Abstract
Acute cerebral infarction (ACI) includes atherosclerotic and cardiogenic ACI and involves a thrombotic state, requiring antithrombotic treatment. However, the thrombotic state in ACI cannot be evaluated using routine hemostatic examinations. Plasma soluble C-type lectin-like receptor 2 (sCLEC-2) and D-dimer levels were measured in patients with ACI. Plasma sCLEC-2 and D-dimer levels were significantly higher in patients with ACI than in those without it. The sCLEC-2 × D-dimer formula was significantly higher in patients with ACI than in those without it. A receiver operating characteristic curve showed a high sensitivity, area under the curve, and odds for diagnosing ACI in the sCLEC-2 × D-dimer formula. Although the sCLEC-2 and D-dimer levels were useful for the differential diagnosis between cardiogenic and atherosclerotic ACI, the sCLEC-2 × D-dimer formula was not useful. sCLEC2 and D-dimer levels are useful for the diagnosis of ACI and the sCLEC2 × D-dimer formula can enhance the diagnostic ability of ACI, and sCLEC2 and D-dimer levels may be useful for differentiating between atherosclerotic and cardioembolic ACI.
Keywords
Introduction
Ischemic stroke (acute cerebral infarction [ACI])1,2 is classified into 3 types: atherosclerotic, 3 cardioembolic, 4 and lacunar 5 ACI. In addition, a few transient ischemic attacks (TIAs), which resolve within 24 h, are included in ACI. 6 Although ACI is usually diagnosed using computed tomography, magnetic resonance imaging, and angiography in core hospitals, the differential diagnosis between cardioembolic ACI and atherosclerotic or lacunar ACI may be difficult. As ACI patients have a thrombotic state, they are usually treated with anti-thrombotic therapy such as antiplatelet agents,1,7 direct anti-thrombin agents 8 for atherosclerotic ACI, and unfractionated heparin, warfarin, or direct oral anticoagulants (DOACs) 9 for cardioembolic ACI. Most Japanese patients with atherosclerotic ACI are generally treated with argatroban, 8 followed by those with atherosclerotic or lacunar ACI, or those with TIA, who are usually treated with aspirin or other antiplatelet agents,1,7 as platelet activation may play an important role in atherosclerosis resulting in ACI or acute myocardial infarction 10 ; in contrast, patients with cardioembolic ACI should be treated with warfarin or DOACs 9 for a hypercoagulable state. 11
Although acute myocardial infarction which is arterial thrombosis as well as ACI, is suggested by laboratory test such as troponin T, 12 or creatine kinase, muscle and brain, 13 no biomarker suggest the onset of ACI. Venous thromboembolism which is venous thrombosis, is also suggested by elevation of D-dimer,14,15 suggesting that biomarker for platelet activation may be useful for the diagnosis of ACI. As biomarkers for platelet activation, platelet factor 4 (PF4),16 β-thromboglobulin (β-TG), 17 and P-selectin 18 have been reported; however, their actual diagnostic specificity for thrombosis due to platelet activation is not high, and they have limited use in clinical laboratories. Platelet activation can be evaluated to detect substances, such as P-selectin 19 or phosphatidylserine, 20 on the platelet membrane using flow cytometry. However, this method using flow cytometry is not a routine laboratory examination. Platelet–leukocyte aggregates 21 are not quantitative. Microparticles containing tissue factors (TFs) from platelets22 are still being researched. Soluble C-type lectin-like receptor 2 (sCLEC-2) has been introduced as a new biomarker for platelet activation.23,24 Elevated sCLEC-2 levels have been reported in patients with thrombotic microangiopathy (TMA) 25 and disseminated intravascular coagulation (DIC), 26 as well as in patients with acute coronary syndrome27,28 and ACI.29,30 The super formula of sCLEC-2 × D-dimer was recently reported to increase the diagnostic ability for DIC. 31
In the present study, plasma sCLEC-2 and D-dimer levels, which were biomarkers for coagulation and platelet activation, were measured in 120 patients with ACI and 130 patients without ACI to examine the usefulness of the sCLEC-2, D-dimer, sCLEC-2 × D-dimer formula, sCLEC-2/D-dimer ratio, and sCLEC-2/platelet count (PLT) ratio for diagnosing ACI and differentiating between atherosclerotic and cardioembolic ACI.
Materials and Methods
Plasma sCLEC-2 and D-dimer levels were measured in 120 patients with ACI, 70 patients with chronic liver disease (
Subjects.
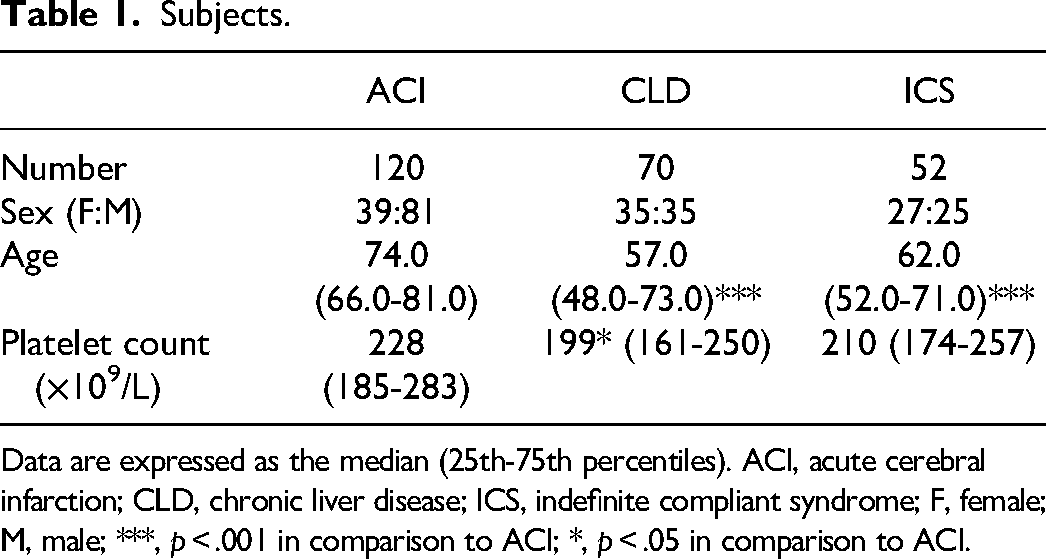
Data are expressed as the median (25th-75th percentiles). ACI, acute cerebral infarction; CLD, chronic liver disease; ICS, indefinite compliant syndrome; F, female; M, male; ***, p < .001 in comparison to ACI; *, p < .05 in comparison to ACI.
Acute Cerebral Infarction and Syncope.
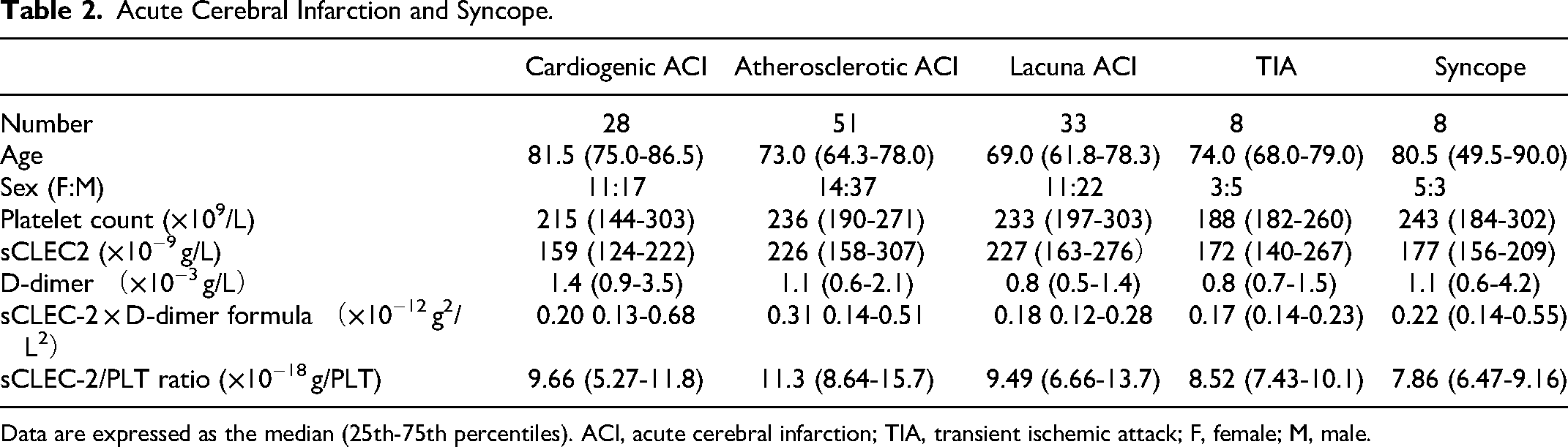
Data are expressed as the median (25th-75th percentiles). ACI, acute cerebral infarction; TIA, transient ischemic attack; F, female; M, male.
Plasma was prepared by 2 centrifugations at 3000 rpm for 15 min (platelet count was ≤0.5 × 1010/L). Plasma sCLEC-2 levels were measured by a chemiluminescent enzyme immunoassay (CLEIA) using previously described monoclonal antibodies and the STACIA-CLEIA system (LSI Medience, Tokyo, Japan).23,24,26 In brief, magnetic particles were coated with the anti-CLEC-2 monoclonal antibody 11D5. The plasma samples were then incubated with antibody-coated magnetic particles, and after being washed, they were incubated further with the alkaline-phosphatase-labeled anti-CLEC-2 monoclonal antibody 11E6. After being washed again, the magnetic particles were incubated with chemiluminescent substrate solution (CDP-Star; Applied BioSystems) and the luminescence was measured using the luminometer installed in the STACIA system. D-dimer levels were measured using the LPIA-Genesis devices (LSI Medience) with the STACIA system (LSI Medience).
Statistical Analyses
Data are expressed as the median (25th-75th percentiles). The significance of the differences between groups was examined using the Mann–Whitney U-test. The cut-off values, areas under the curve (AUCs), sensitivity, specificity, and odds ratios were determined using a receiver operating characteristic (ROC) analysis. In this study of our statistical analysis system, the cut-off value was determined to be the point at which the sensitivity curve crossed the specificity curve. Statistical significance was set at p < .05. All statistical analyses were performed using the Stat-Flex software program (Version 7; Artec Co Ltd, Osaka, Japan).
Results
The patients with ACI (median, 74.0 years old; 25th-75th percentile, 66.0-81.0 years old) were significantly older (p <
The platelet count in patients with CLD (median, 199 × 109/L; 25th-75th percentile, 48.0-73.0 × 109/L) was significantly lower (p < .05) than in patients with ACI (median, 228 × 109/L; 25th-75th percentile, 185-283 × 109/L) and ICS (median, 210 × 109/L; 25th-75th percentile, 174-257 × 109/L) (Table 1); however, there was no significant difference in platelet counts among cardiogenic ACI, atherosclerotic ACI, lacuna ACI, TIA, and syncope patients (Table 2).
The plasma sCLEC-2 levels in patients with ACI (median, 216 × 10−9 g/L; 25th-75th percentile, 150-284 × 10−9 g/L) were significantly higher (p < .001) than in patients with CLD (median, 110 × 10−9 g/L; 25th-75th percentile, 91.9-136 × 10−9 g/L) and in patients with ICS (median, 113 × 10−9 g/L; 25th-75th percentile, 98.7-172 × 10−9 g/L), and the plasma D-dimer levels in patients with ACI (median, 1.1 × 10−3 g/L; 25th-75th percentile, 0.6-1.9 × 10−3 g/L) were significantly higher (p < .001, respectively) than in patients with CLD (median, 0.4 × 10−3 g/L; 25th-75th percentile, 0.3-0.5 × 10−3 g/L) and in patients with ICS (median, 0.4 × 10−3 g/L; 25th-75th percentile, 0.3-0.6 × 10−3 g/L) (Table 3 and Figure 1).
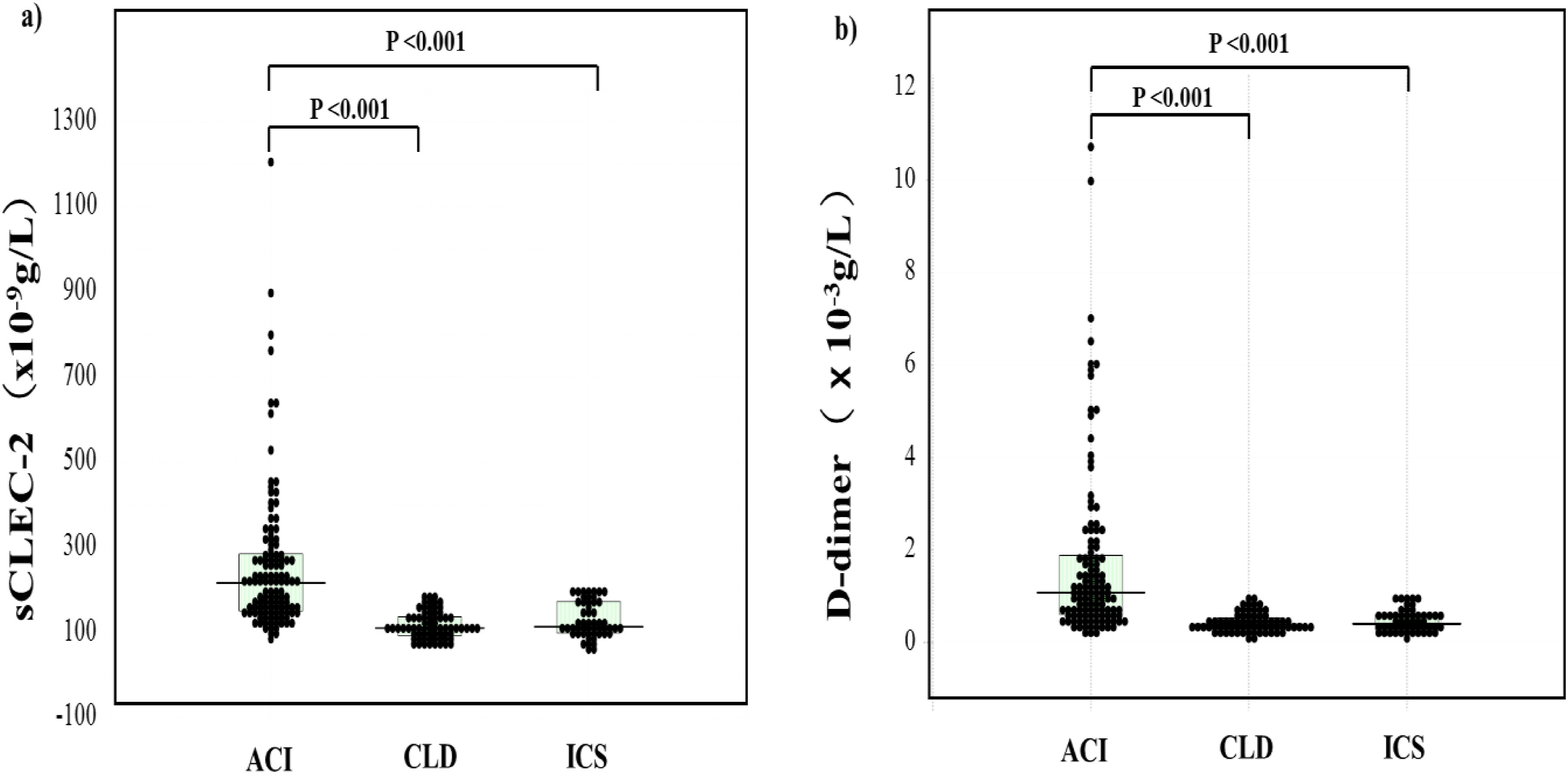
Plasma levels of sCLEC-2 (a) and D-dimer (b) in patients with acute cerebral infarction, patients with chronic liver disease and patients with indefinite complaint syndrome. ACI, acute cerebral infarction; CLD, chronic liver disease; ICS, indefinite complaint syndrome; sCLEC-2, soluble C-type lectin-like receptor 2.
sCLEC-2, D-Dimer and sCLEC-2 × D-Dimer in Patients with ACI, CLD or UCS.

Data are expressed as the median (25th-75th percentiles). ACI, acute cerebral infarction; CLD, chronic liver disease; ICS, indefinite compliant syndrome; ***, p < .001 in comparison to ACI; sCLEC-2, soluble C-type lectin-like receptor 2.
The plasma sCLEC-2 and D-dimer levels were significantly higher (p < .001) in patients with ACI than in those without it (median, 112 × 10−9 g/L; 25th-75th percentile, 94.5-144 × 10−9 g/L and median, 0.4 × 10−3 g/L; 25th-75th percentile, 0.3-0.5 × 10−3 g/L, respectively) (Table 4 and Figure 2). There were no significant differences in sCLEC-2 and D-dimer levels between patients with CLD and ICS.
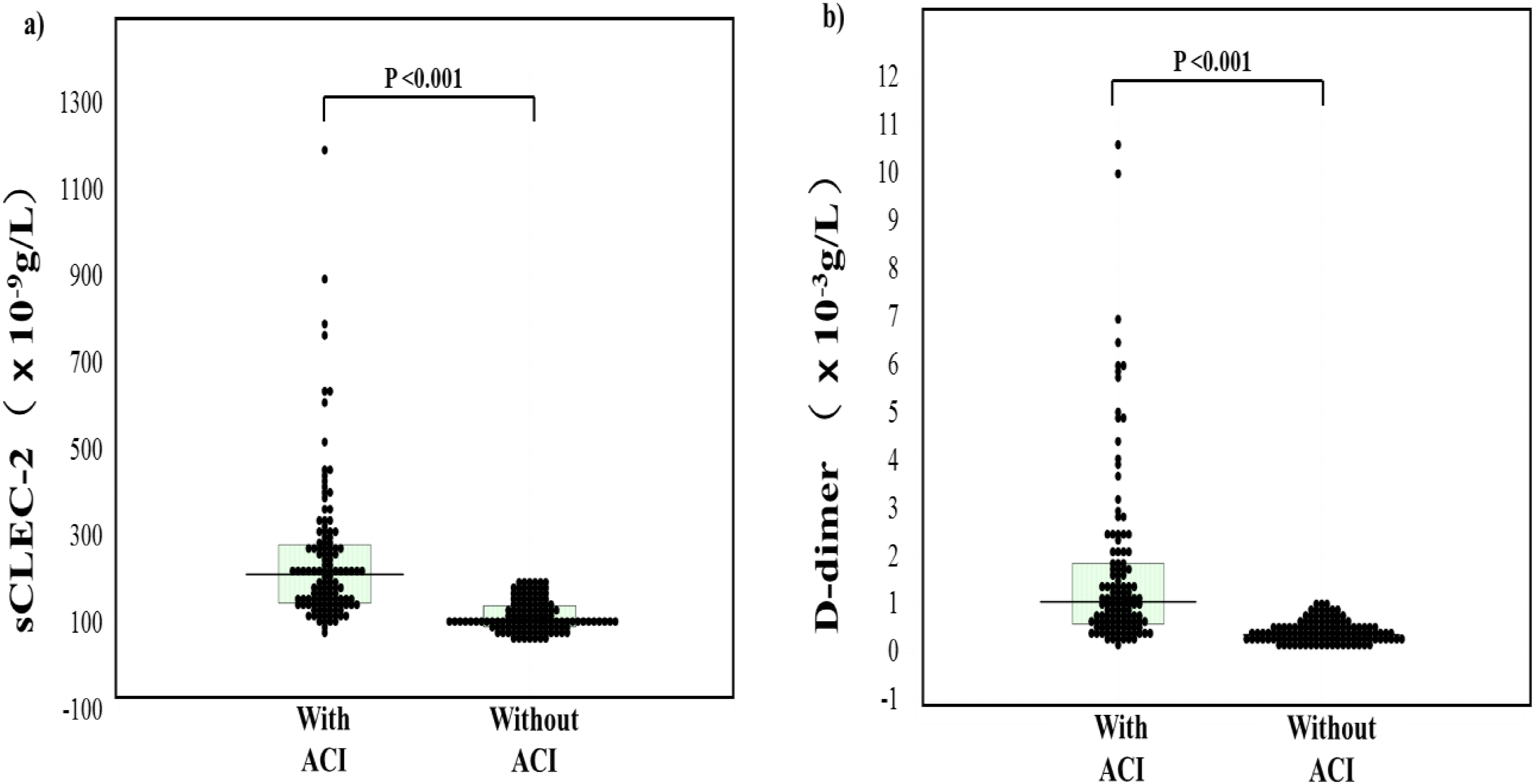
Plasma levels of sCLEC-2 (a) and D-dimer (b) in patients with and without acute cerebral infarction. ACI, acute cerebral infarction; sCLEC-2, soluble C-type lectin-like receptor 2.
sCLEC-2, D-Dimer, sCLEC-2 × D-Dimer and sCLEC-2/PLT Ratio Levels in Patients with and Without ACI.

Data are expressed as the median (25th-75th percentiles). ACI, acute cerebral infarction; sCLEC-2, soluble C-type lectin-like receptor 2; PLT, platelet; ***, p < .001.
The sCLEC-2 × D-dimer formula in patients with ACI (median, 0.22 × 10−9 g/L; 25th-75th percentile, 0.13-0.45 × 10−9 g/L) was significantly higher (p < .001) than in patients with CLD (median, 0.04 × 10−9 g/L; 25th-75th percentile, 0.03-0.07 × 10−9 g/L) and in those with ICS (median, 0.06 × 10−9 g/L; 25th-75th percentile, 0.03-0.09 × 10−9 g/L) (Table 3 and Figure 3). The sCLEC-2 × D-dimer formula was significantly higher (p < .001) in patients with ACI than in those without it (median, 0.06 × 10−9 g/L; 25th-75th percentile, 0.03-0.09 × 10−9 g/L) (Table 4 and Figure 3). There were no significant differences in sCLEC-2 × D-dimer formula between patients with CLD and ICS. The sCLEC-2/D-dimer ratio in patients with ACI were significantly lower (median, 179; 25th-75th percentile, 102-363) than in those without it (median, 304; 25th-75th percentile, 194-435) (p < .001). The sCLEC-2/PLT ratio was significantly higher in patients with ACI than in those with CLD or ICS (p < .001, respectively) (Table 2 and Figure 4) and in patients with ACI than in those without it (Figure 4).
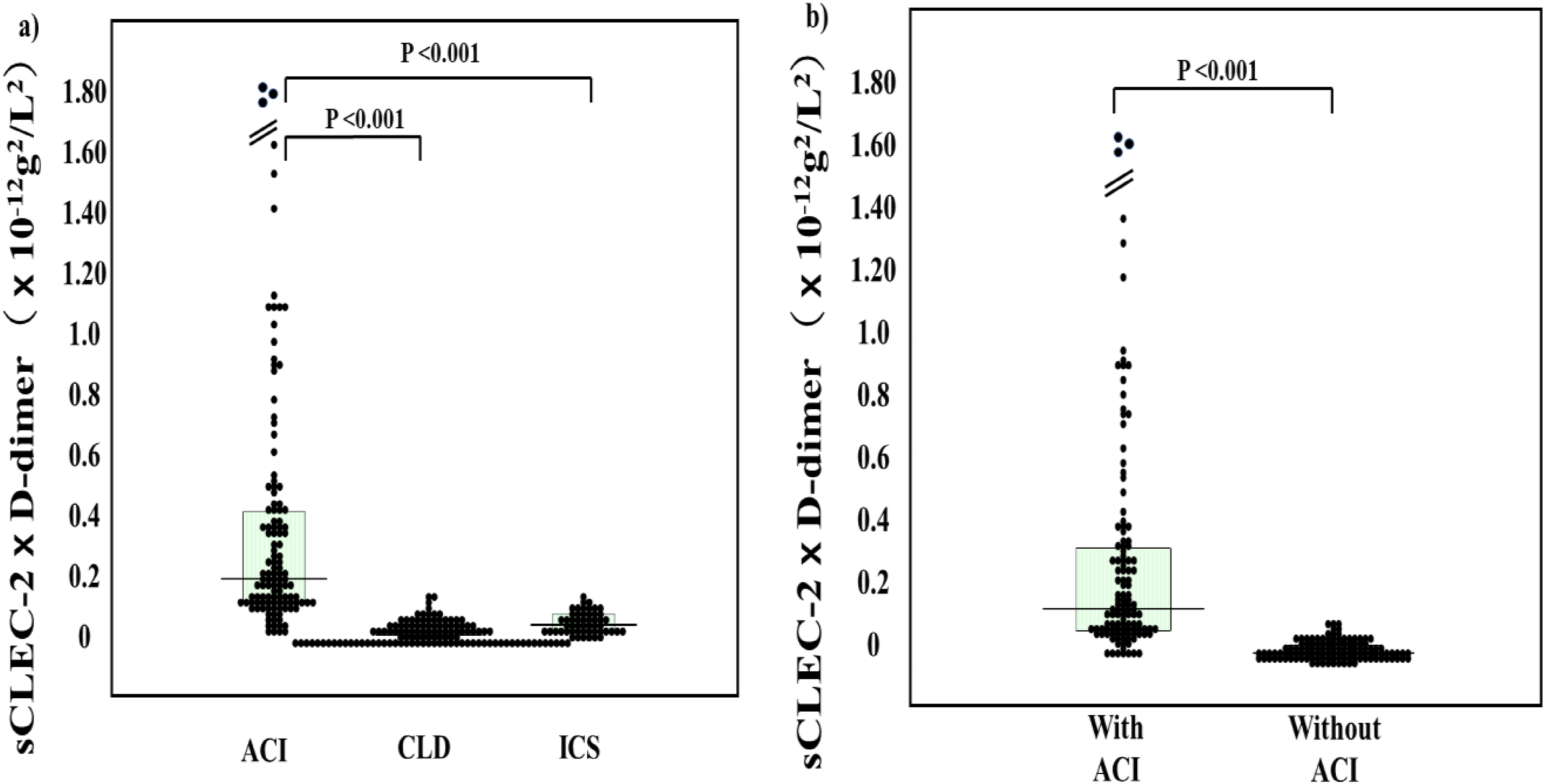
Plasma levels of sCLEC-2 × D-dimer in patients with ACI, those with chronic liver disease and those with ICS (a); and in patients with and without ACI (b). ACI, acute cerebral infarction; CLD, chronic liver disease; ICS, indefinite complaint syndrome; sCLEC-2, soluble C-type lectin-like receptor 2.
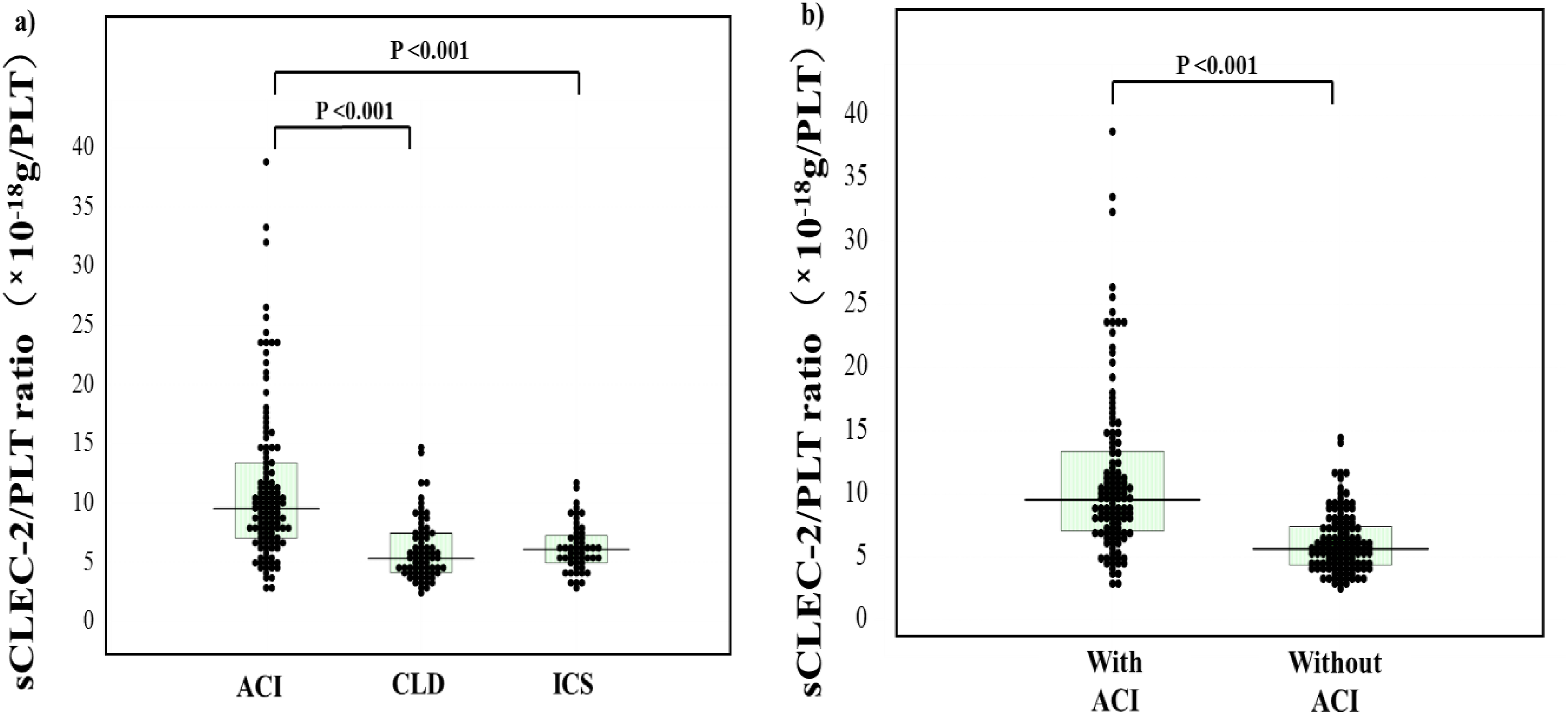
The sCLEC-2/PLT ratio in patients with ACI, those with chronic liver disease and those with ICS (a); and those with and without ACI (b). ACI, acute cerebral infarction; CLD, chronic liver disease; ICS, indefinite complaint syndrome; sCLEC-2, soluble C-type lectin-like receptor 2; PLT, platelet.
Regarding the ROC analysis for patients with ACI versus those without ACI, the cut-off value was 149 × 10−9 g/L in sCLEC-2, 0.6 × 10−3 g/L in D-dimer and 0.10 × 10−9 g/L in sCLEC-2 × D-dimer formula. Although the sensitivity, AUC and odds ratio of the sCLEC-2 or D-dimer level alone, or the sCLEC-2/PLT ratio were <80%, <0.9, and <15.0, respectively, the sensitivity, AUC and odds ratio of sCLEC-2 × D-dimer formula were 88.5%, 0.942, and 61.7, respectively. In contrast, the AUC of sCLEC-2/D-dimer ratio was 0.647 for patients with ACI versus those without it (Table 5 and Figure 5). In ACI, the sCLEC-2 levels in patients with atherosclerotic and lacuna ACI were significantly higher (p < .01 and p < .05, respectively) than in patients with cardiogenic ACI (Figure 6a and Table 2). The D-dimer levels in patients with cardiogenic ACI were significantly higher (p < .05 and p < .01, respectively) than in patients with atherosclerotic and lacuna ACI (Figure 6b). There were no significant difference in the sCLEC-2 × D-dimer formula among cardiogenic ACI, atherosclerotic ACI, lacuna ACI, TIA and syncope patients (Figure 6c). The sCLEC-2 levels in patients with atherosclerotic ACI (median, 191 × 10−6 g/L; 25th-75th percentile, 107-355 × 10−9 g/L) and lacuna ACI (median, 298 × 10−6 g/L; 25th-75th percentile, 156-538 × 10−6 g/L) were significantly higher (p < .05 and p < .001, respectively) than in patients with cardiogenic ACI (median, 105 × 10−6 g/L; 25th-75th percentile, 61.0-162 × 10−6 g/L, Figure 6d). The sCLEC-2/PLT ratio was significantly higher in patients with atherosclerotic ACI than in those with cardiogenic ACI or syncope (p < .05) (Figure 6e).
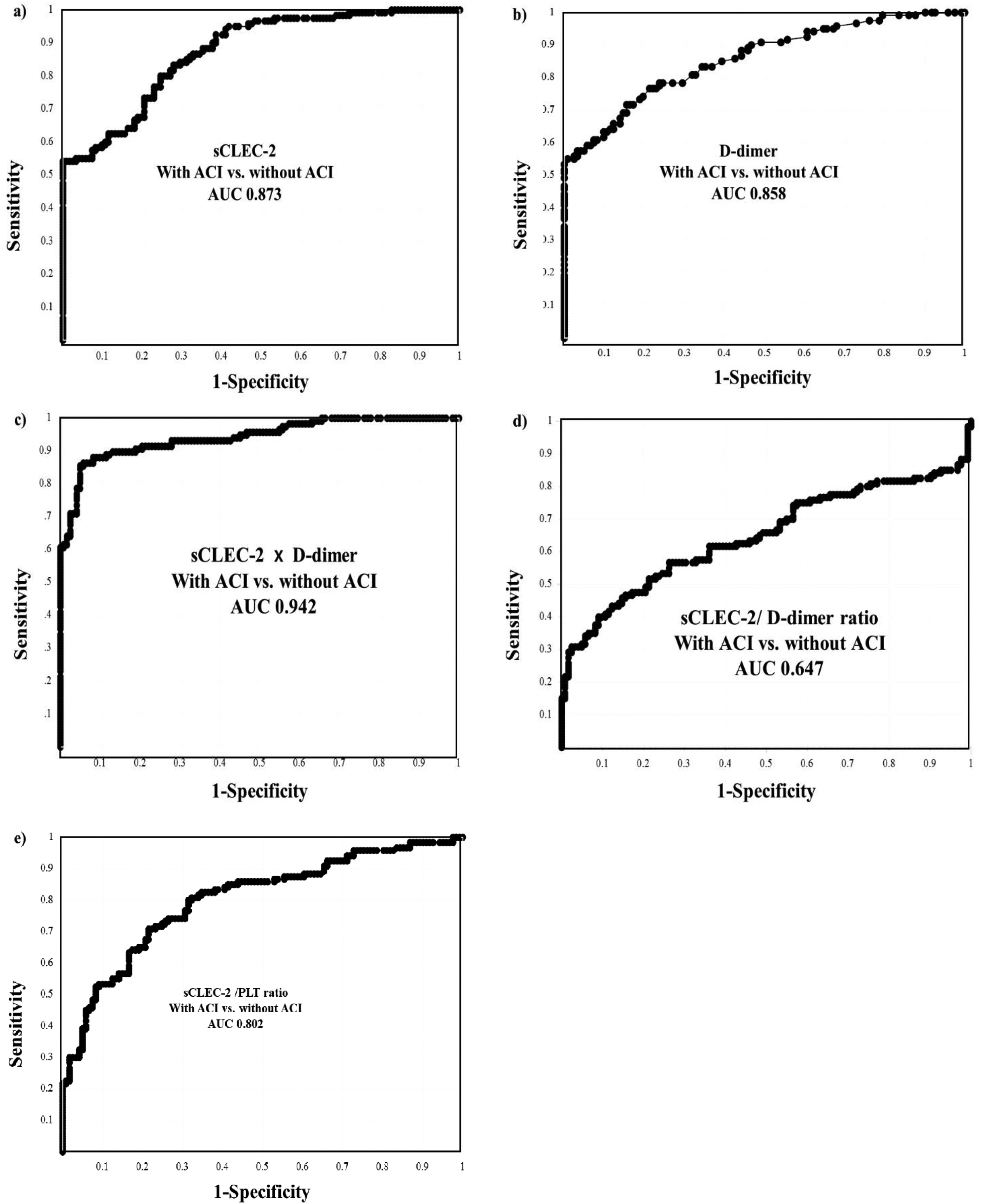
A receiver operating analysis of sCLEC-2 (a), D-dimer (b), sCLEC-2 × D-dimer formula (c), sCLEC-2/D-dimer ratio and sCLEC-2/PLT ratio for patients with acute cerebral infarction (d) versus patients without. ACI, acute cerebral infarction; sCLEC-2, soluble C-type lectin-like receptor 2; PLT, platelet; AUC, area under the curve.
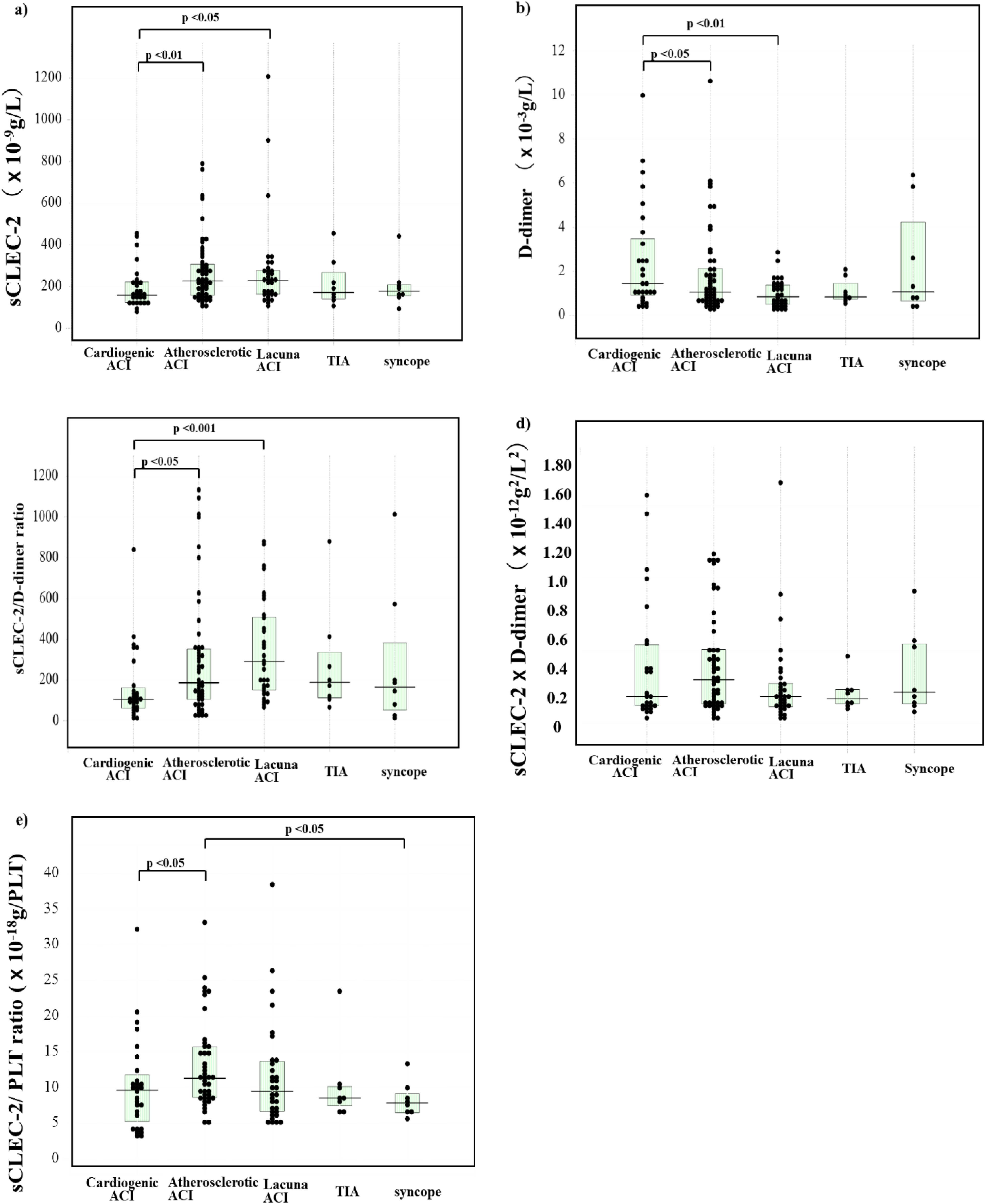
Plasma levels of sCLEC-2 (a), D-dimer (b), sCLEC-2 × D-dimer (c) and sCLEC-2/D-dimer in patients with cardiogenic ACI, patients with atherosclerotic ACI, patients with lacuna, patients with TIA and patients with syncope. ACI, acute cerebral infarction; TIA, transient ischemic attack; sCLEC-2, soluble C-type lectin-like receptor 2.
An ROC Analysis of sCLEC-2, D-Dimer, sCLEC-2 × D-Dimer, sCLEC-2/D-Dimer and sCLEC-2/PLT Ratio Values for the Diagnosis of ACI.

ROC, receiver operating characteristic; ACI, acute cerebral infarction; sCLEC-2, soluble C-type lectin-like receptor 2; PLT, platelet; AUC, area under the curve.
Discussion
ACI occurs when atherosclerotic thrombosis or cardiogenic embolism occludes a cerebral vessel supplying a specific area of the brain resulting in loss of the neurological function. 1 Atherosclerotic thrombosis is caused by atherosclerotic plaque disruption which is the predominant pathogenetic mechanism underlying ACI and acute coronary syndrome. 32 Plaque rupture leads to the exposure of collagen and vessel media including tissue factor and plasminogen activator inhibitor, resulting in platelet and clotting activation, and occlusive thrombus formation. Platelet activation leads to a high rate of re-occlusion of brain tissue.32,33 Therefore, antiplatelet agents have mainly been used for atherosclerotic ACI.34,35 ACI is reportedly associated with hypercoagulability in coronavirus diseases 2019 (COVID-19) 36 and platelet activation has also been reported in COVID-19.37,38 It is recently proposed that platelet activation may cause thrombosis in patients with COVID-19. 39 Cardioembolic ACI is caused by emboli mainly from cardiac thrombi due to hypercoagulability. 40 Therefore, patients with cardioembolic ACI patients are mainly treated with anticoagulant therapy.
ACI is still a fatal disease and often causes severe complications such as hemiplegia, decreased quality of life, and dementia. Thus, an early diagnosis and treatment are required for its management. Regarding routine hemostatic tests, fibrin-related products (FRPs) such as D-dimer, soluble fibrin (SF), and fibrinogen and fibrin degradation products (FDPs) are sensitive but non-specific biomarkers for diagnosing PE/DVT 41 and DIC. 26 Although clot waveform analyses for activated partial thromboplastin time (APTT) and a small amount of TF-induced clotting factor IX activation assay can detect hypercoagulability in ACI patients, routine APTT and prothrombin time cannot detect hypercoagulability. 11 Although plasma D-dimer levels are high in patients with ACI, particularly, cardioembolic ACI, they were low in patients with atherosclerotic or lacunar ACI in this study. Plasma sCLEC-2 levels were extremely high in patients with ACI, particularly, in atherosclerotic and lacuna ACI patients, suggesting that elevation of sCLEC-2 may be useful for diagnosing ACI. 30 However, sCLEC-2 levels were not significantly elevated in patients with cardioembolic ACI. The elevation of sCLEC-2 levels alone may miss cardioembolic ACI in diagnosing ACI.
Regarding diagnosing platelet activation, there are many proposals such as detecting substance due to platelet activation using a flow cytometry,19,20 platelet aggregation using microscopic findings 21 or spontaneous echocardiographic contrast, 42 microparticles using a flow cytometry or enzyme immunosorbent assay (ELISA), 18 PF4 and β-TG16,17 using ELISA. Human platelets and megakaryocytes highly and almost specifically express a CLEC-2 protein on their cell membrane. 23 Although PF4 and β-TG are contained in the α-granule of platelets and are used as classical platelet activation markers, they are easily released upon minimal platelet activation that occurs during sampling and require plasma mixed with citrate.16,17 Furthermore, special techniques are required for blood sampling and sample preparation for PF4 and β-TG measurement, whereas CLEC-2 is a platelet membrane protein, while sCLEC-2 is derived from membrane shedding and microvesicles. sCLEC-2 measurement has several advantages over classical platelet activation markers; its measurement can be used in standard blood collection procedures performed in daily clinical laboratory tests. 43 Patients with ACI sometimes associates with recurrence of stroke or major bleeding 44 suggesting that an adequate antiplatelet therapy is important. Therefore, many physicians want to know the efficacy of antiplatelet therapy for patients with ACI to prevent the recurrence of ACI or bleeding. However, a platelet function test 45 such as the test for platelet aggregation is not easy and time consuming. Therefore, most patients treated with antiplatelet agent are not usually monitored by platelet function test. As the measurement of sCLEC-2 is easy and rapid assay, the patients with ACI treated with antiplatelet agent may able to be monitored by sCLEC-2 level. Future study for sCLEC-2 on patients with ACI will be required the behavior of sCLEC-2 levels in clinical course of ACI and the relationship between sCLEC-2 levels and outcome of ACI including worsening, recurrence, or cerebral bleeding. The sCLEC-2/PLT ratio (called the C2PAC index) was used to determine sCLEC2 concentrations per platelet unit and was considered to be an index of platelet activation that is a useful predictor of the progression and diagnosis of sepsis-induced DIC (SID) in patients with sepsis. 46 Although the sCLEC-2/PLT ratio was useful in patients with thrombocytopenia, this ratio was not superior to sCLEC-2 alone in this study without thrombocytopenia. Similarly, the sCLEC-2 × D-dimer/PLT formula was useful for diagnosing DIC 31 ; however, this formula was not more useful than the sCLEC-2 × D-dimer formula in patients without thrombocytopenia.
As the onset mechanisms between atherosclerotic and cardioembolic ACI differ, 47 the treatments for the 2 types of ACI are also different, suggesting that a differential diagnosis between atherosclerotic and cardioembolic ACI is urgently required. However, it can be difficult to differentiate between these 2 types of ACI using CT or MRI. The present study showed that plasma sCLEC2 levels were high in atherosclerotic and lacuna ACI, whereas plasma D-dimer levels were high in cardioembolic ACI, resulting in a higher sCLEC-2/D-dimer ratio in atherosclerotic and lacuna ACI than in cardioembolic ACI. The sCLEC-2/D-dimer ratio enhanced the difference not only between atherosclerotic and cardioembolic ACI but also between venous and atherosclerotic thromboses. 30 In contrast, the sCLEC-2 × D-dimer formula eliminated the difference between atherosclerotic and cardioembolic ACI, resulting in an increased diagnostic ability for total ACI including atherosclerotic and cardioembolic ACI. Furthermore, the sCLEC-2 × D-dimer formula is not useful for differential diagnosis for DIC from infectious diseases, 38 other thrombotic diseases such as venous thromboembolism. DIC 26 and TMA. 25 The super formula of the sCLEC-2 × D-dimer/platelet count also increases the diagnostic ability of DIC and pre-DIC. 31 These findings suggest that the super formula of the sCLEC-2 × D-dimer can diagnose both platelet activation and hypercoagulability. Several formulae have been developed for diagnosing thrombosis; the sCLEC-2 × D-dimer formula may indicate the activation of coagulation, platelets, and fibrinolysis; the sCLEC-2/PLT ratio may indicate platelet activation and consumption but does not indicate coagulation and fibrinolysis; the sCLEC-2 × D-dimer/PLT formula includes the activation of coagulation, platelets, and fibrinolysis with the consumption of clotting factors48,49; and the sCLEC-2/D-dimer ratio may be useful for making the differential diagnosis between arterial and venous thrombosis. However, these formulae are limited to use for specific underlying diseases; the sCLEC-2/PLT ratio and the sCLEC-2 × D-dimer/PLT are useful for thrombocytopenia, whereas the sCLEC-2 × D-dimer and sCLEC-2 × D-dimer/PLT formulae are useful for thrombotic diseases.
In Japan, soluble C-type lectin-like receptor 2 in stroke (CLECSTRO) study 50 for diagnosing ACI and differentiating atherosclerotic ACI from cardioembolic ACI using sCLEC-2 and D-dimer has been ongoing in multicenter. The establishment of super formula of the sCLEC-2 × D-dimer in diagnosing ACI may change the protocol of CRECSTRO study to increase the diagnostic ability for ACI using biomarker.
In limitation, this formula is useful for the differential diagnosis among limited underlying diseases; although the formula for sCLEC-2 × D-dimer is useful for the differential diagnosis between non-thrombotic and thrombotic disease, it is not useful for differential diagnosis between ACI and infectious diseases or other thrombotic diseases such as venous thromboembolism, DIC and TMA.
In conclusion, sCLEC-2 levels were significantly increased in patients with atherosclerotic ACI, and D-dimer levels were significantly increased in patients with cardioembolic ACI. The super formula of sCLEC-2 × D-dimer enhanced the diagnostic ability for both atherosclerotic and cardioembolic ACI, whereas the super formula sCLEC-2/D-dimer enhanced the differentiation ability between atherosclerotic and cardioembolic ACI.
Footnotes
Acknowledgments
We would like to thank Professor Katsue Suzuki-Inoue (Department of Clinical and Laboratory Medicine, Faculty of Medicine, University of Yamanashi) for support of sCLEC-2 measurement.
Declaration of Conflicting Interests
The measurement of sCLEC-2 and D-dimer levels was partially supported by LSI Medience.
Funding
This research was funded by a Grant-in-Aid from the Ministry of Health, Labour and Welfare of Japan (21FC1008).
