Abstract
This clinical study compares 2 hemostatic agents, a novel combination powder (CP) (HEMOBLAST™ Bellows) and an established polysaccharide starch powder (PP) (Arista™ AH) to assess the usefulness of CP. Retrospective comparative analysis of CP (July 2018 to July 2019, 68 patients) to PP (January 2011 to January 2013, 94 patients) in cardiothoracic patients was performed using linear regression models adjusting for age, sex, and procedure type for the endpoints: blood loss; protamine to skin closure time (hemostasis time); chest tube output and blood products required 48 hours postoperatively; ICU stay; postoperative comorbidities; and 30 day mortality. 162 patients (108 M: 54 F) underwent 162 cardiothoracic surgical procedures including: transplantation (n = 44), placement of ventricular assist device (n = 87), and others (n = 31). Use of CP compared to PP (Estimated Mean Difference [95% CI], P-value) produced significant reductions: blood loss (mL) (−886.51 [−1457.76, −312.26], P = 0.003); protamine to skin closure time (min) (−16.81 [−28.03, −5.59], P = 0.004); chest tube output (48 hrs, mL) (−445.76 [−669.38, −222.14], P < 0.001); packed red blood cell transfusions (units) (−0.98 [−1.56, −0.4], P = 0.001); and postoperative comorbidities (−0.31 [−0.55, −0.07], P = 0.012). There were no differences in the ICU stay (4.07 [−2.01, 10.15], P = 0.188) or 30-day mortality (0.57 [0.20, 1.63], P = 0.291). The use of CP in complex cardiothoracic operations resulted in improved hemostasis and significant clinical benefits in blood loss, transfusion requirements, morbidity, and time in operating room.
Introduction
Complex cardiothoracic surgical operations remain subject to the risks of bleeding as a result of the use of anticoagulation and the cardiopulmonary bypass circuit required to perform these procedures as well as the multiple, possible sequelae of using these modalities and the underlying pathology of each patient including hepatic and renal dysfunction. The required combination of anticoagulation and cardiopulmonary bypass is known to impair the coagulation cascade, activate the fibrinolytic cascade, reduce platelet function, incite an inflammatory reaction, and create hemodilution as well as potentially cause acidosis and hypothermia. All these factors create a coagulopathic substrate. 1 Thus, the risk of bleeding in complex cardiothoracic surgery remains clinically significant and can contribute to: exposure to blood product transfusions; 2 need for re-explorations for bleeding, 3 prolonged lengths of intensive care unit and hospital of stay; 3 increased morbidity and mortality; 3 and larger hospital costs. 4,5 Greater patient acuity and procedural complexity as characterized by the emerging use of aggressive antiplatelet therapy 6 and the placement of mechanical assist devices (MCAD) 7 may also contribute to increased risk of postoperative bleeding. In challenging surgical settings, hemostatic agents can be useful adjuncts to the armamentarium available to surgeons for the control of bleeding 8 –12 and are known to be useful during cardiothoracic surgical operations. 13 –15 Recently, a new powder has been added to the toolbox of available hemostatic agents. 16 This study compares a novel combination powder (CP) hemostatic agent (HEMOBLAST™ Bellows, Biom’Up France SAS, Lyon, France) 16 consisting of porcine collagen, bovine chondroitin sulfate, and human pooled plasma thrombin to an established plant derived, polysaccharide starch powder (PP) hemostatic agent (Arista™ AH, Becton Dickinson/Bard, Warwick, RI, USA) 17 to assess the usefulness of CP. Although both hemostatic agents are provided in the form of powders, the potential enhancements of CP include activation of platelets by collagen, adhesion of the wound to surrounding tissues by chondroitin sulfate, and conversion of fibrinogen to fibrin by thrombin.
Methods
Study Population
This retrospective, de-identified, single site study underwent review and approval by the institutional review board (IRB) of Houston Methodist Hospital (HMH) with patient confidentiality insured. The study design (Figure 1) is observational in nature and utilizes the data available on all patients, sequentially sampled into a single surgical practice, over 2 cohort periods operated upon by senior members from an internationally known group performing over 1600 cardiac operations annually. The CP group consisted of patients undergoing complex cardiothoracic procedures by 2 experienced surgeons (more than 3 years in practice) without surgeons in training from July 2018 until July 2019 (68 patients, Drs. Bruckner and Suarez) and the comparator PP group consisted of patients undergoing complex cardiothoracic procedures by 2 experienced surgeons (more than 3 years in practice) without surgeons in training from January 2011 to January 2013 (94 patients, Drs. Brucker and Loebe), all at the same institution. Subjects were identified in 1of 4 ways: 1. Directly from our practice or inpatient data; 2. Review of medical records within the HMH-Medical Center inpatient list (MethOD); Epic electronic medical records; or archived medical records. The case mix inclusion criteria were based on the clinical practice of a surgical heart failure service that predominantly performs complex heart failure procedures with high bleeding tendencies including consecutive heart transplants, lung transplants, and ventricular assist devices (VADs) as well as a small number of coronary artery bypass graft (CABG) and valvular/aortic procedures. Inclusion criteria included factors that are important to reduce perioperative bleeding: cessation of platelet inhibitors (>5 days) as well as correction of anemia (Hgb < 8.0); hypothrombocytopenia (Plts < 60,000); and coagulopathy (PT/INR > 15/1.5). All patients underwent cardiopulmonary bypass with the exception of 3 patients who required unplanned emergent re-exploration for bleeding within 24 hours.
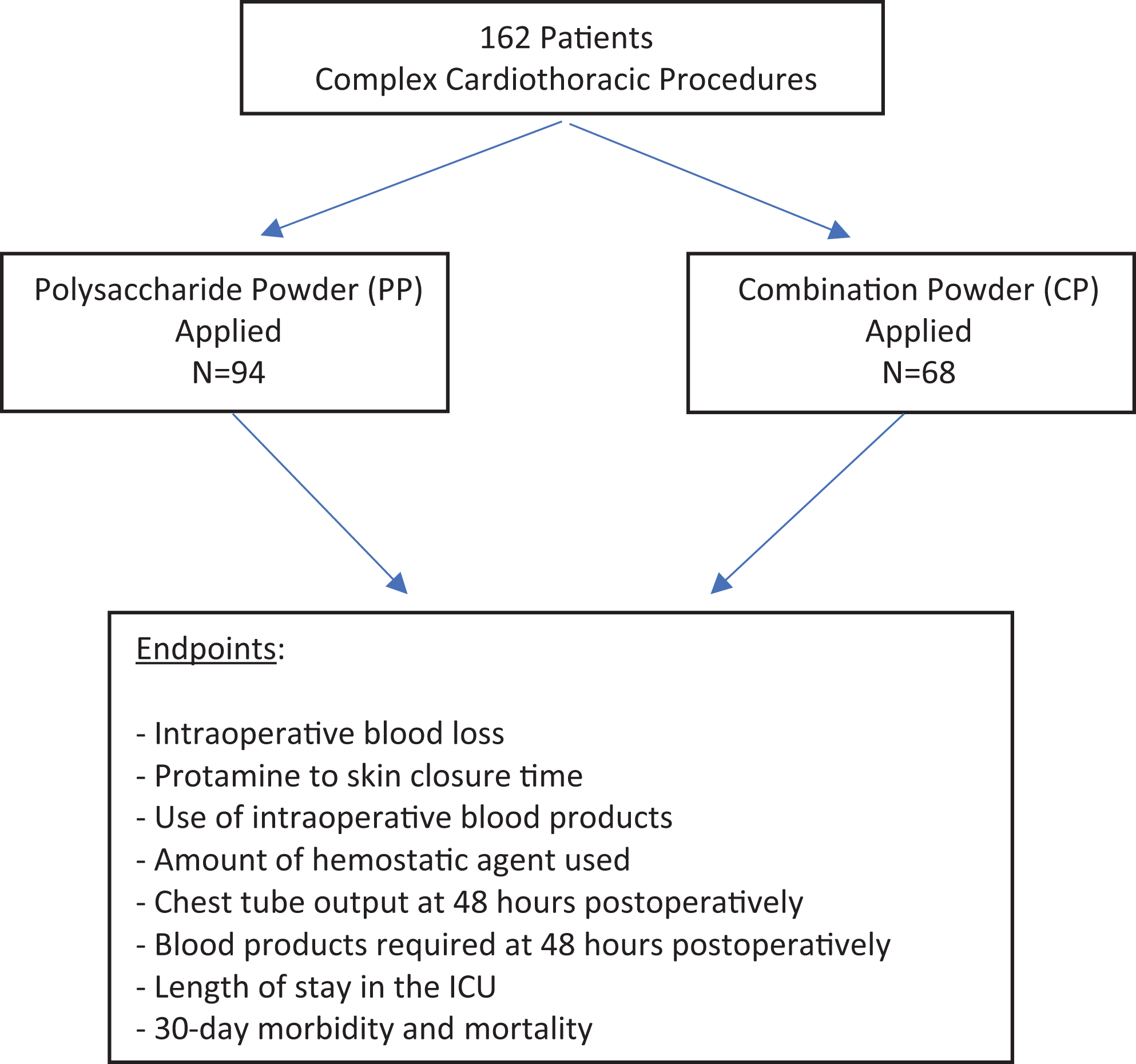
The study design compared 2 groups: a novel combination powder (CP) hemostatic agent (HEMOBLAST™ bellows) and an established polysaccharide starch powder (PP) hemostatic agent (arista™ AH).
Study Endpoints
All patient records were stratified by age, sex, and complexity of the cardiothoracic surgical procedure. The endpoints measured in this retrospective comparison consisted of both intraoperative and postoperative elements. Intraoperative measurements included: blood loss; protamine to skin closure time (hemostasis time); and amount of hemostatic agent used. Postoperative measurements included: chest tube output at 48 hours; blood products required; length of stay in the intensive care unit (ICU); 30-day morbidity; and 30-day mortality. Morbidity included ventilator dependence (ventilator requirement > 7 days), renal failure (new onset requiring dialysis), shock (hypotension requiring 2 or more pressors), sepsis (positive blood cultures and hypotension), and stroke (verified by CT scan or MRI scan).
Surgical Technique
All procedures were performed through a complete median sternotomy except for lung transplants, that were performed through clamshell incisions. After weaning from cardiopulmonary bypass, heparinization was reversed by protamine sulfate. Thereafter, the powdered hemostatic agents were applied as specified in their instructions for use (IFUs). 16,17 Application was performed encompassing the entire operative field to address all instances of active bleeding persisting after employing conventional means including direct pressure, electrocautery, and suture ligation.
Statistical Methods
The distribution of patient and procedure characteristics were described using the sample mean and standard deviation for continuous measures and the observed frequency and percent for discrete measures. Inferential comparisons of continuous intra- and post-operative outcome measures were compared using multiple linear regression. Logistic regression was used to compare the probability of death between patients treated with CP and those treated with PP as well as to examine the association between blood loss and mortality. In all regression analyses, we a priori adjusted for age, sex, and procedure type as potential confounding factors in the comparison between CP and PP. Residuals diagnostics to assess departures from the assumption of homoscedasticity in linear regression models and to assess influential data points were conducted. Any identified influential points could not be determined to have arisen from data entry error and were, hence, kept in all reported analyses. Secondary sensitivity analyses omitting influential points were also conducted but are not presented as results did not qualitatively differ from the primary analysis. While all pre-specified analyses are presented, no adjustments for multiple comparisons have been made and inferential P values should be interpreted accordingly. All statistical analyses were performed using R version 3.4.1.
Results
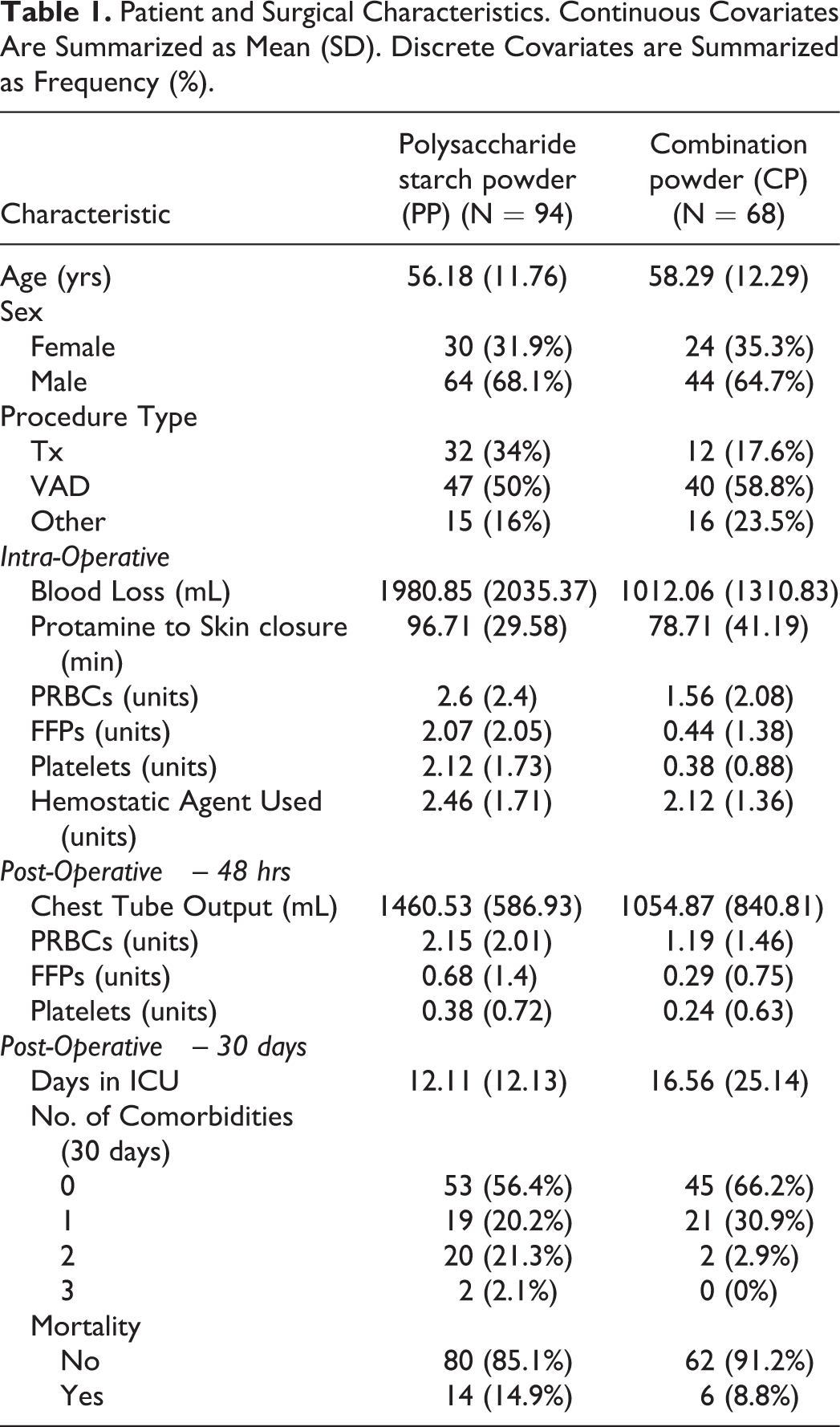
Comparison of baseline demographics (age, sex, procedure type) between the CP and PP groups revealed no statistically significant differences using a 2-sided level .05 test (Table 1). The mean ages of the CP and PP groups were 58.29 (12.29) and 56.18 (11.76), respectively. There was an observed preponderance of men compared to women in both groups. A similar distribution of procedures was observed in both groups with the most frequent procedure being placement of VADs.
Patient and Surgical Characteristics. Continuous Covariates Are Summarized as Mean (SD). Discrete Covariates are Summarized as Frequency (%).
Visual depictions of the distribution of intra- and post-operative measures of interest comparing CP to PP are displays in Figures 2 to 5. After adjustment for age, sex, and procedure type, use of CP compared to PP (Estimated Mean Difference [95% CI], P value) resulted in multiple significant intraoperative endpoint reductions (Table 2) using CP including: blood loss (mL) (−886.51 [−1457.76, −312.26], P = 0.003); protamine to skin closure time (min) (−16.81 [−28.03, −5.59], P = 0.004); and amount of packed red blood cells (units) (−1.10 [−1.83, −0.37], P = 0.003), fresh frozen plasma (units) (−1.60 [−2.18, −1.02], P < 0.001), and platelets (units) (−1.73 [−2.19, −1.27], P < 0.001) transfused. There was no significant difference in the mean amount of intraoperative hemostatic agent used (units) (−0.37 [−0.87, 0.13], P = 0.154). Significant postoperative endpoint reductions at 48 hours included: chest tube output (mL) (−445.76 [−669.38, −222.14], P < 0.001); packed red blood cell transfusions (units) (−0.98 [−1.56, −0.4], P = 0.001); and fresh frozen plasma transfusions (units) (−0.38 [−0.75, −0.01], P = 0.044) as well as postoperative comorbidities (−0.31 [−0.55, −0.07], P = 0.012).
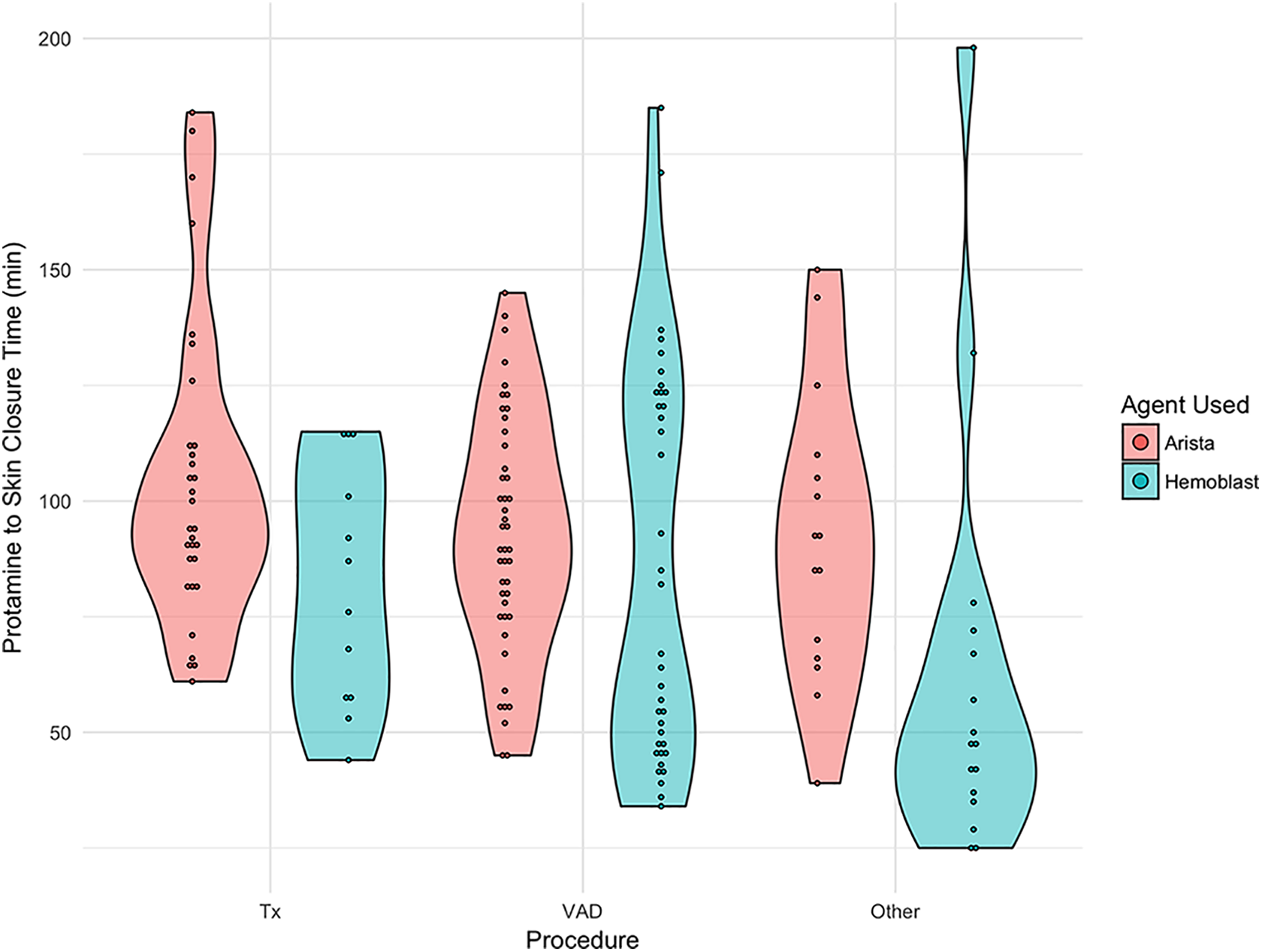
Violin plots depicting the distribution of protamine to skin closure time in minutes for patients treated with combination powder (CP) and those treated with polysaccharide starch powder (PP). Plots are stratified by procedure type (transplant, VAD, and other). Dots within each violin plot depict observed data points.

Empirical density plots for number of intra-operative PRBCs for patients treated with combination powder (CP) and those treated with polysaccharide starch powder (PP). The upper left plot includes all data while the remaining 3 plots are stratified by procedure type (transplant, VAD, and other).

Empirical density plots for number of intra-operative FFPs for patients treated with combination powder (CP) and those treated with polysaccharide starch powder (PP). The upper left plot includes all data while the remaining 3 plots are stratified by procedure type (transplant, VAD, and other).
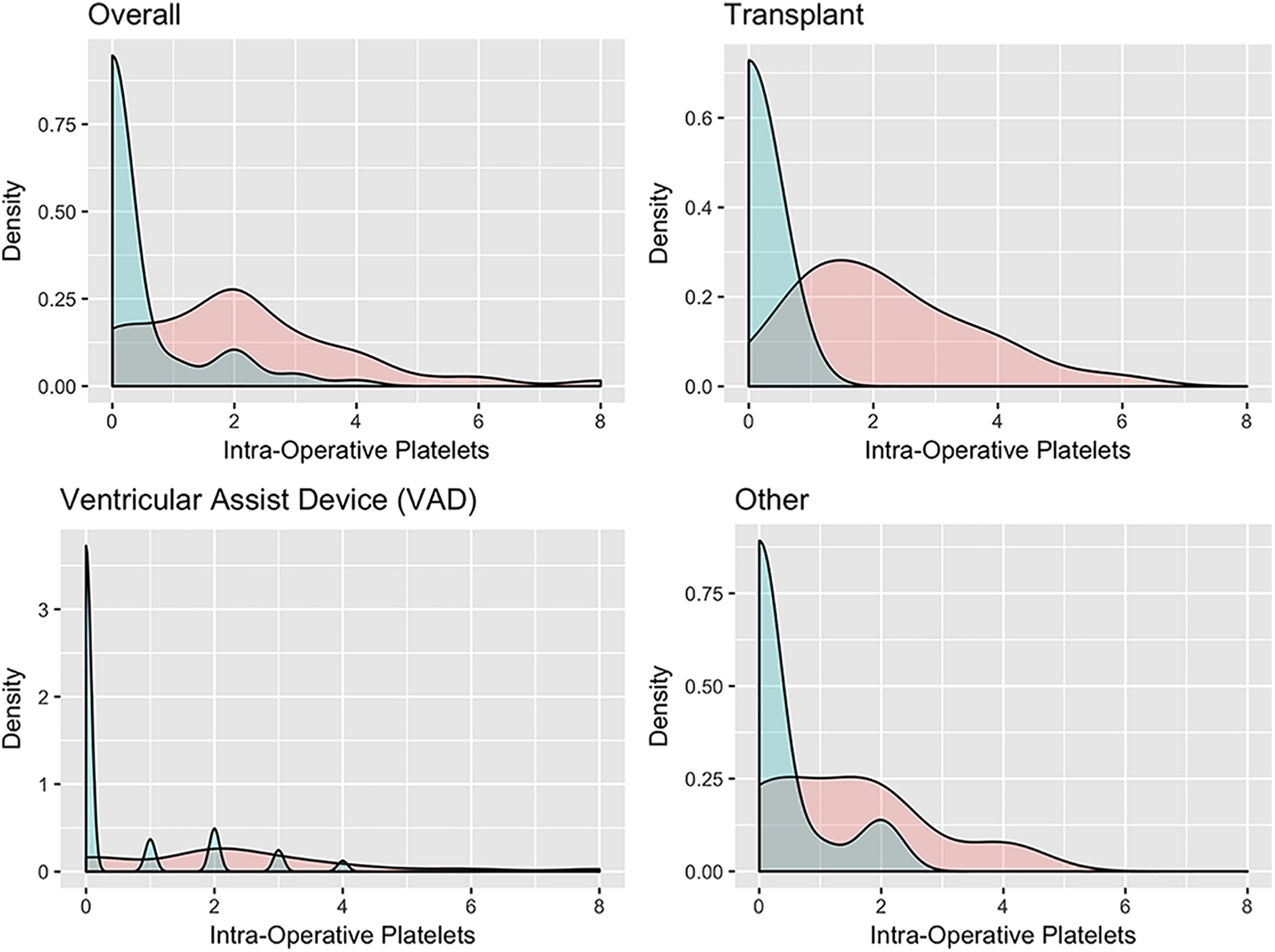
Empirical density plots for number of intra-operative platelets for patients treated with combination powder (CP) and those treated with polysaccharide starch powder (PP). The upper left plot includes all data while the remaining 3 plots are stratified by procedure type (transplant, VAD, and other).
Estimated Mean Difference in Intra- and Post-Operative Outcomes Comparing Combination Powder (CP) to Polysaccharide Starch Powder (PP). Estimates Are Based on Separate Linear Regression Models That Adjust for Age and Sex of the Patient as well as Procedure Type.
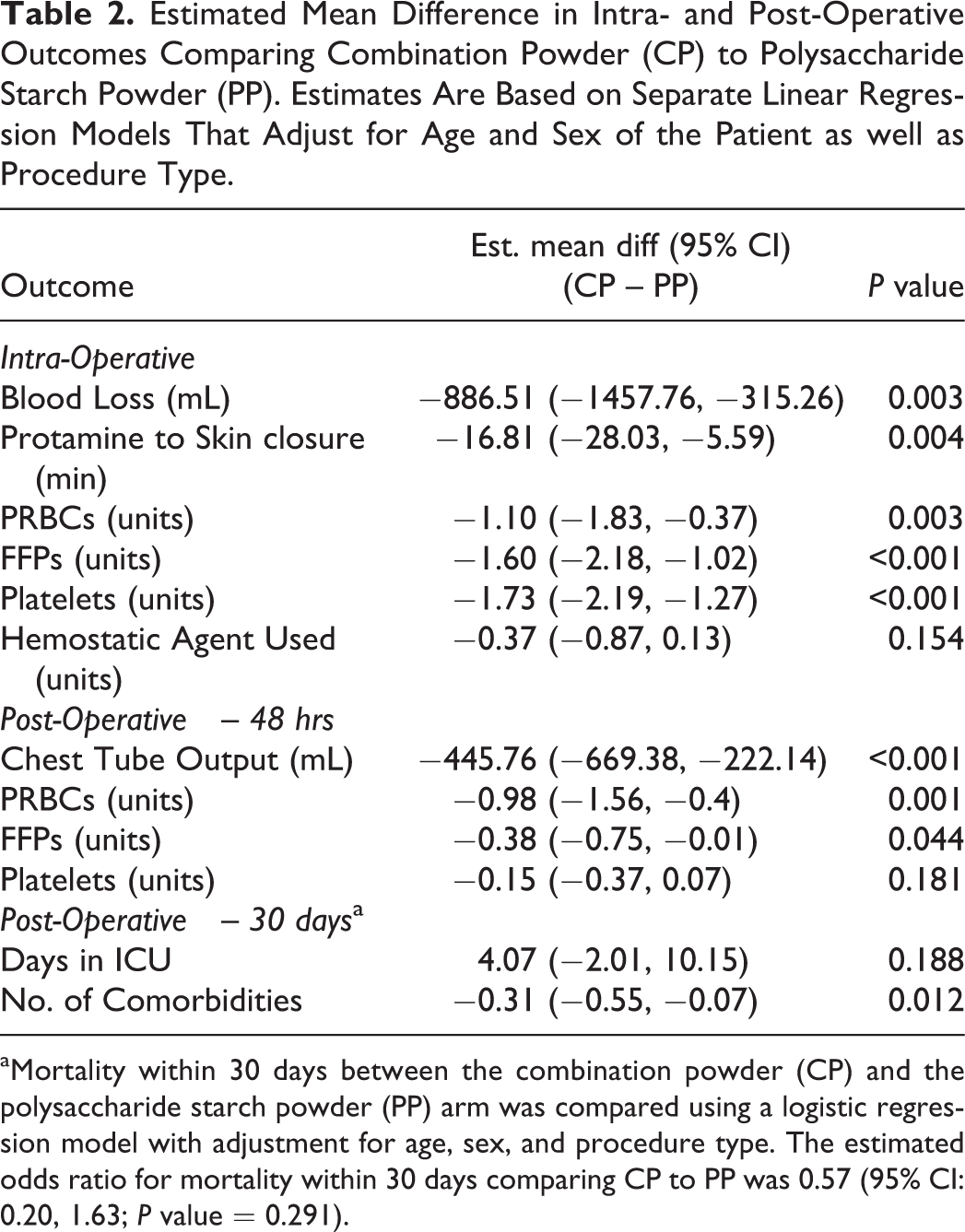
aMortality within 30 days between the combination powder (CP) and the polysaccharide starch powder (PP) arm was compared using a logistic regression model with adjustment for age, sex, and procedure type. The estimated odds ratio for mortality within 30 days comparing CP to PP was 0.57 (95% CI: 0.20, 1.63; P value = 0.291).
After adjustment for age, sex, and procedure type there were no significant differences between the 2 groups with respect to: postoperative platelet transfusions (units) (−0.15 [−0.37, 0.07], P = 0.181); postoperative days in the ICU (4.07 [−2.10, 10.15], P = 0.188); and the odds of mortality within 30 days of surgery (OR = 0.57 [0.20, 1.63], P = 0.291).
Secondary analyses considered the association between total blood loss and 30-day mortality. After adjustment for age, sex, and procedure type it was estimated that each 500 mL of blood loss experienced during surgery was associated with a 21% higher odds of mortality within 30 days (OR = 1.21 [1.35, 3.35], P = 0.008).
There were no adverse events noted to be specifically associated with the use of either CP or PP.
Discussion
The demographics of the 2 groups used for this comparison were similar with respect to age, sex, and procedure type, thereby permitting reasonable comparison of specific endpoints between the 2 retrospective groups.
The endpoints reflecting the degree of bleeding associated with the cardiac surgical procedures in this report strongly supported improvements using CP. These statistically significant differences included, intraoperatively: blood loss (mL); protamine to skin closure time (min); transfusion of packed red blood cells (units), fresh frozen plasma (units), and platelets (units); as well as postoperatively: chest tube output (mL); transfusion of packed red blood cells (units) and fresh frozen plasma (units); and postoperative comorbidities.
Postoperative days in the ICU and 30-day mortality estimated odds ratio did not reach statistically significant values. There were, however, noted to be outliers in ICU days that had moderate influence on the large standard deviation for mean ICU days. As these outlying observations were not concluded to result from data entry errors, they were retained in all analyses presented. Further, precision to estimate the association with 30-day mortality is fairly low in the current study given the total sample size and number of observed deaths. Intraoperative blood loss was, however, found to be significantly associated with 30-day mortality and significant differences between CP and PP were seen with respect to blood loss.
In addition, the observed differences in intraoperative hemostatic agent use for CP and PP, in terms of numbers of units used (units) (−0.37 [−0.87, 0.13], P = 0.154), are actually even larger in terms of amount of powder used because CP units contained 1.65 grams of powder and PP units contained 5 grams of powder. After making calculations for the total amount of product used in grams, the estimated difference was significantly less in the CP group compared to the PP group (−8.86 [−11.01, −6.71], P < 0.001). Thus, it was observed that more than 2 or 3 times as much PP was needed compared CP. The costs of clinically using the 2 different hemostatic powders in this study, particularly when one considers the fact that almost 2 to 3 times as much AristaTM was required to be used as HEMOBLASTTM are similar or more expensive for AristaTM. However, as detailed costs were not tracked in either group, no definitive, precise cost comparisons can be made and have not be provided.
The parameters measured in this study including transfusion requirements for a variety of blood components, chest tube output, and postoperative comorbidity and mortality are well known measures of bleeding severity during cardiac surgical operations.1,19 Protamine to skin closure represents the topical hemostasis time, since the heparin is reversed by the protamine and has been previously used as a measure of hemostatic effectiveness. 18
Previous authors have examined the retrospective performance of a powdered hemostatic agent 18 as well as the prospective comparative performance of hemostatic agents including powders in cardiothoracic procedures. 15,19 –30 However, to the best of our knowledge, this report presents the first direct comparison of 2 powdered hemostatic agents during cardiac surgical procedures.
Postoperative bleeding following cardiothoracic procedures remains an important concern with multiple avenues of investigation continuing to assess factors predisposing and potentially influencing the severity of this adverse event. Recent areas of interest include: development of new antifibrinolytic agents to reduce bleeding; 31 measurement of platelet function to reduce bleeding and mortality; 32 single or dual agent antiplatelet therapy to maximize bypass graft patency, but increase postoperative bleeding; 33 –36 direct linkage of postoperative cardiac surgical bleeding with increased cost; 4,5 safe and efficacious use of postoperative fibrinogen concentrates; 37,38 role of patient blood management in reducing blood product transfusion and improving cardiac surgical outcomes; 39,40 transfusion reductions associated with the use of prothrombin complex concentrate; 41,42 morbidity associated with reoperation for bleeding following cardiac surgery; 3,43,44 methods of employing thromboelastometry (ROTEM) during cardiac surgery; 45,46 contribution of albumin/gelatin to postoperative bleeding; 47 –49 advantages of anticoagulation management using thromboelastography (TEG) during cardiopulmonary bypass; 50 –52 benefit of thrombin inhibitors for pediatric VAD anticoagulation; 53,54 reduction of postoperative blood loss using Unilastin as an antifibrinolytic agent; 55,56 and blood loss leading to multisystem organ failure and hematologic complications following LVAD insertion. 7,57
The CP agent described in this study contains porcine collagen, bovine chondroitin sulfate, and human thrombin. 16 The collagen provides initiation of coagulation and platelet activity, the chondroitin sulfate assists with adherence of the wound to surrounding tissues, and the thrombin adjunctively functions to enhance the conversion of fibrinogen to fibrin. CP is the only currently commercially available, powdered hemostatic agent consisting of multiple components as well as the only one containing thrombin. A scanning electron micrograph (800×) illustrates the clot formation with the interaction of red cells, collagen, and chondroitin sulfate (Figure 6).

Scanning electron micrograph (800×) of the fibrin network trapping red blood cells (A and B). In addition, chondroitin sulfate constitutes a thin layer over the fibrin network (C).
The authors have noted several advantages to using a hemostatic powder such as CP including: immediate availability with essentially no required preparation eliminating time consuming efforts by operating room staff to prepare the agent as well as the possibility that pre-preparing the agent before the case to save time would result in product waste if the agent is not actually needed; ease of application to both local and large areas as the powder can be easily placed focally as well as to large surface areas; clear glaze like appearance of the product after hemostasis is achieved; and versatility permitting use in both open and minimally invasive procedures using a 35 cm nozzle extension. 58
Strengths and Limitations
There are limitations to the use of any retrospective data particularly that comparing more recent clinical information for CP (2018-2019) to that obtained from a historical control group such as PP (2011-2013). The five-year time separation between groups encompasses a variety of changes in surgical techniques and methods that could affect the parameters being measured. During this time period, there have been changes in the way transfusion of blood products are managed (thresholds and triggers for transfusion have changed). Blood conservation protocols are now commonplace in hospitals and a multitude of literature now exists describing the deleterious side effects of transfusion on morbidity, mortality, cost, length of stay, etc. HMH hospital adopted these types of protocols in the 2014/2015-time frame and the overall transfusion of products during cardiac surgery and in the ICU afterward significantly decreased (internal institutional data). Additionally, intravenous agents to aid in blood coagulation status have been developed including non-activated factor concentrates. Prothrombin complex concentrate (K-Centra) was given to at least approximately 70% to 80% of the CP group. In the PP group, blood coagulation factor (Factor VIIa) was used in some patients (approximately 20%-30% of patients). Antifibrinolytic therapy in the form of epsilon aminocaproic acid (EACA) was also used in the majority of both groups of patients (>90%). The use of EACA during the majority of all cardiac procedures requiring cardio-pulmonary bypass has been a standard used at HMH over the time span encompassing both the PP and CP groups. Finally, with respect to potential bleeding related the different LVADs used in this study, all of the pumps in AristaTM earlier group were Heartmate 2TM or HeartwareTM and Heartmate 2TM, while in the HEMOBLASTTM later group the pumps were HeartwareTM, or Heartmate 3TM. All of these pumps in both studies are continuous flow and no pulsatile pumps (i.e. no HeartmateTM 1 or Novacor) were used. The Heartmate 2TM does require creation of a small pump pocket with a potential for bleeding, but not nearly to the degree the earlier pulsatile pumps require. In addition, von Willebrand Factor (vWF) has been studied in the continuous flow pumps used in both groups (i.e. Heartmate 2TM, Heartmate 3TM, and HeartwareTM) and may affect coagulation status/thrombosis especially in the long term setting during pump support. In this study, we are dealing with the intraoperative and immediate postoperative period up to 48 hours (i.e. peri-operative bleeding) and we are not aware that continuous flow vWF has been studied in this short, acute, initial implant period (i.e. 2 days) with respect to its risk of bleeding/thrombosis. 59 Thus, the evolution of cardiac surgical, clinical care has almost certainly affected some of the parameters measured in this study in addition to the use of CP. However, it is believed that the extent of the differences between the CP and PP groups described in the paper are so significant as to not be completely explained by the above-mentioned confounding factors.
Conclusion
Multiple significant differences in parameters used to measure the bleeding associated with complex cardiac surgical procedures have been described in this comparison between the use of CP and PP. The use of CP resulted in important benefits indicative for the achievement of improved hemostasis with no observed safety concerns. Despite the retrospective nature of this study, the data suggests the benefits of using CP in this environment.
Footnotes
Authors' Note
Concept/design: Bruckner, Spotnitz, Gillen, and Manson; Data analysis/interpretation: Bruckner, Spotnitz, Gillen, and Manson; Drafting article: Spotnitz; Critical revision of article: Bruckner, Spotnitz, Suarez, Gillen, and Manson; Data collection: Bruckner, Suarez, Loebe, and Ngo. Data used for this trial is available from the authors upon request.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Bruckner, Mr. Ngo, and Dr. Gillen are consultants of Biom’Up France SAS. Drs. Spotnitz and Manson are employees of Biom’Up France SAS.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The development of this manuscript was supported by Biom’Up France, SAS.
