Abstract
Keywords
Introduction
Platelets play a crucial role in the thrombotic processes within the ruptured plaques in coronary arteries, being responsible for one of the main mechanisms of myocardial infarction (MI).1,2 Reticulated platelets (RPs) are immature platelets, newly released to the circulation, that are usually larger than the mature ones. 3 As they contain mRNA and rough endoplasmic reticulum, they can synthesize proteins; as such their functional potential has been recognized to be more important than that of mature platelets, which would explain their greater reactivity or capacity to aggregate and initiate blood clotting.4,5 Immature platelet fraction (IPF) is one of the possible indices to describe the level of RPs.6–8 Calculated as the ratio of RPs to the total platelet count, IPF can serve as a marker for platelet production and turnover.6–8 With the increased adoption of automated hematology analyzers, it has now become possible to assess IPF levels quickly and inexpensively as part of an extended blood count test, which is a significant advantage enabling IPF measurement to be used as a potential screening test. 9
Several studies have suggested that RPs can be a good indicator of the effectiveness of dual antiplatelet therapy (DAPT): higher levels of RPs have been associated with an increased risk of thrombotic events whereas lower levels of RPs with better platelet inhibition.10–12 It was also demonstrated that RPs were increased in ST-segment elevation myocardial infarction (STEMI) when compared with healthy controls as well as with other types of acute coronary syndrome (ACS).13,14 To date, however, the long-term prognostic value of RPs or IPF quantification among patients presenting with ACS has not been elucidated.
The aim of the presented study was to evaluate the effectiveness of IPF levels in predicting long-term clinical outcomes in patients with ACS.
Methods
This was a prospective, single-center study conducted in the 1st Chair and Department of Cardiology, Medical University of Warsaw. Written informed consent was obtained from each participant included in the study. This study was conducted according to the principles outlined in the Declaration of Helsinki. Collection and management of data was approved by the local medical ethics committee. The clinical trial was registered with ClinicalTrials.gov under the identifier NCT06177587.
Study Population
A total of 140 patients were recruited between July 2017 and May 2018. The inclusion criteria were age > 18 years, diagnosis of ACS, treatment with antiplatelet therapy and the ability to sign an informed consent. Patients were excluded from the study if their life expectancy was shorter than 1 year or if they received GPIIbIIIa antagonists in order to isolate the impact of DAPT on patient outcomes depending on IPF. All the patients were treated with 75 mg of acetylsalicylic acid (ASA) daily and either clopidogrel (75 mg once a day) or ticagrelor (90 mg twice a day). Patients undergoing percutaneous coronary intervention (PCI) received 300 mg of ASA and the loading dose of P2Y12 inhibitor (either 300 mg of clopidogrel or 180 mg of ticagrelor) periprocedurally. Patients diagnosed with ACS were recruited after coronary angiography and, if applicable, a successful PCI. Consecutive patients who met all inclusion criteria were enrolled.
Blood Tests
Blood sampling was obtained from peripheral vein in the first 24 h of hospitalization after coronary angiography and, if performed, PCI. In cases where multiple IPF value measurements were accessible from a single patient, the initial available value was selected. Platelet count, hemoglobin, white blood cell count, platelet distribution width, mean platelet volume (MPV) and IPF were assessed in whole blood anticoagulated with ethylenediaminetetraacetic (K3EDTA) using an automated hematological analyzer (Sysmex XN 2000, Kope, Japan).
Follow-up
The primary endpoint was the occurrence of major adverse cardiovascular events (MACE) defined as all-cause mortality, MI, ischemic stroke, or unplanned revascularization. Data was also collected on the cause of death. Cardiovascular (CV) death was defined as sudden cardiac death including unwitnessed death with no conclusive evidence of other than CV cause, fatal MI, death due to heart failure or cardiogenic shock, fatal stroke or other cerebrovascular event, fatal dysrhythmias, pulmonary embolism or other vascular causes. MI was defined according to the Fourth Universal Definition of Myocardial Infarction. 15 The diagnosis of ischemic stroke had to be based on an imaging study (CT scan or MRI) by an experienced neurologist or radiologist. Unplanned revascularization was defined as ischemic-driven urgent hospitalization and PCI or coronary artery bypass grafting due to ischemic symptoms not meeting criteria for MI. If a patient presented with MI and required PCI, the primary endpoint was classified as MI. In case more than one event occurred, the date of the first one was considered. Patients were contacted in December 2022. The researchers who collected follow-up were blinded to all laboratory tests including IPF. Based on the source documentation including discharge letters or electronic records, 2 physicians independently adjudicated an outcome. Mortality was verified via the public registries. Patients or their families if contact with the patient was not possible were contacted via phone to confirm whether they were hospitalized after the index hospitalization for ACS.
Statistical Analysis
The statistical analysis was performed using SPSS Statistics version 28.0 (IBM SPSS, Inc.). Patients were divided into tertiles according to their IPF levels. The normality of distribution was assessed with Shapiro–Wilk test. Normally distributed variables are presented as mean and standard deviation and compared using analysis of variance. The non-parametric data is presented as median and interquartile range (IQR) and compared with Kruskal–Wallis test. Analyses were stratified according to IPF tertiles. Time-dependent receiver-operating characteristic (ROC) curve analysis was used to determine the IPF cutoff point that may have predictive value for MACE. Cox regression analysis was performed to assess the association between IPF tertile and MACE. Multivariable Cox regression was performed for IPF treated as a continuous value, as well as for the factors identified with P < .1 at univariate analyses. P-values < .05 were considered statistically significant.
Results
A total of 140 patients were enrolled in the study. Ten patients were lost to follow-up despite at least 3 attempts to contact them or their families. The remaining 130 patients (92.9%) were followed up for a median time of 57 months [IQR 55-59 months].
Baseline characteristics by IPF tertiles are presented in Tables 1 and 2. Overall, women constituted almost 30% of all the patients. In the 1st tertile there were 39% of female patients, whereas in the 3rd tertile 14% (P = .029). MI prior to the index hospitalization and smoking were more common among patients in the upper tertile. Apart from these differences, only platelet assay parameters (platelet count P = .002, mean platelet volume P < .001, platelet distribution width P < .001, platelet large cell ratio P < .001) differed significantly between groups. All patients received DAPT, of which 108 (77.1%) were given clopidogrel and 32 (22.9%) ticagrelor. The recruited patients were not using glycoprotein IIb/IIIa inhibitors.
Baseline Characteristics.

Bold values denote statistical significance at the P < .05 level.
Abbreviations: ACEI/ARB, angiotensis converting enzyme inhibitors/angiotensin receptor blockers; BB, beta-blockers; CABG, coronary artery by-pass graft; CCB, calcium channel blockers; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; Cx, the circumflex artery; Dg, diagonal branches of the left anterior descending artery; DM, diabetes mellitus; EF, ejection fraction; HF, heart failure; HL, hyperlipidemia; HT, hypertension; IQR, interquartile range; LAD, the left anterior descending artery; LM, the left main artery; Mg, marginal branches of the circumflex artery; MI, myocardial infarction; MVD, multi-vessel disease; n, number; NSTEMI, non-ST-segment elevation myocardial infarction; PCI, percutaneous coronary intervention; PPI, proton pump inhibitors; RCA, the right coronary artery; SD, standard deviation; STEMI, ST-segment elevation myocardial infarction, UA, unstable angina.
Laboratory Characteristics.
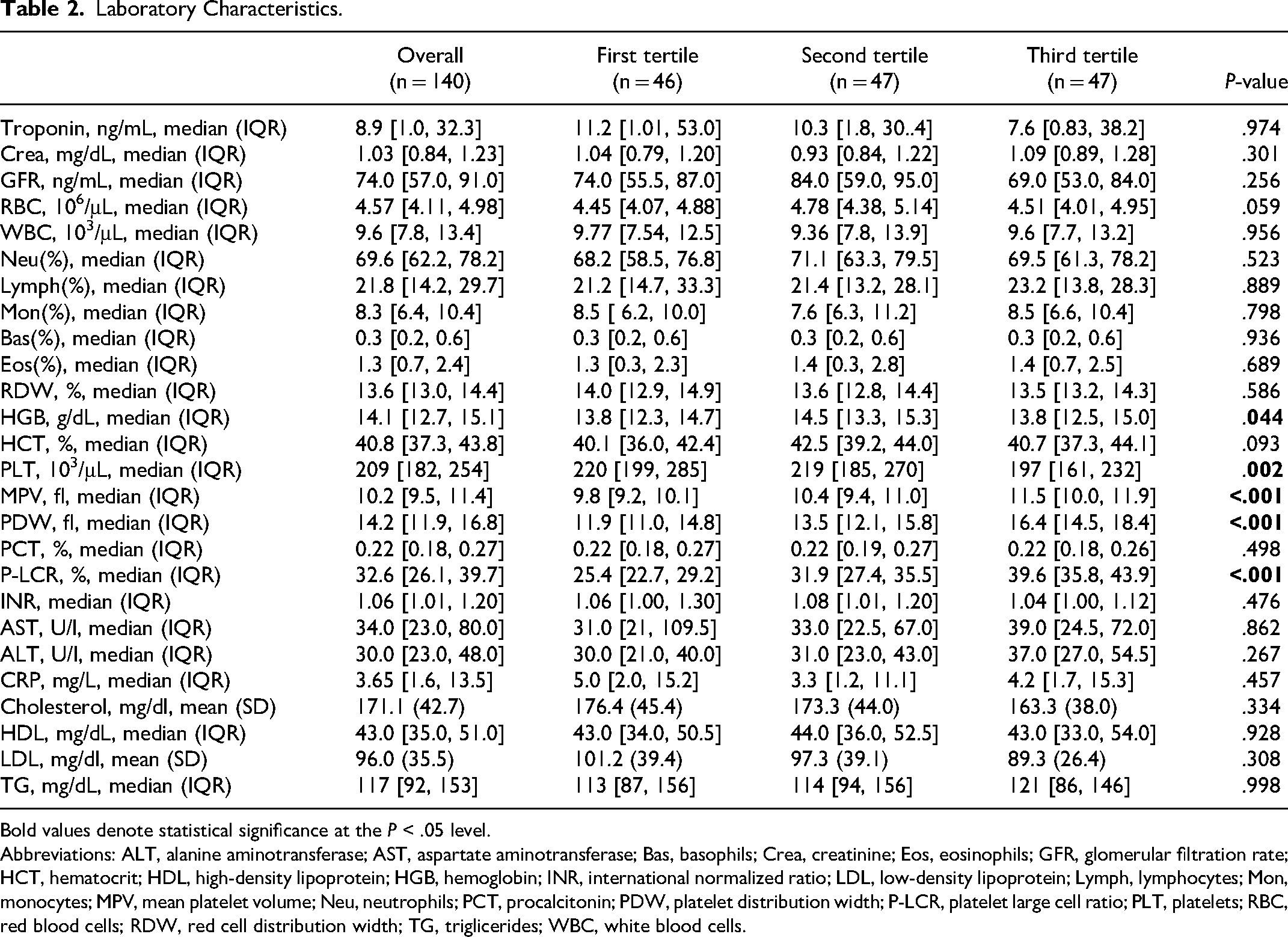
Bold values denote statistical significance at the P < .05 level.
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; Bas, basophils; Crea, creatinine; Eos, eosinophils; GFR, glomerular filtration rate; HCT, hematocrit; HDL, high-density lipoprotein; HGB, hemoglobin; INR, international normalized ratio; LDL, low-density lipoprotein; Lymph, lymphocytes; Mon, monocytes; MPV, mean platelet volume; Neu, neutrophils; PCT, procalcitonin; PDW, platelet distribution width; P-LCR, platelet large cell ratio; PLT, platelets; RBC, red blood cells; RDW, red cell distribution width; TG, triglicerides; WBC, white blood cells.
All patients were diagnosed with ACS. Among them, 78 (55.7%) presented with STEMI and 62 (44.3%) with NSTEMI (non-ST-segment elevation myocardial infarction) or UA (unstable angina). 126 (90%) patients had PCI performed. Overall MACE occurred in 27 patients (20.8%). Patients in the highest tertile had 1.7 times higher relative risk of MACE than patients in the middle tertile, and 4.9 times higher than patients in the lowest tertile. Follow-up events in each IPF tertile are presented in Table 3. The main cause of death was CV mortality and it occurred in 14 patients (93.3%).
Follow-up.

Abbreviations: IPF, immature platelet fraction; MI, myocardial infarction.
Cox proportional hazard model for MACE revealed a significant difference in the occurrence of MACE in the highest compared to the lowest tertile (HR = 5.341 95% CI: 1.546-18.454, P = .008). The risk of MACE was also higher in patients in the middle tertile when compared to the lowest tertile, but it did not reach statistical significance (HR = 3.395 95% CI: 0.919-12.543, P = .067). The survival Kaplan–Meier curve for each tertile is presented in Figure 1. Time-dependent ROC curve analysis revealed AUC of 0.656 for 5-year outcome with an IPF cut point of 3.45% being 63.0% sensitive and 65.0% specific for MACE (Figure 2).
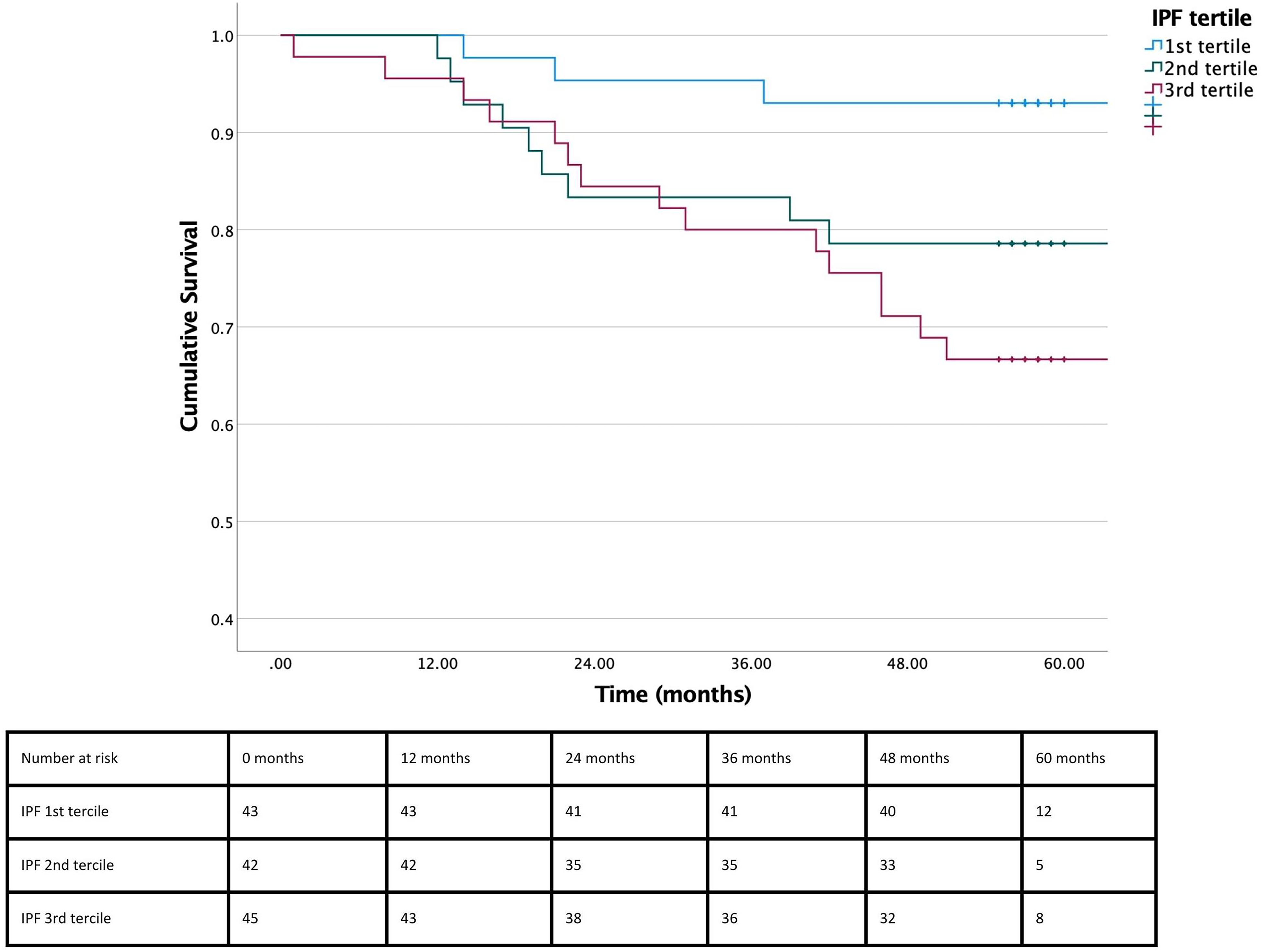
Kaplan–Meier event free survival curve with follow-up duration of 5 years after acute coronary syndrome. Blue, green, and red lines represent 1st, 2nd, and 3rd immature platelet fraction (IPF) tertiles, respectively. The numbers in the table below the graph represent the number of patients at risk at 0, 12, 24, 36, 48, and 60 months. Log rank P = .013.

Receiver-operating characteristic curve for prediction of major adverse cardiovascular events based on the immature platelet fraction (IPF) levels.
Multivariable Cox regression analyses showed that baseline IPF level was independently associated with MACE (HR = 1.279, 95% CI: 1.056-1.549, P = .012). Other tested variables did not reach statistical significance. Results are presented in Table 4.
Multivariable Cox Regression.

Bold values denote statistical significance at the P < .05 level.
Abbreviations: CI95%, 95% confidence interval; IPF, immature platelet fraction; MI, myocardial infarction; RBC, red blood cells.
In order to evaluate the importance of particular drugs, the analysis inside subgroups receiving various P2Y12 inhibitors was also designed. Similarly to the overall study population, Cox regression analysis further confirmed a significantly higher risk of MACE in patients in the highest versus lowest IPF tertile in clopidogrel-treated cohort (HR 6.25 CI: 1.4-27.7, P = .016), while no statistical significance was achieved in the ticagrelor-treated group despite the numerically higher risk (HR 3.54 CI: 0.37-34.03, P = .274).
Discussion
To the best knowledge of the authors, the presented study was the first study with such a long-term follow-up to assess the relationship between IPF level and the risk of MACE in patients who presented with ACS and were treated with DAPT. We demonstrated that the higher IPF values were associated with significantly increased occurrence of MACE, corroborating the preliminary findings of prior reports with shorter observation.
Platelets are known to contribute to the complex and multifactorial pathomechanism of CV events.16,17 Their younger fraction, namely RPs, may reflect underlying disturbances in the process of platelet production, maturation and clearance, which can lead to an increased risk of thrombosis.18,19 The hyperreactivity of RPs could be one of the possible explanations for the increased risk of CV events in patients with higher IPF values. In research involving patients with ACS receiving DAPT (aspirin and clopidogrel), it was found that individuals with a high platelet turnover, indicated by a larger number of RPs, experienced a significant reduction in the effectiveness of the antiplatelet treatment. 6 The findings were confirmed in different patients’ groups, also treated with prasugrel. 12 One may suggest that the impaired response to DAPT associated with increased baseline IPF may be one of the factors underlying the increased risk of subsequent CV events.
It is presumed that RPs may affect ADP-induced platelet aggregation in patients on therapy with thienopyridines. Bongiovanni et al investigated the existence of an association of high IPF with high MACE risk in patients treated with either prasugrel or ticagrelor. In addition, the association of IPF with ADP-induced platelet aggregation was compared. The risk of MACE was higher in patients with elevated IPF values regardless of the drug used, yet the association between ADP-induced platelet aggregation and IPF existed only in patients treated with prasugrel. 20 Another important feature of RPs is their ability to produce cyclooxygenase 2 (COX-2). Increased COX2 makes low doses of ASA insufficient to inhibit thromboxane production, and thus the risk of thrombotic events is reportedly higher despite proper intake of the medications. 21 The phenomena outlined above seem to partially explain our findings, however, further research is needed to fully understand the potential mechanism of RPs in predicting adverse CV events.
We observed that the risk of MACE in patients with IPF ≥ 3.7% was 4.9 times higher as compared with the group in the lowest tertile of IPF, that is, < 2.2%. Our finding is consistent with previously conducted research with shorter follow-up time. It was first shown in 2013 by Cesari et al that RPs can predict CV death in ACS patients in 1-year follow-up with optimal cutoff point of 3.3%. This is similar to the value obtained in our study (3.45%). However, both the previous and our study showed unsatisfactory sensitivity (63.6% and 63.0%, respectively) and specificity (61.8% and 65.0%, respectively). 22 Another study by Freynhofer et al performed on a greater number of patients undergoing PCI also showed association between IPF level and the risk of MACE with the cutoff point at 3.35% in 6-month follow-up. 23 The other parameters dependent form the number of RPs were also identified as predictors of MACE including immature platelet count (IPC) 24 and mean platelet volume (MPV). 23
The studies presented above differ from ours in terms of early mortality or MACE rate. Cesari et al showed that there was a significant difference in survival rate early after the incident, which was not observed in our population. However, it should be pointed out that the mortality rate in the first year was generally higher in that study. It may be due to multiple factors, the most important of which we find much higher mean age of patients included in the previous research, that is, 11 years difference in overall population. Moreover, in the group with CV death the mean age was 87 years, being significantly different than in no CV death group and over 20 years higher than in our population. Furthermore, in our study, a higher percentage of patients received optimal therapy including ACE-inhibitor and statin (92.9% and 97.9%, respectively, in our study vs 26.2% and 20.5% in the previously described). Ibrahim et al on the other hand presented similar results for the 1-month MACE, yet the difference between low and high IPC groups also occurred earlier. However, the inclusion criteria in that study were more liberal including both ACS and CCS patients. Therefore, the treatment differed, meaning only 33% of patients were treated with stent implantation. Also, the median baseline IPF value in that study was higher than in ours (4.3% vs 2.85%, respectively). That is consistent with the findings of IPF's predictive value. Knowledge regarding the relationship between RP levels and early complications of ACS is sparse. Previous works evaluated the association between IPF and MACE over various observation periods, from 6 to about 30 months.20,22–24 However, studies assessing the early risk of complications after ACS in relation to MACE are lacking. Studies also suggest that the risk of CV complications after cardiac surgery is higher for higher IPF values. 25 Therefore, the aspect of the relationship between IPF and early complications is a gap in knowledge that should be addressed.
CAD is the most common type of heart disease, causing hundreds of millions of deaths each year. 26 As the response to antiplatelet therapy is not identical, it is believed that there should be a personalization of treatment including the choice of drug, the duration of treatment and the dosing. Some evidence exists that identifying patients with higher bleeding or ischemic risk among patients treated with antiplatelet therapy using genetic testing or advanced methods determining platelet activity was beneficial, 27 though it is not recommended in routine clinical practice, partially considering its costs. Taking into account the lack of effective and accessible methods to select patients requiring intensified treatment, IPF appears to be a potentially useful marker to identify the individuals at an increased risk of ischemic events. Further research powered for clinical outcomes in that field is warranted.
The presented study should be viewed at in light of the following limitations. First, the method of collecting follow-up by interviewing patients or their families by telephone calls carries the risk of bias. However, we made every effort to verify the information obtained as far as possible by confirming death records in a public database and reviewing information on all hospitalizations due to MACE in the source documentation. Second, despite the accurate and reliable collection of patient medical history, the existence of unidentified confounders cannot be excluded. Moreover, the ratio of conservatively treated patients compared to the invasively managed group does not allow a comparison of the predictive value of IPF in the 2 groups and would require the collection of a larger cohort. The low event rate in the first few months could also be viewed as a limitation, not providing the insights into the impact of IPF on early mortality and MACE. Finally, the population was relatively small. Further studies on a larger cohort are warranted to confirm presented findings. Future research should focus on understanding of potential value of therapy intensification among patients identified with higher IPF values. In addition, studies to date have primarily assessed baseline IPF values, while it may be necessary to assess the variability of this parameter over time and in relation to the treatment used in different patients’ groups.
Conclusions
IPF may be an independent predictor of long-term mortality and MACE in patients with ACS treated with DAPT. Its accessibility and ease of assessment by flow cytometry may facilitate its more common use in CV risk assessment in this group of patients, provided its accuracy is proven on a larger population.
Footnotes
Author Contributions
All authors contributed to the study conception and design. M.T., K.G., G.S., and A.W. contributed to methodology; M.T. G.S., and D.K.T contributed to validation; A.B. and K.G. contributed to formal analysis; K.G., P.B., J.Z., and A.P. contributed to investigation and data collection; K.G., A.B., J.Z., and M.T. contributed to writing the original draft preparation; K.G., G.S., D.K.T., M.G., J.K., and M.T. contributed to writing the review and editing; K.G., A.B., and J.Z. contributed to visualization; M.T., J.K., and M.G. contributed to supervision. All authors edited and approved the final version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
This study was conducted according to the principles outlined in the Declaration of Helsinki. Collection and management of data was approved by the Ethics Committee of Medical University of Warsaw.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
