Abstract
Limited available evidence comparing DOACs with warfarin suggests efficacy and safety of DOACs for CVT. We aimed to evaluate whether a specific DOAC is preferred for the treatment of CVT. This retrospective cohort study included adult patients with CVTs between September 2018 and September 2022 treated with a DOAC. The primary outcome was rate of partial or complete recanalization. Secondary outcomes included rate of recurrent VTE, CVT extension, major or clinically relevant non-major bleeding, and death within 180 days after DOAC initiation. Of 31 patients with CVT, 21 received apixaban, 7 received rivaroxaban, and 3 received dabigatran. Among those with repeat imaging, the primary composite outcome occurred in 100%, 80%, and 100% for each group, respectively (P = 0.34). One patient had extension of CVT while on apixaban and one patient had increased midline shift while on rivaroxaban. No other secondary outcomes were observed. There do not appear to be significant efficacy or safety differences between DOACs when used for CVT treatment, though larger studies are needed to validate these findings.
Introduction
Cerebral venous thrombosis (CVT) is a rare form of stroke that is associated with the thrombosis of the dural sinus and/or cerebral veins, affecting approximately 5 people per million annually and representing about 0.5-1% of all stroke cases. 1 CVT occurs more frequently in young individuals (age less than 50 years).1,2
For the treatment of venous thromboembolism (VTE), guidelines3–7 support the use of direct oral anticoagulants (DOACs) as the preferred agents over warfarin, but recommendations for DOACs in CVT treatment are less clear. The 2017 European Stroke Organization guideline recommended against the use of DOACs in CVT treatment due to the low quality of evidence from two observational case series. 8 The 2021 American College of Chest Physicians and 2021 European Society for Vascular Surgery VTE guidelines do not mention use of DOACs for CVT treatment and recommend parenteral anticoagulation such as low molecular weight heparin (LMWH) and unfractionated heparin or warfarin.6,7
Available evidence supporting DOAC use in CVT is limited. Cases-series suggested that DOACs may be effective and safe options for treating CVT.9,10 Observational studies evaluating CVT treatment with rivaroxaban or apixaban compared with warfarin or LMWH have found similar efficacy without increased safety risks.11–13 A randomized controlled trial (RCT) 14 and a large multicentered international observational study 15 demonstrated similar efficacy and similar or improved safety with DOACs versus warfarin among patients with CVT. Meta-analyses have further supported the use of DOACs over warfarin or LMWH for CVT treatment as results illustrated that DOACs are similarly efficacious from the standpoint of recanalization rate or modified Rankin score and similarly safe in terms of rates of recurrent VTE, all-cause mortality, or major and minor hemorrhage.16,17
Large RCTs are challenging to conduct due to the low incidence of CVT and the available evidence lacks comparison between DOACs. In this study, we aimed to determine whether a specific DOAC is preferable for the treatment of CVT.
Study Design and Methods
This retrospective cohort study was approved by the Loma Linda University Institutional Review Board (#5220336). Adults at least 18 years of age with a CVT diagnosis during their hospital admission at our large academic medical center who received DOAC treatment between September 2018 and September 2022 were screened for inclusion. The original data pull identified CVT via ICD-9 codes (325.0, 437.6, and 671.5), ICD-10 codes (G08.X, I63.6, I67.6, O22.5, and O87.3), and CVT diagnosis documented within medical progress notes. Patients with antiphospholipid syndrome, a mechanical heart valve, and severe renal impairment (creatinine clearance below 30 mL/min) were excluded.
The primary composite outcome was the rate of partial and complete recanalization on magnetic resonance imaging (MRI). 15 Recanalization status on latest MRI study was abstracted from radiology reports and was classified as no recanalization, partial recanalization, or complete recanalization. Complete recanalization was defined as full recanalization of the thrombosed vein or sinus without any residual thrombus. Partial recanalization was defined as improved opacification or flow in the affected cerebral sinus or vein, but with residual thrombus present on follow-up imaging. No recanalization was defined as no change or worsening in opacification or flow in the affected cerebral sinus or vein from baseline imaging. Secondary outcomes included individual rates of partial and complete recanalization, recurrent VTE, extension of CVT, major and clinically relevant non-major bleed (as defined by the International Society of Thrombosis and Haemostasis 18 ), and death within 180 days of DOAC initiation. Major bleeding was defined as having a symptomatic presentation and (1) fatal bleeding; (2) bleeding in a critical area or organ; and/or (3) bleeding causing a fall in hemoglobin level of at least 2 g/dL or leading to transfusion of at least two units of whole blood or red cells. 18 Clinically relevant non-major bleeding was defined as any sign or symptom of hemorrhage that did not fit the criteria for major bleeding but met at least one of the following criteria: (1) requiring mechanical intervention by a healthcare professional; (2) leading to hospitalization or increase level of care; and (3) prompting a face-to-face evaluation. 18
Descriptive statistics were employed with nonparametric continuous data presented as median with interquartile range and categorical data reported as number with percentage. Nonparametric variables were compared using the Kruskal-Wallis test, and categorical variables were compared with Fisher's exact test. P < 0.05 was considered statistically significant.
To address missing data in cases without repeat imaging, we used a conservative imputation approach whereby the absence of recanalization data was categorized as no recanalization. This worst case scenario assumption was used to avoid overestimating recanalization rates. A subgroup analysis of the primary outcome was performed for only those with a follow-up MRI to evaluate recanalization rates among a more reliable subset. A univariate logistic regression analysis was used to determine the effect of full DOAC loading dose (LD), parenteral anticoagulation prior to DOAC initiation, and duration of parenteral anticoagulation on the primary outcome. The data were analyzed using SAS version 9.4 (Cary, NC).
Results
A total of 31 patients were included: 21 (68%) received apixaban, 7 (22%) received rivaroxaban, and 3 (10%) received dabigatran (Figure 1). Baseline characteristics were similar between groups (Table 1) with a median age of 44 years, 71% of the patients being female, and most identifying as White (55%) or Hispanic (32%). No patients received concurrent P2Y12 inhibitors, only 3 patients (all in the apixaban group) received concomitant aspirin, and one patient in the apixaban group also received a non-steroidal anti-inflammatory drug.
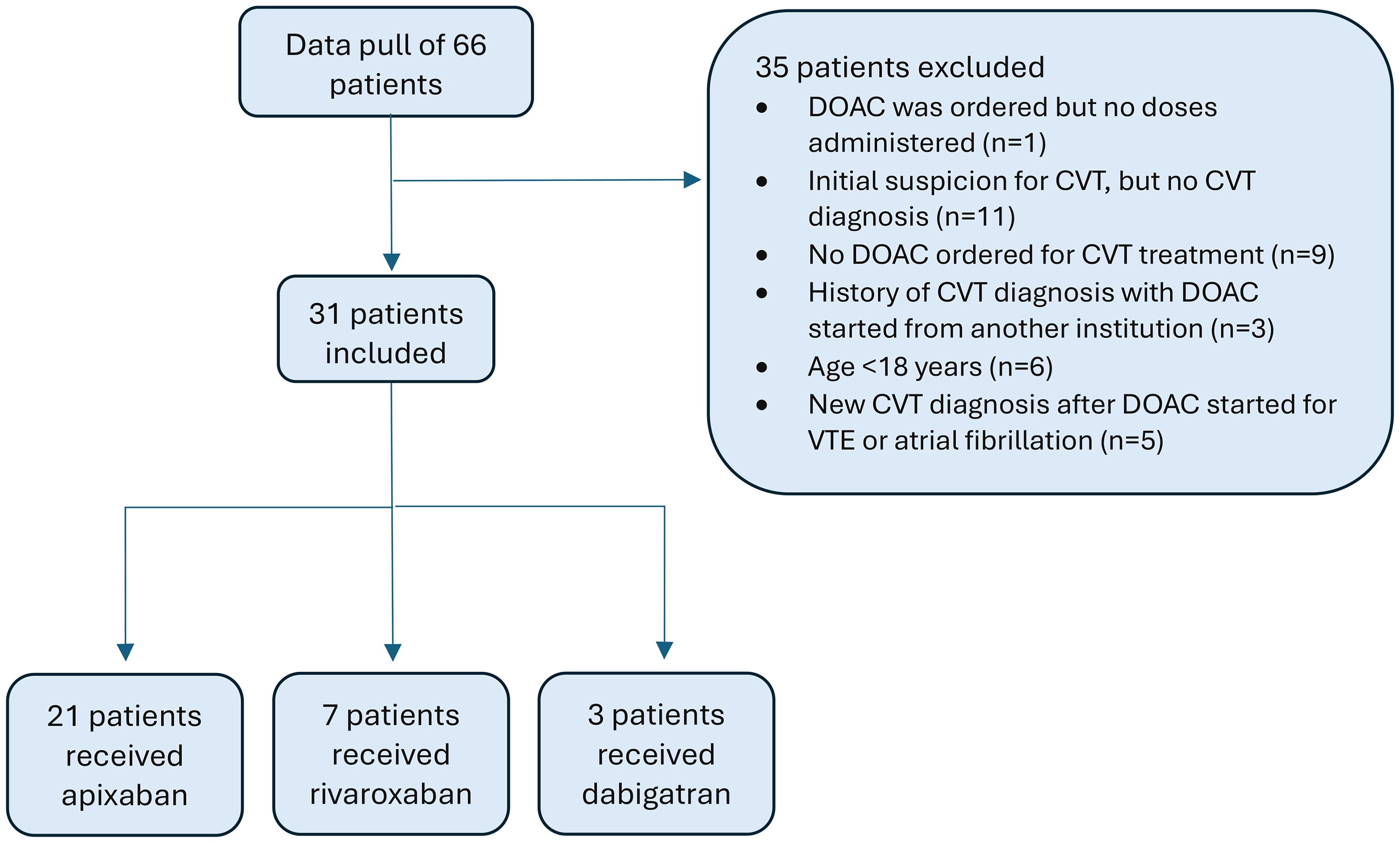
Patient inclusion and exclusion.
Baseline Characteristics.
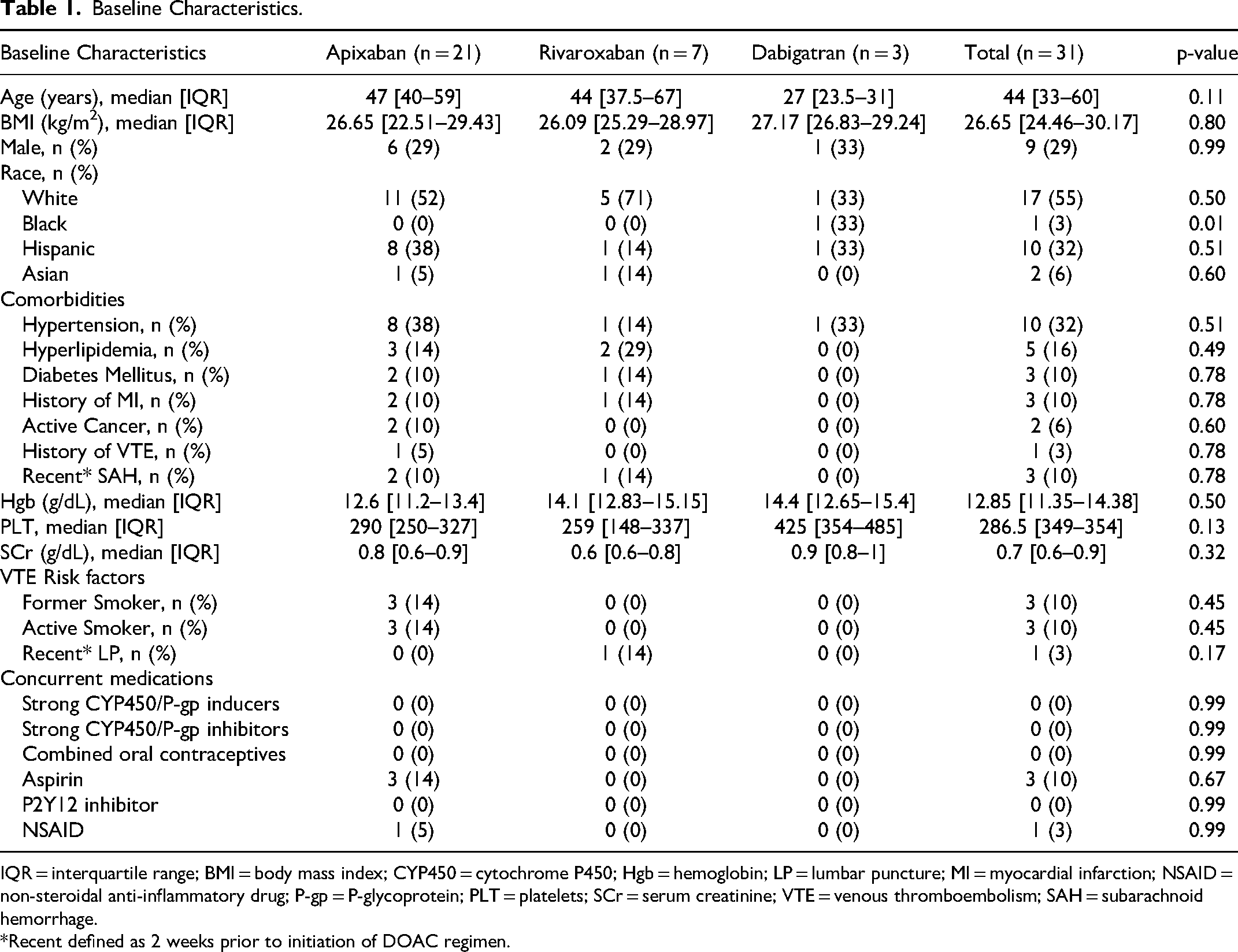
IQR = interquartile range; BMI = body mass index; CYP450 = cytochrome P450; Hgb = hemoglobin; LP = lumbar puncture; MI = myocardial infarction; NSAID = non-steroidal anti-inflammatory drug; P-gp = P-glycoprotein; PLT = platelets; SCr = serum creatinine; VTE = venous thromboembolism; SAH = subarachnoid hemorrhage.
*Recent defined as 2 weeks prior to initiation of DOAC regimen.
Receipt of parenteral anticoagulation (81% vs 57% vs 100%, P = 0.263) was similar between groups and the median duration of parenteral anticoagulation was 3 days (IQR 1-5 days); 2 (IQR 1-6) for apixaban, 1.5 (IQR 0-4) for rivaroxaban, and 4 (IQR 3.5-4.5) for dabigatran, P = 0.475. While not statistically significant, a full DOAC LD was given to fewer patients in the apixaban and rivaroxaban groups (29% vs 43% vs 100%, P = 0.058). All patients received maintenance doses concordant with recommendations for VTE treatment and the median duration of DOAC therapy was 180 days for each group.
When comparing all patients, regardless of reimaging status, dabigatran had higher rates of composite partial and complete recanalization (apixaban 33% vs rivaroxaban 57% vs dabigatran 100%, P = 0.02). No significant differences were observed in the frequency of partial recanalization (14% vs 43% vs 33%, P = 0.13) or complete recanalization (19% vs 14% vs 67%, P = 0.15) (Table 2). The median time to recanalization was 170 days [IQR 85.75–209.5 days], though this was mostly dependent on timing of scheduled follow-up imaging. The proportion of patients with repeat imaging differed between groups (33% vs 71% vs 100%, P = 0.034) with the apixaban group having the least follow-up MRIs. A subgroup analysis including only patients with repeat imaging found no significant difference in composite recanalization (100% vs 80% vs 100%, P = 0.34). Similarly, the incidence of partial recanalization (43% vs 60% vs 33%, P = 0.74) and complete recanalization (57% vs 20% vs 67%, P = 0.33) were not different between groups. Baseline demographics were compared between patients with and without repeat imaging (Table S1) to address any potential biases related to who was selected for repeat imaging and encouragingly, no significant differences were observed.
Study Outcomes.
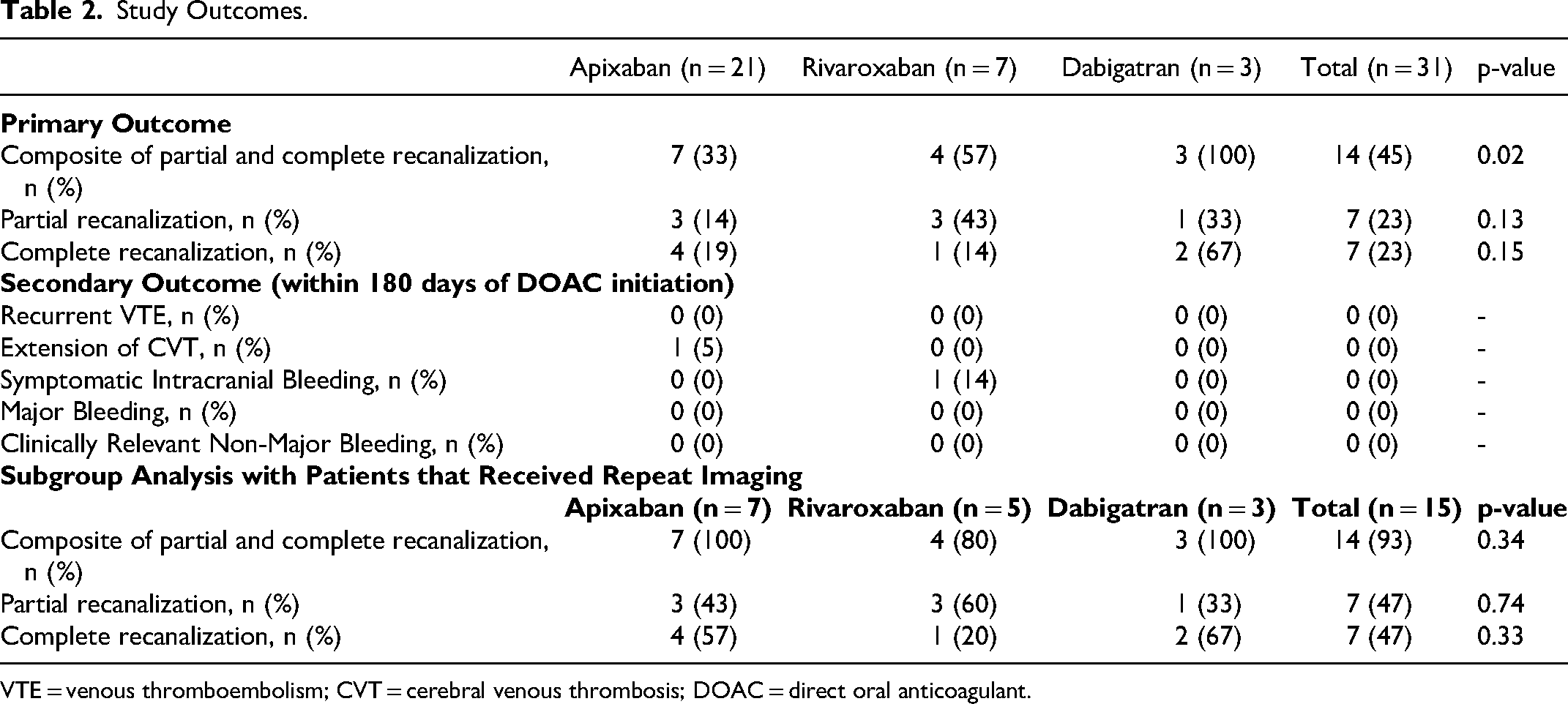
VTE = venous thromboembolism; CVT = cerebral venous thrombosis; DOAC = direct oral anticoagulant.
No differences were observed in the rates of bleeding or death. One patient had extension of CVT on MRI. This patient did not receive parenteral anticoagulation or a LD prior to initiating apixaban 5 mg orally twice daily. Another patient in the rivaroxaban group had an increased midline shift on MRI. This patient was transferred from an outside hospital where head computed tomography (CT) noted right parietal and occipital intraparenchymal hemorrhage with 2 mm midline shift. Intravenous argatroban was initiated for CVT treatment after a repeat head CT noted that the hemorrhage was stabilized. Information from the outside hospital detailing the rational for argatroban was unavailable and argatroban was continued for 5 days during which partial thromboplastin time (PTT) remained therapeutic except for one supratherapeutic PTT value. Afterwards, they were transitioned to rivaroxaban 15 mg twice daily for 6 days before a repeat head CT demonstrated an increased midline shift and rivaroxaban was discontinued. No concurrent non-steroidal anti-inflammatory drugs or antiplatelets were administered in both cases. It is unclear whether the choice or dosing of DOACs contributed to these outcomes.
Univariate logistic regression analyses showed that patients who received a full DOAC LD prior to starting their maintenance regimen had 2.2-fold increased likelihood of achieving the primary outcome. However, this result did not reach statistical significance (OR 2.2; 95% CI 0.5–9.8). Use of parenteral anticoagulation before DOAC initiation did not predict recanalization (OR 0.5; 95% CI 0.1–2.9), and neither did the duration of parenteral anticoagulation prior to DOAC initiation (OR 0.965; 95% CI 0.864–1.078).
Discussion
In our conservative analysis, composite recanalization was achieved in 45% of patients, which is lower than the 86% recanalization rate shown in a large international multicenter study. 15 Individual rates of partial recanalization and complete recanalization was also lower in our study (23% each) compared to a meta-analysis that reported partial recanalization in 46% of patients across nine studies and complete recanalization in 59% of patients across fourteen studies. 16 Our recanalization rates are likely underestimated due to our conservative approach using the worst-case assumption. Surprisingly, dabigatran had a significantly higher recanalization rate, while the apixaban group had the lowest, despite the general emergence of apixaban as the preferred DOAC in clinical practice based on efficacy and/or safety for indications such as VTE or atrial fibrillation. 19 However, when exclusively analyzing patients with repeat imaging, no difference between DOACs was observed. Additionally, 93% of all patients with follow-up imaging achieved composite recanalization after the initiation of a DOAC, aligning with the aforementioned studies. The low incidence of bleeding and clotting was also consistent with previous literature.
Though the over two-fold increased likelihood of achieving the primary outcome among patients given a full DOAC LD (ie, 7 days of apixaban 10 mg orally twice daily) before starting maintenance therapy was not statistically significant, it highlights the importance of investigating whether a full LD improves recanalization rate. Given only 39% of patients received a full DOAC LD, optimizing computerized physician order entry by including full LDs for CVT treatment in DOAC order sets could be beneficial until more concrete evidence on CVT treatment emerges. The nearly two-fold decreased odds of recanalization among those receiving parenteral anticoagulation (though the confidence interval was wide), warrants further research to explore the role for initial parenteral anticoagulation, particularly as the duration of parenteral anticoagulation did not appear to significantly predict recanalization and DOACs have relatively short half-lives. It is probable that all patients in the dabigatran group received parenteral anticoagulation because this dosing strategy is recommended in the package labeling. Notably, the lack of initial parenteral anticoagulation for patients in the apixaban and rivaroxaban groups did not appear to be associated with increased VTE recurrence. Further investigation is necessary to address the following questions: 1) Is a full LD necessary? 2) Is initial parenteral anticoagulation necessary? 3) If parenteral anticoagulation is required, what is the optimal duration?
Several limitations stem from the retrospective nature of this study. Patient data were limited to what was documented within the electronic medical record. Thus, rationale for specific DOAC selection, use of parenteral anticoagulation, and the decision to initiate or forego a DOAC LD could not be determined. Accordingly, decisions regarding repeat imaging were driven by individual clinician preference, and a significant proportion of patients, particularly those in the apixaban group, did not undergo follow-up imaging. One plausible explanation could be the impact of the COVID-19 pandemic (eg, reduced in-person visits and patient concerns about visiting a medical facility) as most patients admitted during the year 2020 received apixaban. The potential financial burden associated with obtaining a repeat MRI may also have hindered patients from returning for follow-up. Regardless, we attempted to mitigate any potential bias related to this by performing a subgroup analysis exclusively among patients with repeat imaging to reduce potential confounding factors and by comparing baseline characteristics between patients with and without repeat imaging, which reassured us that the decision to repeat imaging was not systematically influenced by patient-specific factors. Other limitations include a small sample size of 31 patients over 4 years at a single institution (though this is expected due to the rarity of this disease state), and inconsistent DOAC LD use, parenteral anticoagulation, and duration of parenteral anticoagulation. Currently, the exact duration of parenteral therapy, or whether it is necessary prior to DOAC initiation, has yet to be elucidated. If future evidence substantiates the effectiveness of parenteral anticoagulation for a specific duration, a protocolized approach to ordering this therapy may enhance consistency. A DOAC order panel to help ensure selection of the appropriate DOAC LD and duration followed by the correct maintenance regimen was implemented at our institution in December 2021 and was only in effect for 5 (16%) of our patients.
Conclusion
DOACs continue to be a viable agent for the treatment of CVT with individual DOACs appearing to be similar in efficacy and safety. However, the preferred agent cannot be elucidated from this study, and larger, randomized, multicentered studies are needed to validate these findings as well as to determine the most appropriate dosing and duration.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251316869 - Supplemental material for Comparison of Direct Oral Anticoagulants for Treatment of Cerebral Venous Thrombosis – A Retrospective Cohort Study
Supplemental material, sj-docx-1-cat-10.1177_10760296251316869 for Comparison of Direct Oral Anticoagulants for Treatment of Cerebral Venous Thrombosis – A Retrospective Cohort Study by Paul Phan and Lisa T. Hong in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Abbreviations
Author Contributions
Conceptualization (PP and LH), Investigation (PP and LH), Methodology (PP and LH), Formal analysis (PP), Data curation (PP), Validation (PP and LH), Writing – original draft (PP), Writi- review & editing (PP and LH), Visualization (PP and LH), Project administration (LH), Supervision (LH).
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Considerations
The study was conducted according to the guidelines of the Declaration of Helsinki.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Institutional Review Board Statement
The study was approved by the Institutional Review Board of Loma Linda University (#5220336, October 12, 2022).
Informed Consent
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
