Abstract
Background
Cerebral venous thrombosis (CVT), a rare cause of stroke, primarily occurs in young individuals. The established treatment regimen involves anticoagulation with low molecular weight heparin (LMWH) and vitamin K antagonists (VKA). Although direct oral anticoagulants (DOACs) have emerged as a promising alternative, their efficacy and safety remain unclear. This meta-analysis compared the efficacy and safety of DOACs versus VKA in managing CVT.
Methods
Electronic databases, including PubMed, Cochrane Library, and ScienceDirect, were searched from inception until April 2025. Risk ratios (RR) with a 95% Confidence interval (CI) were pooled under the random effects model in the Review Manager 5.4.1. Quality assessment was done through the Cochrane risk of bias (RoB 2.0) tool and the Newcastle Ottawa Scale (NOS). Subgroup analyses based on study design and different types of DOACs were carried out.
Results
Thirty-one studies, including five randomized controlled trials (RCTs) and 26 observational studies, were included in this meta-analysis. Our analysis showed a significant reduction in the risk of recurrent venous thromboembolism (VTE) in the DOACs arm compared to VKA (RR = 0.84; 95%CI: [0.71,0.99]; p = 0.04; I2 = 0%). Similarly, the DOACs showed significant superiority over VKA regarding the intracranial hemorrhage (ICH) (RR = 0.67; 95%CI: [0.50,0.89]; p = 0.007; I2 = 0%). Other endpoints, including major hemorrhage (RR = 0.70; 95%CI:[0.42,1.15]; p = 0.16; I2 = 0%), all-cause mortality (RR = 0.96; 95%CI:[0.68,1.35]; p = 0.81; I2 = 0%), and full recanalization (RR = 0.92; 95%CI:[0.82,1.03]; p = 0.16; I2 = 21%), are comparable between the two arms.
Conclusion
DOACs showed a significant reduction in the risk of recurrent VTE and ICH compared to VKA, whereas other endpoints are comparable. Further RCTs with a robust sample size are required to validate and confirm these findings.
Keywords
Introduction
A rare cause of stroke that mainly affects young people is cerebral venous thrombosis (CVT). 1 It is thought to affect 13 to 20 out of every million people annually, making up between 0.5% and 3% of all strokes.2,3 A blood clot in the cerebral veins, dural venous sinuses, or both can cause CVT, which impairs venous drainage and may result in neurological issues..4,5 Majority of the cases affect people under the age of 55, with approximately two-thirds occurring in women of childbearing age, which is frequently associated with temporary prothrombotic conditions like pregnancy, puerperium, or oral contraceptive use.. 4
Current guidelines recommend systemic anticoagulation therapy for 3 to 12 months in patients with CVT to promote venous recanalization and reduce the risk of recurrent venous thromboembolism (VTE).6,7 Traditionally, low-molecular-weight heparin (LMWH) followed by vitamin K antagonists (VKA), such as warfarin, have been the standard of care.6–8 However, LMWH needs to be administered subcutaneously and has concerns such as heparin-induced thrombocytopenia, whereas VKA need to be monitored often, have several drug and food interactions, and raise the risk of cerebral bleeding.9–12 Direct oral anticoagulants (DOACs) have become viable substitutes with benefits like reduced risk of cerebral hemorrhage, easier clinical control, and enhanced patient compliance.13,14 Nevertheless, DOACs continue to be more costly and may increase the risk of bleeding in the gastrointestinal tract.15,16 Although they have demonstrated usefulness in treating disorders such as peripheral VTE and atrial fibrillation, the safety and efficacy of DOACs in CVT remain unknown and require further investigation.
Comparing DOACs and VKA in large-scale randomized controlled trials (RCTs) is difficult because CVT is uncommon. The available information comes from small observational studies and RCTs, which frequently have low event rates and thus less statistical power. The safety and efficacy of the two anticoagulation techniques are similar, according to meta-analyses that combine data from observational studies and RCTs.3,17 The American Heart Association (AHA) and other organizations recognize DOACs as a legitimate choice, but they emphasize the need for high-quality research, which is why the current guidelines are still cautious. 4 Therefore, we conducted a systematic review and meta-analysis of clinical trials and cohorts to evaluate the efficacy and safety of DOACs compared to VKA in patients with CVT. This analysis focused on multiple clinically relevant outcomes to provide a comprehensive understanding of DOAC use in routine clinical practice.
Methods
This study follows the guidelines of Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 18 and the Cochrane Handbook for Systematic Reviews of Interventions. 19
Data Sources and Search Strategy
A comprehensive literature search was conducted from inception until April 2025. We thoroughly searched electronic databases, including PubMed, Cochrane Library, and ScienceDirect, for studies comparing DOACs versus VKA in patients with CVT. Additionally, we checked clinicaltrials.gov for any unpublished trials. The Following MeSH headings and keywords were utilized for the screening process “Factor Xa Inhibitors”, “Direct-Acting Oral Anticoagulant”, “Dabigatran”, “Edoxaban”, “Rivaroxaban”, “Apixaban”, “Warfarin”, “Vitamin K antagonist”, “Cerebro-venous thrombosis”, and “cerebral venous sinus thrombosis”. A comprehensive search strategy for each database is summarized in Supplementary Table S1.
Study Selection and Criteria
The relevant articles identified using specific keywords were imported into the EndNote Reference Manager (Version X7.5; Clarivate Analytics, Philadelphia, Pennsylvania, 2016) for screening, where duplicates were flagged and removed. Studies were initially reviewed at the title and abstract level, followed by full-text screening. There was no restriction on the study timeline, and only English-language articles were considered. Additionally, the bibliographies of the articles included were checked for any additional relevant studies. Two authors (AS and NR) established and assessed the eligibility criteria, with any disagreements resolved by the third author (MHW). The Inclusion criteria included: patients above 18 years old, who have acute or sub-acute CVT, and have undergone treatment with DOACs and VKA. Exclusion criteria included participants under 18 years, those with infection, malignancy, or pregnancy, or those with increased risk of bleeding. Studies included in the review encompassed observational studies, RCTs, case-control studies, and cohorts, while case reports, systematic reviews, protocols, and editorials were excluded. The screening process is summarized in Figure 1 (PRISMA flowchart).

PRISMA Diagram Showing the Study Selection Process.
Data Extraction and Quality Assessment
The relevant data was reviewed and entered into a standardized Excel sheet. The extracted baseline characteristics included study ID, study design, location, patient comorbidities, sample size, and mean age. The extracted outcomes included recurrent VTE, major hemorrhage, intracranial hemorrhage, all-cause mortality, and full recanalization.
Two authors (MO and ZUA) performed data extraction, with disparities being resolved by a third author (MHW). To assess the quality of the included trials and observational studies, tools such as, Cochrane Risk of Bias (RoB 2.0) tool was employed, which consists of five domains that help evaluate the robustness of the included studies, and the Newcastle Ottawa Scale (NOS) to assess the quality of the studies based on selection, comparability, and outcomes, respectively.20,21 According to the standardized star rating system, studies with seven or more stars were considered high quality. The included studies’ publication bias risk was assessed visually through funnel plots and statistically through Egger's regression. We also performed the GRADE assessment to determine the certainty of evidence using GRADEpro GDT. 22
Statistical Analysis
The forest plots were constructed utilizing the latest version of Review Manager software (version 5.4.1). The random effects model accounted for any variability among the studies. Risk ratio (RR) was used to draw a comparison between the two interventions for dichotomous outcomes. A P-value of ≤0.05 was defined as statistically significant. The confidence interval (CI) of 95% was calculated for all the outcomes. Heterogeneity was determined using Higgins’ I2 Test, where studies reporting I2 of more than 50% were regarded as significantly heterogeneous and were further evaluated by performing the sensitivity analysis (leave one-out method). 23 We performed a subgroup analysis based on the study design (RCTs and observational studies), and depending on the data available, we also performed a subgroup analysis by stratifying DOACs into subtypes, including rivaroxaban, apixaban, dabigatran, and unspecified DOAC type. The category of ``unspecified DOAC typè` was employed when studies did not clearly mention the type of DOAC used or when a combination of DOACs was used.
Results
Search Results
The literature searches initially retrieved 545 articles. After removing duplicates (n = 109), 436 articles remained. The title and the abstract screening excluded 370 articles, leaving 66 studies for full-text screening against the eligibility criteria. Eventually, 31 studies were included in this meta-analysis.24–48 The selection of studies is summarized in Figure 1.
Study Characteristics
This meta-analysis included 31 studies that met the eligibility criteria. Five RCTs and 26 observational studies were included. The publication years ranged from 2014 to 2025. Table 1 provides the baseline characteristics for included studies. The GRADE summary of findings are provided in Table 2.
Baseline Characteristics of Included Studies.

Note: RCT: Randomized Controlled Trial; DOACs: Direct oral anticoagulants; VKA: Vitamin K antagonist.
GRADE Summary of Findings.

Note: a. The overall risk of bias for the Randomized controlled trials (RCTs) ranged from high to some concerns. b. The 95% Confidence Interval (CI) is wide and crossing the 1.
Bias Assessment
A risk of bias assessment was conducted for the included studies using two validated tools appropriate for their respective study designs. RCTs were assessed using the Cochrane ROB 2.0 tool,
20
which evaluates five key domains: randomization process, deviations from intended interventions, missing outcome data, measurement of the outcome, and selection of the reported result. Among the RCTs, two studies24,40 demonstrated a high risk of bias. Three studies25–27 had some concerns, particularly related to deviation from intended intervention and completeness of outcome data. Observational studies were evaluated using NOS, which assesses quality based on selection, comparability, and outcome assessment.
21
Several studies28–33 received high scores,8,9 reflecting strong methodology and minimal bias. Most other studies scored in the moderate range,6,7 with minor limitations in follow-up or comparability. How ever, a few studies34–36,41,45,47,51,56 were rated lower 4,5 due to notable deficiencies in participant selection, adjustment for confounding variables, and outcome ascertainment. Overall, while many of the included studies demonstrated acceptable methodological quality, the presence of high-risk studies highlights the need for cautious interpretation of pooled findings. The RoB 2.0 quality assessment traffic plot and NOS quality assessment table are provided in Figure 2 and Supplementary Table S2

Risk of Bias (RoB 2.0) Tool Traffic Light Plot.
Recurrent Venous Thromboembolism
The analysis included 24 studies (4 RCTs, 20 observational studies) involving 4,470 patients (1,923 DOACs vs 2,547 VKA). The analysis revealed that the risk of recurrent VTE is significantly less with DOACs compared to VKA (RR = 0.84; 95% CI: [0.71, 0.99], p = 0.04, I2= 0%) Figure 3.

Forest Plot for Recurrent Venous Thromboembolism (VTE).
Subgroup analysis by study design showed that RCTs reported no significant difference between DOACs and VKA (RR: 2.78; 95% CI: [0.86,8.95]; p = 0.09; I² = 0%), whereas observational studies favored DOACs with a significant risk reduction (RR: 0.81; 95% CI: 0.69-0.97; p = 0.02; I² = 0%).
Subgroup analysis by DOAC subtypes indicated that only the group with unspecified DOAC type (RR: 0.83; 95% CI: 0.70- 0.99; p = 0.04) demonstrated a statistically significant reduction in recurrent VTE compared to VKA. Rivaroxaban (RR: 0.38; 95% CI: 0.10-1.41; p = 0.15) and apixaban (RR: 0.35; 95% CI: 0.02-5.81; p = 0.46) demonstrated decreased recurrent VTE rates, whereas dabigatran (RR: 2.73; 95% CI: 0.77-9.62; p = 0.12) demonstrated increased rates; however, the results were not statistically significant. (Supplementary Figure S1).
Major Hemorrhage
The analysis included 19 studies (3 RCTs, 16 observational studies) involving 2,659 patients (1,201 DOACs vs 1,458 VKA). The analysis revealed no statistically significant difference in the risk of the major hemorrhage between DOACs and VKA (RR = 0.70; 95% CI: [0.42, 1.15], p = 0.16, I² = 0%) Figure 4.

Forest Plot for Major Hemorrhage.
In the RCTs subgroup, DOACs showed a non-significant lower risk (RR = 0.50; 95% CI: 0.05–5.37; p = 0.57), and similar effects were observed in the observational studies subgroup (RR = 0.71; 95% CI: 0.43 to 1.19; p = 0.19).
Although a comparable association between all DOAC subtypes was observed during the subgroup analysis, dabigatran demonstrated the lowest rates of major hemorrhage (RR = 0.50; 95% CI: 0.05–5.37; p = 0.57), Supplementary Figure S2.
Intracranial Hemorrhage
The analysis included 26 studies (4 RCTs, 22 observational studies) involving 4,386 patients (2,034 DOACs vs 2,352 VKA). The results showed that DOACs were associated with a significantly lower risk of intracranial hemorrhage compared to VKA (RR = 0.67; 95% CI: [0.50, 0.89], p = 0.007, I² = 0%), Figure 5.

Forest Plot for Intracranial Hemorrhage.
In the RCTs subgroup, the difference was not statistically significant (RR = 1.03; 95% CI: [0.19, 5.45], p = 0.97), while observational studies demonstrated a significant reduction in intracranial hemorrhage with DOACs (RR = 0.66; 95% CI: [0.50, 0.89], p = 0.006).
During subgroup analysis, dabigatran (RR = 0.50; 95% CI: 0.05–5.37; p = 0.57) subgroup demonstrated lower occurrence of intracranial hemorrhage, while rivaroxaban (RR = 1.11; 95% CI: 0.26- 4.70; p = 0.88) and apixaban (RR = 1.02; 95% CI: 0.18–5.94; p = 0.98) showed a higher incidence although the results were statistically non-significant. The ‘unspecified DOAC subtype’ group indicated a significantly lower association with intracranial hemorrhage (RR = 0.65; 95% CI: 0.47–0.88; p = 0.005), Supplementary Figure S3.
All-Cause Mortality
The analysis included 22 studies (4 RCTs, 18 observational studies) involving 4,368 patients (1,916 DOACs vs 2,452 VKA). The results showed no statistically significant difference in all-cause mortality between DOACs and VKA (RR = 0.96; 95% CI: [0.68, 1.35], p = 0.81, I² = 0%), Figure 6.
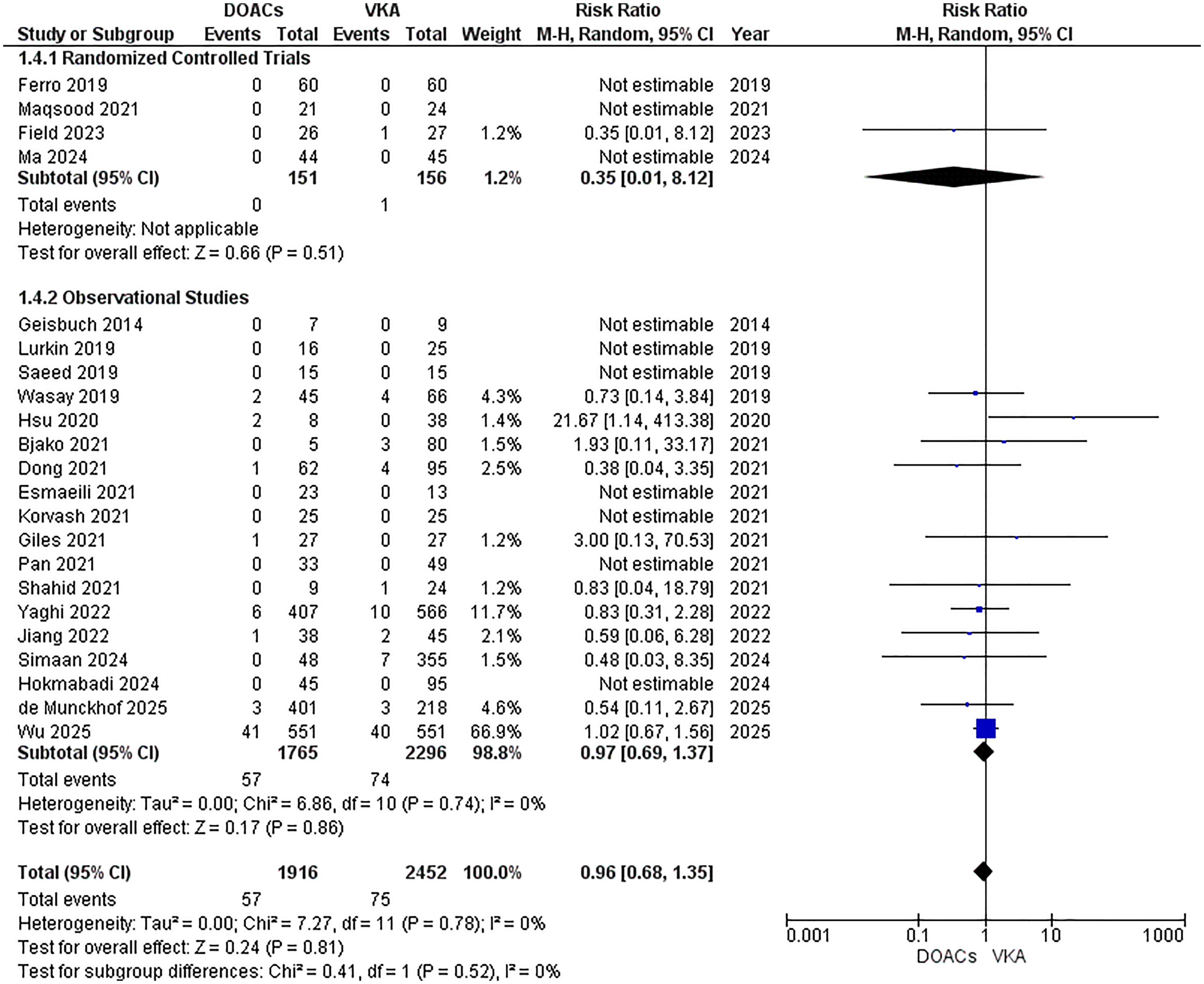
Forest Plot for All-Cause Mortality.
RCTs demonstrated a non-significant reduction in mortality with DOACs (RR = 0.35; 95% CI: [0.01, 8.12], p = 0.51), while observational studies also showed no significant difference (RR = 0.97; 95% CI: [0.69, 1.37], p = 0.86).
The subgroup analysis by DOAC subtypes demonstrated that rivaroxaban (RR = 0.74; 95% CI: 0.15- 3.59; p = 0.71), apixaban (RR = 0.42; 95% CI: 0.07–2.34; p = 0.32) and ‘unspecified DOAC type’ (RR = 1.01; 95% CI: 0.70- 1.44; p = 0.97) illustrated comparable outcomes in terms of all-cause mortality Supplementary Figure S4.
Full Recanalization
This analysis included 16 studies (3 RCTs, 13 observational studies) with 1,709 patients (824 receiving DOACs and 885 receiving VKA). There was no significant difference in full recanalization between DOACs and VKA (RR = 0.92; 95% CI: [0.82, 1.03], p = 0.16, I² = 21%), Figure 7.
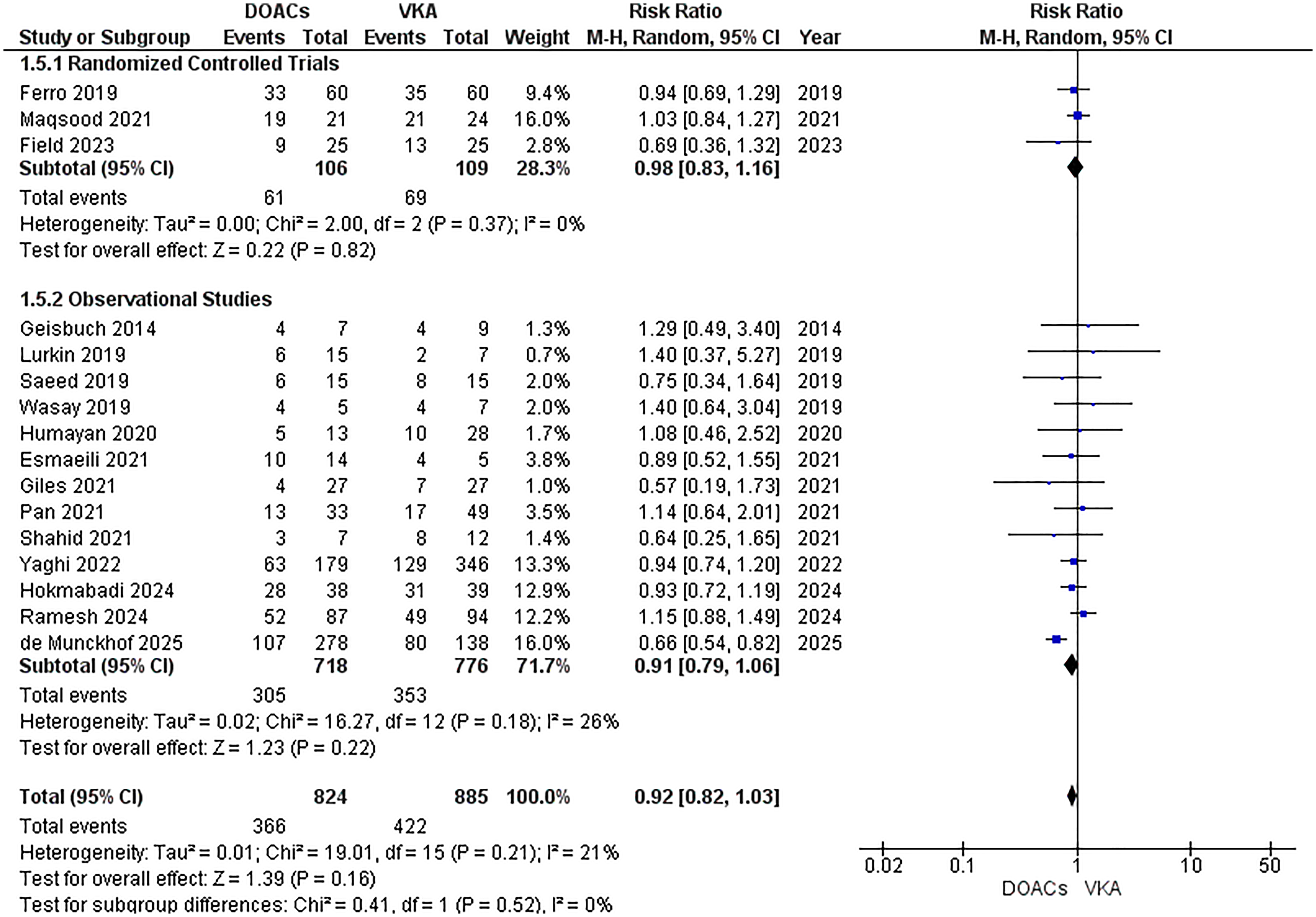
Forest Plot for Full Recanalization.
RCTs showed no significant difference in full recanalization (RR = 0.98; 95% CI: [0.83, 1.16], p = 0.82), and observational studies also reported a non-significant effect (RR = 0.91; 95% CI: [0.79, 1.06], p = 0.22).
The subgroup analysis by DOAC subtypes demonstrated that rivaroxaban (RR = 0.99; 95% CI: 0.84- 1.17; p = 0.92), dabigatran (RR = 0.94; 95% CI: 0.69–1.29; p = 0.71) and ‘unspecified DOAC type’ (RR = 0.91; 95% CI: 0.75- 1.09; p = 0.31) illustrated comparable risk in context of full recanalization Supplementary Figure S5.
Publication Bias
The publication bias was assessed visually by analyzing the funnel plots and statistically through Egger's regression test. No significant bias was detected for any of the assessed endpoints Supplementary Table S3 and Supplementary Figure S6–10.
Discussion
This meta-analysis pooled results of the 26 observational studies and 5 RCTs to compare the safety and efficacy profile of DOACs in contrast to VKA. The results suggest that DOACs were associated with a reduced incidence of recurrent VTE and intracranial hemorrhage in patients with CVT. Comparable findings were elucidated between the two treatment groups for major hemorrhage, all-cause mortality, and full recanalization. This meta-analysis evaluated DOACs by stratifying outcomes according to the specific subtypes—rivaroxaban, apixaban, dabigatran, and an ‘unspecified DOAC type’ where studies employed a combination of agents. In this systematic review, we also illustrate the avenues for future research while highlighting areas where evidence is currently lacking.
Concerning the occurrence of recurrent VTE, the findings of our meta-analysis are corroborated by existing literature.33,57 Our pooled analysis aligns with the real-world study by Wu et al. that probed into anonymized individual patient-level data of over 123 million patients, demonstrating a reduced recurrence of VTE with DOACs compared with warfarin.
33
Wu et al. conducted their study using the TriNetX platform and employed propensity score matching to ensure comparability between the DOAC and warfarin groups. The sensitivity analysis was carried out during the assessment of CVT recurrence to confirm that the observed events were true recurrences. Additionally, intracranial hemorrhage was carefully monitored over a one-year follow-up. Although the large sample size in Wu et al's study has a substantial impact on the overall results, their rigorous methodology, use of a large patient cohort, and reliance on real-world data enhance the generalizability and external validity of the findings of this meta-analysis
The outcomes of our meta-analysis underscore that DOACs confer superior safety profiles when compared with VKA in terms of lower intracranial hemorrhage, which aligns with existing literature. 33 Previous studies show the use of DOACs is associated with a lower risk of intracranial hemorrhage across several clinically relevant subgroups, including patients who are neither underweight nor obese, as well as those treated with apixaban. 33 The current evidence from other VTE populations highlights the association of rivaroxaban and dabigatran with comparable rates of major hemorrhage, wherein they offered a notable reduction in the incidence of intracranial hemorrhage.59,60 The subgroup analysis of our study employing rivaroxaban and dabigatran in the CVT population demonstrated consistent findings regarding major hemorrhage. However, the two DOACs illustrated comparable incidence rates of intracerebral hemorrhage, consistent with the landmark RE-SPECT and SECRET trials.24,26 It must, however, be noted that the trials were underpowered to test the superiority or non-inferiority of DOACs or to illustrate differences in intracranial hemorrhage rates between the employed treatment modalities. In sync with existing evidence, our results elucidate comparable safety and efficacy outcomes regarding major hemorrhage58–61 and full recanalization.24,29 Of note, a previously published meta-analysis reported an elevated incidence of full recanalization in the patients treated with DOACs compared to those treated with VKA. 62 However, our pooled analysis does not establish any evidence of this result. It is important to note that the included studies constituted variations in the definition and assessment of hemorrhage, including heterogeneities in imaging, evaluation metrics, and assessment scales to categorize and calibrate bleeding events. This mandates that the findings be interpreted against the backdrop of these constraints while weighing the risks of treatment modalities against benefits.
The demonstration of a contracted incidence of recurrent VTEs and intracranial hemorrhage in the patients treated with DOACs may be explained by the enhanced drug profile of DOACs. Exhibiting their effects either as direct thrombin inhibitors (dabigatran etexilate) or Factor Xa inhibitors (apixaban, edoxaban, and rivaroxaban), the DOACs have been identified as potent and efficacious alternatives to VKA in emerging research.
63
In contrast to VKA, which can yield subtherapeutic periods and hypercoagulability, DOACs have stable pharmacokinetics and pharmacodynamics, thereby featuring consistent anticoagulant parameters while bypassing the requirement of INR monitoring.64–66 They constitute an enhanced safety profile with a reduced rate of intracranial hemorrhage, fewer interactions with other drugs and food, and a lower burden of dietary restrictions
Of note, the reduction in ICH with DOACs in our study did not translate into a statistically significant difference between the two treatment populations in terms of major hemorrhage. This could potentially stem from the inclusion of systemic hemorrhage incidences in the outcomes for major hemorrhagic events, which demonstrated comparable results in previous studies.27,39,4042–44,46,49 Our findings could also reflect a redistribution of bleeding risk while employing DOACs. Except for apixaban, DOACs are associated with higher rates of gastrointestinal bleeding than VKA, which counterbalance their cerebral safety profile.60,73 In addition, extracranial bleeding events may be subject to underreporting or inconsistent categorization across various studies, whereas intracranial hemorrhage, due to its severity and well-established reporting standards, might have been documented with higher precision.
Although the emerging research elucidates a better safety and efficacy profile of DOACs in comparison to VKA, the guidelines by the European Stroke Organization (ESO) recommend VKA as long-term anticoagulation therapy after the initial treatment with heparin for 3–12 months.6,7 Despite the accumulating comparative data demonstrating the potential superiority of DOACs over warfarin, their incorporation in the CVT management guidelines is constrained by a paucity of high-quality, evidence-based data.6,7,74 Nevertheless, DOACs are now the preferred pharmacologic agents in the settings of stroke prevention in atrial fibrillation, and stable cardiovascular and peripheral artery disease. 4 74–77 The results from the recently conducted SECRET and DOAC-CVT trials, when contextualized with previous studies through our meta-analysis, may potentially facilitate a foundational base for synthesizing updated clinical guidelines to optimize patient-related outcomes in CVT.
The appropriate duration of anticoagulation therapy following CVT constitutes a domain of clinical uncertainty due to the paucity of conclusive evidence guiding best practices. The studies included in our meta-analysis utilized anticoagulation therapy for a duration of 3 months to an indefinite period, with most studies employing a duration of 6 to 12 months. The emerging research, including the DOAC-CVT study, demonstrated comparable safety and efficacy profiles between DOACs and VKA over a period of 6 months. 49 In corroboration of these observations, a recent meta-analysis incorporating studies of ≥6 months’ duration demonstrated no significant discrepancies between DOACs and VKA regarding functional recovery, CVT recurrence, or vessel recanalization. 57
It is also indicated that DOACs may offer practical advantages owing to their ease of clinical management, potentially yielding enhanced treatment adherence and better patient-related outcomes. 57 These findings are consistent with current guideline recommendations elucidating a minimum treatment duration of 3–6 months for provoked CVT associated with transient risk factors. An extended therapy spanning 6–12 months is recommended for unprovoked cases, with consideration of indefinite treatment for patients with recurrent CVT or underlying thrombophilia disorders.63,78 The heterogeneity in treatment durations across studies serves as an impetus to establish standardized evidence-based protocols that optimize long-term clinical management of CVT, ensuring consistent therapeutic decision-making while balancing the risks of recurrence and anticoagulation-related complications.
Our meta-analysis is not free from limitations. Firstly, most of the included studies were observational, which introduced selection bias, and confounding variables were not adjusted for. Secondly, the included RCTs constituted a small sample size. Thirdly, the included studies featured a paucity of data regarding apixaban, dabigatran, and edoxaban, which might have limited the scope of our subgroup analysis, thereby constraining a comprehensive evaluation of each agent's differential efficacy and safety profiles. Also, the larger sample size studies, while strengthening the findings by enhancing generalizability and external validity, may have disproportionately influenced the overall results. Finally, discrepancies in CVT management protocols, including variations in drug initiation and duration, assessment timelines, and outcome definitions, could have possibly introduced clinical heterogeneity; however, our study demonstrated a low statistical heterogeneity.
Conclusion
This meta-analysis illustrated a reduced incidence of recurrent VTEs and intracranial hemorrhage with DOACs compared to VKA, while comparable profiles were observed for all-cause mortality, major hemorrhage, and vessel recanalization. These findings align with the growing rationale for considering DOACs as a safer, convenient, and more effective alternative to VKA for optimizing the clinical management of patients with CVT. However, inconsistencies across studies regarding initiation protocols, therapeutic durations, and outcome measures mandate future research to standardize the evaluation metrics.
Well-powered studies incorporating subgroup analyses based on key clinical variables, including underlying prothrombotic states, severity of venous occlusion, and hemorrhagic complications, are crucial to enhancing therapeutic decision-making. Head-to-head comparisons of individual DOACs are warranted to illustrate more nuanced and drug-specific efficacy and safety profiles in the treatment of patients with CVT.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251405417 - Supplemental material for Direct Oral Anticoagulants Versus Vitamin K Antagonists in Cerebral Venous Thrombosis: A Systematic Review and Meta-Analysis of 4,929 Patients
Supplemental material, sj-docx-1-cat-10.1177_10760296251405417 for Direct Oral Anticoagulants Versus Vitamin K Antagonists in Cerebral Venous Thrombosis: A Systematic Review and Meta-Analysis of 4,929 Patients by Muhammad Hassan Waseem, Zain ul Abideen, Areeba Shoaib, Nohela Rehman, Muhammad Osama, Barka Sajid, Muhammad Mukhlis, Sania Aimen, Muhammad Wajih Ansari, Rowaid Ahmad, Zara Fahim, Ameer Haider Cheema, Muhammad Bilal Zahid and Pawan Kumar Thada in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Ethical Approval
Not applicable.
Patient Consent
Not applicable.
Authors’ contributions CRediT roles
Study concept and design: MHW and ZUA; acquisition of data: ZUA, PWT and AS; analysis and interpretation of data: AS, NR, SA, MBZ and MO; drafting of the manuscript: RA, ZF, WA, and MM; critical revision of the manuscript: MHW and AHC.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data will be made available upon reasonable request to the authors.
Status
This manuscript has not been published previously and is not under consideration for publication elsewhere.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
