immature platelets as a marker for disease progression and severity.11,12
COVID-19 is also characterized by immune dysregulation and hyperinflammation, with elevated interleukin-6 (IL-6) levels strongly associated with severe disease and mortality.13,14 IL-6 plays a central role in the acute-phase response, stimulating hepatic production of C-reactive protein (CRP).15,16 Tocilizumab, a recombinant humanized monoclonal antibody targeting the IL-6 receptor, has been introduced as a treatment for severe COVID-19, effectively reducing CRP levels. However, this reduction renders CRP less reliable as an inflammatory marker in patients receiving this therapy. 17 Thus, alternative biomarkers less influenced by IL-6 blockade are essential for accurately evaluating inflammation and guiding clinical management in these patients.
The goal of the current study is to assess platelet turnover and reactivity by measuring the immature platelet fraction (IPF) and other platelet indices (platelet count, mean platelet volume, and platelet distribution width) in severe COVID-19 patients admitted to the intensive care unit (ICU) in comparison to non-COVID-19 septic patients. IPF measures the percentage of immature platelets, which are newly produced and typically larger and more reactive than mature platelets. 11 Elevated levels of IPF indicate a higher rate of platelet production, which is associated with increased platelet reactivity and a greater risk of thrombotic events regardless of disease severity.11,18,19 This provides valuable information on platelet production and turnover, especially in conditions associated with increased thrombopoiesis, such as severe infections.18,19
Demographic, clinical and laboratory findings of patients following admission to the emergency room (ER).

CAD: Coronary Artery Disease; CHF: Chronic Heart Failure; CKD: Chronic Kidney Disease; CABG: Coronary Artery Bypass Surgery.
Methods
In this prospective cohort study, we compared consecutive patients with COVID-19 (study group) to a control group, consisting of non-COVID-19 septic patients (were all tested negative for COVID-19). The study group included consecutive patients admitted to the ICU at Assuta Ashdod Medical Center in Israel, from September 14, 2020 to February 19, 2021, with confirmed novel corona virus (SARS-CoV-2). Patients’ disease severity was defined according to the WHO criteria.
The control group consisted of patients with septic shock admitted to the ICU. Our COVID-19 patient cohort was selected based on disease severity, mild and moderate COVID-19 cases were excluded. For the sepsis group, we used the Sepsis-3 criteria, which define septic shock as a subset of sepsis characterized by underlying circulatory, cellular, and metabolic abnormalities, and clinically identified by the need for vasopressors to maintain a mean arterial pressure (MAP) ≥ 65 mm Hg and a serum lactate level >2 mmol/L despite adequate fluid resuscitation.
The study group was divided into two subgroups: COVID-19 patients who were treated with Tocilizumab (n = 11) and those that did not receive this medication (n = 42). The decision to administer Tocilizumab was based on clinical assessments of disease severity and progression, following institutional protocols aligned with WHO recommendations. It should be noted that Tocilizumab became part of the standard COVID-19 treatment protocol in Israel during later waves of the pandemic, and a portion of the study cohort includes patients from earlier waves in 2020, prior to its widespread use.
Diagnosis of COVID-19 was made by at least one positive real time RT-PCR assay, accompanied by respiratory symptoms and radiological findings on the chest x-ray. Blood samples for IPF assessment were drawn for both study groups on admission to the ICU, three days afterwards and one week post hospitalization. The control group included patients hospitalized at Assuta Ashdod Medical Center, Israel, with sepsis from another origin (not COVID-19). The study was approved by the investigational review board of the Assuta Ashdod Medical Center, Israel. All subjects provided written informed consent.
Blood samples were evaluated for immature platelet fraction (IPF) using an automated analyzer (Sysmex XN-3000, Sysmex America Inc. Mundelein, Illinois), which uses fluorescent dyes containing oxazine and ethylene glycol. This system discriminates between mature and immature platelets and reports the immature platelet fraction as percentage. IPC (immature platelet count) was calculated by multiplying IPF by total platelet count, representing the absolute count of immature platelets (×10⁹/ L). All groups’ IPF and IPC results are presented as baseline value and maximal value (from all blood samples taken).
Statistical Analysis
Categorical variables were described as frequency rates and percentages. Continuous variables were described using median, and interquartile range (IQR) values. Differences between the two groups (COVID-19 vs Control) according to continuous parameters were tested by Mann-Whitney test. For categorical parameters we used Fisher exact tests. P < 0.05 was considered significant. SPSS version 28 was used for statistical analysis.
Results
The study included 53 consecutive patients with confirmed COVID-19 (the study group) and 41 patients with sepsis from non-Covid origin (sepsis control group). The median age of the COVID-19 group was 73 years (IQR, 65–80; range, 32–87 years), and 34 (64.2%) were men. In the sepsis control group, median age was 69 years (IQR, 54–74); range 34–86); 24 (58.5%) patients were men. 25 (47.2%) and 18 (43.9%) patients had diabetes type 2 in the COVID-19 group and sepsis group, respectively. The median ICU length of stay was similar between the COVID-19 and sepsis groups (12 days [IQR 10-15] vs 13 days [IQR 7-23], P = 0.75). Two thromboembolic events (both cerebrovascular) were recorded in the COVID-19 group (3.77%), with no events in the sepsis group (P = 0.5). Mortality rates were comparable across the groups (COVID-19: 22.6% vs Sepsis: 21.9%, P = 1.0) [Table 1].
The IPF percentage on admission was similar in the COVID-19 group compared to the sepsis group (5.4% [IQR 3.6–8.3] in the COVID-19 group vs 5.2% [IQR 3.3–9.5] in the sepsis group, P = 0.98) [Figure 1]. The IPC levels on admission were also similar in the two groups (11.2 × 10⁹/ L [IQR 8–16.4] in the COVID group vs (10.8 × 10⁹/L [IQR 6.5–15.7] in the sepsis group, P = 0.68) [Figure 2]. Furthermore, the COVID-19 group showed no significant difference in platelet count upon ICU admission compared to the sepsis group (221 × 10⁹/ L [IQR 157.5–265] in the COVID group vs 225 × 10⁹/ L [IQR 147–297.5] in the sepsis group, P = 0.67) [Table 2].

Immature platelets fraction on admission (IPF1), three days after admission (IPF2) and a week after admission (IPF3) in COVID-19 group compared to sepsis group. Medians and 95% CI.

Immature platelets count in admission (IPC1), three days after admission (IPC2) and a week after admission (IPC3) in COVID-19 group compared to sepsis group. Medians and 95% CI.
Immature Platelets Fraction, Immature Platelets Count and Platelets Count in the COVID-19 and Sepsis Cohort Following Admission to the ICU.

p values were calculated using Mann-Whitney test.
At three days post-admission, the IPF percentage was comparable in the COVID-19 group and the sepsis group (5.5% [IQR 3.6–8.3] in the COVID-19 group vs 5.4% [IQR 3.4–10.3] in the sepsis group, P = 0.97) [Figure 1]. However, the IPC levels were significantly higher in the COVID-19 group compared to the sepsis group (13.4 × 10⁹/ L [IQR 9.1–18.5] in the COVID group vs 9 × 10⁹/ L [5.5–14.7] in the sepsis group, P = 0.007) [Figure 2]. Furthermore, platelet count three days post admission was also significantly higher in the COVID-19 group compared to the sepsis group (256 × 10⁹/ L [IQR 177–369] in the COVID group vs 179 × 10⁹/ L [128.5–229.5] in the sepsis group, P = 0.002) [Table 2].
Similar level of IPF% was observed among the COVID-19 and sepsis groups one week after admission (5.8% [IQR 3.1–8.5] in the COVID group vs 5.0% [IQR 3.8–9.3] in the sepsis group, P = 0.94) [Figure 1]. Additionally, the IPC a week after admission in the COVID-19 group was similar to the sepsis group (14.2 × 10⁹/ L [IQR 10.2–21.7] in the COVID-19 group vs 13.1 × 10⁹/ L [IQR 7.85–19.8] in the sepsis group, P = 0.11) [Figure 2]. The platelet count in the COVID-19 group was higher than that in the sepsis group, although this difference was not statistically significant (313 × 10⁹/ L [IQR 219.5–371] in the COVID group vs 261 × 10⁹/ L [IQR 169–338.5] in the sepsis group, P = 0.058) [Table 2].
Upon admission, CRP levels did not differ significantly between the COVID-19 and sepsis groups (90.05 mg/L [IQR 42.1–151.3] vs 106.8 mg/L [IQR 48.4–232.1], respectively; P = 0.085). However, three days and a week after admission, the COVID-19 group had significantly lower CRP levels compared to the sepsis group (58.2 mg/L [IQR 22.5–103.9] vs 134.3 mg/L [IQR 49.3–223.8], P = 0.001 and 31 mg/L [IQR 10.9–89.1] vs 54.5 mg/L [IQR 34.05–144.2], P = 0.02, respectively) [Table 2].
In a mortality subgroup analysis, COVID-19 patients exhibited significantly higher IPF levels compared to sepsis patients at admission (7.1% [IQR 4.8-9.4] vs 3.8% [IQR 2.5-8.7], P = 0.0069) and at day 3 (8.7% [IQR 6.2-10.7] vs 5.7% [IQR 3.2-9.6], P = 0.0172). By day 7, IPF differences were not statistically significant (6.7% [IQR 3.7-10] vs 5.6% [IQR 4-10.3], P = 0.393) [Table 2].
Among the COVID-19 cohort, TCZ was administered to 11 patients. The percentage of IPF on admission was similar in the TCZ group and the non-TCZ group (n = 42), with no significant difference observed (5.2% [IQR 4.6–14.2] in the TCZ group vs 5.4% [IQR 3.5–8.2] in the non-TCZ group, P = 0.35). Similarly, the level of IPC on admission was similar in the TCZ group and the non-TCZ group (14.7 × 10⁹/ L [IQR 9.4–18.2] in the TCZ group vs 9.8 × 10⁹/ L [IQR 7.95–14.98] in the non-TCZ group, P = 0.11). Furthermore, there was no significant difference in platelet count on admission between the TCZ group and the non-TCZ group (218 × 10⁹/ L [IQR 153–262] in the TCZ group vs 225 × 10⁹/ L [IQR 172–272.3] in the non-TCZ group, P = 0.77) [Table 3].
Immature Platelets Fraction, Immature Platelets Count and Platelets Count in the Group That Received TCZ and the Group That Didn’t Receive This Medication Following Admission to the ICU.

p values were calculated using Mann-Whitney test.
Three days after admission, we observed no significant differences between patients who received TCZ and those who did not in IPF percentage (respectively 5.4% [IQR 4.2–12.3] vs 5.65% [IQR (3.6–7.8)], P = 0.28), IPC (respectively 14.4 × 10⁹/ L [7.2–19.9] vs 13.3 × 10⁹/ L [IQR 9.3–17.0], P = 0.81), or platelet count (respectively 199 × 10⁹/ L [IQR 129–343] vs 288 × 10⁹/ L [IQR 186–371], P = 0.15).
Similarly, a week after admission, the TCZ treated and non-treated had similar IPF percentage and IPC. Specifically, the TCZ group had an IPF percentage of 6.4% [IQR 3.4–10.8] compared to 5.65% [IQR 2.93–8.1] in the non-TCZ group (P = 0.28), and an IPC of 13.4 × 10⁹/L [IQR 8.8–19.4] compared to 14.6 × 10⁹/L [IQR 10.5–21.9] in the non-TCZ group (P = 0.42). However, the platelet count was significantly lower in the group that received TCZ compared to the non-TCZ group 210 × 10⁹/L [IQR 180–359] compared to 325.5 × 10⁹/L [IQR 262.8–373.8] in the non-TCZ group (P = 0.041) [Table 3].
The CRP measurements were taken on admission, three days after admission, and a week after admission for both the TCZ and non-TCZ groups. The TCZ group had significantly lower CRP levels compared to the non-TCZ group across all three time points. On admission, the TCZ group had significantly lower CRP levels compared to the group that did not receive TCZ (32.3 mg/L [28.2–87.7] vs 95.9 mg/L [IQR 69.9–165.4], P = 0.016). Similarly, at three days after admission, the TCZ group had significantly lower CRP levels compared to the group that did not receive TCZ (21.0 mg/L [IQR 12.4–49.9] vs 78.5 mg/L [IQR 36.1–119.9], p < 0.001). Furthermore, a week after admission, the TCZ group had significantly lower CRP levels compared to the group that did not receive TCZ (14.5 mg/L [IQR 2.3–20.9] vs 45.0 mg/L [IQR 15.3–126.7], p = 0.004) [Table 3, Figure 3].
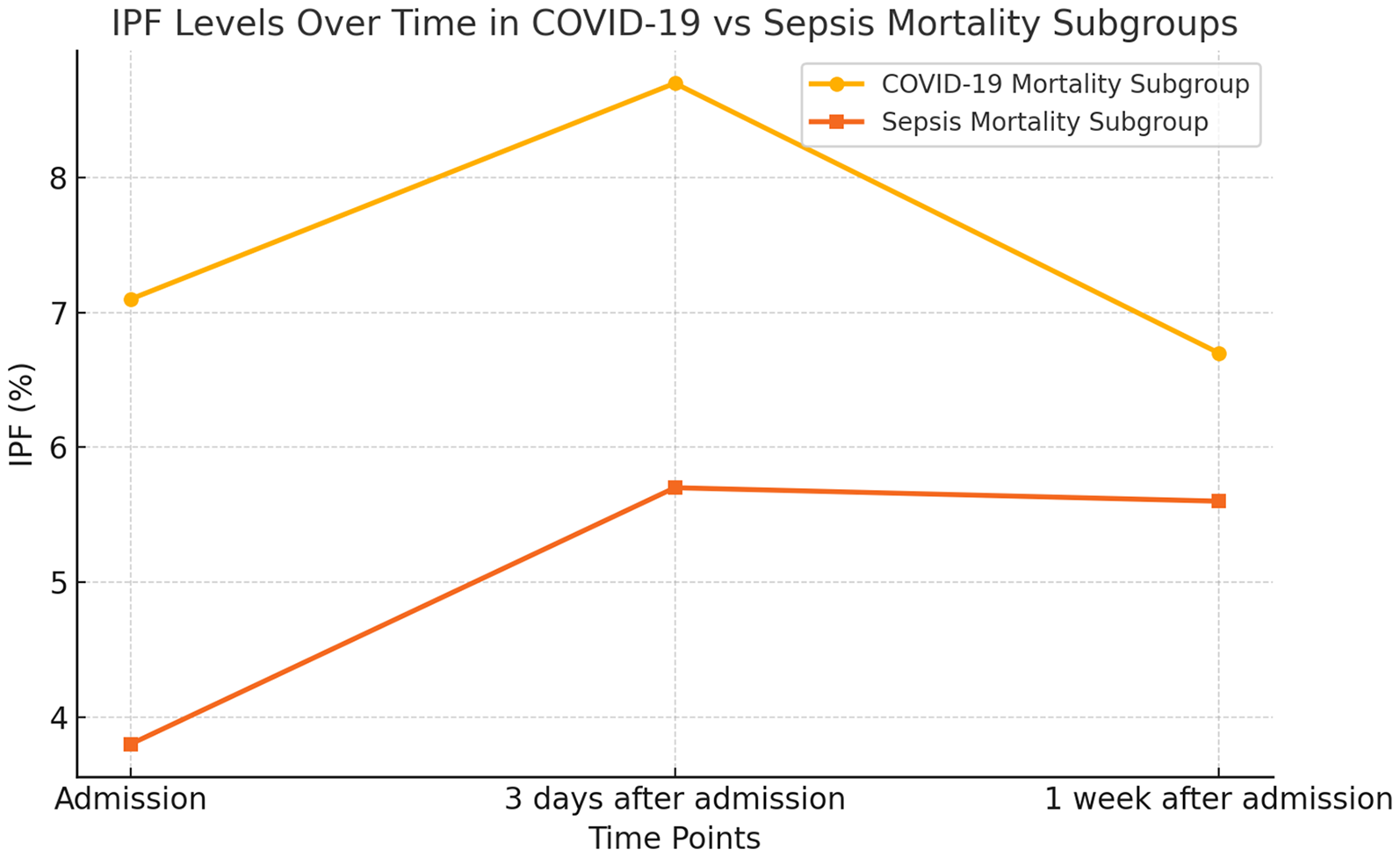
Immature platelet fraction (IPF) at admission, three days after admission, and one week after admission in the COVID-19 mortality subgroup compared to the sepsis mortality subgroup. The graph displays median IPF values for both groups at each time point.
Discussion
The current study assessed the role of immature platelet indices (IPF and IPC) in severe COVID-19 patients compared to non-COVID-19 septic patients, focusing on their potential association with disease severity and thrombotic risk. COVID-19 is characterized by a prothrombotic state driven by systemic inflammation, endothelial dysfunction, and platelet activation. 19 Elevated immature platelet indices have been linked to increased platelet turnover and worse clinical outcomes in other hypercoagulable states, such as septic shock. 20
In our study, IPC levels were higher in the COVID-19 group three days after admission compared to the sepsis group. Previous studies have highlighted similar trends, showing that elevated IPC values are associated with severe outcomes such as ICU admission, disease severity, increased length of hospital stay and mechanical ventilation requirements.11,12 This may reflect a heightened thrombopoietic response during the acute inflammatory phase, consistent with the pro-inflammatory and pro-thrombotic nature of severe COVID-19. Elevated IPC may represent increased platelet production in response to systemic inflammation and platelet consumption, processes frequently observed in hypercoagulable states. 21 Despite statistically higher platelet counts in the COVID-19 group three days post-admission, this difference is unlikely to be clinically relevant, as both values remained within the normal range.
Significantly higher IPF levels at admission and three days post-admission in the COVID-19 group were observed in our mortality subgroup analysis. These findings suggest that immature platelet indices may serve as markers of early disease severity and adverse outcomes in COVID-19 patients. This conclusion is consistent with a recent study that was able to demonstrate that IPF% may be predictive of disease severity measured by need for ICU admission or ventilator use. 11
Tocilizumab (TCZ), an IL-6 receptor antagonist, effectively reduced CRP levels in our cohort without corresponding clinical improvement. However, IPF, IPC, and platelet levels remained elevated, highlighting the potential of IPF as a reliable biomarker for inflammation and thrombopoiesis even when traditional markers are modulated by targeted therapies.
The management of severe COVID-19 is further complicated by thrombocytopenia, coagulopathy, and an elevated risk of venous and arterial thrombotic events. Current guidelines recommend pharmacological VTE prophylaxis in all hospitalized COVID-19 patients unless contraindicated. 21 However, the role of antiplatelet therapy remains uncertain. Immature platelet indices could help refine risk stratification, enabling tailored antithrombotic strategies.
The current study have several limitations. The relatively small sample size may have limited the statistical power to detect significant differences in clinical outcomes such as thrombotic events and mortality. In our study, only two thromboembolic events occurred in the COVID-19 group. Both events were cerebrovascular in nature. The first event occurred during the acute hospitalization phase, while the second was documented within the first 30 days post-discharge, and there were no significant differences in mortality between the groups. Furthermore, baseline differences in age, hemoglobin, and WBC counts between the COVID-19 and sepsis groups represent potential confounding factors, though they are unlikely to have influenced the primary findings.
In conclusion, patients with severe COVID-19 exhibit enhanced platelet turnover, as reflected by elevated IPF and IPC levels. These indices may serve as early biomarkers for disease severity and thrombotic risk, improving risk stratification and potentially guiding antithrombotic management. Further studies are needed to confirm these findings, explore correlations with coagulation markers like fibrinogen or D-dimer, and validate their clinical utility in guiding management decisions.
Footnotes
Acknowledgements
None.
Author Contributions
YB, TM and AM prepared the manuscript, including tables and figures, in consultation with EL and AC and assisted by EA. MC and IB performed all laboratory measurements.
AC collected the clinical data and YB collected the control group data. All authors have read and approved the final version of the manuscript.
Data Availability
The data that support the findings of this study are available on request from the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the investigational review board of the Assuta Ashdod Medical Center, Israel. All subjects provided written informed consent.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
