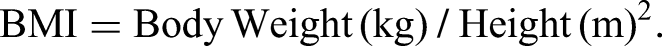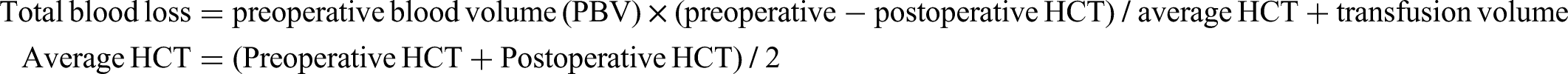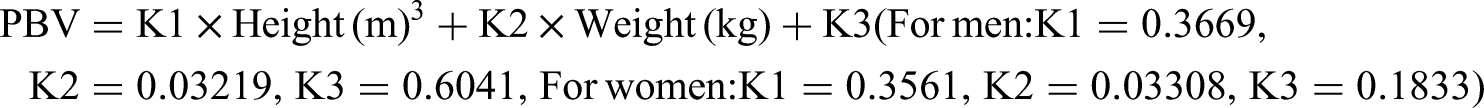Abstract
This retrospective study evaluated the safety and efficacy of SURGICEL® Powder–Absorbable Hemostatic Powder (SP) made from oxidized regenerated cellulose (ORC) in total knee arthroplasty (TKA). Ninety-one patients who underwent TKA at Peking University Third Hospital between January 2024 and July 2024 were retrospectively analyzed. Hemostatic effectiveness was evaluated by comparing perioperative blood loss, hemoglobin (Hb) levels, hematocrit (HCT), and transfusion rates. Clinical safety was assessed by comparing complication rates, operation time, visual analog scale (VAS) scores, and postoperative fever. Finally, 85 patients were included: 43 in the SP group (using SP) and 42 in the control group (not using SP). There were no significant differences in age, sex, height, weight, preoperative HCT, or Hb levels. The volume of perioperative blood loss within 3 days in the SP group was reduced by approximately 154 ml compared with that in the control group. The Hb and HCT levels in the SP group were higher on the first, second, and third days after surgery, and there were significant differences between the two groups in terms of repeated measurements of HCT and Hb levels within 3 days postoperatively. There were no statistically significant differences in the operation time, postoperative VAS scores, complications, transfusion rates, or postoperative fever. In conclusion, this study confirmed that SP can significantly reduce perioperative blood loss in TKA without negatively affecting clinical outcomes.
Introduction
Total knee arthroplasty (TKA) is currently recognized as one of the most effective treatments for advanced osteoarthritis of the knee. By restoring limb alignment and improving soft tissue balance, TKA significantly enhances postoperative mobility, greatly improving quality of life.1,2 However, surgeons face the challenge of substantial perioperative blood loss during TKA. According to data from the public welfare research project of the National Health and Family Planning Commission in China (“Evaluation of Safety and Effectiveness of Joint Replacement”), which involved 20,308 patients, the preoperative anemia rates in men undergoing TKA were 30.2% in men and 25.3% in women, while postoperative rates were 82.5% in men and 84.3% in women. 3 Intraoperative bleeding is mainly associated with bone cuts and exposure of large areas of cancellous bone, especially when there is a mismatch between the prosthesis size and the exposed cancellous bone area. Additionally, soft tissue release around the joint and synovectomy can further exacerbate bleeding.4,5 Moreover, trauma and the use of tourniquets may induce hyperfibrinolysis, contributing to increased hidden blood loss.6,7 Postoperative anemia not only delays functional recovery and wound healing in patients undergoing TKA but also increases the risk of periprosthetic infections and other complications, prolongs hospital stays, and imposes additional financial burdens. 8 Therefore, perioperative blood management is crucial in TKA. Novel hemostatic materials have been rapidly developed to reduce perioperative blood loss. These materials include sponges, gels, powders, and platelet-like substances, which reduce blood loss through physical, chemical, or biological mechanisms. 9 Oxidized regenerated cellulose (ORC) is a biodegradable topical hemostatic material. New ORC-based hemostatic materials have been proven to be effective in clinical practice, including neurosurgery, thoracic surgery, general surgery, urology, and gynecology.10,11 However, the application of ORC in orthopedics remains limited. Some studies have demonstrated its ability to reduce perioperative blood loss in TKA. For instance, in a randomized controlled trial (RCT) with 70 patients using Surgicel FibrillarTM, an ORC-based hemostatic pad, the Surgicel group showed significantly lower total perioperative blood loss and postoperative bleeding than the control group. 12
However, there have been no clinical studies on the use of the latest ORC-based product in the SURGICEL® series (SURGICEL® Powder–Absorbable Hemostatic Powder [SP]) in TKA. MacDonald et al demonstrated through two porcine bleeding models that SP has higher efficiency than traditional hemostatic gauze. 13 A European multicenter prospective clinical study involving 103 adult patients showed that SP provided excellent hemostatic outcomes in general, gynecological, urological, and cardiothoracic surgeries. 10 Another multicenter RCT conducted in China compared the hemostatic efficacy of SP with that of regular gauze and showed that the new hemostatic powder was superior in general surgery, neurosurgery, gynecology, and cardiothoracic surgery. 11
Although studies have indicated that SP has good results in various surgical fields, to date, no clinical studies have been conducted on its application in TKA. Therefore, we conducted a retrospective cohort study to explore the hemostatic efficacy of SP in TKA, in order to provide guidance and evidence for its clinical application.
Materials and Methods
Study Design
This retrospective study analyzed the data of patients who underwent TKA in the Department of Orthopedics at Peking University Third Hospital between January 2024 and July 2024. The study was approved by the Institutional Ethics Committee of Peking University Third Hospital (Registration Number: NCT06608992-M2024683). The enrolled patients consented to the use of their de-identified clinical data for research purposes. This study was conducted in full conformity with the appropriate local laws and regulations, and with tenets of the Declaration of Helsinki.
SP was obtained from Ethicon, Inc. (Somerville, NJ, USA). SP is a novel ORC-based product designed for capillary, venous, or small arterial bleeding in soft tissues with uneven tissue topography, near critical nerves and blood vessels, or for oozing over broad areas where conventional methods such as suturing, ligature, or electrocautery may be impractical.
Inclusion and Exclusion Criteria
Inclusion Criteria
Patients diagnosed with knee osteoarthritis.
Patients that underwent unilateral primary TKA performed by the same surgeon.
Patients undergoing manual surgery with traditional instruments.
Patients with minimum data available for analysis (ie, at least one set of baseline and post-treatment data and one safety data record within 3 days postoperatively).
Exclusion Criteria
Patients that underwent TKA surgery with robotic or navigation-assisted digital technology, patient specific instrumentation and other extramedullary alignment technologies.
Patients requiring secondary orthopedic surgery.
Patients with three or more postoperative variables missing.
Patients whose clinical data contained outliers in terms of blood loss or safety.
Data Collection
All medical records and examination results were obtained from electronic medical systems. The following data were retrieved:
Baseline information included age, height, weight, body mass index (BMI), preoperative hematocrit (HCT), and preoperative hemoglobin level (Hb).
Pain and joint function indicators: Visual analog scale (VAS) score on the day after surgery.
The postoperative outcomes included perioperative blood loss, transfusion rate and volume, postoperative Hb and HCT decline, operation time, complication rate, and body temperature within 3 days postoperatively.
Total perioperative blood loss was the primary outcome measure, while safety indicators included surgery duration, complication rate, VAS score on postoperative day 1, and highest body temperature within the first 3 days postoperatively. Other measures were considered secondary outcome indicators.
Quantitative and Variable Calculation Methods
The total postoperative blood loss was calculated using the Gross linear formula:
Perioperative Management and Surgical Methods
All patients included in the study underwent TKA performed by the same surgical team with the same perioperative management and rehabilitation treatment.
All patients were administered spinal or general anesthesia. A tourniquet was routinely used during surgery and was inflated to 150 mm Hg above the patient's intraoperative systolic pressure. TKA was performed according to standard procedures. After the prosthesis was implanted, the tourniquet was released, and monopolar cautery was used for hemostasis. In the SP group, hemostasis was achieved by spraying the powder into areas such as the posterior joint capsule, medial gap, lateral gap, suprapatellar pouch, and infrapatellar fat pad (Figure 1). After the powder had fully reacted (indicated by a color change), an irrigation gun was used to wash away residual powder.
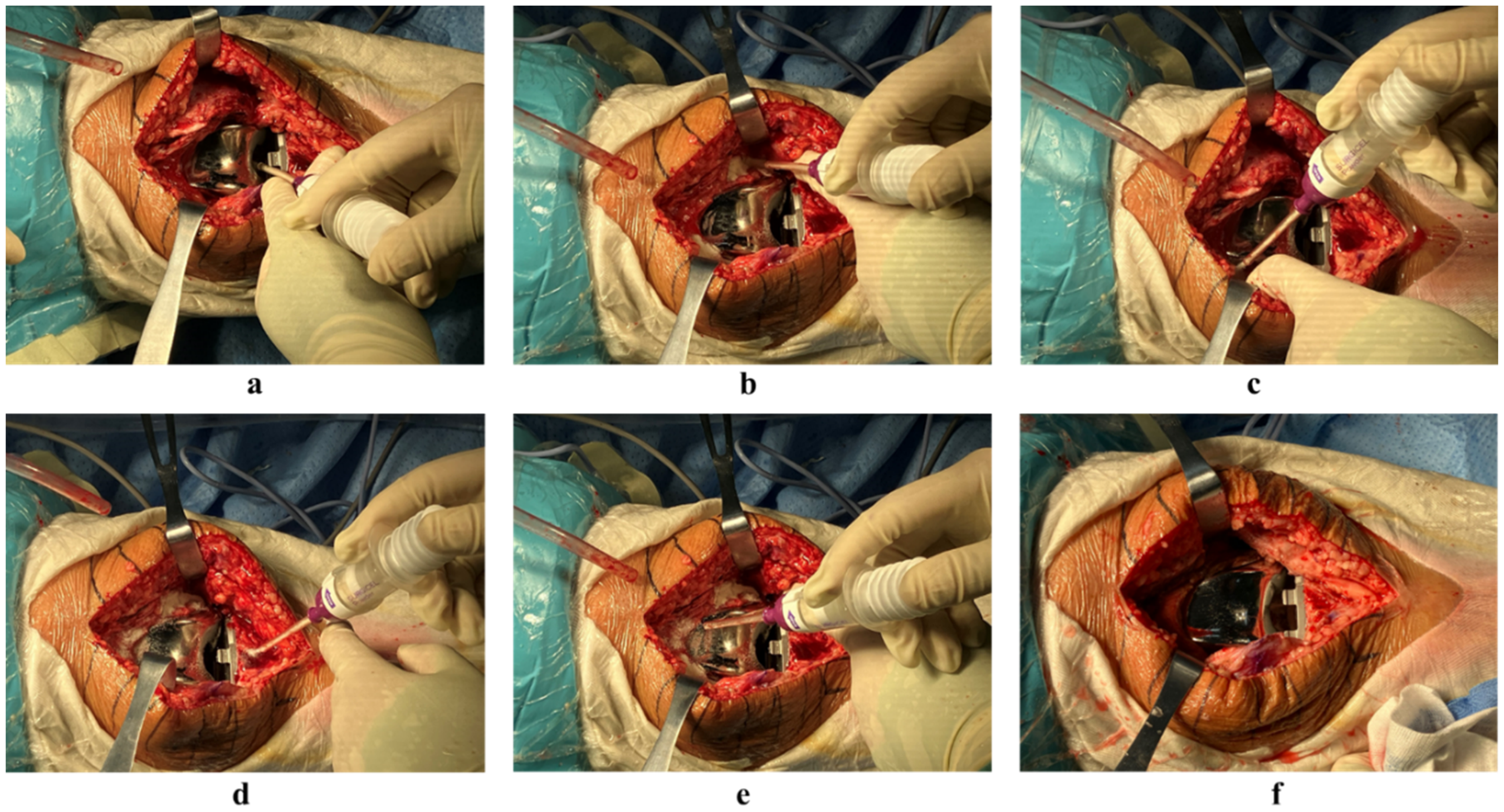
The application of hemostatic powder: (a) posterior joint capsule (b) medial gap (c) lateral gap (d) suprapatellar pouch (e) infrapatellar fat pad (f) the joint capsule after washing away the residual powder.
All patients received 1 gram of tranexamic acid administered intravenously before skin incision and skin closure, and a local injection (1 g) after joint capsule closure. All patients also received a single femoral nerve block preoperatively without a continuous femoral nerve catheter or postoperative wound drainage. After wound closure, the affected limb was compressed with an elastic bandage applied from the ankle to the proximal thigh, which was removed 24 h postoperatively. For anticoagulation, oral rivaroxaban (10 mg, once daily) was administered starting 12 h after surgery in combination with active and passive ankle pump exercises (initiated immediately after surgery). The patients began active and passive functional rehabilitation exercises 24–36 h postoperatively under the guidance of medical staff, and ice packs were applied locally to the affected area for 15 min at a time.
Statistical Analysis
Statistical analyses were conducted using SPSS 27.0. Normally distributed continuous variables are presented as mean ± standard deviation (x̅ ± s), and between-group comparisons were performed using the two-sample t-test. Continuous variables with a non-normal distribution are presented as medians (p25, p75), and between-group comparisons were performed using the non-parametric rank-sum test. Categorical variables are presented as n (%), and between-group comparisons were performed using the chi-square test.
Results
Patient Participation and Demographics
Between January 2024 and July 2024, 91 patients underwent TKA performed by the same surgical team at Peking University Third Hospital. Outliers appeared in the data of two patients, and several indicators were missing in four patients. Therefore, the remaining 85 patients were included in this study. Based on the use of SP, patients were divided into a control group (without hemostatic powder) and an SP group (with hemostatic powder). There were 43 and 42 patients in the control and SP groups, respectively. There were no statistical differences between the two groups in terms of age, height or weight. The baseline Hb and HCT values of the two groups were comparable (Table 1).
Preoperative Demographics
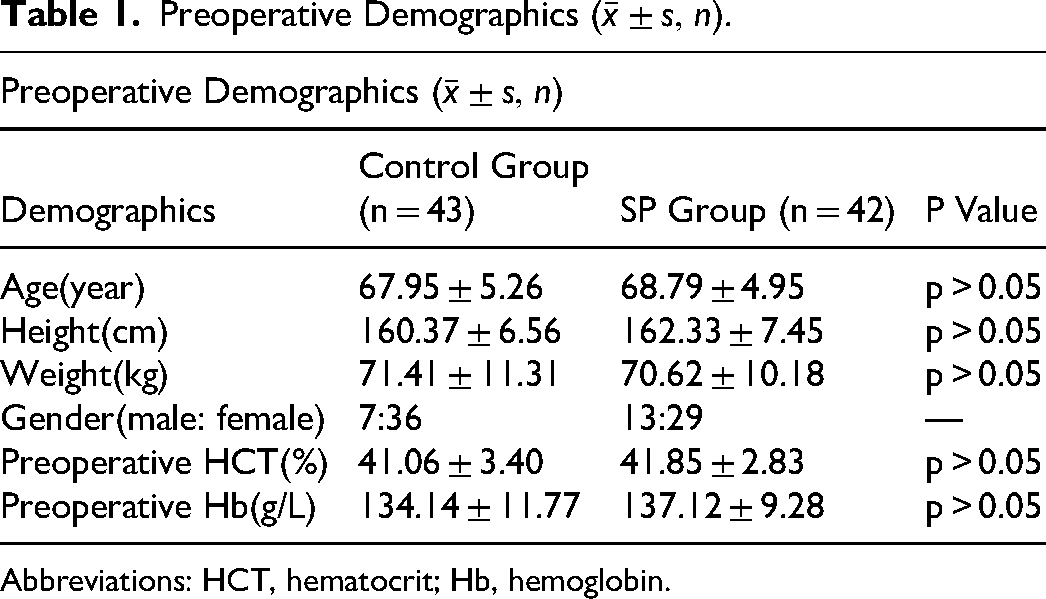
Abbreviations: HCT, hematocrit; Hb, hemoglobin.
Variables Related to Clinical Safety
Comparison of Operation Time
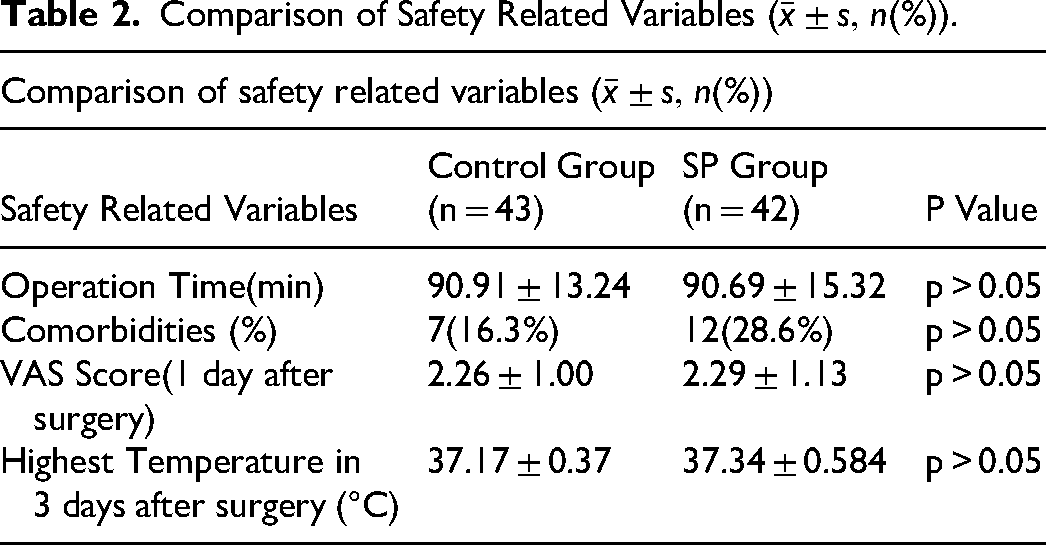
The operation time was 90.91 ± 13.24 min in the control group and 90.69 ± 15.32 min in the SP group. No significant differences were found in the operative time between the two groups (Table 2).
Comparison of Safety Related Variables
Comparison of Complication Rates
There were seven cases of postoperative complications in the control group, all of which corresponded to lower limb intermuscular vein thrombosis, and 12 cases of postoperative complications in the SP group; of these, 11 had lower limb intermuscular vein thrombosis and one had ischemic bowel disease. There was no significant difference in the incidence of complications between the two groups within 2 months postoperatively (Table 2).
Comparison of VAS Scores
There was no significant difference in VAS scores between the two groups on the first day after surgery, with the control group having a mean score of 2.26 ± 1.00, whereas that of the SP group was 2.29 ± 1.13 (Table 2).
Comparison of Postoperative Fever
Comparison of the highest body temperature within 3 days after surgery between the two groups showed no statistically significant difference. The highest body temperature was 37.17 ± 0.37°C in the control group, and 37.3 ± 0.584°C in the SP group (Table 2).
Variables Related to Blood Loss
Comparison of Changes in Hb and HCT Between the Groups Within 3 Days Postoperatively
There were statistically significant differences in the Hb and HCT levels between the two groups within 3 days. The overall Hb and HCT levels in the SP group were higher than those in the control group within three days (Table 3). The average Hb levels in both groups decreased most noticeably on the first day after surgery, and were the lowest on the third day after surgery (Figure 2). The Hb and HCT levels of the two groups on the first, second, and third days after surgery differed significantly. The Hb and HCT levels in the SP group were higher on the first, second, and third days after surgery (Table 2).
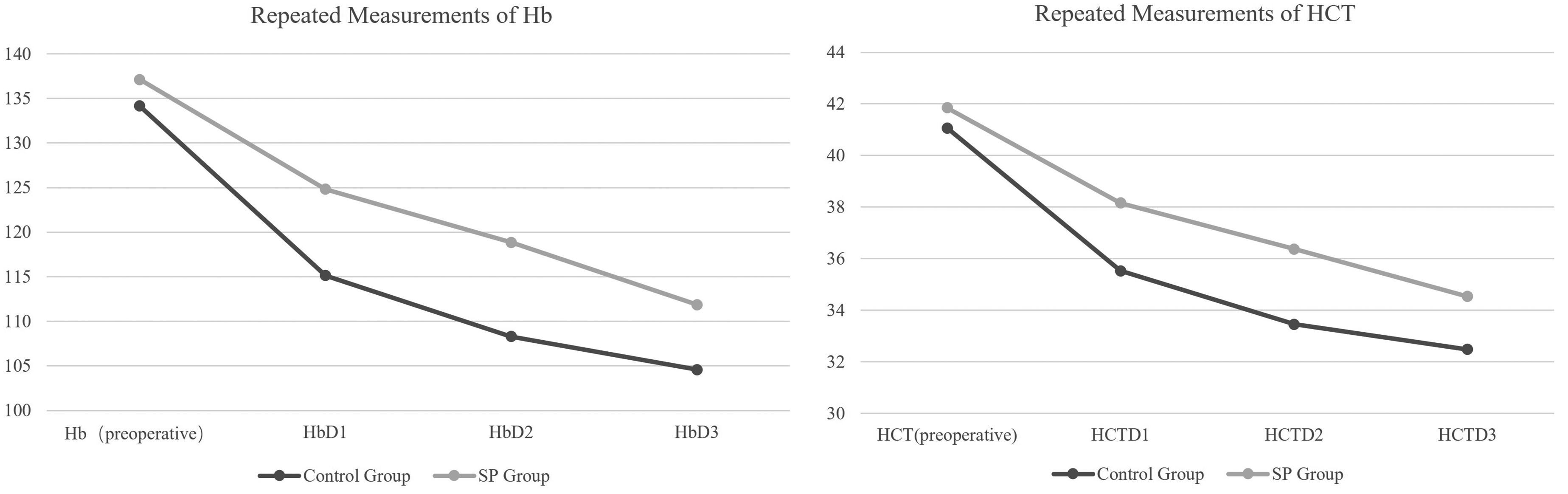
Comparison of Hb and HCT repeated measurements. Abbreviations: HCT, hematocrit; Hb, hemoglobin.
Comparison of Repeated Measurement Variables.
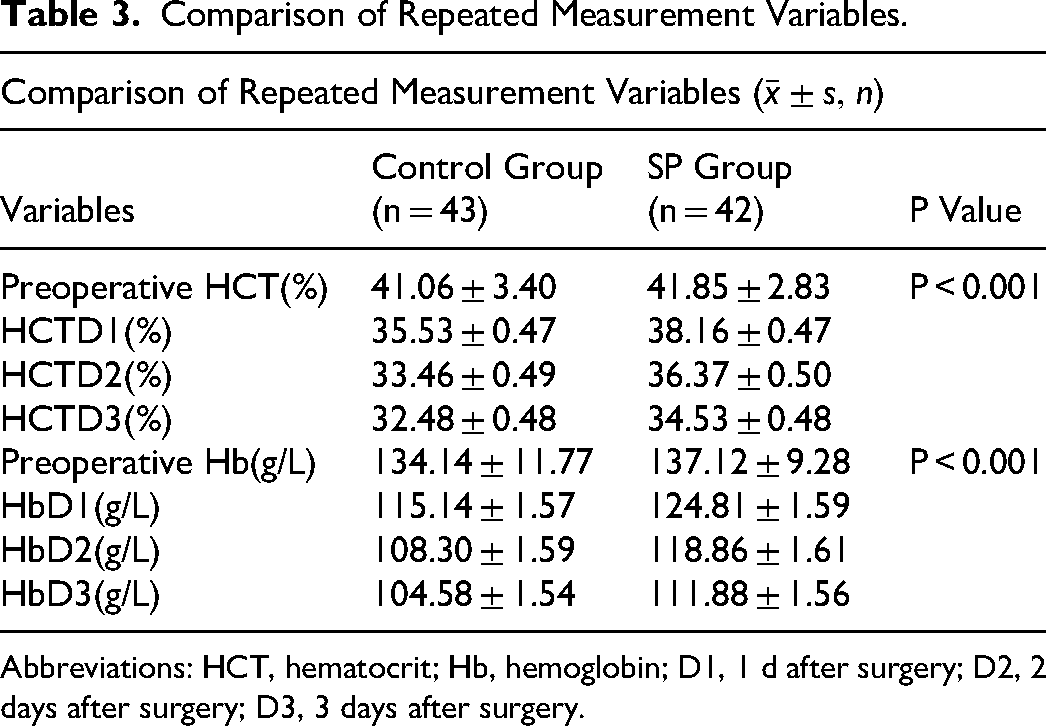
Abbreviations: HCT, hematocrit; Hb, hemoglobin; D1, 1 d after surgery; D2, 2 days after surgery; D3, 3 days after surgery.
Comparison of Perioperative Blood Loss and Transfusion Rates Within 3 Days
The volume of perioperative blood loss within 3 days was 957.22 ± 284.95 ml in the control group and 802.70 ± 299.68 ml in the experimental group. The average blood loss in the SP group was 154 ml less than that in the control group, with a significant difference between the two groups. None of the patients required blood transfusions (Table 4).
Comparison of Variables Related to Blood Loss
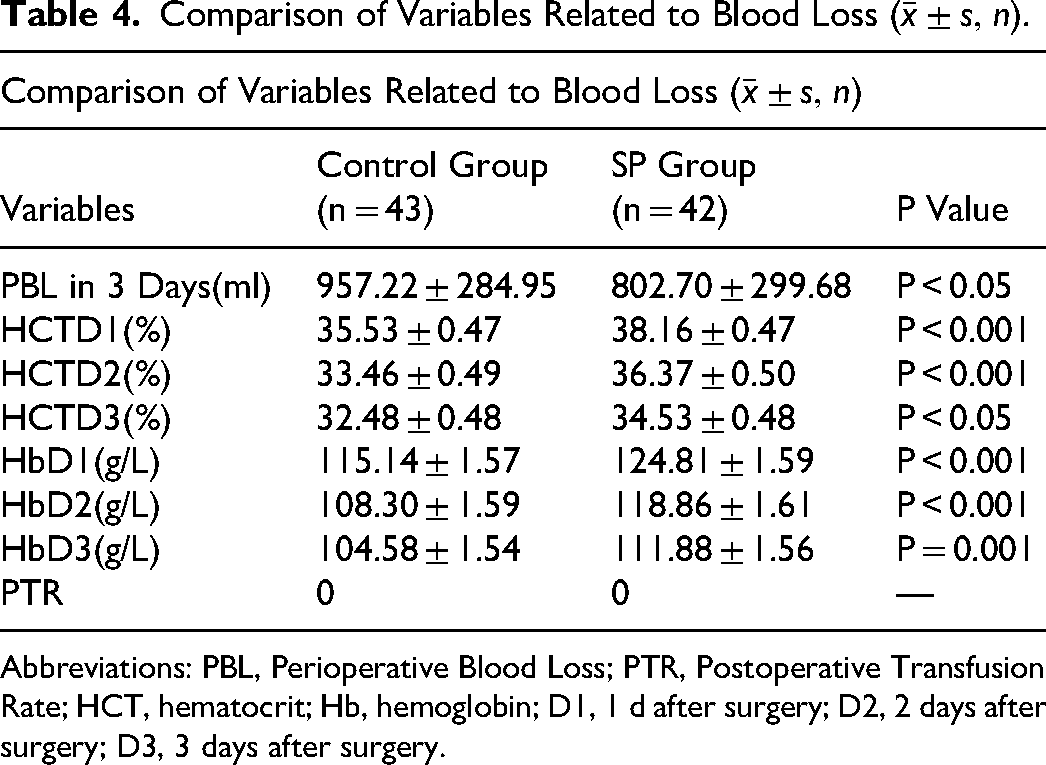
Abbreviations: PBL, Perioperative Blood Loss; PTR, Postoperative Transfusion Rate; HCT, hematocrit; Hb, hemoglobin; D1, 1 d after surgery; D2, 2 days after surgery; D3, 3 days after surgery.
Discussion
Among the SURGICEL hemostatic products available in the market, SP is the most recent. Although animal experiments 13 and clinical trials10,11 have demonstrated its hemostatic effectiveness, research has been limited to neurosurgery, thoracic and general surgery, urology, obstetrics, and gynecology. However, the clinical application of hemostatic powder is not restricted to these fields, and its application in orthopedic surgeries and in joint surgery particularly demands further research.
This study retrospectively analyzed the effectiveness of SP in 85 patients, and evaluated its hemostatic efficacy and safety. Data analysis showed that the perioperative blood loss within 3 days of TKA was reduced by approximately 154 ml in the SP group compared with that in the control group. Additionally, Hb and HCT levels in the SP group were higher than those in the control group, indicating that applying hemostatic powder can reduce perioperative blood loss. Furthermore, the use of hemostatic powder did not prolong the surgical procedure, as there was no statistically significant difference in the operation time between the two groups. This demonstrates that the design of SP is surgeon-friendly. In terms of clinical outcomes such as VAS scores, complication rates, and postoperative body temperature, there were no statistically significant differences between the two groups. This suggests that the hemostatic powder can reduce blood loss without negatively affecting the clinical outcomes of TKA, thereby supporting its clinical safety.
Hemostatic Effectiveness
The volume of perioperative blood loss within 3 days in the SP group was reduced by 154 ml compared to that in the control group, which aligns with previous reports on the use of SP in other surgical fields. 10 SP is derived from ORC, which was developed to assist in the control of oozing bleeding over broad areas where application of common fabric hemostatic products could be difficult. 14 When applied locally in the joint capsule and the gaps between the remnants of the medial and lateral menisci, the degradation of red blood cells and the subsequent procoagulant substances cause the ORC to change into a dark-colored gelatinous substance. 12 Additionally, the ORC maintains a low pH environment that promotes vasoconstriction. 15 As a foreign substance, hemostatic powder provides a scaffold for platelet aggregation and adhesion, thereby enhancing coagulation, which explains the favorable hemostatic effects observed in this study.16–18
Traditional hemostatic materials include foam, fabric, and gel structures 9 ; however, the granular structure of SP offers improved conformability and adhesion to broad surfaces and to the anatomically complex geometry of the joint capsule. Composed of ORC fiber aggregates, the powder-structured SP has superior in vitro clotting performance compared to the constituent ORC fibers: The clotting efficacies for high-sphericity ORC aggregates and ORC fibers were observed to be 95% and 26%, respectively. 16 When the hemostatic powder changes into a gelatinous structure, its volume increases, enhancing hemostasis through both procoagulant chemicals and physical compression.
Clinical Safety
In this study, variables related to safety included operation time, complication rate, VAS score on the first postoperative day, and highest body temperature within 3 days after surgery. Comparisons between the two groups revealed no statistically significant differences, suggesting that the use of hemostatic powder in TKA does not result in additional surgical risks. However, the use of hemostatic powders is not entirely without risk. Owing to expansion of its volume, it may cause neural compression syndrome if not thoroughly rinsed during neurosurgical procedures.19,20 In thoracic surgeries, the implantation of an ORC may change surgical scars. 21 Additionally, residual ORC particles may interfere with imaging results. 22 In TKA, owing to the anatomical characteristics of the knee joint, surgeons can rinse off the residual hemostatic powder thoroughly, and there is little concern about nerve compression, as the distribution of motor nerves is sparse within the joint cavity. In contrast, using hemostatic powder can reduce the need for an electrocoagulation knife, which has a positive impact on infection control and patient prognosis. 23
Limitations
This was a retrospective observational study with a comparatively short follow-up period and a limited number of observational indicators. However, based on the results of this study, a subsequent randomized clinical trial can be designed for prospective research, which will include a 6-month follow-up to observe the long-term clinical effects of SP in joint replacement surgery. Additionally, the sample size in this study was relatively small, and future research should aim to expand it. In addition, the main indicator of this study was perioperative blood loss, which was approximated using a formula that inevitably involved inaccuracies. Moreover, this study did not distinguish between intraoperative and hidden blood loss. 24 Future research should adopt more refined approaches, such as measuring limb swelling rates and using the intraoperative gauze-weighing method, to further improve indicator accuracy.
Conclusion
This study confirms that the use of SURGICEL® Powder can significantly reduce perioperative blood loss in patients undergoing TKA. Additionally, SURGICEL® Absorbable Hemostat Powder does not increase operation time or the complication rate, nor does it exacerbate postoperative limb pain, demonstrating acceptable safety and efficacy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical Approval and Informed Consent Statements
This study was approved by the Institutional Ethics Committee of the Peking University Third Hospital (Study Registration Number: NCT06608992-M2024683). The enrolled patients consented to the use of their de-identified clinical data for research purposes. This study was conducted in full conformity with the appropriate local laws and regulations and the Declaration of Helsinki.
Funding
This study was funded by Clinical Cohort Construction Program of Peking University Third Hospital, No.BYSYDL2023007.