Abstract
Objective
This study aimed to investigate the association between the triglyceride-glucose (TyG) index and cryptogenic stroke (CS) in patients diagnosed with patent foramen ovale (PFO) using transesophageal echocardiography (TEE).
Methods
A retrospective, single-center study was conducted at a tertiary education and research hospital from January 2015 to December 2023. The study population included 1017 consecutive patients with sinus rhythm diagnosed with PFO, of whom 210 had CS. The TyG Index was calculated using triglyceride and fasting glucose levels.
Results
Patients with CS were older (47.53 ± 12.34 years) compared to control group (44.40 ± 17.82 years, p = .005). The proportion of males was higher in the CS group (56.2%) compared to the control group (48.3%, p = .042). Laboratory findings revealed higher TyG Index (8.87 ± 0.51 vs 8.63 ± 0.55, p < .001) in patients with CS. TyG Index was an independent predictor of CS in patients with PFO (OR: 2.832, 95%CI: 1.979-4.053 p < .001).
Conclusion
Elevated TyG Index levels was associated with CS in patients with PFO. The TyG index may serve as a useful biomarker for assessing CS risk in this population.
Keywords
Introduction
Cryptogenic stroke (CS), defined as a stroke of unknown origin after thorough diagnostic evaluation, remains a significant clinical challenge.1,2 Patent foramen ovale (PFO) is a potential etiological factor in a subset of these patients. 3 PFO is a common congenital cardiac anomaly. It is characterised by an incomplete closure of the atrial septum, which facilitates paradoxical embolism and subsequent stroke. 4 Despite the established association between PFO and CS, the precise mechanisms and additional risk factors contributing to stroke in these patients remain unclear. 5
The triglyceride-glucose (TyG) index, a surrogate marker of insulin resistance, is a reliable predictor of cardiovascular events.6,7 It is well established that insulin resistance contributes to both atherosclerosis and thrombogenesis, which are critical in the pathophysiology of stroke.8,9 However, there is a clear need for further research into the relationship between the TyG Index and CS in patients with PFO. Understanding this association will undoubtedly provide valuable insights into the metabolic contributions to stroke risk and identify potential biomarkers for risk stratification.10,11 This study set out to investigate the relationship between the TyG Index and CS in patients diagnosed with PFO using TEE.
Methods
Study Design and Population
This retrospective, single-center study was conducted at a tertiary education and research hospital from January 2015 to December 2022. The study included 1017 consecutive patients with sinus rhythm diagnosed with PFO via TEE. Of these patients, 210 had CS and 807 did not. Patients were excluded if they were younger than 18 years, had a known cause of stroke other than CS, had incomplete medical records, or had significant comorbid conditions that could confound the results, such as severe renal or hepatic disease and active malignancy.
The data were extracted from the hospital's electronic medical records. The data collected included the following: demographic characteristics (age, sex), clinical history (hypertension (HT), diabetes mellitus (DM), hyperlipidaemia (HPL), smoking status), laboratory results (fasting blood glucose, fasting triglycerides, total cholesterol), and anatomical features assessed by TEE (PFO tunnel diameter, PFO tunnel length, presence of interatrial septal aneurysm (ISA), prominent eustachian valve, left ventricular ejection fraction (LV EF)). This study was conducted in accordance with the principles outlined in the Declaration of Helsinki and received approval from the Institutional Review Board of the tertiary education and research hospital (Ethics Committee Approval No: 0049/2024). As the study was retrospective, the Medical Ethics Committee waived the need for informed consent.
Cryptogenic stroke (CS) is a diagnosis made by the neurology department based on clinical evaluation and imaging studies, following the exclusion of other known causes of stroke. CS was diagnosed when all other known causes of stroke were excluded and the absence of large artery atherosclerosis, cardioembolism, small artery disease, and other specific etiologies was confirmed after a comprehensive workup, including brain imaging (computed tomography or magnetic resonance imaging), vascular imaging (computed tomography angiography, magnetic resonance angiography, Doppler ultrasound), cardiac evaluation (electrocardiogram, echocardiography), and routine laboratory tests. Furthermore, atrial fibrillation was definitively excluded through 24-h Holter monitoring.
TEE Protocol
The TEE protocol involved a comprehensive assessment of cardiac structures, including the examination of the interatrial septum for the presence of PFO and atrial septal aneurysm (ASA). The TEE examinations were performed by experienced cardiologists using the latest echocardiography equipment, including a Philips iE33 system (manufactured in the Netherlands). The examination lasted, on average, 10 min per patient. All images were digitally recorded for subsequent analysis.
We diagnosed the patient with a PFO using TEE. TEE is the gold standard for diagnosing PFO. Its high sensitivity and specificity make it the best method available. A contrast study was performed by injecting agitated saline into a peripheral vein to clearly visualise the right-to-left shunt across the atrial septum. The presence of bubbles in the left atrium within three cardiac cycles of their appearance in the right atrium definitively confirmed the diagnosis of PFO. We measured the diameter of the PFO tunnel at its widest point during diastole. We measured the length of the PFO tunnel from the entrance to the exit during the cardiac cycle. An IAS was present if there was a phasic septal excursion of over 10 mm from the midline into either the left or right atrium during the cardiac cycle. We used colour Doppler imaging to visualise the blood flow across the PFO and assess the shunt size and direction. We calculated the Risk of Paradoxical Embolism (ROPE) Score for all patients with PFO to estimate the probability of a CS being associated with PFO. 5 Blood samples were collected from all patients upon admission to the hospital. We measured fasting triglyceride and fasting blood glucose levels using standard enzymatic methods. The TyG index is a reliable marker of insulin resistance. The TyG index was calculated as the natural logarithm (Ln) of the product of plasma glucose and TG using the formula: Ln (TG [mg/dL] × glucose [mg/dL]/2).
Statistical Analysis
Continuous variables were presented as the mean ± standard deviation and were compared using t-tests. Categorical variables were expressed as frequencies and percentages and were compared using chi-square tests. A logistic regression analysis was conducted to identify the most significant predictors of CS. We estimated the predictive value of the TyG Index, triglyceride, glucose, and ROPE score by calculating the areas under the receiver operating characteristic curve (ROC). We used the DeLong test to compare the area under the curve (AUC) with each of these parameters. 12 Furthermore, we estimated the increased discriminative value of the TyG Index using net reclassification improvement (NRI) and integrated discrimination improvement (IDI). 13 A p-value of less than .05 was considered statistically significant. All statistical analyses were conducted using SPSS software, version 26 (SPSS Inc., Chicago, IL, USA), and R software.
Results
The study included 1017 patients diagnosed with PFO via TEE. Of these patients, 210 had CS and 807 did not. The mean age of patients with CS was higher than that of patients in the control group (47.53 ± 12.34 years vs 44.40 ± 17.82 years, p = .005). There was a higher proportion of males in the CS group compared to the control group (56.2% vs 48.3%, p = .042). Furthermore, the CS group had a higher prevalence of hypertension, diabetes mellitus, hyperlipidaemia, and smoking compared to the control group (Table 1). There was a difference in the prevalence of hypertension, diabetes mellitus, hyperlipidemia, and smoking between the two groups (2.9% vs 0.035%, p = .035; 20.5% vs 10.3%, p < .001; 42.9% vs 25.9%, p < .001, respectively).
Baseline Demographic and Clinical Characteristics of the Study Groups.
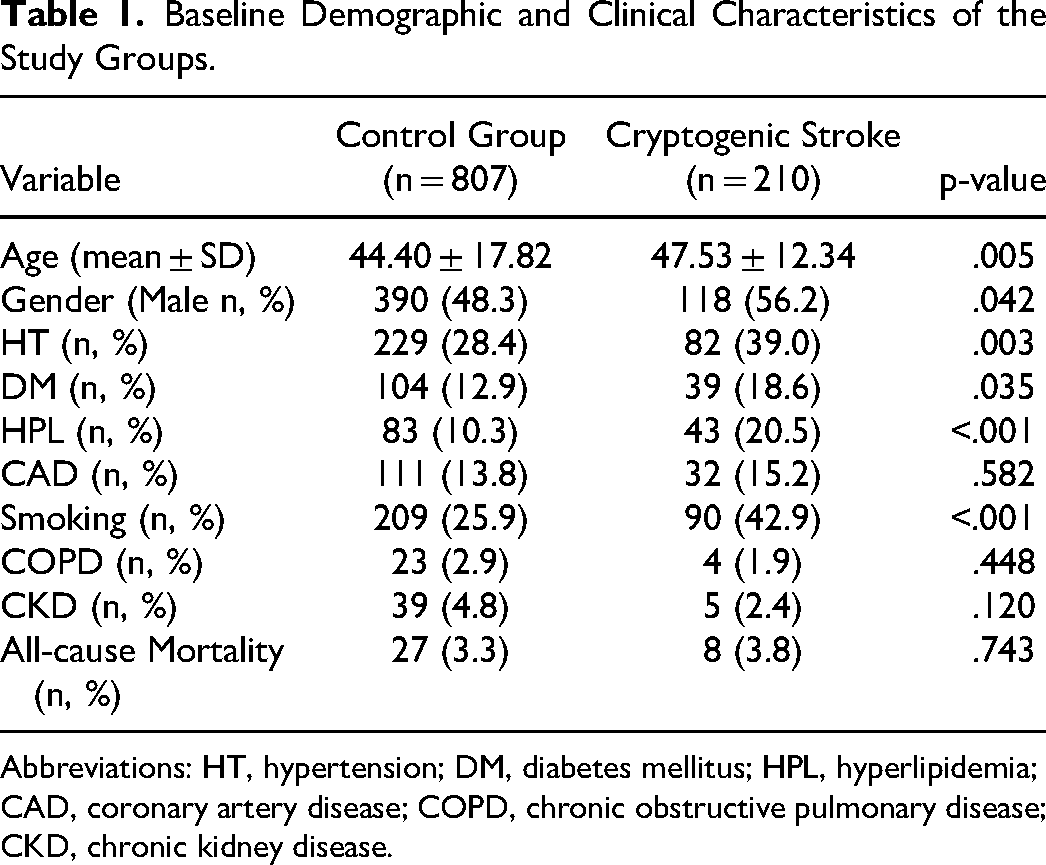
Abbreviations: HT, hypertension; DM, diabetes mellitus; HPL, hyperlipidemia; CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; CKD, chronic kidney disease.
Laboratory results showed higher levels of total cholesterol, triglycerides, and TyG Index in patients with CS compared to the control group. The mean total cholesterol was 199.46 ± 47.89 mg/dL in the CS group versus 188.42 ± 47.76 mg/dL in the control group (p = .007). The CS group had higher triglyceride levels (156.49 ± 71.81 mg/dL vs 128.98 ± 69.49 mg/dL, p < .001). The mean TyG Index was also higher in the CS group than in the control group (8.87 ± 0.51 vs 8.63 ± 0.55, p < .001).
The TEE findings clearly indicated differences between the two groups (Table 2). Patients with CS had a larger mean PFO tunnel diameter (2.18 ± 1.18 mm vs 1.79 ± 1.11 mm, p = .020) and a higher prevalence of ISA (49.0% vs 34.3%, p < .001). However, there were no statistically significant differences in PFO tunnel length (11.48 ± 3.88 mm vs 11.31 ± 4.48 mm, p = .654) or LV EF % (59.17 ± 5.22% vs 58.32 ± 6.71%, p = .086). The mean ROPE score was higher in patients with CS compared to the control group (6.47 ± 1.74 vs 4.76 ± 2.10, p < .001, Table 3). The ROPE Score is an invaluable tool for identifying patients at an elevated risk of CS. Furthermore, 128 patients underwent PFO closure during the study period.
Echocardiographic Findings.
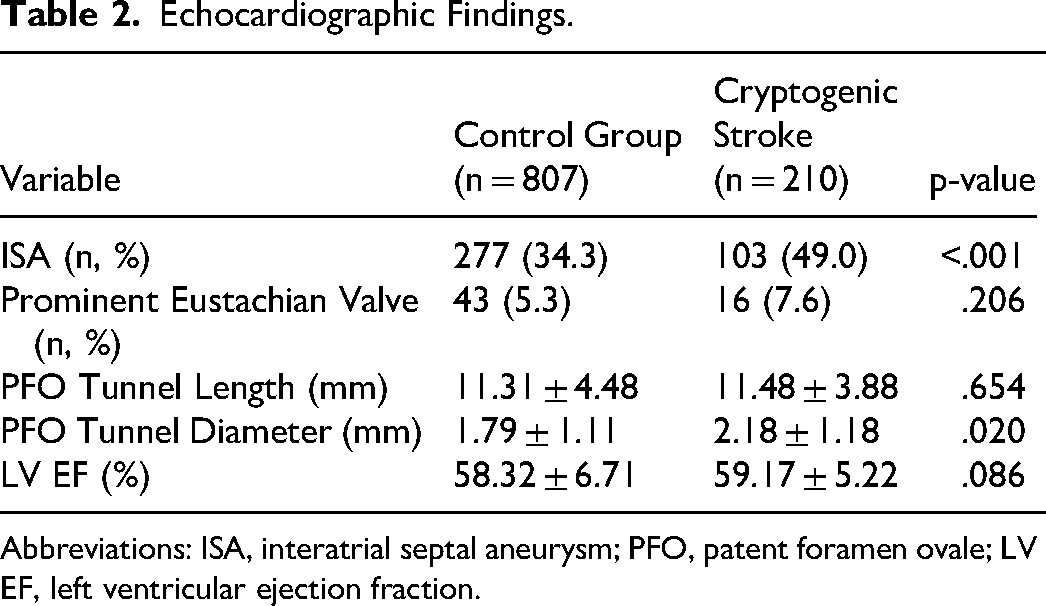
Abbreviations: ISA, interatrial septal aneurysm; PFO, patent foramen ovale; LV EF, left ventricular ejection fraction.
Laboratory and Index Parameters of the Study Groups.
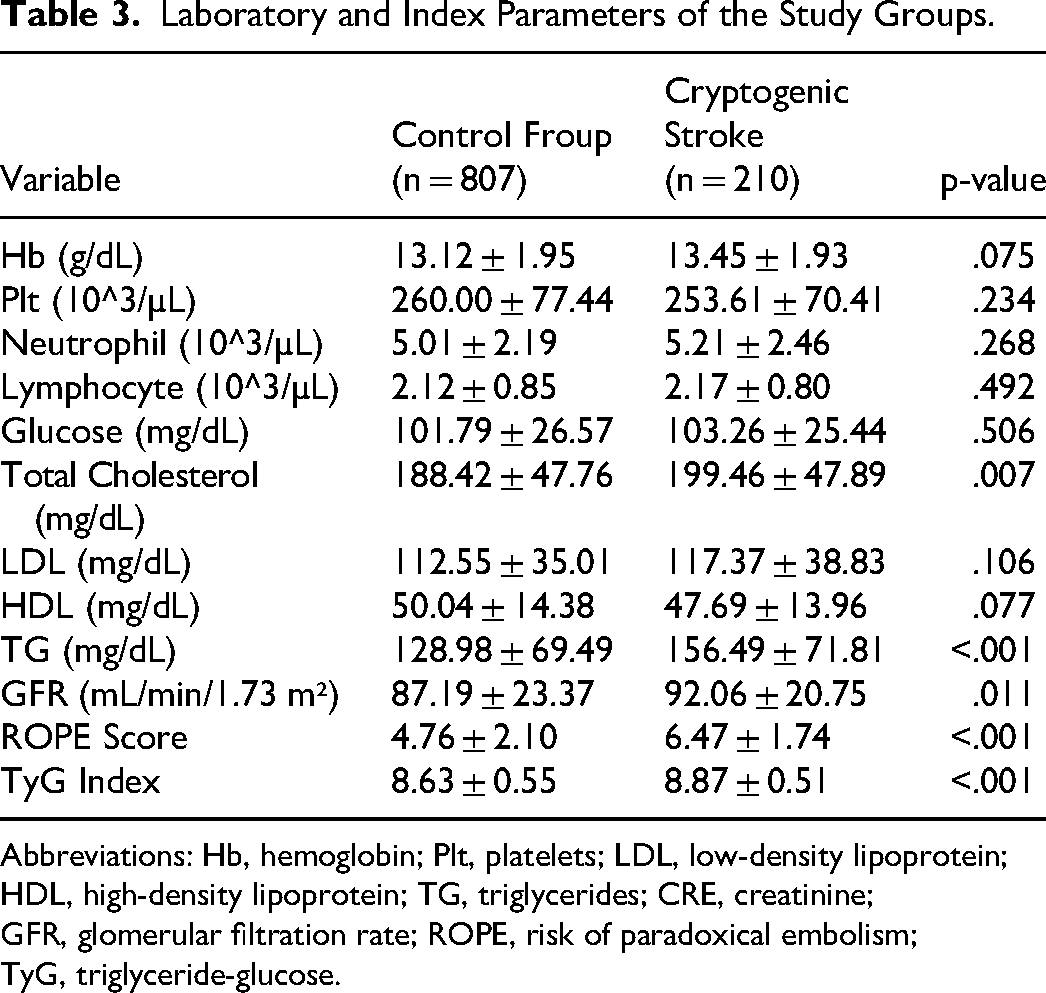
Abbreviations: Hb, hemoglobin; Plt, platelets; LDL, low-density lipoprotein; HDL, high-density lipoprotein; TG, triglycerides; CRE, creatinine; GFR, glomerular filtration rate; ROPE, risk of paradoxical embolism; TyG, triglyceride-glucose.
Logistic regression analysis identified several predictors of CS. An elevated TyG Index was an independent predictor of an increased risk of CS (OR: 2.832, 95% CI: 1.979-4.053, p < .001, Table 4). The TyG Index has an AUC of 0.647 (95% CI: 0.606-0.687, p < .001, Figure 1) for predicting CS. A TyG Index of 8.75 or above was a clear indicator of severe CS, with an impressive 61% sensitivity and 60% specificity. The TyG Index is more accurate than glucose alone in predicting CS. AUC: The TyG Index was more accurate than glucose alone in predicting CS (AUC: 0.647 vs 0.553, z = 3.883, difference p < .001; NRI: 29.8%, p < .001; IDI: 0.026, p < .05, Figure 1). Furthermore, the TyG Index outperformed triglycerides in predicting CS. AUC: The TyG Index was more accurate than glucose alone (AUC: 0.647 vs 0.628, z = 2.031, p = .042; NRI: 35.7%, z = 4.612, p < .001; IDI: 0.020, p < .05, Figure 1). Furthermore, the addition of the TyG Index to the ROPE score resulted in an AUC of 0.776 (95% CI: 0.741-0.811, z = 4.561, p < .001, Figure 2). The addition of the TyG Index to the ROPE score resulted in a NRI of 48.1% (z = 6.212, p < .001) and an IDI of 0.061 (p < .05).
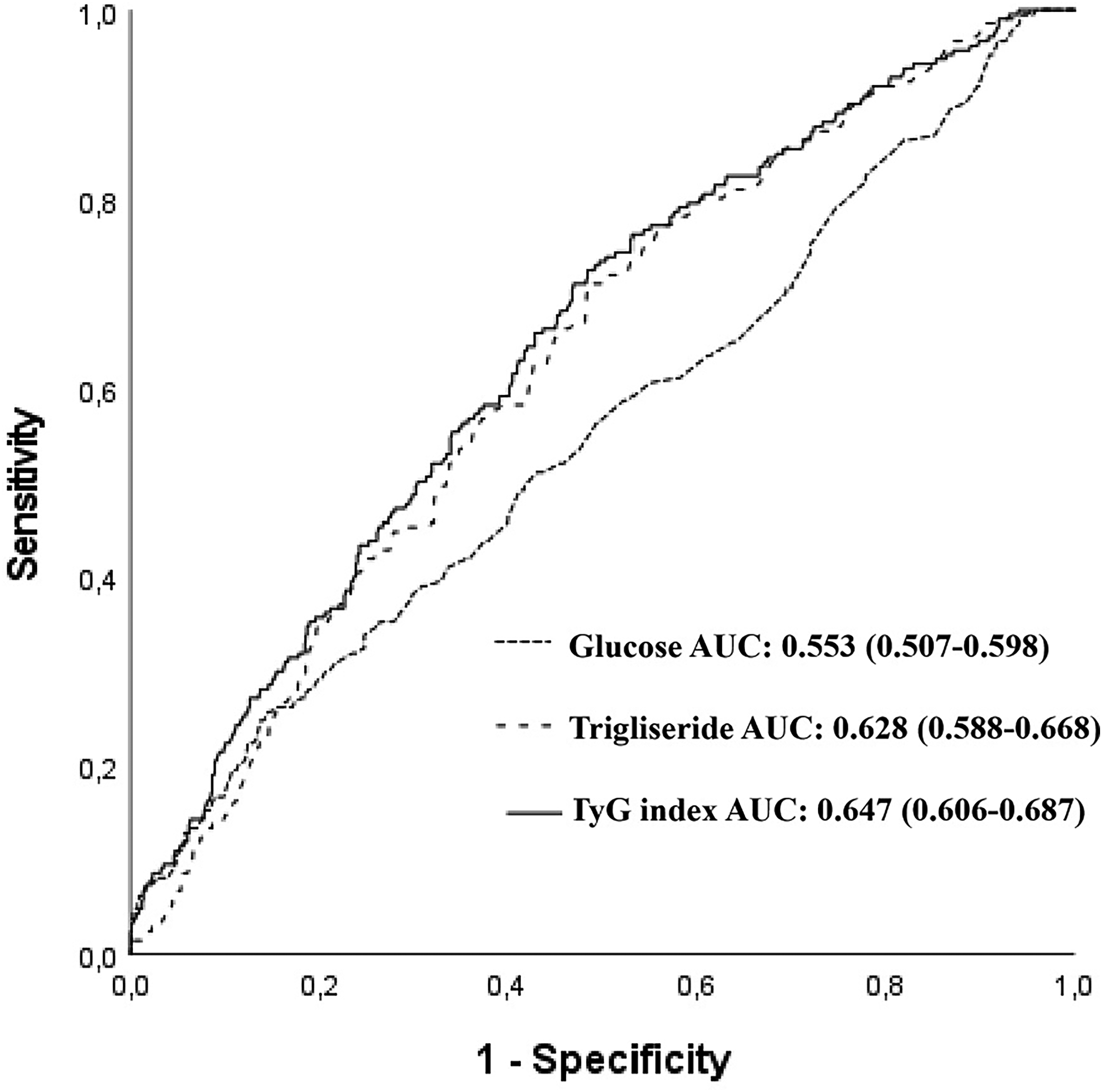
Receiver operating characteristic (ROC) curves for the glucose, triglyceride, and triglyceride glucose index for predicting cryptogenic stroke.
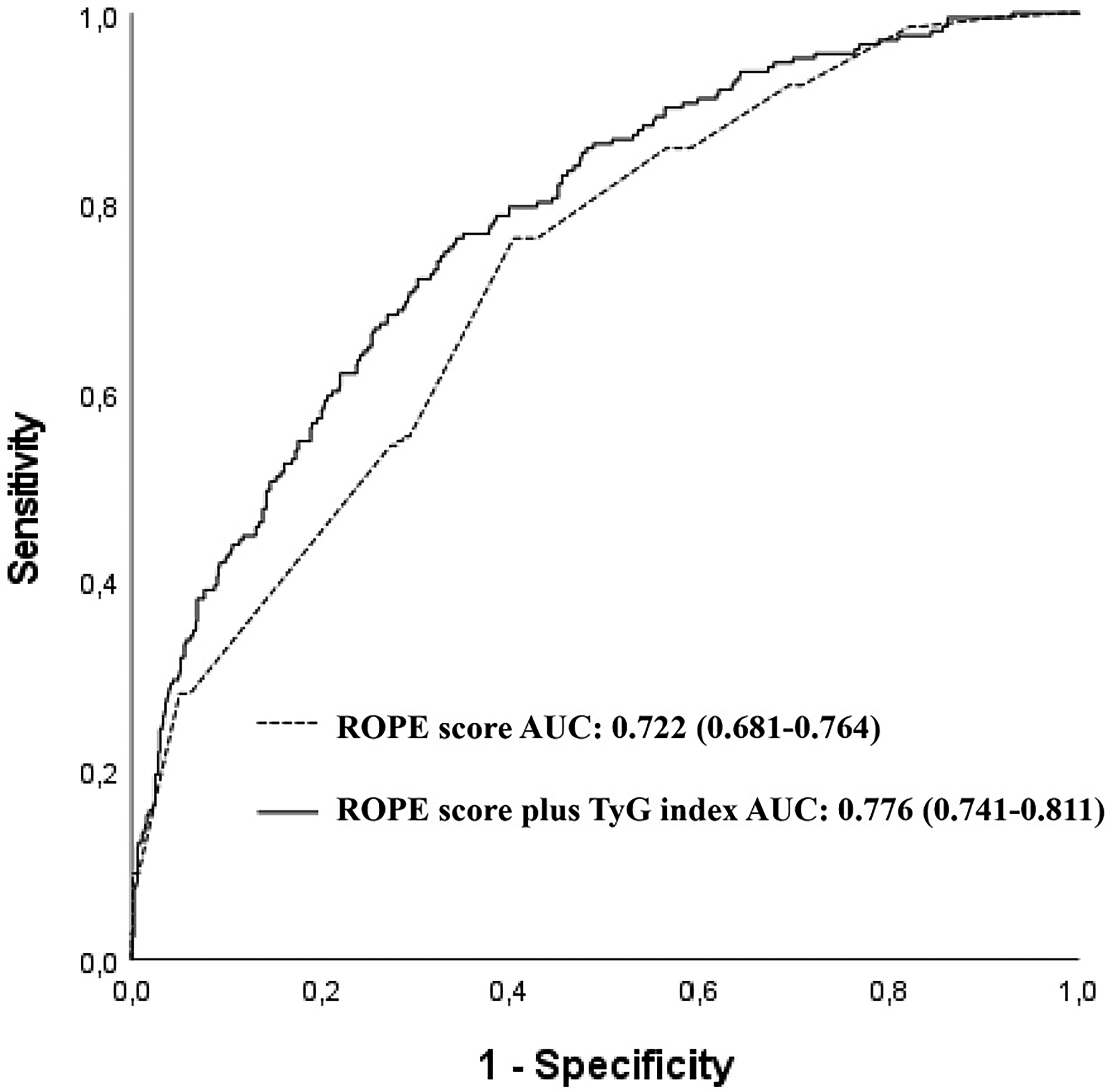
Receiver operating characteristic (ROC) curves for the risk of paradoxical embolism score, and the risk of paradoxical embolism plus trigliseride glucose index for predicting cryptogenic stroke.
Univariate and Multivariate Analysis for Prediction of CS.
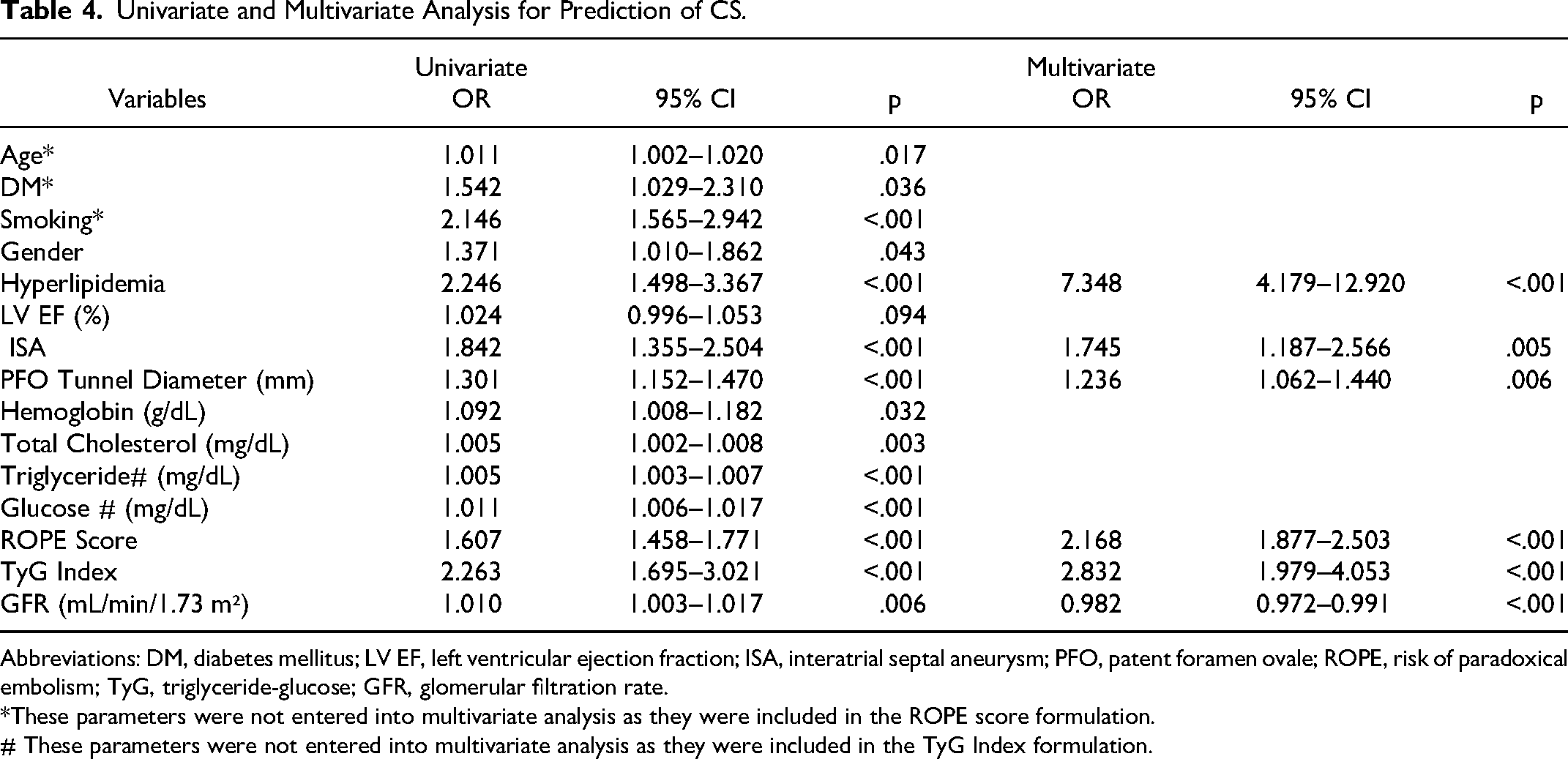
Abbreviations: DM, diabetes mellitus; LV EF, left ventricular ejection fraction; ISA, interatrial septal aneurysm; PFO, patent foramen ovale; ROPE, risk of paradoxical embolism; TyG, triglyceride-glucose; GFR, glomerular filtration rate.
*These parameters were not entered into multivariate analysis as they were included in the ROPE score formulation.
# These parameters were not entered into multivariate analysis as they were included in the TyG Index formulation.
Discussion
This is the first study to investigate the relationship between the TyG index and CS in patients diagnosed with PFO. Our study showed that the TyG index was an independent predictor of CS in these patients.
CS presents a considerable clinical challenge due to its unknown etiology, despite comprehensive diagnostic evaluations. Emerging evidence definitively shows that potential causes of CS include paradoxical embolism through a PFO, small vessel disease, occult atrial fibrillation, and other undiagnosed cardiac sources of emboli. PFO is a clear culprit in a subset of CS cases, as it allows venous thrombi to traverse into the arterial system, leading to cerebral embolism.14,15 However, we can be sure that the presence of a PFO alone does not account for all cases of CS. This indicates that additional risk factors, such as genetic predispositions, coagulopathies, and other cardiovascular anomalies, play a significant role.16,17 Recent studies have shown that multi-faceted diagnostic approaches are essential for identifying the complex etiology of CS. Comprehensive cardiovascular assessments and long-term monitoring are therefore vital. 18
The TyG Index is a reliable predictor of cardiovascular events, including stroke.19,20 Insulin resistance undoubtedly contributes to atherosclerosis and thrombogenesis through various mechanisms, including the promotion of endothelial dysfunction, the enhancement of inflammatory responses, and the increase of platelet aggregation.21,22 An elevated TyG Index level is a clear sign of underlying metabolic dysfunction, which predisposes patients to thrombosis and embolism. 23 In patients with PFO, this metabolic dysfunction undoubtedly elevates the risk of thrombus formation in the venous system, which can then embolise through the PFO to the cerebral circulation, resulting in CS.3,24 Our findings demonstrate that the TyG Index is a valuable biomarker for identifying patients at heightened risk of CS, thereby facilitating targeted prevention and management strategies.
Our study definitively showed that patients with CS were significantly older than the control group. This is in line with previous research that has identified older age as a risk factor for CS in PFO patients.11,25 Furthermore, our findings revealed a higher proportion of males in the CS group compared to the control group, which aligns with the results of previous studies that reported a male predominance in CS cases.26,27 Our study clearly showed that the prevalence of hypertension and diabetes mellitus was significantly higher in CS patients. This is in line with the findings of recent meta-analyses which identified hypertension and diabetes as common comorbidities associated with an increased risk of stroke.28,29 These conditions are well-established as significant contributors to the risk profile for CS in patients with PFO.30,31 Our study showed a s higher prevalence of hyperlipidaemia and smoking in the CS group. These findings align with the existing literature, which has established hyperlipidaemia and smoking as significant risk factors for stroke.32,33 It is clear that managing lipids is crucial in reducing the incidence of CS. Elevated lipid levels are associated with an increased risk of stroke, which highlights the importance of lipid management in preventing CS. CS patients had higher total cholesterol and triglycerides. The elevated TyG Index levels in CS patients are in line with previous research that has identified the TyG Index as a reliable predictor of cardiovascular events and insulin resistance.6,34
Our study clearly demonstrated significant anatomical differences between the CS and control groups. CS patients had a significantly larger mean PFO tunnel diameter and a greater presence of ISA. 35 These findings are corroborated by Overell et al 36 and Turc et al, 37 who definitively reported that larger PFO sizes and the presence of ISA are associated with an increased risk of stroke. Furthermore, Zhu et al 38 definitively stated that specific anatomic features of PFO, such as tunnel diameter and presence of ISA, significantly impact the right-to-left shunt and stroke risk.
This study has several limitations that must be acknowledged. Firstly, as a retrospective, single-centre study, there will undoubtedly be inherent biases in patient selection and data collection. Our findings may not be generalisable to other populations and settings. Secondly, the study relied on medical records for data extraction, which undoubtedly resulted in missing or incomplete information. Third, CS was diagnosed based on the absence of identifiable causes, which can vary between clinicians and institutions. Fourthly, the study did not account for potential confounders such as medication use, lifestyle factors, and genetic predispositions. Finally, the TyG Index was identified as an independent predictor of CS. Further prospective studies are needed to validate its utility as a biomarker for CS risk in patients with PFO.
Conclusion
This study demonstrates that elevated TyG Index levels was associated with CS in patients with PFO. The findings suggest that the TyG Index may serve as a valuable biomarker for assessing the risk of CS in these population.
Footnotes
Acknowledgements
None.
Author Contributions
All authors contributed to the study's conception and design. Material preparation, data collection, and analysis were performed by [TK, HSI, SA, FTC, FE, AC]. The primary draft of the manuscript was written by [FE], [TK], [AC], [SA] and [UK]. Writing-review and editing of the final version of the manuscript were carried out by [TK], [FE], [HSI], [AC], [FTC], and [UK]. [MK], and [TK] add critical points to the study. All authors read and approved the final manuscript.
Availability of Data and Materials
The data supporting this study's findings are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
