Abstract
Introduction
Stroke is the leading cause of death and disability among Chinese residents, with the highest incidence globally. 1 Acute ischemic stroke (AIS) is the most common stroke type in China, accounting for more than 70% of cases.2,3 According to the widely used TOAST classification of AIS, atherosclerotic cerebral infarction is the predominant ischemic stroke cases. 4 Approximately 20%–40% of AIS patients experience early neurological deterioration, which significantly increases the risk of death and disability. 5
Restoring blood flow promptly through thrombolytic therapy is the most effective way to salvage ischemic but not infarcted brain tissue. 5 Intravenous thrombolysis (IVT) can be administered within 4.5 h of symptom onset, while mechanical thrombectomy can be performed within 24 h. 6 In addition to reperfusion therapy, antiplatelet drugs and anticoagulants prevent recurrent ischemic stroke. 7 Glycoprotein IIb/IIIa inhibitors, which have been successful in treating acute coronary syndrome, may also inhibit activated platelet-mediated thrombosis in acute stroke. 8 Tirofiban, a rapid-acting, selective glycoprotein IIb/IIIa inhibitor, with a short half-life, has shown varying benefits in AIS patients.9,10 As a platelet receptor antagonist, Tirofiban blocks platelet aggregation. However, prior studies on its use in cerebral infarction have been inconclusive. This study aims to provide a clearer understanding of Tirofiban's efficacy by analyzing its effect and exploring potential new biomarkers.
Recent studies have used complex inflammatory markers, such as neutrophil-to-lymphocyte ratio(NLR), platelet-to-lymphocyte ratio(PLR), systemic immune-inflammation index (SII), platelet-to-albumin ratio(PAR), neutrophil-to-albumin ratio(NAR), fibrinogen (FIB)-to-albumin ratio (FAR), derived NLR (dNLR), and C-reactive protein (CRP)-to-albumin ratio (CAR), to predict the cerebral infarction prognosis.11,12 However, it remains unclear whether the D-dimer/albumin ratio can predict the efficacy of tirofiban in cerebral infarction. This study aims to investigate this relationship to better guide the selection of anticoagulation therapy.
Materials and Methods
Study Subjects
This study retrospectively selected patients diagnosed with cerebral infarction through neurosurgery at our hospital between January 2021 and January 2023. The patients were divided into 4 groups: Group A: unsuitable for reperfusion therapy and within 24 h after stroke onset; Group B: unsuitable for reperfusion therapy and progressing 24-96 h after stroke onset; Group C: experienced early neurological deterioration following IVT; Group D: no neurological improvement after IVT. The decision that a patient is unsuitable for further reperfusion therapy is based on factors such as the patient's condition, physical status, time of onset and progression of symptoms. All patients received tirofiban hydrochloride sodium chloride injection, administered intravenously at a rate of 0.4 μg·kg−1·min−1 for 30 min, followed by a continuous infusion of 0.1 μg·kg−1·min−1 for 2 days. This was then sequentially combined with enteric-coated aspirin tablets and clopidogrel hydrochloride tablets (overlapping for 4 h). Detailed medical history and clinical characteristics were collected, and all patients were followed up for 90 days (a relatively short but feasible follow-up period due to the preliminary nature of the study and resource constraints). The modified Rankin scale (mRS) and the National Institutes of Health Stroke Scale (NIHSS) were used to evaluate the efficacy of tirofiban, while the modified Heidelberg bleeding classification was applied to assess the safety of symptomatic intracranial hemorrhage within 4 to 8 h after treatment.
Research Methods
Collection of Clinical Data and Laboratory Test Indicators
General information about the patients was collected, including gender, age, BMI, smoking history, drinking history, past medical history, and current medication use. Laboratory test results, including blood routine and biochemistry indicators, were obtained from our hospital's laboratory. These tests were performed using commercial ELISA kits or other testing methods routinely used by the hospital.
Collection of Efficacy and Safety Indicators
Modified Rankin Scale
The mRS is one of the most commonly used tools for evaluating stroke outcomes. It is derived from the original Rankin scale, 13 which was designed by Rankin in 1957 to evaluate stroke outcomes based on the patient's ability to live independently. The scale was initially divided into five levels. In 1988, Warlow modified the scale to enhance its comprehensiveness, resulting in the creation of the modified Rankin scale. The version evaluates a patient's ability to live independently across three categories: physical function, mobility, and daily participation. The modified scale consists of seven levels, with 0 indicating no symptoms, 5 indicating severe disability, and 6 indicating death. A change of 1 point on the scale tipically represents a significant clinical change.
NIH Stroke Scale (NIHSS)
The NIHSS is a neurological assessment tool developed by Thomos et al in 1989 for evaluating acute stroke. It consists of 15 items 14 and combines meaningful elements selected from three scales: the Toronto Stroke Scale, the Oxbury Initial Severity Scale, and the Cincinnati Stroke Scale. The NIHSS covers neurological functions associated with lesions in the major cerebral arteries. Additionally, it includes two mental status assessments, sensory function and pupillary response, and plantar reflex. The NIHSS scale provides a sensitive tool for monitoring changes in patients’ neurological function. The scale is easy to use and can be quickly mastered by medical staff after simple training. There is no significant difference in the test-retest reliability between assessors with different cultural working backgrounds. Healthcare staff can quickly and accurately assess patients’ stroke-related neurological deficits and facilitate communication between doctors about the condition. The NIHSS is the most commonly used scale for assessing stroke severity in both routine clinical practice and clinical trials.
Heidelberg Classification (HBC)
In 2015, researchers developed a formal classification standard for intracranial hemorrhage after reperfusion therapy for acute ischemic stroke based on previous classifications, known as the Heidelberg Classification (HBC). 15 This classification is based solely on imaging features, and includes three primary types: Types 1 represents hemorrhagic transformation, Type 2 indicates intracerebral hemorrhage limited to the infarct area, Type 3 encompasses parenchymal hemorrhage outside the infarcted area or intracranial-extracranial hemorrhage. Type 1 is further divided into three subtypes, 1a, 1b, and 1c, corresponding to the previous classifications of hemorrhagic infarction-1, hemorrhagic infarction-2, and parenchymal hematoma-1, respectively. Type 3 is further divided into four subtypes: 3a, defined as a parenchymal hematoma distant from the infarct area; 3b, as intraventricular hemorrhage; 3c, as subarachnoid hemorrhage; and 3d, as subdural hemorrhage. The HBC suggests that an increase in NIHSS score by ≥4 points, or a single item increase of ≥2 points on the NIHSS, indicates a change in neurological status that may be associated with poor long-term prognosis. Therefore, it is recommended that patients undergo daily NIHSS assessments for at least 5 days after reperfusion therapy. If the NIHSS score increases by ≥4 points or if a single item increases by ≥2 points, a head CT or MRI should be performed.
Statistical Analysis
Statistical analysis was performed using IBM SPSS Statistics 26.0. For continuous variables that follow normal distribution, the results are reported as mean ± standard deviation, and comparisons between groups were made using independent sample t test or analysis of variance (ANOVA). For variables that do not meet the normality or homogeneity of variance, results are reported as medians, and non-parametric tests were used to compare differences between groups. Categorical variables are reported as frequency (percentage), and chi-square test was used to analyze statistical significance between groups. The impact of biomarkers on the efficacy of tirofiban in patients with cerebral infarction was analyzed using logistic regression models. Receiver operating characteristic (ROC) curve was used to evaluate the diagnostic value of relevant biomarkers in predicting the efficacy of tirofiban. A P value less than 0.05 was considered statistically significant.
Result
Study Population
A total of 359 patients diagnosed with cerebral infarction in the neurosurgery department of our hospital were screened for this study. Among them, 59 cases did not meet the inclusion criteria, leaving 300 patients included in the study. These patients were divided into four groups: 68 patients in Group A, 73 patients in Group B, 65 patients in Group C, and 94 patients in Group D. Six patients were lost to follow-up at 90 days. Among the remaining 294 patients, efficacy evaluation at 90 days were completed either by telephone or through on-site assessment. A detailed flow chart is shown in Figure 1.

Patient enrollment flow chart.
Comparison of Baseline Clinical Characteristics among the Four Groups
The demographic information, medical history, concurrent medications, NIHSS score, ASPECTS score, and laboratory test results of each group were statistically analyzed. The results showed that there was no statistically significant difference in clinical data and laboratory test outcomes among the four groups (P > .05), Table 1.
Comparison of Baseline Clinical Characteristics among the Four Groups.

Comparison of mRS and NIHSS Scores and Adverse Events among the Four Groups at 90 Days Follow-up
Patients were followed up for 90 days, with evaluations conducted at admission, 30 days post-discharge, and 90 days post-discharge through telephone interviews or on-site visits. Follow-up assessments included the evaluation of all four groups using the mRS and NIHSS. In addition, adverse events, such as symptomatic intracranial hemorrhage and death, were collected within the 90-day period. The results are shown in Table 2, revealed no significant differences among the four groups.
Comparison of mRS and NIHSS Scores and Adverse Events among the Four Groups.

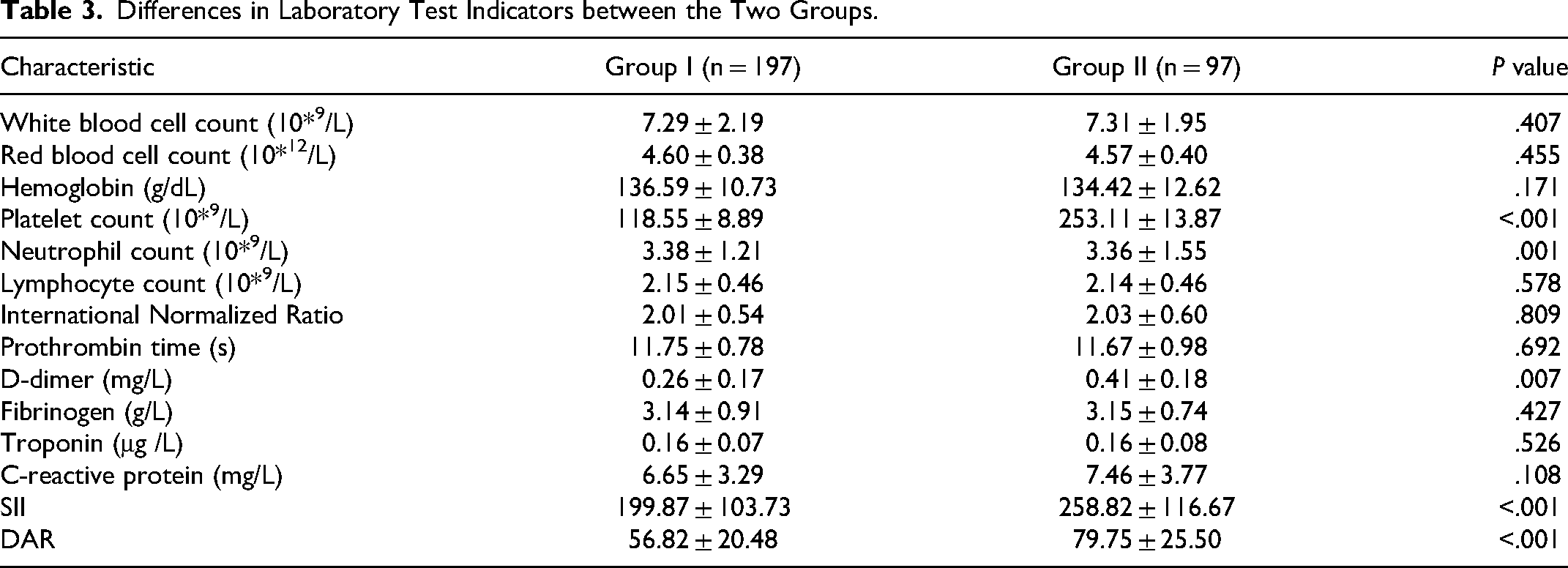
Differences in Laboratory Test Indicators
Based on the mRS scores at 90 days of follow-up, all patients were divided into two groups: Group I (scores of 0-2) and Group II (score of 3-6). Laboratory test indicators were compared between the two groups. As shown in the table below, there were significant differences in platelet count, neutrophil count, D-dimer, D-dimer/albumin ratio (DAR), and SII between the two groups. Specifically: Platelet count: Group I 118.55 ± 8.89 10* 9 /L, Group II 253.11 ± 13.87 10* 9 /L, P < .001; Neutrophil count: Group I 3.38 ± 1.21 10* 9 /L, Group II 3.36 ± 1.55 10* 9 /L, P = .001; D-dimer: Group I 0.26 ± 0.17 mg/L, Group II 0.31 ± 0.15 mg/L, P = .007; DAR : Group I 56.82 ± 20.48, Group II 79.75 ± 25.50, P < .001; SII: Group I 199.87 ± 103.73, Group II 258.82 ± 116.67, P < .001. Detailed results are shown in Table 3.
Differences in Laboratory Test Indicators between the Two Groups.
Univariate and Multivariate Logistic Regression Analysis of Laboratory Indicators and Patient Efficacy
Logistic regression was used to analyze the relationship between laboratory indicators and patient efficacy scores at the 90-day follow-up. The results showed that DAR (OR: 1.047, 95% CIs: 1.032-1.061, P < .001), SII (OR: 1.004, 95% CIs: 1.002-1.007, P < .001), and D-dimer (OR: 8.827, 95% CIs: 1.695-45.984, P = .010) levels were independent factors affecting the efficacy of tirofiban in patients with cerebral infarction (Table 4).
Logistic Analysis of Laboratory Indicators and Patient Efficacy Scores at 90-Day Follow-up.

Laboratory Indicators Alone or in Combination Predict Good Outcomes in Patients with Cerebral Infarction After Oral Tirofiban
The ROC curve was used to evaluate the ability of laboratory indicators to predict favorable outcomes in patients with cerebral infarction receiving oral tirofiban, either alone or in combination. The results showed that DAR, SII and D-dimer each have predictive value and diagnostic efficacy in assessing the efficacy of oral tirofiban in patients with cerebral infarction. Among them, the combination of DAR, SII and D-dimer exhibited the highest diagnostic efficacy. The areas under the curve (AUC) were 0.760, 0.681, 0.608, and 0.797, respectively, with all the P values < .001, indicating statistically significant differences, as shown in Figure 2.
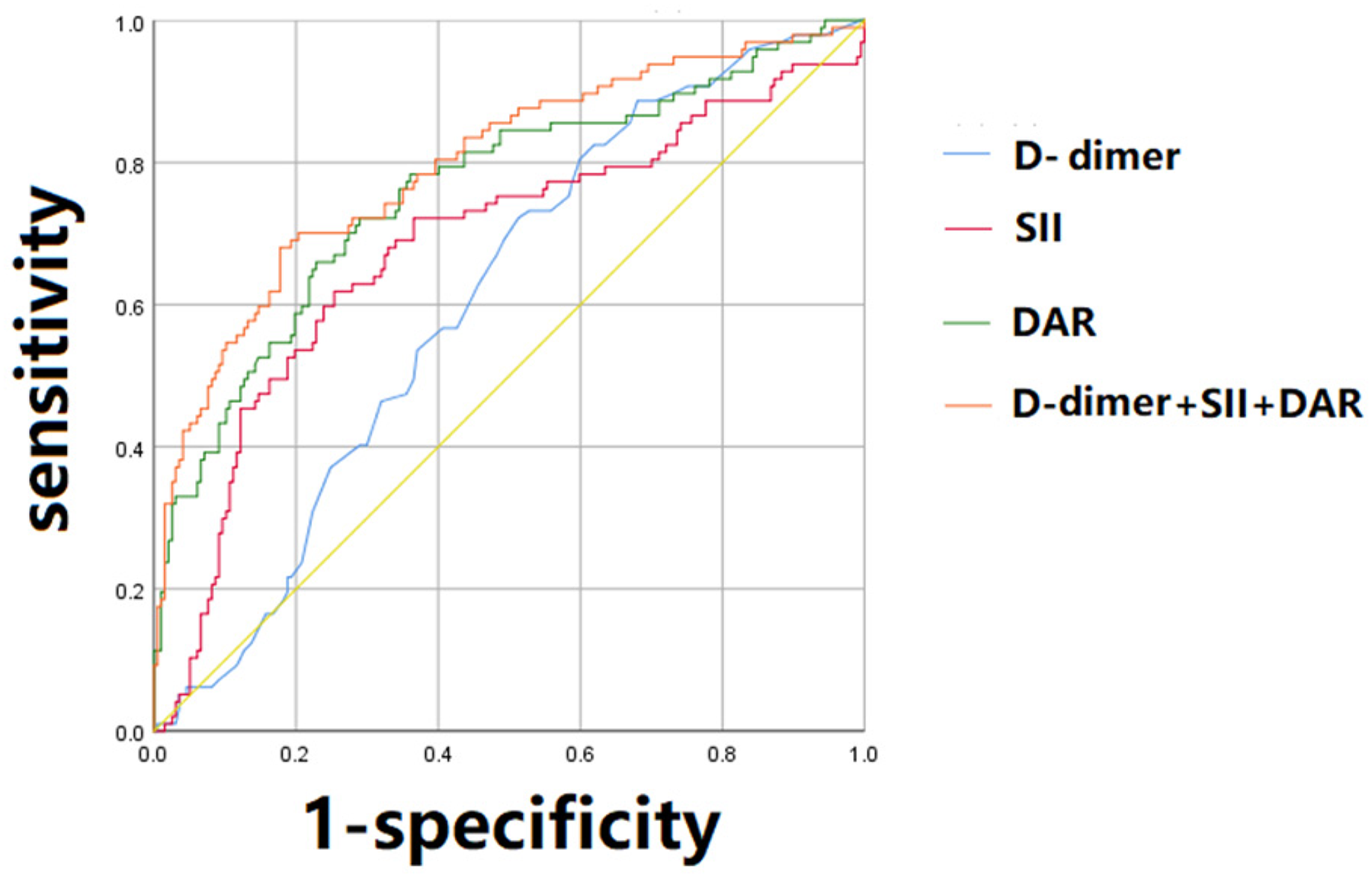
ROC curve analysis of laboratory indicators for predicting the efficacy of oral tirofiban in patients with cerebral infarction.
Discussion
Stroke is a leading cause of disability and cognitive impairment worldwide. In 2015, ischemic stroke accounted for 5.2% of all global deaths. 16 The high incidence, recurrence, and mortality of cerebral infarction place a significant burden on patients and their families. Early prediction of tirofiban's efficacy is clinically important. This study found that D-dimer, DAR, and SII may be associated with treatment outcomes, providing valuable insights for future treatment strategies in infarction patients.
Tirofiban has been demonstrated efficacy in the treatment of acute ischemic stroke and is increasingly used in the clinical practice.17,18 However, its effectiveness in treating cerebral infarction remains a topic of controversy. With the growing body of research and new clinical trials, it has become evident that not all patients benefit from tirofiban. In a randomized study of cerebral vascular occlusion, 90-day outcomes were similar for patients given tirofiban before thrombectomy and those given a placebo. 19 Additionally, Kleinschnitz et al compared the pathogenic effects of GPIb, VI, and IIb/IIIa receptors in a mouse model of transient middle cerebral artery occlusion. The results suggested that selectively inhibition of the GPIIb /IIIa receptor, the final common pathway of platelet aggregation, did not significantly reduce infarct volume or improve prognosis. Instead, it was associated with an increased risk of cerebral hemorrhage and higher mortality rates. 20 Therefore, it is of great clinical significance to identify a simple, convenient, specific, and cost-effective laboratory marker to predict the efficacy of tirofiban in patients with cerebral infarction.
D-dimer is a substance produced by the degradation of fibrin by plasmin during the coagulation process. 21 Increased D-dimer levels may reflect thrombosis formation/dissolution and serve as a key noninvasive marker for predicting thrombosis. 22 Currently, D-dimer has been widely used to predict diseases such as deep vein thrombosis, cerebral hemorrhage, and acute aortic dissection.23,24 Previous studies have shown that patients with cerebral infarction who have high D-dimer levels tend to have poor outcomes. This is similar to the results of this study. Patients with higher D-dimer levels had a relatively high modified Rankin score at 90 days. However, although D-dimer has a high sensitivity, its specificity is relatively low, and its level in the body increases with age. Given that patients with cerebral infarction are often older, D-dimer remains a useful marker for predicting clinical efficacy in this patients population.
Inflammation increases capillary permeability, causing dilation of the interstitial space and expansion of albumin distribution, which can lead to hypoalbuminemia. 25 Since albumin is exclusively produced in the liver, low albumin levels in the body may also indicate insufficient liver function and a deficiency of anticoagulant factors. 26 Therefore, SII and albumin levels may play a significant role in thrombosis. 27 DAR is a novel combination selected in this study and applied for the first time to predict the efficacy of tirofiban in patients with cerebral infarction. D-dimer, SII, and albumin are the most common laboratory test indicators in clinical practice, providing a simple and convenient way for predicting treatment efficacy. This study found that patients with poor efficacy from tirofiban after cerebral infarction generally had higher DAR, SII, and D-dimer levels. Further analysis revealed that these markers are independent predictors of treatment efficacy, demonstrating significant predictive value and diagnostic performance. Additionally, this study is the first to compare the effects of different stages and types of cerebral infarction on the efficacy of tirofiban by using the mRS and NIHSS and adverse events among patients in different groups. The results showed that there was no significant difference among the four groups, which further verified the universality of DAR, SII, and D-dimer as biomarkers for predicting the efficacy of tirofiban.
However, this study has certain limitations. First, it is based on a small sample size, and small samples are more susceptible to random error, which can reduce the reliability of the findings. Secondly, the study was conducted at a single-center hospital, which may limit the generalizability of the results to other geographic regions or hospital settings. Finally, we chose both the mRS and the NIHSS as scales based primarily on subjective criteria. Despite some controversies and inconsistent results in the literature regarding the effectiveness of tirofiban in the treatment of cerebral infarcts, these inconsistencies highlight the need for further in-depth study to clarify its efficacy and the specific conditions for which it is indicated. Therefore, future research should involve prospective, multicenter, and large-sample studies to better evaluate the role of D-dimer, DAR, and SII as predictors of tirofiban efficacy.
Conclusions
Laboratory markers DAR, SII and D-dimer are effective diagnostic indicators for predicting the efficacy of tirofiban in patients with cerebral infarction. These markers can assist clinicians in selecting appropriate treatment strategies for patients with cerebral infarction, ultimately helping to improve patients outcomes and reduce the economic burden on patients.
Footnotes
Acknowledgments
The authors would like to thank the study participants and clinical staff for their support and contributions to this study.
Data Availability
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The study was approved by the Ethics Committee of Heji Hospital Affiliated to Changzhi Medical College (HJCMC-2020-1206), and it was conducted in accordance with the ethical standards as laid out in the 1964 Declaration of Helsinki. Informed consent was obtained from all individual participants included in the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
