Abstract
Objective
This study aims to explore the optimal predictors of HBV-associated HCC using Lasso, and establish a prediction model.
Methods
A retrospective analysis was conducted on patients who underwent CBC and CRP testing between January 2016 and March 2024. The study population comprised 5441 cases divided into three cohorts: non-HBV-infected (1333 cases), HBV-infected (1023 cases), and HBV-associated HCC (3085 cases). A value of CRP <10 mg/L was used to exclude cases of acute bacterial infections. Baseline data and blood parameters were compared across the three groups (control group (n = 1049), the HBV-infected group (n = 789), and the HBV-associated HCC group (n = 1367)). HBV-infected group and the HBV-associated HCC group were used as modeling subjects which 70% were classified as training set (n = 1512) and 30% were classified as validation set (n = 644). Lasso regression and logistic regression were employed to identify the most effective predictors of HBV-associated HCC, which were subsequently incorporated into a predictive model by training set.
Results
Significant variations in age, gender, and blood parameter indices were observed between individuals with acute bacterial infections and non-infections in the study population, and also between three groups. The optimal predictors identified for HBV-associated HCC included gender, age, MONO, EO%, MCHC, MPV, and PCT.
Conclusions
The study highlights the significant impact of acute bacterial infections on immune status, erythrocyte system, and platelet system. After excluding acute bacterial infections, factors such as gender, age, MONO, EO%, MCHC, MPV, and PCT are effective predictors for clinical prediction of HCC development in HBV-infected patients.
Keywords
Introduction
Hepatocellular carcinoma (HCC) poses a significant clinical challenge due to its multifactorial etiology, which includes hepatitis B virus (HBV), hepatitis C virus (HCV), and drug-induced liver injury. In China, HBV is the predominant cause of HCC, contributing to a high incidence and mortality rate. 1 While early-stage HCC patients can achieve a 5-year survival rate of up to 70% with hepatectomy, the prognosis for advanced-stage HCC patients remains poor, with a survival rate of only 10%.2–4 The asymptomatic nature of early HCC and the lack of effective diagnostic biomarkers led to most cases being diagnosed at an advanced stage. This underscores the urgent need for reliable early detection methods and predictive models to improve clinical outcomes for HBV-associated HCC.
Routine blood tests are widely used in clinical settings to assess an individual's immune status and overall health. They have been shown to be closely related to the development of various diseases, including HCC.4,5 However, previous studies have often overlooked the impact of acute bacterial infections on immune responses, which can confound the analysis of chronic diseases such as HBV. C-reactive protein (CRP) is a sensitive marker for infections, with levels above 10 mg/L indicating acute bacterial infections. 6 It has been shown7,8 that acute bacterial infections are associated with high CRP concentrations and that low CRP ranges (<10 mg/L) are characteristic of cardiovascular disease and viral infections. By focusing on patients with CRP levels below this threshold, our study aimed to eliminate the confounding effects of acute bacterial infections and more accurately identify predictors of chronic HBV-associated HCC.
Our study utilized least absolute shrinkage and selection operator (LASSO) logistic regression to identify optimal predictors of HBV-associated HCC from clinical data of patients at Jingzhou Hospital Affiliated to Yangtze University. By excluding patients with acute bacterial infections (CRP <10 mg/L), we focused on the effects of HBV infection. The identified predictors: gender, age, monocyte count (MONO), eosinophil percentage (EO%), mean corpuscular hemoglobin (MCH) concentration (MCHC), mean platelet volume (MPV), and plateletcrit (PCT) were used to construct a nomogram prediction model. The model has good discrimination and accuracy in both training set and validation set, making it a potentially valuable tool for clinical prediction of HCC development in HBV-infected patients.
Our findings suggest that acute bacterial infections significantly impacts the immune status, erythrocyte system, and platelet system, thereby justifying the exclusion of individuals with acute bacterial infections from studies examining the effects of HBV. The predictors identified in our study, such as MONO and EO%, align with previous research on immune system markers, while novel predictors like MCHC, MPV and PCT offer new insights into the pathophysiology of HBV-associated HCC. These findings highlight the importance of considering inflammation markers when developing predictive models for HCC. Future studies should concentrate on multicenter collaborations to validate and refine these predictors, potentially incorporating additional biomarkers to enhance the model's predictive power and applicability across diverse populations.
Materials and Methods
Study Subjects
A retrospective analysis was conducted on hospitalized patients at Jingzhou Hospital Affiliated to Yangtze University, blood routine and CRP were detected for the first time in the same period, who underwent routine blood and CRP testing between January 2016 and March 2024. Our study focused on identifying cases of HBV infection (1023 cases) and HBV-associated HCC (3085 cases) by reviewing medical records and clinical diagnoses. Additionally, 1333 cases of patients without a history of HBV infection who had undergone physical examinations were included, all enrolled patients had no history of HBV infection and were over 18 years old by retrieving medical records. The blood parameters assessed in this study included white blood cell count (WBC), neutrophil count (NEU), lymphocyte count (LYM), monocyte count (MONO), eosinophil count (EO), basophil count (BA), percentage of neutrophils (NEU%), percentage of lymphocytes (LYM%), percentage of monocytes (MONO%), percentage of eosinophilic (EO%), percentage of basophilic (BA%), red blood cell count (RBC), hemoglobin (HGB), hematocrit (HCT), mean corpuscular volume (MCV), MCH, mean corpuscular hemoglobin concentration (MCHC), red cell distribution width (RDW), platelet count (PLT), MPV, PCT, platelet distribution width (PDW) and CRP. The diagnostic criteria for HBV infection and HBV-associated HCC were based on guidelines outlined in “The Management of Hepatocellular Carcinoma". 5 Cases involving hepatitis A, C, autoimmune liver disease, drug-induced liver injury, and other forms of hepatitis were excluded from the study. Ethical approval for the study protocol was obtained from the Ethics Committee of Jingzhou Hospital Affiliated to Yangtze University, under approval number 2024-095-01.
Predictive Model Construction
In this research, the population of 5441 individuals was stratified into three cohorts: non-HBV-infected (1333 cases), HBV-infected (1023 cases), and HBV-associated HCC (3085 cases). A value of CRP <10 mg/L was used to exclude cases of acute bacterial infections. Baseline data and blood parameters were compared across the three groups (control group (n = 1049), the HBV-infected group (n = 789), and the HBV-associated HCC group (n = 1367)). A predictive model was developed using clinical data from the HBV-infected group (789 cases) and HBV-associated HCC group (1367 cases). It were used as modeling subjects which 70% were classified as training set (n = 1512) and 30% were classified as validation set (n = 644). Lasso regression and logistic regression analyses were employed on the train data set to identify the most predictive factors of HCC in patients infected with HBV. The model's assessment of discrimination and accuracy was conducted using ROC curves and calibration curves for both the training and the validation sets. The final predictive model was represented in the form of a Nomogram. The method of inclusion and exclusion of participants in this study is shown in Figure 1.
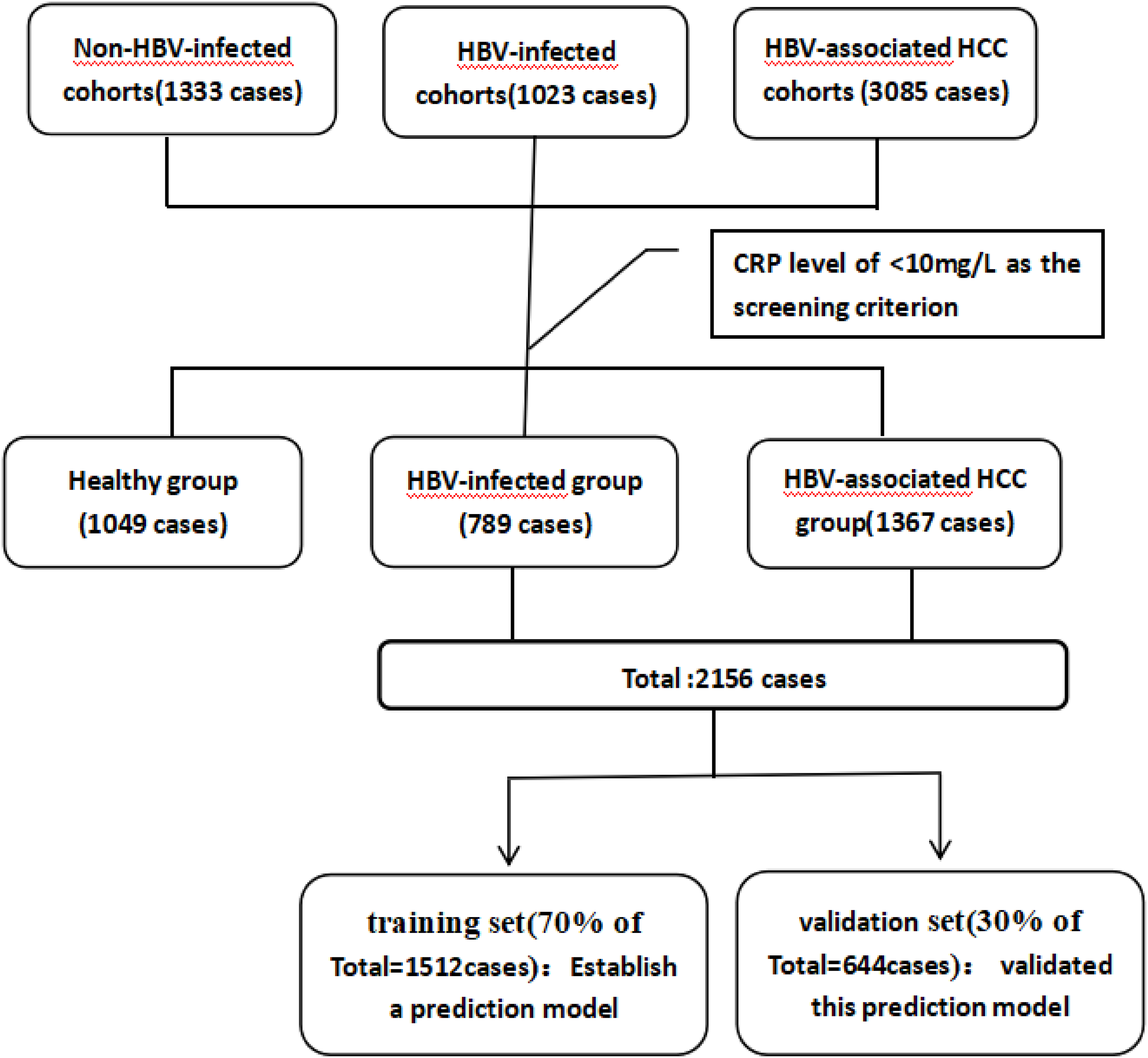
Flowchart of the participant selection procedure.
Statistical Analysis
The data analysis was conducted using SPSS 26.0, with non-normally distributed measures presented as median (interquartile range) [M(P25,P75)]. The comparison of two groups was performed using the Mann-Whitney U test, while the comparison of three groups was conducted using the Kruskal-Wallis H test. Enumeration data was assessed by Chi-square (χ2) test. The best predictors were identified through logistic regression and Lasso regression. The model's discrimination and accuracy were assessed using ROC curve and calibration curve. Predictive models were developed and validated using R 4.2.4.
Results
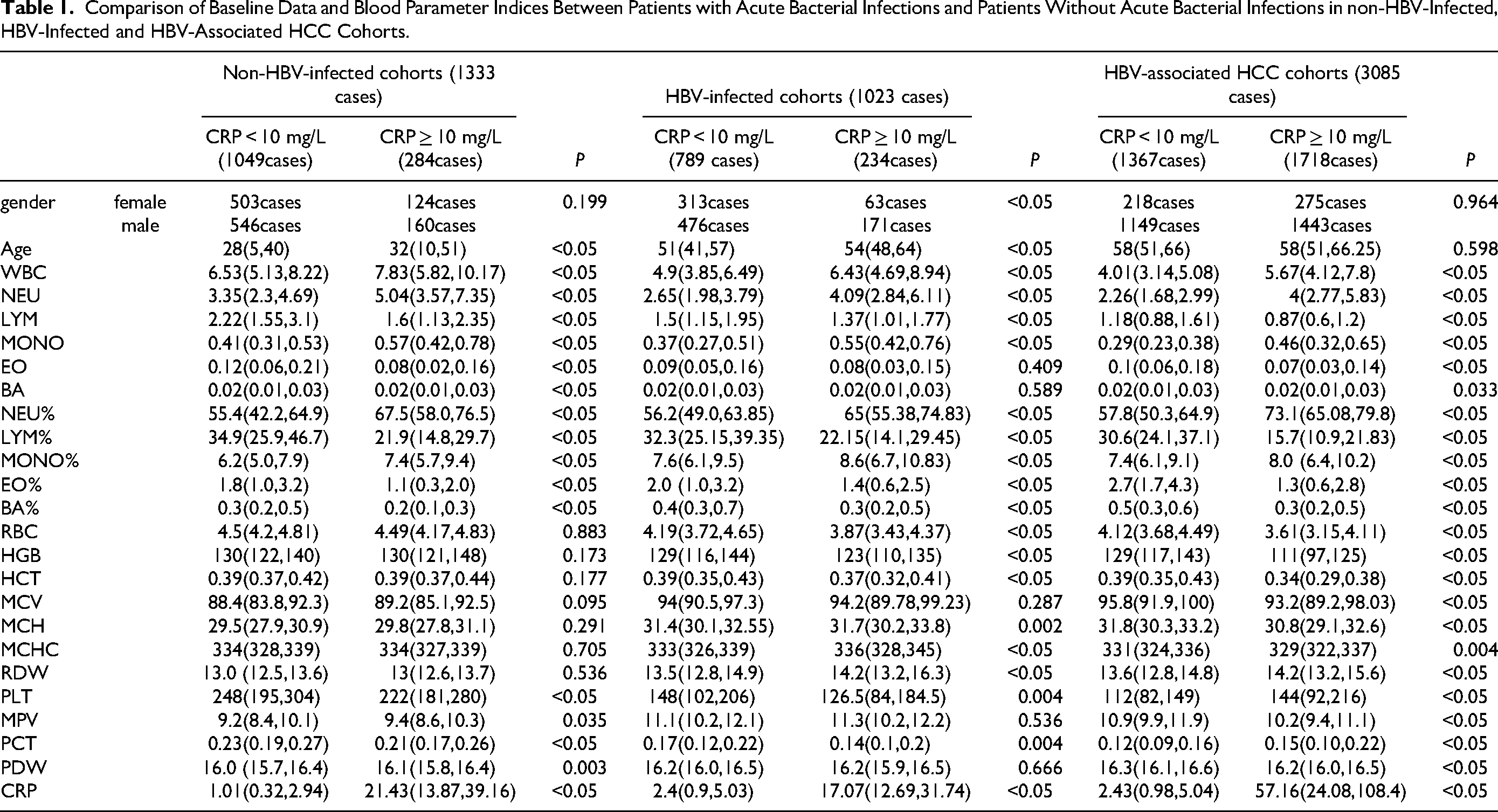
Comparison of Baseline Data and Blood Parameter Indexes Between Acute Bacterial Infections and Non-Acute Bacterial Infections in Non-HBV-Infected, HBV-Infected and HBV-Associated HCC Cohorts
Utilizing a level of CRP 10 mg/L, acute and non-acute bacterial infections were discerned within these cohorts. The study aims to analyze variances in baseline characteristics and hematological parameters between acute and non-acute bacterial infections within these three cohorts. For the analysis, three cohorts with CRP levels less than 10 mg/L were chosen for inclusion in our study. A total of 1049 individuals were categorized as the control group, 799 as the HBV-infected group, and 1367 as the HBV-associated HCC group. Baseline characteristics and blood parameters were compared across these three groups.
The findings indicate that age and gender do not have a significant impact on distinguishing between acute and chronic inflammation. acute bacterial infections notably affects the immune system in these cohorts, leading to elevated levels of NEU and MONO, and decreased levels of LYM and EO. Additionally, acute bacterial infections has implications for the erythrocyte and platelet systems (Table 1).
Comparison of Baseline Data and Blood Parameter Indices Between Patients with Acute Bacterial Infections and Patients Without Acute Bacterial Infections in non-HBV-Infected, HBV-Infected and HBV-Associated HCC Cohorts.
Comparison of Baseline Data and Blood Parameter Indicators among Healthy, HBV-Infected and HBV-Associated HCC Groups in Non-Acute Population (CRP < 10 mg/L)
The analysis of the three groups revealed that individuals with HBV-infected and HBV-associated HCC were predominantly older and predominantly males. Furthermore, HBV-infected and HBV-associated HCC groups are associate with HBV infection. HBV infection was found to impact the immune, erythrocyte, and platelet systems, as evidenced in Table 2.
Comparison of Baseline Data and Blood Parameter Indicators Between Healthy, HBV-Infected, and HBV-Associated HCC Groups.

Lasso Regression and Logistic Regression Were Adopted to Explore the Optimal Predictors of Transformation to HBV-Associated HCC in HBV-Infected Patients
It has been divided 70% to training set and 30% to validation set with clinical data (2156 cases),which from the HBV-infected group (789 cases) and HBV-associated HCC group (1367 cases). Subsequently, a predictive model was developed using training set and validated using validation set. Lasso regression was used to select independent risk factors for the predictive model (Figure 2). Factors including gender, age, WBC, MONO, EO%, MCHC, MPV, and PCT were selected based on the LASSO regression. These factors were incorporated in the Logistic regression predictive model. Finally gender, age, MONO, EO%, MCHC, MPV, and PCT were screened as valid predictors. This model assessed for its discriminative and accuracy by training set and validation set. The model has good discrimination and accuracy in both training set (ROC curves: AUC = 0.834, P < 0.05; calibration curves: C = 0.834, P = 0.708) (Figure 3) and validation set (ROC curves: AUC = 0.821, P < 0.05; calibration curves: C = 0.821, P = 0.940) (Figure 4).
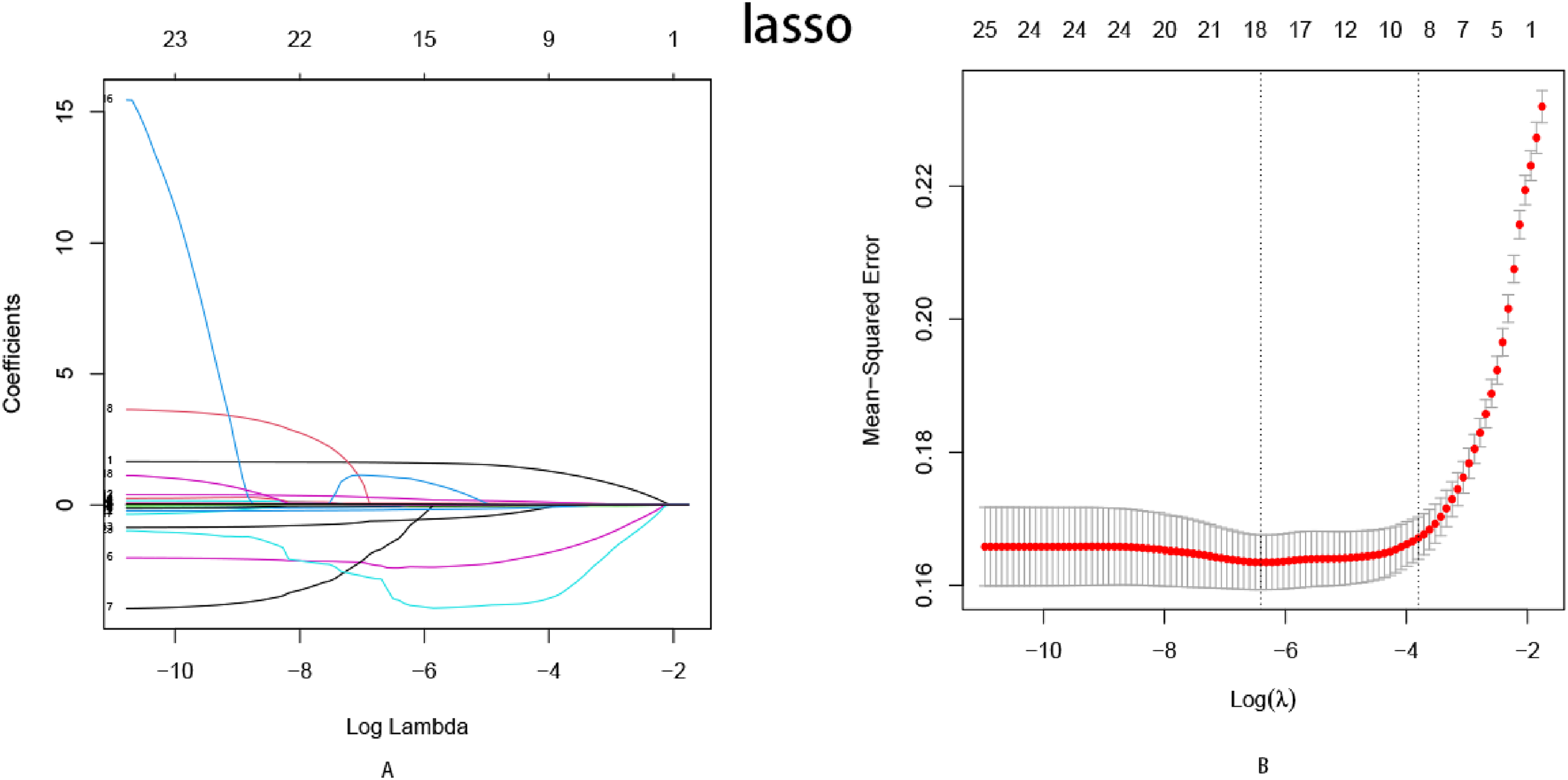
Lasso regression screening for optimal predictors.
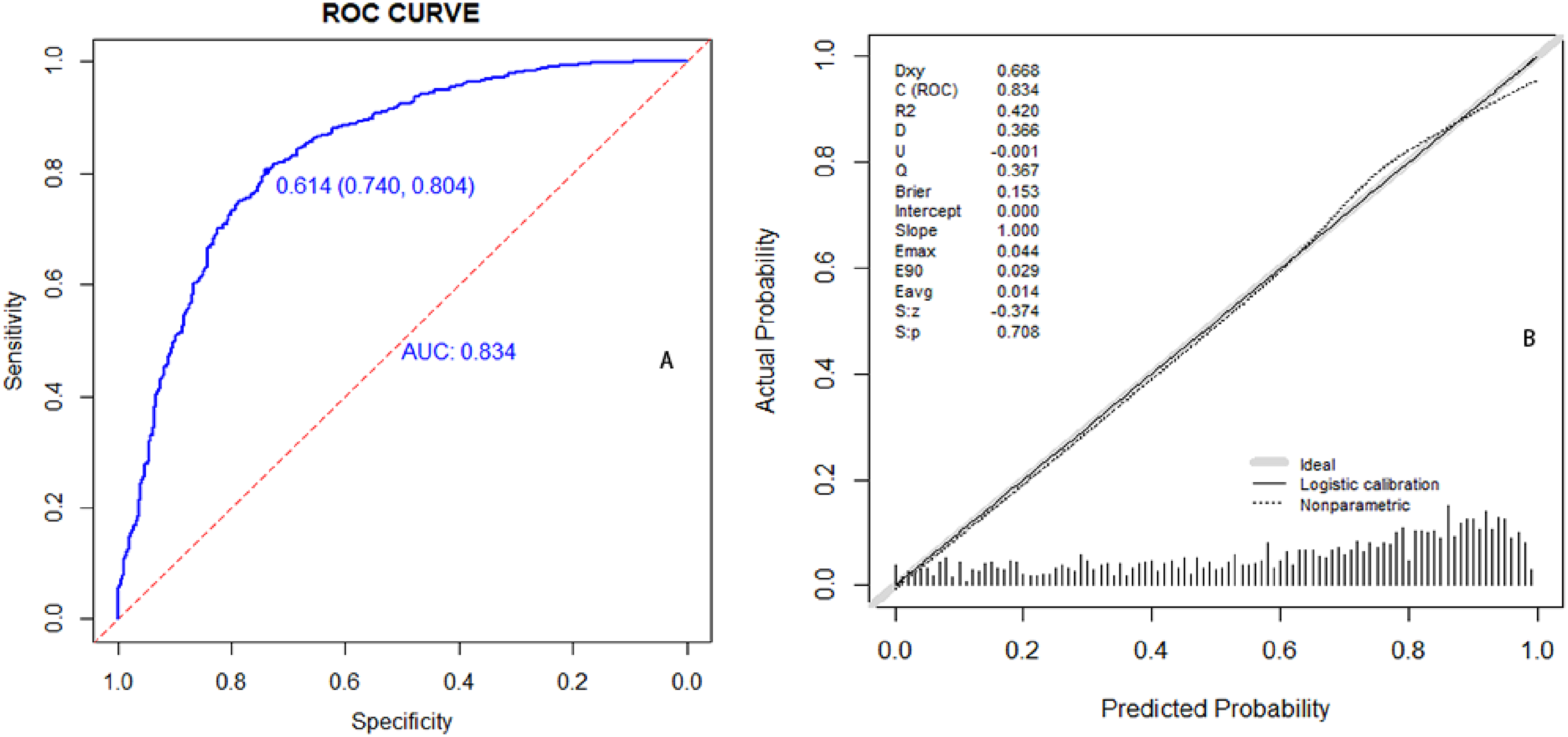
Model validation (training set): discrimination test (A, ROC curves), accuracy (B, calibration curves).
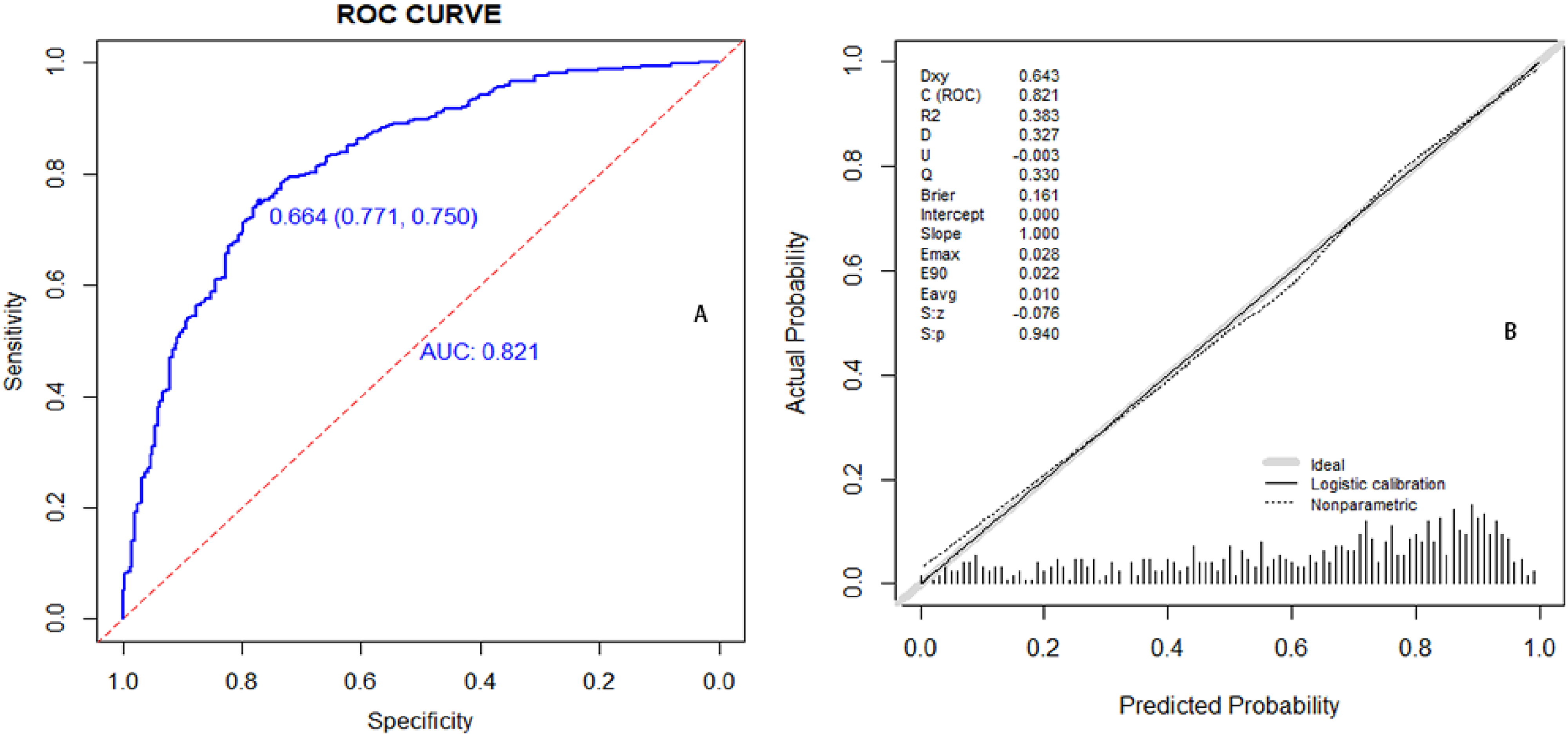
Model validation (validation set): discrimination test (A, ROC curves), accuracy (B, calibration curves).
Development and Application of a Nomogram for Predicting HCC Risk in HBV-Infected Patients
We also developed a normogram (Figure 5) to predict probability of HCC and to facilitate the practical application of the model. Analysis of the model revealed that male individuals infected with HBV exhibited an elevated susceptibility to HCC compared to females. Moreover, advancing age and elevated EO% levels were identified as risk factors for HCC, while decreased MONO, MCHC, MPV, and PCT levels were associated with an increased risk of developing HCC.
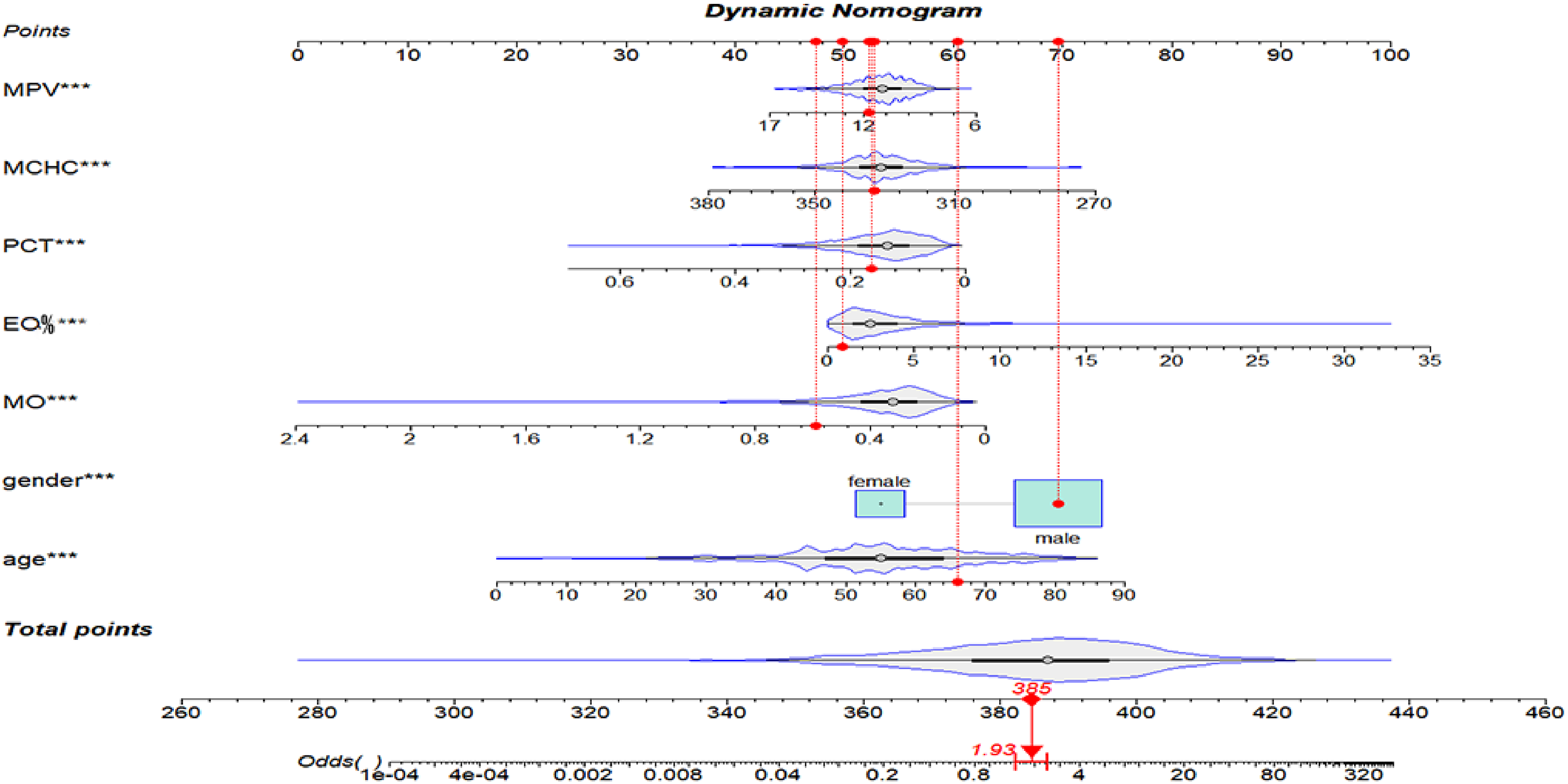
The nomogram for the estimation of the risk of HCC.
Discussion
The clinical outcome of patients infected with HBV is influenced by multiple factors. On the one hand, after the virus invades the hepatocytes, it will form covalently closed circular DNA (cccDNA), which can integrate with the hepatocytes and continuously synthesize serum HBV DNA, HBV RNA, HBeAg, and HBsAg,9,10 etc. These HBV antigen derivatives can cause the patient's body to be in a state of chronic inflammation for a long time, resulting in hepatocyte damage and an increased risk of liver fibrosis and HCC. On the other hand, host lymphocytes and other cells in the peripheral blood play an important role in the clearance of HBV.11,12 acute bacterial infections has been identified as having pronounced effect on patients’ immune status. 9 To understand the role of chronic inflammation caused by HBV infection in the development of HCC, it is essential to exclude the effect of acute bacterial infections on patients. CRP13–16 is an acute-phase response protein, a pentamer synthesized by the liver, whose concentration tends to be stable in healthy populations and is not affected by diurnal and seasonal variations. CRP level rises and falls rapidly in response to inflammatory stimuli, such as bacterial invasion or trauma, with a half-life of 19 h. Therefore, CRP was chosen as the defining condition of acute and chronic inflammation in this study, with CRP ≥10 mg/L as acute bacterial infections and CRP < 10 mg/L as non-acute bacterial infections. 9 Our investigation into acute and non-acute bacterial infections in three cohorts revealed that acute bacterial infections significantly impacted the immune status, erythrocyte system, and platelet system, as outlined in Table 1. Consequently, individuals with acute bacterial infections were excluded from our study. Three cohorts with CRP levels less than 10 mg/L were selected for further analysis. A total of 1049 cases were classified as control group, 799 cases as HBV-infected group, and 1367 cases as HBV-associated HCC group, respectively. These three groups were used to investigate the influence of chronic inflammation due to HBV infection in the development of HCC. In this study, the impact of HBV on the immune, erythrocyte, and platelet systems is evident from the data presented in Table 2. These findings suggest that age, gender, immune status, erythrocyte parameters, and platelet function may play a role in the progression of disease in patients with HBV infection.
Our study aimed to identify the optimal predictors of HBV-associated HCC using Lasso regression and logistic regression techniques, focusing on patients without acute bacterial infections (CRP <10 mg/L). In this research, we utilized clinical data from patients in the HBV-infected group and the HBV-associated HCC group to develop a predictive model for the progression of HBV infection to HBV-associated HCC. The significant predictors identified were gender, age, monocyte count (MONO), eosinophil percentage (EO%), MCHC, MPV, and PCT. These predictors were incorporated into a nomogram prediction model, as illustrated in Figure 5. The model demonstrated that male, advanced age, and elevated EO% levels were significant factors associated with HCC, while decreased MONO, MCHC, MPV, and PCT levels were linked to HBV-associated HCC, as evidenced in Figure 5. The model demonstrated good discrimination and accuracy, as evidenced in Figure 3 and 4. This model provides a robust tool for predicting the development of HCC in HBV-infected patients and highlights the importance of excluding acute bacterial infections when studying chronic inflammatory conditions like HBV.
The significance of hematological parameters in predicting HCC has been explored in various studies. The role of gender and age in HCC risk has been well-documented. HCC is more likely to occur in older individuals,17,18 as well as men and postmenopausal women.19–24 Consistently, our study confirms these trends by identifying male gender and older age as significant predictors of HBV-associated HCC. The inclusion of these demographic factors in our prediction model enhances its clinical utility by accounting for well-established risk factors.
The impact of chronic HBV infection on the immune system and its role in HCC development has been extensively studied. Chronic HBV infection leads to continuous immune system activation and inflammation, which can cause liver damage and increase the risk of HCC. 1 The findings from our study align with previous research that highlights the role of immune cells and inflammatory markers in HCC development. With HCC patients, decreased lymphocytes and increased MONO are indicative of a worse overall survival. 25 Our study corroborates this by identifying MONO as a significant predictor of HBV-associated HCC. Our findings suggest that reduced MONO could serve as an indicator for the emergence of HBV-associated HCC. This contrasts with some studies that have associated elevated MONO with poor outcomes. The possible reasons may be that other studies have not excluded the effect of acute bacterial infections on the immune system. That effect is expressed as an increase in the number of NEU and MONO and a decrease in the number of LYM in the acutely inflamed population.
Additionally, our study offers a prognostic indicator for the emergence of HBV-associated HCC in individuals with HBV infection, distinguishing it from previous researches. High EO 26 or low NER 27 is associated with better prognosis after HCC surgery, but there are no studies linking EO% to HCC. The identification of EO% as a predictor in our study suggests that eosinophils may play a complicated role in HBV-associated HCC. In our study, elevated EO% is associated with an increased risk of HCC. An elevation in EO% may be attributed to the inhibition of various other immune cells, leading to a rise in the EO%. MCHC has been linked to prognosis in HCC patients, with lower MCHC levels associated with poorer outcomes. 28 Our research identified an association between lower MCHC levels and an increased risk of HCC, which supports these findings, demonstrating that MCHC is a valuable predictor of HBV-associated HCC. Platelet-related parameters such as MPV and PCT have also been studied in the context of HCC. MPV is a marker of platelet activation, and studies have shown that low MPV levels are associated with shorter survival in HCC patients with comorbidities like diabetes.29–31 To our knowledge, no studies have correlated PCT with HCC. In our study, lower MPV was associated with increased risk of HCC, and also found that PCT can be used for the prediction of HBV-associated HCC. Our identification of MPV and PCT as predictors highlights the importance of platelet function in the pathogenesis of HBV-associated HCC, possibly due to the fact that PCT is associated with platelet activation and therefore can influence the course of disease in HBV-infected patients, which needs to be confirmed by further studies in the future.
Despite the valuable insights provided by our study into the predictors of HBV-associated HCC, there are several limitations that need to be acknowledged. Firstly, our study relies on a single-center dataset, which may limit the generalizability of our findings. Future studies should involve multicenter collaborations to validate and refine our predictive model across diverse populations. Secondly, the retrospective nature of our study may introduce biases related to data collection and patient selection. Prospective studies are needed to confirm our findings and explore additional predictors. Thirdly, our study focused on routine blood parameters and did not include other potential biomarkers, such as genetic or molecular markers, which could enhance the predictive power of our model. Lastly, while our model demonstrated good discrimination and accuracy, further validation in different clinical settings is necessary to ensure its applicability in routine practice.
Conclusions
CRP is widely used as a marker for inflammation, and its levels can reflect inflammatory states.6–8 By excluding patients with CRP levels ≥10 mg/L, we aimed to minimize the confounding effects of acute bacterial infections. Our study identifies significant predictors (gender, age, MO, EO%, MCHC, MPV, and PCT) of HBV-associated HCC, and provides a robust predictive model that can aid in early detection and intervention. Future research should aim to validate and expand upon these predictors, incorporating a wider range of biomarkers and involving diverse populations to improve the early detection and management of HBV-associated HCC.
Footnotes
Acknowledgements
This study was supported by the Department of Laboratory Medicine, Jingzhou Central Hospital, Medical Immunological Laboratory, and all colleagues.
Author Contributions
Yafeng Tan and Wei Xia were involved in the paper drafting, responsible for data collection and data analysis; Fenglan Sun, Bing Mei, Yaoling Ouyang, Linyun Li, Zhenxia Chen, Jufang Tan, Zhaxi pubu, Bu Sang and Song Wu contributed to the critical revision of the manuscript for important linguistic content; Tao Jiang was responsible for the research design and critical revision of the manuscript. All authors approved the final version of the manuscript for publication.
Data Availability
All the data released to this work are available at the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by Yangtze University Science and Technology Aid to Tibet Medical Talent Training Program Project (2023YZ04).
