Abstract
Introduction
Cerebral venous thrombosis (CVT) is an uncommon but life-threatening neurological condition that disproportionally afflicts younger patients and females. 1 Prompt recognition and treatment are key to improving the outcome. The standard of care is treatment with anticoagulation to prevent the propagation of the thrombus even in the presence of intracerebral hemorrhage. However, the type of anticoagulation and the duration of treatment are controversial. Patients with CVT have traditionally been treated with unfractionated heparin (UFH) infusion or low molecular weight heparin (LMWH) as a bridge to anticoagulation with vitamin K antagonist (VKA), since the establishment of UFH efficacy in a randomized clinical trial in 1991. 2 Both the European guidelines and American Heart Association (AHA)/ American Stroke Association (ASA) guidelines recommend VKA as a long-term anticoagulant for CVT.1,3 Direct oral anticoagulants (DOAC) are recommended as first-line therapy over VKA for the management of venous thromboembolism (VTE) other than CVT. 4 Apixaban, a factor Xa inhibitor, is commonly used in the treatment of VTE. The AMPLIFY trial showed apixaban to be non-inferior to conventional therapy (enoxaparin/warfarin) in VTE treatment (pulmonary embolism or deep venous thrombosis), with less frequent major bleeding. 5 There is some evidence for the use DOAC in the treatment of CVT. In a small randomized clinical trial, dabigatran, a direct thrombin inhibitor, was found to be equal to warfarin in efficacy and safety for the treatment of CVT. 6 However, there is a paucity of data for the use of apixaban in this patient population. 7 There are a few observational studies but no randomized clinical trials. We present a case series of patients with CVT who were treated with apixaban for long-term anticoagulation. The aim of this study was to evaluate the efficacy and safety of apixaban, the most common used DOAC for CVT at our institution.
Methods
This case series was designed as a retrospective cohort study. We identified and screened all patients diagnosed with CVT at our institution from November 2018 to December 2020. These patients were screened for inclusion criteria that consisted of the following: Radiologically diagnosed CVT with CT venography and/or MR venography and immediate initial treatment with intravenous (IV) anticoagulation for at least twenty-four hours followed by the transition to oral apixaban for long-term anticoagulation. Additional treatment with mechanical thrombectomy was not an exclusion criteria. A total of eleven patients with CVT were identified. Of these, one patient was excluded due to the transition from rivaroxaban to apixaban for recurrent CVT without IV anticoagulation treatment as a bridge. Another patient was excluded due to the transition from IV anticoagulation to dabigatran for long-term anticoagulation. The data for these nine patients were collected by chart review, and include the following: demographics, clinical presentation, radiographical findings, initial treatment, dosing of apixaban, reported adverse events, recanalization rate, time to recanalization, and duration of follow-up.
Results
Patient characteristics are provided in Table 1. Of the nine patients included, five were female (56%). The mean age was 36 years (ranging from 24 to 60). Four patients (44%) had risk factors for CVT (ulcerative colitis, oral contraceptive pill use, anti-thrombin III deficiency, obesity). None of the patients had a history of or active infection with COVID-19. All the patients presented with typical symptoms of CVT. Seven patients (78%) presented with headache, three patients (33%) presented with new onset seizures, and five patients (56%) had additional focal deficits. The diagnosis was confirmed with CT venogram and/or MR venogram. Additional radiological findings were identified and are summarized in Table 2. Radiographic complications of CVT were seen in 56% of patients. These complications include intraparenchymal hemorrhage (IPH), subarachnoid hemorrhage (SAH), and venous infarcts. Two patients (22%) had both IPH and SAH, one patient (11%) had only IPH, and another patient (11%) had only SAH. There were three patients (33%) who had venous infarcts.
Patient Demographics, Comorbidities, and Clinical Presentation.
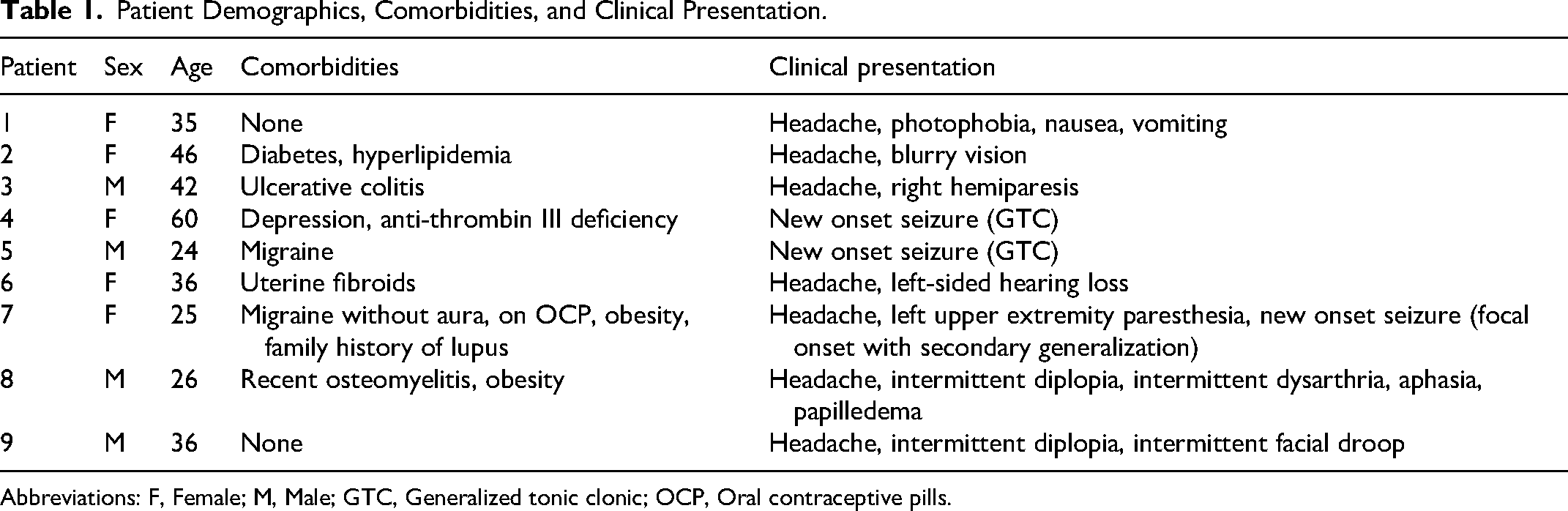
Abbreviations: F, Female; M, Male; GTC, Generalized tonic clonic; OCP, Oral contraceptive pills.
Radiological Findings.

Abbreviations: IPH, Intraparenchymal hemorrhage; SAH, Subarachnoid hemorrhage.
Immediately after confirmed diagnosis, all the patients were initially anticoagulated with UFH infusion for at least twenty-four hours, except for one patient (patient number 4), who had anti-thrombin III deficiency and was treated with argatroban infusion, a direct thrombin inhibitor. Eight patients were eventually transitioned to apixaban prior to discharge. One patient (patient number 6) was initially transitioned to dabigatran but was later switched to apixaban after five days due to health insurance coverage. Five patients (56%) received apixaban 10 mg twice daily for the initial five to seven days followed by 5 mg twice daily, based on the stroke neurologist's preference. The remaining four patients (44%) were transitioned from IV anticoagulation to Apixaban 5 mg twice daily. Anticoagulation treatment details are provided in Table 3. Interestingly, there was no worsening of intracerebral hemorrhage and no hemorrhagic conversion of ischemic strokes in any of the patients after the initiation of apixaban, even with the use of a higher dose of apixaban ie, 10 mg twice daily for the first five to seven days.
Anticoagulation Treatment.
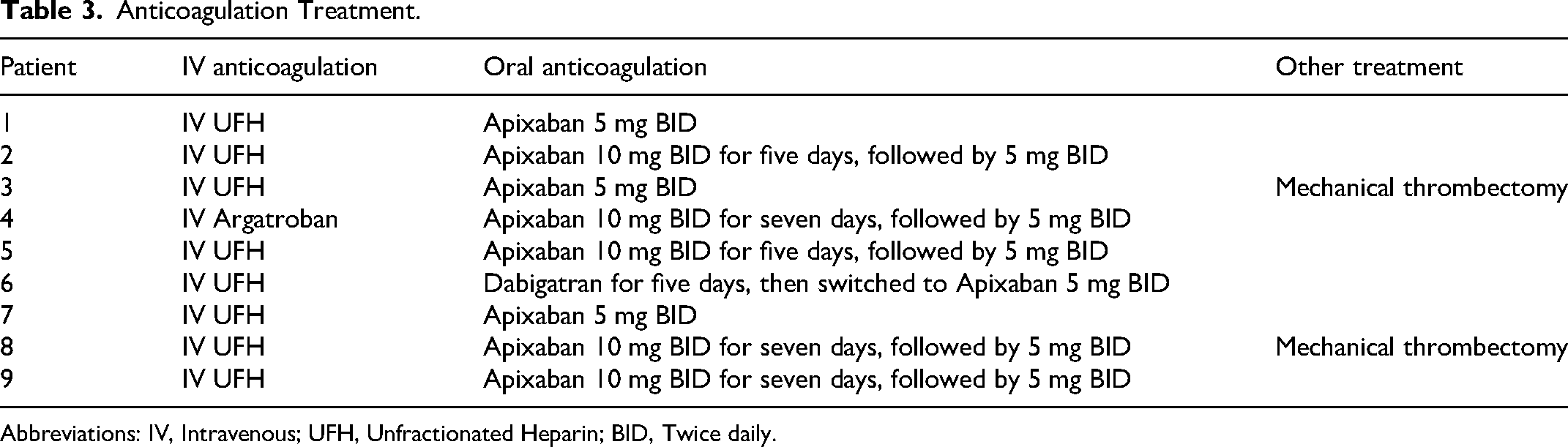
Abbreviations: IV, Intravenous; UFH, Unfractionated Heparin; BID, Twice daily.
Follow-up time ranged from six to twenty-three months. There was no progression of the CVT in any of the patients treated with apixaban. There was only one patient who developed anemia after seven months of treatment and required blood transfusion (patient number 6). This patient had a history of uterine fibroids. She was subsequently switched to aspirin as there was already recanalization on the follow-up imaging. None of the other patients reported any adverse effects of apixaban. There were no patients who experienced life-threatening hemorrhage or recurrent VTE. Complete recanalization was achieved in seven patients (78%) while two patients (22%) had partial recanalization and were still on apixaban twenty-three months and eight months after the initiation of treatment, respectively. The outcomes and adverse events are summarized in Table 4. Time to complete recanalization varied from four to sixteen months. However, many patients followed up only after several reminders from the clinic, leading to a delay in the initial follow-up imaging.
Outcome and Adverse Effects.

Abbreviation: N/a, Not applicable.
Anemia after 6 months on treatment, required 2 units of packed red blood cells.
Recanalized with underlying non-occlusive thrombus.
Discussion
CVT is an uncommon but life-threatening condition with improved outcomes and decreased mortality if correctly diagnosed and promptly treated with anticoagulation. There is limited evidence of the efficacy of DOAC in CVT and variations exist in the clinical practice because there are no large studies or randomized trials evaluating the dosage and duration with the outcomes. The AMPLIFY trial compared apixaban 10 mg twice daily for one week followed by 5 mg twice daily to enoxaparin and warfarin for the treatment of VTE (pulmonary embolism or deep venous thrombosis) and found similar efficacy with less frequent major bleeding in apixaban group. 5 To evaluate long term anticoagulation, AMPLIFY-EXT trial compared apixaban 5 mg twice daily with apixaban 2.5 mg twice daily for VTE treatment (pulmonary embolism or deep venous thrombosis) after six to twelve months of anticoagulation and found no difference. 8 However, there is no comparable trial evaluating safety and efficacy of these two doses in CVT after six to twelve months of treatment.
This case series of patients with cerebral venous thrombosis treated with apixaban demonstrated lack of recurrent thromboembolic events and no major bleeding complications. Apixaban was safe in the treatment of cerebral venous thrombosis, even in the presence of SAH and/or IPH, despite treatment with a higher dose of apixaban, ie, 10 mg twice daily for the first five to seven days in 56% of patients. Anticoagulation with apixaban in CVT prevented recurrent thromboembolic events without life-threatening bleeding complications and promoted cerebral venous recanalization in all of the patients in this case series.
Many authors have reported small case series with three to eight CVT patients treated with apixaban.9–12 Covut et al reported incomplete recanalization in their case series of five patients after the treatment with apixaban. 9 In this series, two patients (40%) achieved partial recanalization and three patients (60%) achieved no recanalization after 1–18 months follow-up. 9 Another case series by Rao et al reported complete resolution in one patient and partial resolution in two patients. 10 Rusin et al included eight patients in their case series and reported complete recanalization in 50% after three to six months of treatment. 12 Recently, a large retrospective observational study compared DOAC (271 patients on apixaban) with warfarin and found a similar rate of recurrent VTE, death, and recanalization but a lower risk of major hemorrhage. 13 The rate of partial or complete recanalization was reported to be 86% in patients on DOAC without specifying the recanalization rate for apixaban with a median follow-up of 345 days. Our case series is unique with 100% recanalization rate (78% complete recanalization and 22% partial recanalization). Complete recanalization was achieved within four to sixteen months (n: 7). The time to complete recanalization might have been shorter than reported here, as several patients had delayed imaging due to missed follow-up appointments. However, the relatively high rate of recanalization in this case series may be attributed to a longer follow-up time overall.
Conclusion
Our single-center case series is limited by its retrospective nature, small sample size, and lack of systematic follow up. It is difficult to draw any conclusions due to these limitations and a heterogeneous group. This case series adds to the emerging studies demonstrating the utility of apixaban for CVT. Larger randomized clinical trials are warranted to further evaluate the safety and efficacy of apixaban in the treatment of cerebral venous thrombosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
