Abstract
Prevention of venous thromboembolism (VTE) is essential in neurological patients. Little is known about the optimal duration, efficacy, and safety of prolonged off-label use of low-molecular-weight heparin (LMWH). We enrolled n = 1176 early neurological rehabilitation cases in a retrospective study. In most cases (n = 1151, 97.9%), 4000 anti-Xa (activated coagulation factor X [factor Xa]) units enoxaparin were administered, only 25 received 2000 units for approximately 2 months (mean of 57.5 days). In 969 cases, enoxaparin was administered for more than 2 weeks. Incidence of symptomatic deep vein thrombosis (DVT) and pulmonary embolism (PE) were 0.43% (n = 5) and 1.11% (n = 13), respectively. Hemorrhages during enoxaparin therapy were more frequent. Bleeding occurred in 1.96% (n = 23) of cases, mainly gastrointestinal, urinary tract, and vitreous body bleeding. In short-term (up to 2 weeks) treatment, bleeding and VTE were more frequent than in long-term treatment. Results from this study suggest that prolonged enoxaparin thromboprophylaxis in neurological rehabilitation is safe and effective.
Introduction
Low-molecular-weight heparin (LMWH) offers an effective prevention of venous thromboembolic events in neurological patients, in particular, after ischemic stroke. 1 –4 Reviewing the prevention of venous thromboembolism (VTE) after acute ischemic stroke with LMWH enoxaparin (PREVAIL) study data, 3 Kase and coworkers 4 found that clinical benefits of thromboprophylaxis with enoxaparin in patients with acute ischemic stroke were not diminished by a poorer 3-month neurologic outcome or an increased rate of symptomatic intracranial hemorrhage compared to unfractioned heparin (UFH).
In the absence of heparin prophylaxis, the incidence of deep vein thrombosis (DVT) in patients with stroke is high (up to 50%). 5,6 As a result, clinically apparent pulmonary embolism (PE) seems to be very common (10%-13%). 6 A small study by Dickmann et al even revealed evidence of PE in 39% of patients 10 days after stroke. 7 It is believed that PE accounts for 13% to 25% of early deaths after stroke. 6 The risk of PE also extends to neurological rehabilitation. In a retrospective study of 363 patients who did not receive heparin, 4% developed PE up to 4 weeks after stroke (on average 11 days after beginning of rehabilitation). 8 Due to immobility, up to 33% of patients admitted to a rehabilitation unit had DVT revealed by bilateral venography 9 Even with heparin prophylaxis (enoxaparin and nadroparin), DVT and PE in neurological rehabilitation patients occur frequently. A recent Polish study (n = 498) showed a PE incidence of 1.9% up to 3 months after stroke, and DVT was observed in 2% of cases. 10
Enoxaparin is also effective in neurological rehabilitation of patients after spinal cord injury and Guillain-Barré syndrome. 11 –13 Comparing enoxaparin and UFH, there was a lower rate of thrombotic and thromboembolic events (8.5% vs 21.7%) up to a 6-week rehabilitation phase. 11 Another study showed a DVT and PE incidence of 2.0% among spinal cord injury patients treated with twice-daily enoxaparin injections. 12 Patients with Guillain-Barré syndrome frequently have severe immobilization due to tetraparesis. In a study with 73 patients, DVT was observed in 7% (n = 5) patients. 13 Among these patients who developed their DVT, 2 had no anticoagulation at all and 3 were on enoxaparin (1 died from PE). 13 Most DVT cases (4 of 5) occurred during the first 2 months of rehabilitation.
Although enoxaparin and other LMWH have shown to be effective in thromboprophylaxis in a broad spectrum of neurological diseases, only few data are available on long-term treatment. In an orthopedic study (n = 238), enoxaparin was given for a mean of 36.8 days during rehabilitation of patients after major orthopedic surgery. 14 Rate of new proximal DVT was 3.2% in this cohort. 14 Among 64 early neurological rehabilitation patients, only minimal bleeding events were found in 6 patients (2 under tinzaparin and 4 under enoxaparin) over a 2-month observation period. 15 Efficacy and safety in long-term treatment of proximal DVT with enoxaparin were shown by Veiga and coworkers. 16 In all, 100 elderly patients (>75 years) after DVT were given enoxaparin or oral anticoagulation (OAC) for a 1-year surveillance period. Only 8 (16%) of 50 enoxaparin patients had new episodes of VTE compared to 6 (12%) of 50 patients receiving OAC. 16 In each treatment group, 1 patient died from complications related to therapy or thromboembolism (1 case of PE in the enoxaparin cohort, 1 intracranial hemorrhage in the OAC group). 16
Regardless of these promising study results, prolonged use (more than 2 weeks) of LMWH in early neurological rehabilitation patients is off-label, yet. However, there is no doubt that this cohort has an extremely high risk of venous thromboembolic events 17 due to a high degree of immobility and morbidity. 18 This dilemma motivated the authors of this study to carefully review retrospectively their early neurological rehabilitation cases in which enoxaparin was used as thromboprophylaxis. The focus of this analysis was on thrombotic and thromboembolic events (DVT and PE) and adverse events during prolonged enoxaparin therapy (eg, clinically relevant bleeding, heparin-induced thrombocytopenia [HIT]).
Methods
Cases
Retrospectively, early neurological rehabilitation cases (inpatients n = 1176 [682 males, 494 females]) of the BDH—Neurological Center Hessisch Oldendorf, Germany, treated from 2004 to 2008, were evaluated. Mean age (whole sample) was 61.1 years (SD = 17.7; range 14-94).
In most cases (97.9%, n = 1151), thromboembolic risk was considered so high that 4000 anti-Xa units (0.4 mL enoxaparin) were administered subcutaneously once daily. In 25 (2.1%) cases, only 2000 anti-Xa units (0.2 mL) enoxaparin were given.
There was a broad spectrum of neurological diseases (Table 1 ). Most cases were admitted to the Neurological Center because of ischemic stroke (31.0%), head trauma (22.6%), intracerebral hemorrhage (16.6%), and subarachnoidal bleeding (9.0%).
List of Main Diagnoses
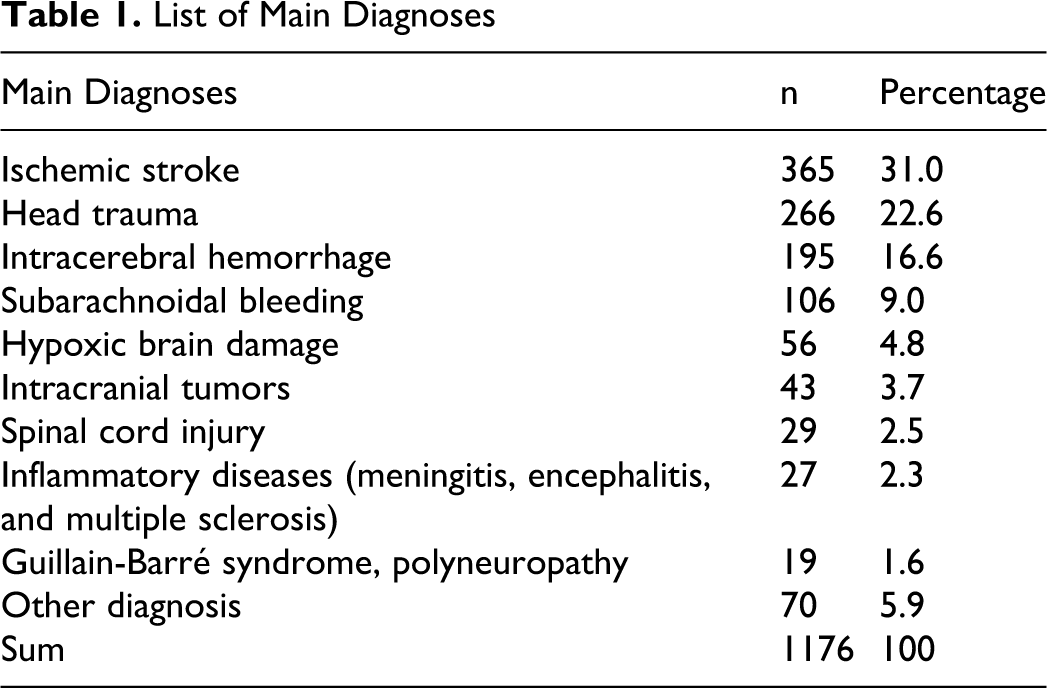
Morbidity was high. Most cases (n = 845, 71.9%) had a patient complication and complexity level ([PCCL]; 0 = minimum, 4 = maximum) of 4 and 22.1% (n = 260) had a level of 3. Immobility was measured with the Early Rehabilitation Barthel Index ([ERBI]; −325 = minimum, +100 = maximum) 19 in half of the cases. Of these, 50.3% (n = 294) had a score between −200 and −75; 45.5% (n = 266) between −75 and 30, and 4.1% (n = 24) less than −200 points. To define the degree of morbidity, the number of concomitant diagnoses (eg, diabetes, arterial hypertension) was recorded. We found a mean of 15.9 concomitant diagnoses (SD = 5.6; range 0-36).
Mean duration of treatment with enoxaparin (whole sample) was approximately 2 months (57.5 days; SD = 49.3; range 1-353). Median duration was 45 days (25th percentile 21.3 and 75th percentile 79 days). Of the 1176 cases, 969 were treated with enoxaparin for more than 2 weeks (prolonged treatment group—group 2). The cases with up to 2 weeks of enoxaparin treatment were named group 1. Characteristics of both groups are presented in Table 2 . Mean duration of enoxaparin prophylaxis in group 1 was 6.9 (4.1) days; and in group 2, it was 68.3 (47.7) days.
Sample Characteristics of Group 1 (≤2 Weeks on Enoxaparin), Group 2 (>2 Weeks on Enoxaparin), and the Whole Population a
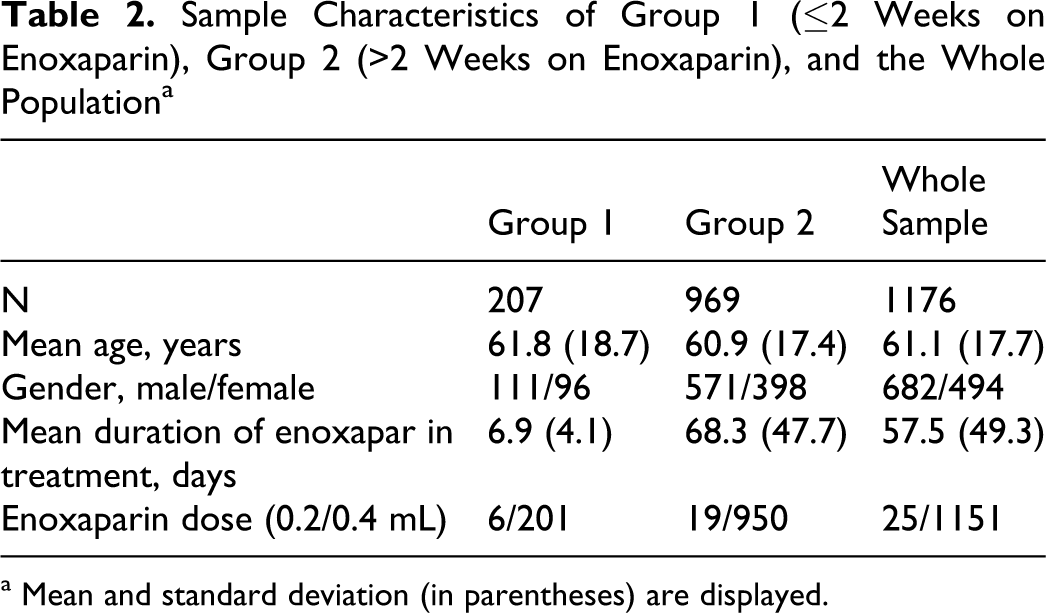
a Mean and standard deviation (in parentheses) are displayed.
Statistics
The data set were analyzed using SPSS software (SPSS Inc, Chicago, Illinois), version 16.0. Mainly, descriptive statistics were calculated. The mean value and standard deviation (in parentheses) are reported in the results section.
Results
Deep Vein Thrombosis
Clinically relevant DVT was observed in 5 cases (incidence 0.43%) only. Deep vein thrombosis was taken into consideration when clinical signs of DVT occurred (pain, swelling) and confirmed by ultrasonography. All patients were on 0.4 mL enoxaparin for a mean of 12.4 (8.0) days when DVT occurred. Patient characteristics are displayed in Table 3 . Mean age of patients with DVT was 56.2 years (SD = 9.3; range 47-71). Of these 5 patients, 2 patients also experienced a clinically relevant PE (confirmed by computed tomography). One male patient (DVT + PE) had a coagulation abnormality (low antithrombin III [AT-III] and protein C), further he was on cortisone medication. All patients with VTE were immobile and bedridden (range of ERBI: −175 to +20). None of these VTEs was fatal.
Deep Vein Thrombosis a
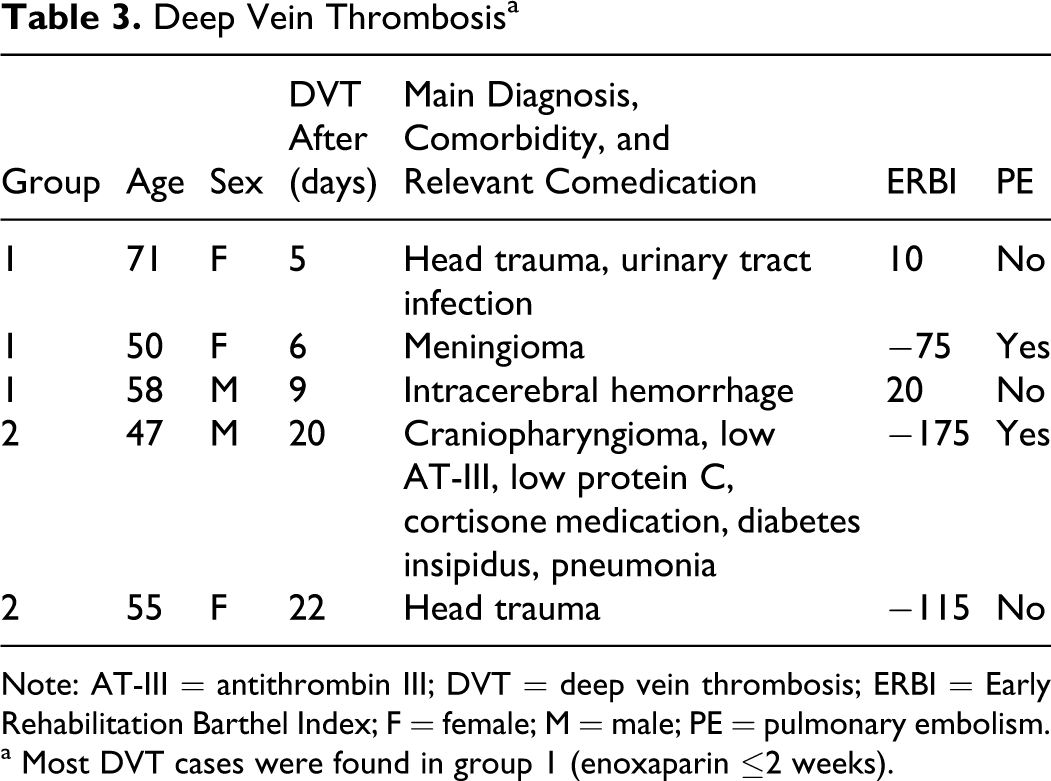
Note: AT-III = antithrombin III; DVT = deep vein thrombosis; ERBI = Early Rehabilitation Barthel Index; F = female; M = male; PE = pulmonary embolism.
a Most DVT cases were found in group 1 (enoxaparin ≤2 weeks).
Most DVT cases were observed in group 1 (3 of 5), only 2 DVT occurred in the prolonged treatment group (after 20 respectively 22 days on enoxaparin prophylaxis).
Pulmonary Embolism
Pulmonary embolism was observed in 13 cases (incidence 1.11%). All patients received 0.4 mL enoxaparin, mean duration of treatment was 15.5 days (12.6). Of 13 patients with PE, 9 died. It has to be taken into account that 3 of 9 fatalities suffered from cancer (glioblastoma WHO grade 4, lung cancer, and rectum carcinoma). Mortality related to thromboembolism was 0.77% for the whole population. Characteristics of PE cases are shown in Table 4 . All patients were immobile and bedridden (range of ERBI: −325 to +5).
Pulmonary Embolism a
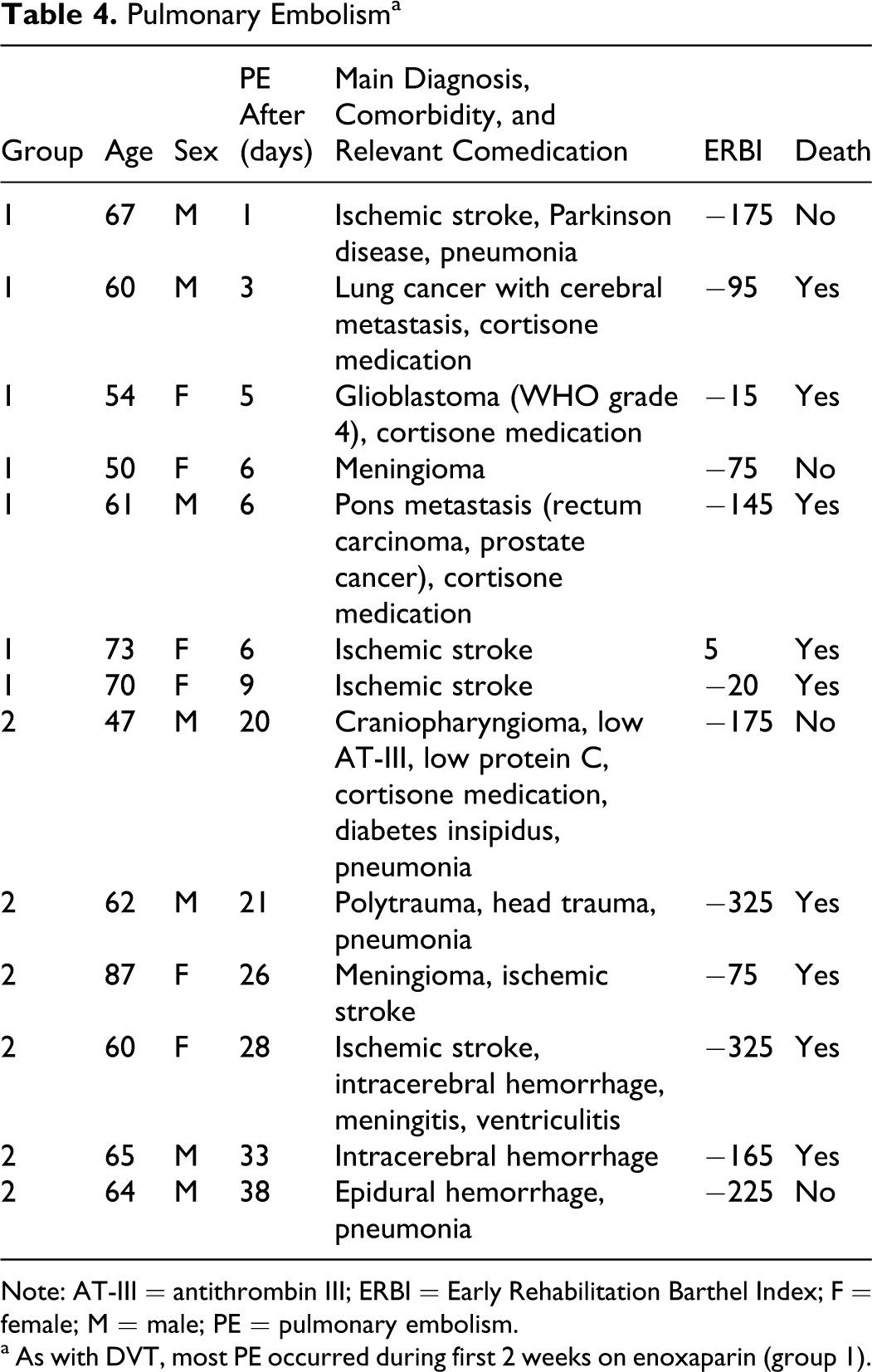
Note: AT-III = antithrombin III; ERBI = Early Rehabilitation Barthel Index; F = female; M = male; PE = pulmonary embolism.
a As with DVT, most PE occurred during first 2 weeks on enoxaparin (group 1).
As with DVT, most PE cases occurred in group 1 (7 of 13, 53.8%). In the prolonged treatment group (>2 weeks), only 6 cases with PE were observed.
Hemorrhagic Adverse Events
Hemorrhagic adverse events were observed (whole sample) in 23 (1.96%) cases, most of them were gastrointestinal (n = 8), urinary tract (n = 5), and vitreous body bleeding (n = 4), Table 5 . Two patients had more than 1 bleeding event. None of them were fatal, but in 6 cases transfusion was vital (range of hemoglobin [Hb] 7.1-7.6 g/dL). Bleeding occurred after a mean of 19.3 (28.3) days. Most of the patients were treated with 0.4 mL enoxaparin (n = 21), only 2 patients received 0.2 mL (Table 5). Mean age of patients with bleeding was 66.0 (15.8) years. It has to be pointed out that 11 of 23 cases (47.8% of hemorrhages) had a comedication consisting of salicylate or clopidogrel. Thus, the bleeding risk of these patients was elevated anyway. Furthermore, it has to be taken into account that patients with subarachnoidal hemorrhage had vitreous body bleeding frequently (so-called Terson syndrome). Of the 4 cases with vitreous body bleeding, 2 patients had subarachnoidal hemorrhage.
Hemorrhagic Adverse Events a
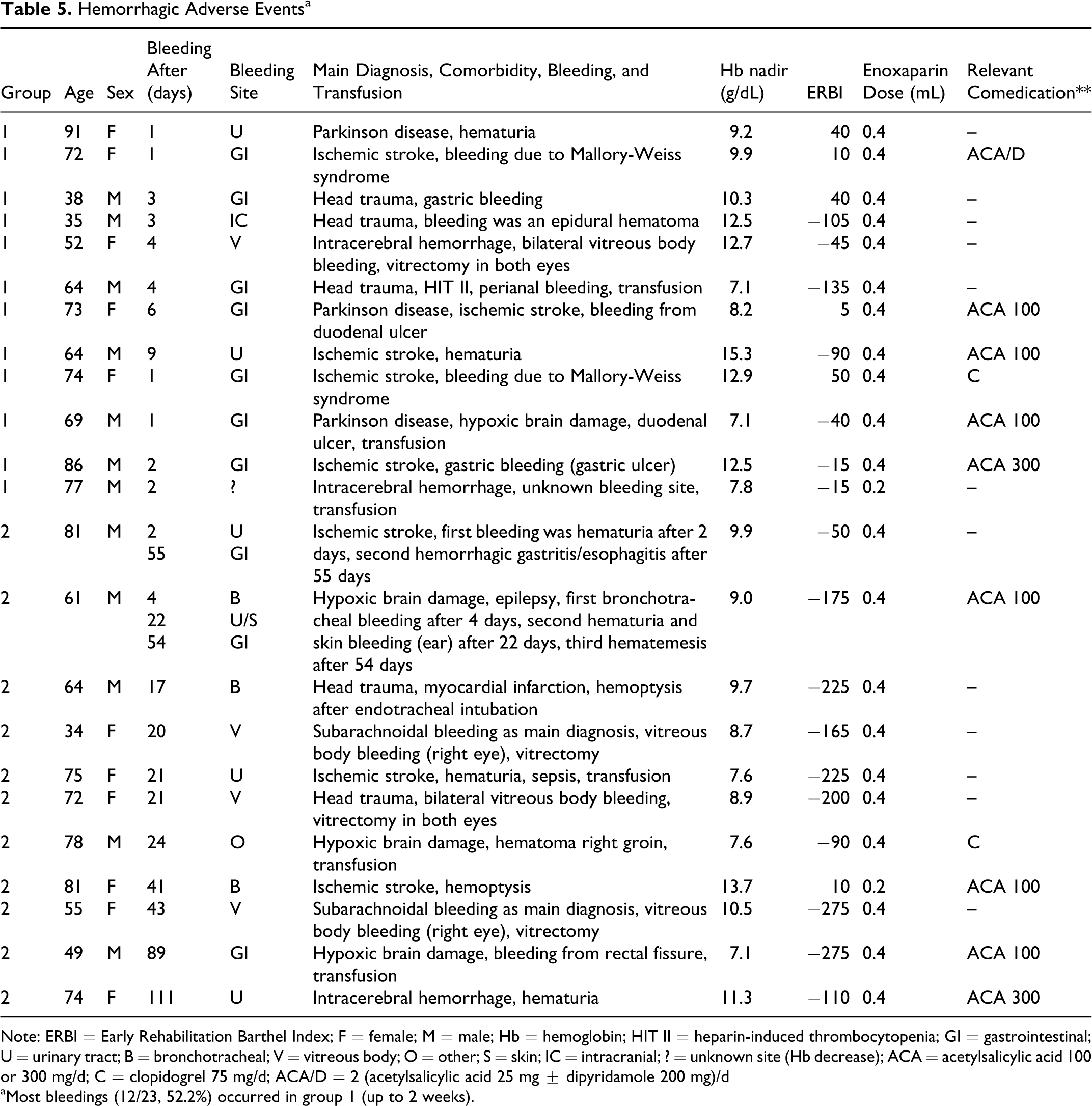
Note: ERBI = Early Rehabilitation Barthel Index; F = female; M = male; Hb = hemoglobin; HIT II = heparin-induced thrombocytopenia; GI = gastrointestinal; U = urinary tract; B = bronchotracheal; V = vitreous body; O = other; S = skin; IC = intracranial; ? = unknown site (Hb decrease); ACA = acetylsalicylic acid 100 or 300 mg/d; C = clopidogrel 75 mg/d; ACA/D = 2 (acetylsalicylic acid 25 mg ± dipyridamole 200 mg)/d
aMost bleedings (12/23, 52.2%) occurred in group 1 (up to 2 weeks).
Most bleedings (12 of 23, 52.2%) were observed in group 1, only 11 of 23 hemorrhages occurred in group 2 (prolonged enoxaparin treatment).
Heparin-Induced Thrombocytopenia
Only 1 patient (male, 64 years, head trauma) had HIT II after 4 days of treatment with 0.4 mL enoxaparin. Thrombocyte count nadir in this case was 60 000/μL (initial count 151 000/μL). After diagnosis of HIT II, thromboprophylaxis was continued with danaparoid. Four more patients had a thrombocytopenia (thrombocyte count 8000-86000/μL) but no HIT was diagnosed. Enoxaparin dose was 0.4 mL in all 4 cases. However, it has to be taken into account that thrombocytopenia might have been caused by another comedication. Of the 4 cases, 3 received anticonvulsant drugs (valproic acid + levetiracatam in 2 cases and carbamazepine in 1 case) known to cause thrombocytopenia.
Discussion
Neurological patients, in particular after stroke, do benefit from a thromboprophylaxis with LMWH. 1 –4 These patients have a substantial risk of experiencing thromboembolic events due to immobility and high comorbidity. Without heparin prophylaxis, the incidence of DVT among these patients may rise up to 50%, clinically significant PE occurs in 10% to 13%. 5,6 We reviewed 1176 early neurological rehabilitation cases, 969 cases were on enoxaparin for more than 2 weeks (prolonged use). Morbidity among this cohort was considerably high. Most of our cases had an ERBI of less than −75 points. This means that they were immobile, bedridden, and dependent on nursing. Although the prolonged use of enoxaparin is off-label in most of these cases, the authors do believe that—due to the high risk of thrombotic and thromboembolic events in this cohort—there is no alternative to heparin prophylaxis. In the majority of cases, 4000 anti-Xa units of enoxaparin were administered for a mean of 2 months (58.6 days). There are only a few studies on safety and efficacy of prolonged use of LMWH over an observational period of one respective 2 months. 14,15 Thus, the current study contributes further knowledge to the use of prolonged thromboprophylaxis with LMWH.
In our cohort, the incidence of clinically relevant DVT was 0.43% only. Pulmonary embolism was diagnosed in 13 cases (incidence 1.11%). Thromboembolic events occurred after a mean of approximately 2 weeks. Mortality (due to PE) was 0.77%. It has to be mentioned that most DVT respectively PE cases were observed in the short-term treatment group (group 1, enoxaparin ≤2 weeks). In the group with prolonged enoxaparin thromboprophylaxis (>2 weeks), only 2 of 5 DVT and 6 of 13 PE cases were found. The authors are well aware that clinically relevant events are just the “tip of the iceberg” because prospective studies have shown much higher incidences of thromboembolic events, 7,9 if asymptomatic VTEs are also taken into account. The retrospective data analysis is a clear limitation of the current investigation. Nevertheless, our clinical data are in line with the results of a previous study on complications in neurological rehabilitation. 10 The authors found a PE incidence of 1.9% up to 3 months after stroke. Deep vein thrombosis was observed in 2%. 10 In our cohort, these incidences were even lower although the cases were more severe.
Clinically relevant—mainly gastrointestinal and urinary—bleeding were recorded with an incidence of 1.96% (whole sample). None of these bleeding was fatal. As with VTE, most bleeding were detected in the short-term treatment group (52.2% of all bleeding). This finding suggests that bleeding and thromboembolic events are more likely to occur during the first 2 weeks of enoxaparin use. Analyzing the whole sample (both groups), bleeding occurred after a mean of 19 days.
Heparin-induced thrombocytopenia II was diagnosed in 1 case only.
Although prolonged use of enoxaparin in early neurological rehabilitation is off-label, it appears to be efficient and safe over a mean period of approximately 2 months. Furthermore, randomized, prospective, and controlled studies are welcome in neurological rehabilitation.
Footnotes
Acknowledgment
The authors would like to thank Ms Angela Baumbach (Study Nurse) for her support.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Sanofi-Aventis Germany.
