Abstract
Background
Chronic hepatitis B virus (HBV) infection remains highly prevalent worldwide, particularly in China. However, its influence on the risk of venous thromboembolism (VTE) following orthopedic trauma has not been well characterized. This study aimed to investigate the incidence and risk factors of deep vein thrombosis (DVT) in HBV-infected patients after lower extremity fractures.
Methods
We retrospectively reviewed the medical records of adult patients with chronic HBV infection who were hospitalized for lower extremity fractures at a tertiary trauma center between December 2013 and January 2024. Demographic, clinical, and laboratory data were collected. DVT was diagnosed using bilateral lower extremity Doppler ultrasonography. Univariate and multivariate logistic regression analyses were performed to identify independent risk factors. Receiver operating characteristic (ROC) curve analysis was used to determine optimal cutoff values for continuous predictors.
Results
Among 579 eligible patients, the overall incidence of post-fracture DVT was 30.4%. Independent risk factors identified in multivariate analysis included multiple fractures (P < .001, OR = 4.009, 95% CI:2.080 ∼ 7.725), a history of cerebral infarction (P = .009, OR = .753, 95% CI:1.287 ∼ 5.886), a history of cirrhosis (P < .001, OR = 5.211, 95% CI:3.273 ∼ 8.297),and elevated HBsAg levels (P < .001, OR = 1.002, 95% CI: 1.001 ∼1.003). Higher hemoglobin levels (P < .001, OR = 0.979, 95% CI:0.970 ∼ 0.988) were independently associated with reduced DVT risk. In contrast, commonly used biomarkers such as D-dimer, fibrin degradation products (FDP), and liver function scores, including the Child–Pugh score and the Model for End-Stage Liver Disease (MELD) score, were not significant predictors in multivariate analysis.
Conclusions
DVT is a common complication after lower extremity fractures in patients with chronic HBV infection. Multiple fractures, elevated HBsAg levels, prior stroke, and cirrhosis were independently associated with a higher risk of DVT, whereas higher hemoglobin levels were associated with a lower risk. These findings suggest that individualized thromboprophylaxis strategies may be considered in HBV-infected trauma patients.
Keywords
Introduction
Deep vein thrombosis (DVT) is a frequent and serious complication following lower extremity fractures. 1 In patients with major lower limb fractures, DVT incidence has been reported to be as high as 24%-28%.2,3 The chief danger of post-fracture DVT is its potential progression to pulmonary embolism (PE), as a dislodged thrombus can travel to the lungs and cause PE leading to sudden death. Indeed, up to 300,000 people die from PE each year in the United States. 4 Even when not immediately fatal, DVT and subsequent venous thromboembolism (VTE) events impose significant long-term morbidity and healthcare burdens. 5 Short-term prognosis is also poor: one study reported that the one-month mortality after lower-extremity DVT was approximately 7.1%. 6 Early identification and prevention of DVT in fracture patients are therefore critical to improving outcomes.
Risk factors for DVT following orthopedic trauma have been the subject of numerous studies. 7 Advanced age, prolonged immobility, obesity, heart failure, and previous venous thromboembolism are known risk factors. 8 However, these known factors do not fully explain all DVT cases, and analyses suggest that additional predictors have been overlooked. Notably, although chronic hepatitis B virus (HBV) infection is highly prevalent worldwide—particularly in East Asia—the risk factors for deep vein thrombosis (DVT) following orthopedic trauma in patients with chronic HBV infection have been scarcely investigated. This represents a significant gap in the current body of literature.
There are several plausible ways that HBV infection could affect thrombotic risk and coagulation. Long-term HBV can cause endothelial dysfunction and systemic inflammation, which can upset the coagulation factor balance and possibly result in hypercoagulability. 9 The balance between procoagulant and anticoagulant pathways is frequently disrupted in patients with chronic liver disease linked to HBV, which is believed to put patients at risk for thromboembolic events rather than shield them from them. 10 Additionally, long-term antiviral treatment is frequently necessary for patients with chronic HBV, especially when using nucleos(t)ide analogues like entecavir (ETV) or tenofovir disoproxil fumarate (TDF). Prolonged use of these agents has been linked to bone mineral density loss and an increased risk of osteoporosis. Over time, this complication can lead to fragility fractures in weight-bearing bones, and any resultant lower extremity fracture necessitates immobilization—thereby further elevating the likelihood of DVT. 11 Taken together, these pathophysiological and treatment-related factors suggest that HBV-positive individuals might have a unique risk profile for DVT after trauma.
Clarifying HBV infection's role in post-fracture DVT is clinically significant due to its high prevalence. About 296 million people worldwide suffer from chronic HBV infection, 12 and the virus is still very common in many places (for instance, there are an estimated 75 million carriers in China). 13 As a result, HBV may be a comorbidity in many orthopedic trauma patients. Finding out if HBV increases the risk of DVT following a fracture will close a significant knowledge gap and guide targeted thromboprophylaxis measures. In order to determine risk factors for lower-limb DVT after fractures in patients with HBV, this retrospective study was created. These results may help direct tailored DVT prevention and enhance patient outcomes.
Patients and Methods
Ethics Statement
In this study, we reviewed the electronic medical records of all adult patients with chronic HBV infection who were treated for lower extremity fractures at our hospital between December 2013 and January 2024. The study was approved by the Institutional Review Board of our hospital (Approval No. S2023-002-1) and conducted in accordance with the ethical principles of the 1964 Declaration of Helsinki. The trial was registered with ClinicalTrials.gov (Identifier: NCT04529330) on August 26, 2020. Given the retrospective nature of the study, the requirement for informed consent was waived.
Patients
This retrospective study was conducted at the Third Hospital of Hebei Medical University, a tertiary care center equipped with a Level I trauma facility. A total of 579 electronic medical records were reviewed based on predefined inclusion and exclusion criteria. The inclusion criteria were: (1) patients with chronic HBV infection who sustained lower extremity fractures; and (2) age ≥18 years. Exclusion criteria included: (1) incomplete clinical data; (2) pathological fractures; (3) prior history of DVT; (4) old fractures (>21 days from injury to admission); and (5) co-infection with other types of viral hepatitis; (6) history of long-term anticoagulant or antiplatelet therapy (Figure 1).

Exclusion criteria and the eligible cases included in this study.
A total of 579 patients (412 men and 167 women) were included in this study. Upon admission, all patients underwent bilateral lower extremity and abdominal color Doppler ultrasonography, performed by at least two experienced sonographers. Based on these Doppler findings, patients were classified as DVT-positive or DVT-negative (non-DVT). Thrombi identified by ultrasound were categorized anatomically as proximal (involving the iliac, femoral, or popliteal veins), distal (tibial or fibular veins), or mixed (involving both proximal and distal segments). At the same time, cirrhosis was diagnosed based on characteristic ultrasonographic features, including coarse or nodular liver echotexture, irregular liver surface, blunt liver edge, and signs of portal hypertension such as splenomegaly and dilated portal vein. Ultrasonography was performed using a blinded method. Sonographers only knew the patient's fracture site and were not informed of clinical information such as HBV infection status, cirrhosis/cerebral infarction history to reduce diagnostic bias Cirrhosis was diagnosed noninvasively, and no histopathological confirmation was required. For prophylaxis, subcutaneous low-molecular-weight heparin (4250 IU) was administered once daily from admission until 12 h before surgery and then every 12 h postoperatively. In addition, bilateral intermittent pneumatic compression devices were applied throughout hospitalization for mechanical thromboprophylaxis; compression was avoided in any limb at risk of acute compartment syndrome. Notably, no patient developed symptomatic pulmonary embolism, and accordingly no CT pulmonary angiography was required.
Demographic, clinical, and laboratory data were extracted retrospectively from the medical records of HBV-infected patients with lower extremity fractures who underwent surgical treatment. Demographic variables included patient age, sex, body mass index (BMI), fracture site, the presence of multiple fractures, duration of hepatitis B, timing of surgery, postoperative bed rest duration, history of prior antiviral therapy (eg, entecavir [ETV] or tenofovir disoproxil fumarate [TDF]), and duration of antiviral treatment, as well as American Society of Anesthesiologists (ASA) physical status (dichotomized as grades I-II vs III-IV). In patients with multiple fracture sites, the anatomical location deemed most clinically relevant—typically the one associated with higher thrombotic risk or requiring primary surgical intervention—was selected as the representative fracture site for classification purposes. Based on nucleos(t)ide analogue (NA) exposure, patients were then classified into three groups: those treated with entecavir (ETV), those treated with tenofovir disoproxil fumarate (TDF), and those who were nucleos(t)ide analogue–naive (NA-naive). Patients who had received both agents were categorized according to the most recent or predominant regimen administered before surgery. We also recorded smoking status, alcohol use, mechanism of injury (traffic accident, fall, crush injury, sprain, or other), and duration of chronic HBV infection. Comorbid conditions noted in the history included hypertension, diabetes mellitus, arrhythmia, coronary heart (artery) disease, cerebral infarction (stroke), cirrhosis, and hepatocellular carcinoma. All laboratory tests performed on the day of admission were collected, encompassing hematologic indices (basophils [BAS], eosinophils [EOS], hematocrit [HCT], hemoglobin [HGB], immature granulocytes [IMM], lymphocytes [LYM], mean corpuscular hemoglobin concentration [MCHC], monocytes [MON], mean platelet volume [MPV], neutrophils [NEU], platelets [PLT], red blood cells [RBC], white blood cells [WBC]), coagulation parameters (activated partial thromboplastin time [APTT], antithrombin III [AT III], D-dimer, fibrin degradation products [FDP], fibrinogen [FIB], thrombin time [TT]), biochemical panels (albumin [ALB], alkaline phosphatase [ALP], aspartate aminotransferase [AST], alanine aminotransferase [ALT], calcium [Ca], potassium [K], sodium [Na], magnesium [Mg], phosphorus [P], chloride [Cl], globulin [GLOB], cholinesterase [CHE], creatine kinase [CK], creatinine [CREA], direct bilirubin [DBIL], glucose [GLU], C-reactive protein [CRP], lactate dehydrogenase [LDH], triglycerides [TG], total cholesterol [TC], total carbon dioxide [TCO₂], urea [UREA], and uric acid [UA]), and Hepatitis B virus serological markers(Hepatitis B surface antigen [HBsAg], Antibody to HBsAg [Anti-HBs], Hepatitis B e antigen [HBeAg], Antibody to HBeAg [Anti-HBe], and Antibody to Hepatitis B core antigen [Anti-HBc]). From these admission laboratory values, the Aspartate Aminotransferase-to-Platelet Ratio Index (APRI), Child–Pugh classification, Model for End-Stage Liver Disease (MELD) scores, Neutrophil-to-Lymphocyte Ratio (NLR) and Platelet-to-Lymphocyte Ratio (PLR) were computed for each patient. Variables achieving significance in univariate analyses were entered into multivariate regression models for further analysis.
Statistics
All statistical analyses were performed using SPSS software (version 25.0; SPSS Inc., New York, USA), with a two-tailed P value <.05 considered statistically significant. The Shapiro–Wilk test was used to assess the normality of continuous variables. Normally distributed data were presented as mean ± standard deviation (SD) and compared using the independent samples t-test. For non-normally distributed data, the Mann–Whitney U test was applied. Categorical variables were expressed as frequencies and percentages, and intergroup comparisons were conducted using the Chi-square test or Fisher's exact test, as appropriate. In addition, to identify the most reliable predictors of DVT, binary logistic regression analysis was performed to determine independent risk factors for DVT in patients with chronic hepatitis B virus infection following lower extremity fractures.
Receiver operating characteristic (ROC) curve analysis was employed to determine the optimal cutoff values for continuous variables (eg, BMI), based on the maximum Youden index (sensitivity + specificity − 1). Variables were subsequently dichotomized into low- and high-risk groups according to these cutoffs. The area under the ROC curve (AUC) was used to evaluate the discriminatory power of each variable, with values ranging from 0.0 to 1.0, where a higher AUC indicates better predictive performance. The 95% confidence intervals (CI) for the AUC values were calculated using the DeLong method.
Results
A total of 579 patients were included in this study, comprising 412 males and 167 females. The incidence of DVT following lower extremity fractures was 30.4%, with 176 patients diagnosed with DVT and 403 without. Among those who developed DVT, 14 patients (8.0%) had proximal DVT, 152 patients (86.4%) had distal DVT, and 10 patients (5.6%) had mixed-type DVT.
As shown in Table 1, significant differences were observed between the DVT and non-DVT groups in terms of duration of chronic hepatitis B infection (P = .040), timing of surgery (P = .049), postoperative bed rest duration (P = .049) and presence of multiple fractures (P < .001). Specifically, the DVT group exhibited a longer hepatitis B infection history, extended preoperative waiting and postoperative bed rest periods, and a higher incidence of multiple fractures. In contrast, no significant differences were found between the two groups regarding age, BMI, time from injury to hospital admission, sex, ASA classification, smoking status, alcohol consumption, or injury mechanism (all P > .05).
Demographic Data of Patients with and Without DVT.

Abbreviations: BMI, body mass index; NA, nucleos(t)ide analogue; ETV, entecavir; TDF, tenofovir disoproxil fumarate; ASA, American Society of Anesthesiologists.
Values are presented as n (%) for categorical variables and as mean ± SD or median (interquartile range [IQR]) for continuous variables, as appropriate. *P < .05 indicates statistical significance.
Table 2 compares the comorbidities between the two groups. Patients with a history of cerebral infarction (P < .001) was found to have a significantly increased risk of developing DVT. Notably, the presence of liver cirrhosis was associated with a significantly increased risk of DVT (P < .001). No significant differences were observed between the DVT and non-DVT groups in terms of other comorbidities, including hypertension and coronary artery disease, (all P > .05). Table 3 summarizes the laboratory parameters of the two groups. Patients in the DVT group had significantly higher levels of CRP (P = .033) and HBsAg (P < .001) compared to the non-DVT group. Conversely, they exhibited significantly lower levels of the HGB (P = .017), ALP (P = .012), Ca (P < .001) and DBIL (P = .001). No statistically significant differences were noted in the remaining laboratory indicators (all P > .05).
Comorbidity Data of Patients with and Without DVT.
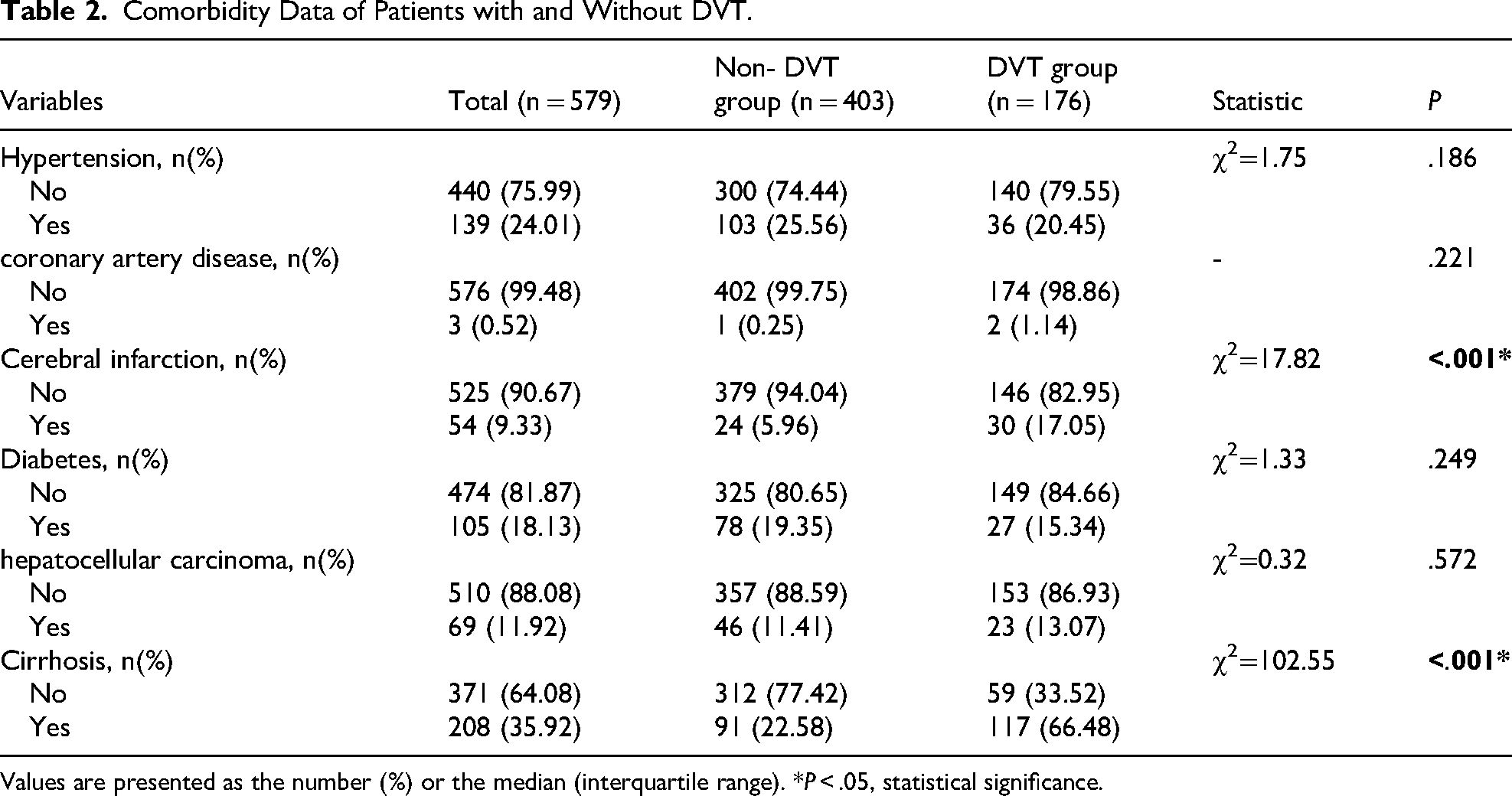
Values are presented as the number (%) or the median (interquartile range). *P < .05, statistical significance.
Laboratory Results of Patients with and Without DVT.

Abbreviations: APRI, Aspartate Aminotransferase-to-Platelet Ratio Index; MELD, Model for End-Stage Liver Disease scores; NLR, neutrophil-to-lymphocyte ratio; PLR, Platelet-to-Lymphocyte Ratio; BAS, basophil; EOS, eosinophil; HCT, hematocrit; HGB, hemoglobin; LYM, lymphocyte; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; MON, monocyte; NEU, neutrophil; PLT, platelet; RBC, red blood cell; WBC, white blood cell; AST, aspartate aminotransferase; ALT, alanine aminotransferase; AST/ALT, aspartate aminotransferase / alanine aminotransferase; ALB, albumin; ALP, alkaline phosphatase; CHE, cholinesterase; CK, creatine kinase; CKMB, creatine kinase-MB; CREA, creatinine; DBIL, direct bilirubin; GAP, anion gap; GGT, gamma-glutamyl transferase; GLOB, globulin; GLU, glucose; CRP, C-reactive protein; HDL-C, high-density lipoprotein cholesterol; IBIL, indirect bilirubin; LDH, lactate dehydrogenase; LDL-C, low-density lipoprotein cholesterol; OSM, osmolality; TBA, total bile acid; TBIL, total bilirubin; TC, total cholesterol; TCO2, total carbon dioxide; TG, triglyceride; TP, total protein; UA, uric acid; UREA, urea; VLDL, very low-density lipoprotein; APTT, activated partial thromboplastin time; APTT-R, APTT ratio; AT III, antithrombin III; FDP, Fibrin degradation products, FIB, fibrinogen; INR, international normalized ratio; PT, prothrombin time; PTA, prothrombin activity; TT, thrombin time; TT-R, thrombin time ratio; HBsAg, Hepatitis B surface antigen; Anti-HBs, Antibody to hepatitis B surface antigen; HBeAg, Hepatitis B e antigen; Anti-HBe, Antibody to hepatitis B e antigen; Anti-HBc, Antibody to hepatitis B core antigen.
Values are presented as the number (%) or the median (interquartile range). *P < .05, statistical significance.
According to the logistic regression analysis, the presence of multiple fractures (P < .001, OR = 4.009, 95% CI:2.080 ∼ 7.725), a history of cerebral infarction (P = .009, OR = 2.753, 95% CI:1.287 ∼ 5.886), a history of cirrhosis (P < .001, OR = 5.211, 95% CI:3.273 ∼ 8.297), and elevated HBsAg levels (P < .001, OR = 1.002, 95% CI: 1.001 ∼1.003) were independently associated with an increased risk of DVT in patients with lower extremity fractures and chronic HBV infection. Conversely, higher hemoglobin levels (P = <.001, OR = 0.979, 95% CI:0.970 ∼ 0.988) were identified as protective factors against DVT in this patient population. (Table 4)
ROC curve analysis demonstrated that HBsAg level was an independent predictor of DVT in patients with lower extremity fractures and chronic hepatitis B infection (P < .001, AUC = 0.719, 95% CI: 0.662-0.776). The optimal cutoff value for HBsAg was identified as 826.35 IU/mL, yielding a sensitivity of 0.625, a specificity of 0.782, and a Youden index of 0.407 (Figure 2).
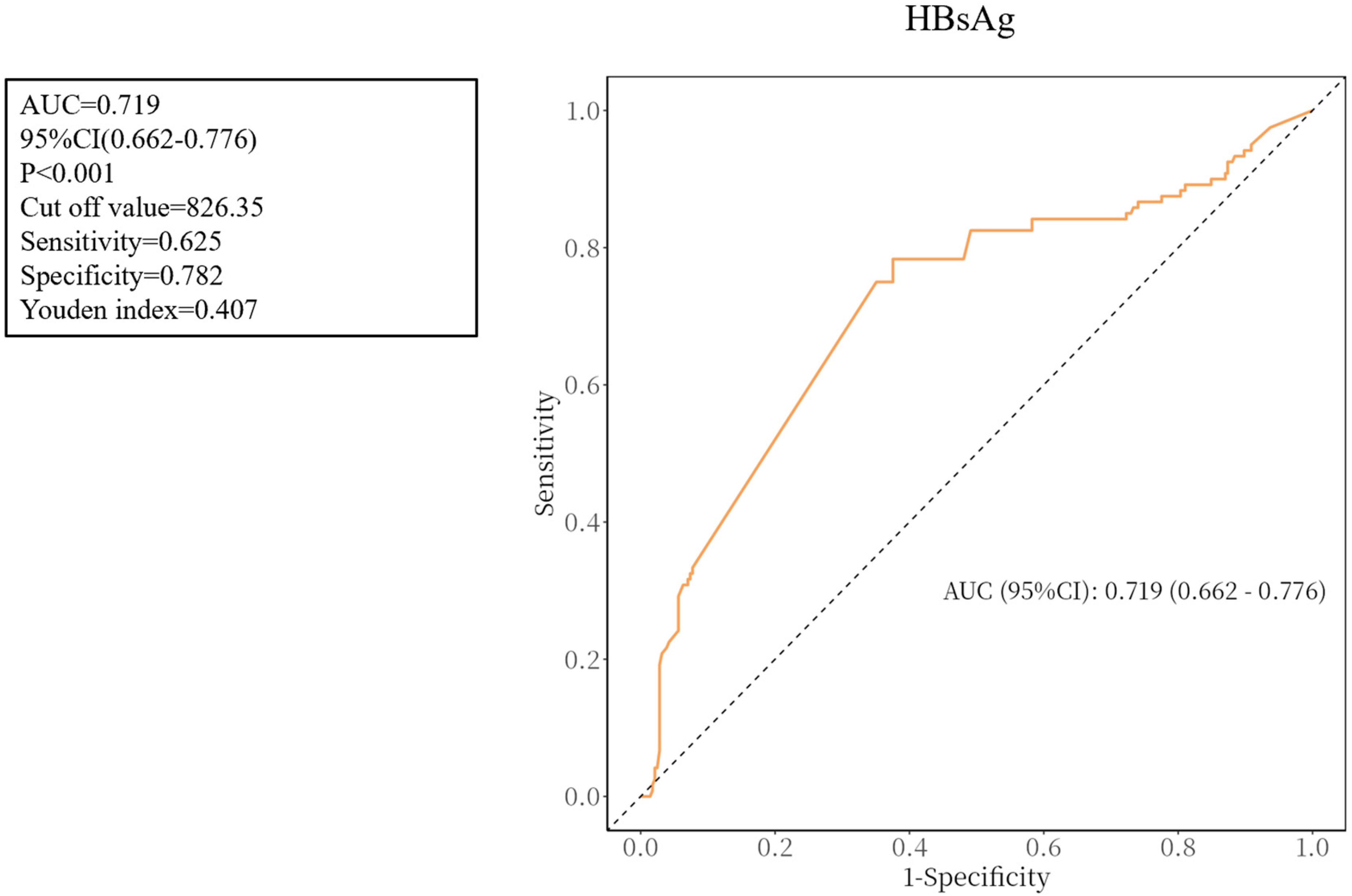
ROC curves for HBsAg.
Discussion
A significant global public health burden is imposed by chronic HBV infection, one of the most common and significant chronic viral diseases in the world. More attention should be paid to the burden of underlying HBV infection among patients receiving orthopedic trauma care. DVT is still one of the most frequent complications among hospitalized patients, especially those who have fractures to their lower extremities, where it can cause serious disability or even death. The high prevalence of HBV and its systemic pathophysiological impact have been highlighted by extensive epidemiological studies and multicenter data.14,15 In trauma patients, immobilization and tissue damage already meet crucial Virchow's triad requirements. The prothrombotic state caused by a chronic HBV infection may further complete this triad through mechanisms such as endothelial dysfunction, inflammation, and coagulation imbalance, greatly raising the risk of DVT. However, previous research has focused primarily on DVT following lower limb fractures, with little attention paid to patients with chronic HBV infection. Determining the risk factors for the development of DVT in patients with lower extremity fractures and a chronic HBV infection was the aim of this study.
To the best of our knowledge, this is the first cohort study specifically investigating the risk factors for DVT in patients with chronic HBV infection following lower extremity fractures. In this study, the incidence of DVT was 30.4%, suggesting a higher occurrence of DVT in patients with HBV infection compared to the general population.2,3 Multivariate analysis identified multiple fractures, a history of cerebral infarction, a history of cirrhosis and elevated HBsAg levels were independently associated with a higher risk of DVT in this population. ROC curve analysis further revealed that the optimal HBsAg threshold for predicting DVT was 826.35 IU/mL.
In patients with lower extremity fractures, multiple fractures have consistently been associated with an increased risk of DVT. According to epidemiological research, patients who have multiple fracture sites or polytrauma are much more likely to develop DVT than those who only have one fracture.1,16 Consistently, in our cohort, patients with hepatitis B who had multiple fractures resulting in lower extremity fractures were approximately two times more likely to develop DVT (P < .001, OR = 4.009, 95% CI: 2.080 ∼ 7.725), underscoring its role as an independent predictor. Mechanistically, tissue damage from multiple fractures causes systemic release of prothrombotic factors. Damaged tissues, in particular, release large quantities of tissue factor, which triggers the extrinsic coagulation cascade and leads to thrombin generation. 17 Via cytokine-mediated pathways, the concomitant trauma-induced inflammatory response intensifies coagulation even more. 18 Additionally, prolonged surgical procedures that are followed by immobilization or bed rest promote venous stasis in these patients, 19 completing Virchow's triad and raising the risk of thrombosis. Crucially, these prothrombotic mechanisms may worsen in patients with a persistent HBV infection. Persistent inflammation and coagulation abnormalities are hallmarks of chronic HBV infection, which can exacerbate coagulation activation and reduce anticoagulant reserves, further predisposing HBV-infected individuals to DVT. 20 To improve clinical outcomes, HBV-positive patients with multiple fractures require increased vigilance and thromboprophylaxis, as evidenced by the synergy between polytrauma and HBV-related coagulopathy.
In patients with HBV who had lower extremity fractures, our study found that a history of cerebral infarction was an independently associated with a higher risk of DVT (P = .009, OR = 2.753, 95% CI:1.287 ∼ 5.886).This finding supports earlier epidemiological evidence showing that prolonged immobility, venous stasis, and systemic inflammatory responses significantly increase the risk of DVT in stroke survivors, particularly those with limited mobility. 21 According to a comprehensive study, in acute stroke patients who do not receive prophylactic treatment, the incidence of DVT can rise to 75%. 22 The underlying mechanisms may be explained by Virchow's triad, which is commonly seen in post-stroke individuals and includes endothelial dysfunction, reduced venous return due to paralysis or immobility, and a hypercoagulable state.The coagulation profile becomes even more complex in patients who have both chronic liver disease and an HBV infection.A fragile hemostatic balance results from chronic hepatic dysfunction, which modifies the synthesis of procoagulant and anticoagulant proteins. 23 The risk of thrombus formation can also be increased by systemic inflammation following cerebrovascular infarction, which can increase the expression of tissue factor and prothrombotic cytokines. 24 These factors may work in concert to increase the thrombogenic burden when paired with orthopedic trauma, which on its own triggers a coagulation cascade through endothelial damage and inflammatory activation. 25 Our findings underscore the potential clinical importance of early risk stratification and tailored anticoagulant management in HBV-infected patients with a history of stroke who are having surgery for lower extremity fractures because of these overlapping risks.Prompt monitoring and intervention may effectively reduce thromboembolic complications and improve postoperative outcomes in this high-risk population.
New evidence shows that even in chronic HBV patients, cirrhosis is associated with a markedly increased risk of thrombosis. According to recent studies, cirrhosis increases the incidence of DVT and pulmonary embolism. A meta-analysis revealed that the odds of venous thromboembolism (VTE) were about 70% higher than those of non-cirrhotic controls, 26 and a 2022 systematic review discovered that the rate of VTE was twice as high in patients with chronic liver disease. 27 In our cohort, cirrhotic patients had a DVT incidence of 66.48% (OR = 5.211), which is higher than the summary estimates from these meta-analyses. This stronger association may reflect the “double hit” of acute orthopedic trauma plus chronic HBV infection superimposed on cirrhosis, creating a particularly prothrombotic milieu. Mechanistically, this paradox results from the “rebalanced hemostasis” model, which states that cirrhosis reduces procoagulant and anticoagulant factors at the same time, resulting in an unstable equilibrium as opposed to a hypocoagulable state. 23 Notably, when combined with inflammation, trauma, or surgery, decreased production of important liver-synthesised anticoagulants such as protein C, protein S, and antithrombin III can tip this balance in favor of thrombosis.In cirrhotic patients, endothelial activation promotes platelet aggregation and microvascular thrombosis, as evidenced by increased von Willebrand factor levels and decreased ADAMTS13 activity. 28 Because coagulation pathways are continuously activated by hepatic damage and ongoing viral activity, these processes are particularly significant in HBV-associated cirrhosis. Clinically, our results support earlier research that questioned the standard practice of avoiding anticoagulation in cirrhosis. Low-molecular-weight heparin and even direct oral anticoagulants can be safely administered to certain cirrhotic patients with acceptable bleeding profiles, according to recent data. 29 Undertreatment for orthopedic trauma may result from the false belief that cirrhosis prevents thrombosis.Our findings suggest that thromboprophylaxis strategies for HBV-related cirrhosis may need to be reevaluated, incorporating prothrombotic risk and personalized bleeding assessment instead of depending only on platelet count or INR.
This study found that elevated HBsAg levels were independently associated with a higher risk of DVT in patients with chronic HBV and lower extremity fractures, consistent with previous findings. Wang et al reported that HBV infection increased the risk of postoperative complications—including DVT—by approximately 18% in elective joint arthroplasty patients, 30 which appears higher than the effect size observed in our study (OR = 1.002). This discrepancy may be explained by differences in study populations and outcomes: our cohort consisted of acute orthopedic trauma patients, who typically experience stronger stress responses, more pronounced inflammation and endothelial injury, and longer periods of immobilization, while our analysis focused specifically on thrombotic events. These factors may contribute to the variation in effect estimates between the two settings. In contrast to cirrhosis, where hepatic synthetic dysfunction is the primary cause of the coagulation imbalance, the prothrombotic state in an active HBV infection seems to involve systemic endothelial activation, immune dysregulation, and persistent viral inflammation.It has been demonstrated that damaged hepatocytes increase the expression of tissue factor (TF), which starts the extrinsic coagulation cascade and encourages the release of procoagulant microparticles. 31 Moreover, HBV-associated inflammation stimulates platelet and monocyte activation, which raises thrombin production independent of liver synthetic capacity. 23 In this situation, elevated HBsAg titers could be direct contributors to the hypercoagulable environment as well as indicators of viral burden.This primed thrombo-inflammatory state, when layered on top of orthopedic trauma, produces a “second hit” situation that significantly raises the risk of thrombosis. In addition to HBV-induced coagulopathy, trauma itself causes endothelial disruption and inflammatory cytokine surges, 32 creating a synergistic risk environment for DVT. Clinically, these results imply that elevated HBsAg levels, especially above the 826.35 IU/mL threshold, may be a valid indicator for identifying patients at high risk for DVT and may help direct more focused thromboprophylaxis treatments for chronic HBV patients.
Binary Logistic Regression Analysis of Variables Associated with DVT.

Abbreviations: HGB, hemoglobin; HBsAg, Hepatitis B surface antigen; CI, confidence interval.
*P < .05, statistical significance.
In our cohort of HBV-infected patients, higher HGB levels were associated with a lower risk of DVT, which is consistent with existing literature. A recent cohort study revealed a strong inverse relationship between HGB concentration and the risk of VTE, where anemia (Hb < 12-13 g/dL) significantly increased the risk of DVT and PE. 33 Additionally, previous studies have shown a U-shaped relationship between HGB levels and DVT incidence, with moderate HGB levels providing a protective effect, while both low and high HGB levels increase thrombosis risk. 34 Therefore, the protective effect of higher HGB levels observed in our study should be interpreted with caution, as it may apply only within a physiological range of HGB levels. Mechanistically, low HGB may promote thrombosis via iron-deficiency–driven reactive thrombocytosis and hypoxia-induced endothelial dysfunction with upregulation of procoagulant pathways, 35 whereas high HGB may favor thrombosis primarily through elevated viscosity, altered shear stress, and impaired venous return. 36 Importantly, we did not measure iron metabolism indices (eg, ferritin, transferrin saturation) or classify anemia subtypes, which limits mechanistic inference at the low-HGB end. Future studies should incorporate iron panels, anemia phenotyping, and viscosity-related measures (eg, hematocrit/whole-blood viscosity), and apply stratified or non-linear modeling to delineate thresholds and inflection points, thereby informing individualized thromboprophylaxis in HBV-infected patients with fractures.
In advanced chronic liver disease (especially HBV-related cirrhosis), continuous low-grade coagulation activation and impaired fibrinolytic clearance lead to elevated D-dimer and FDP levels even in the absence of overt thrombosis. 37 Mechanistically, factors such as endotoxemia and systemic inflammation drive ongoing thrombin generation and fibrin turnover, while reduced hepatic clearance of coagulation byproducts further elevates baseline D-dimer. 38 Consequently, these conventional thrombosis markers lack specificity in cirrhosis. 39 Conversely, their sensitivity is also compromised: clinical studies have reported that, in patients with liver cirrhosis, mean D-dimer and FDP levels in those with thrombosis are often comparable to those without thrombosis. Consistent with these findings, our study also showed no significant between-group differences in D-dimer levels (1.78 ± 4.18 vs 1.86 ± 4.66 μg/mL) or FDP levels (10.58 ± 20.77 vs 13.83 ± 27.32 μg/mL), further indicating a substantial risk of false-negative results in this population. 40 Standard D-dimer cut-offs have had to be raised in cirrhosis to improve specificity, but this comes at the cost of sensitivity. 37 Therefore, recent literature emphasizes the limitations of traditional biomarkers in chronic liver disease and advocates for adjunctive tools – for example, viscoelastic tests, including thromboelastography (TEG) and rotational thromboelastometry (ROTEM) or anti-factor Xa activity assays – to more accurately evaluate coagulation status and thrombosis risk in these patients.41,42 Given that our study found HBsAg levels to have predictive value in distinguishing DVT status in HBV-infected patients, we suggest that HBsAg be used as a supplementary predictor of DVT in this population to address the limitations of conventional markers in terms of sensitivity and specificity.
Several limitations should be acknowledged. First, this was a retrospective, single-center study from a tertiary trauma hospital in China with a moderate sample size and multiple subgroup/risk factor analyses, which limits statistical power and generalizability, especially to settings with different HBV prevalence, trauma patterns, or thromboprophylaxis practices. Second, although we collected pre-admission antiviral treatment history and surgery-related variables (such as surgery timing and postoperative bed rest) and adjusted for them, treatment information came from retrospective records and may be incomplete, and DVT diagnosis relied on duplex ultrasonography without routine CT venography, so small or asymptomatic distal DVT may have been missed, leading to underestimation of the true incidence. Third, in cirrhotic patients we did not measure natural anticoagulant proteins (protein C, protein S, antithrombin III) or perform detailed coagulation profiling, limiting mechanistic interpretation of “cirrhosis-induced DVT.” Finally, because of the retrospective design, our findings demonstrate associations rather than causality, and larger multicenter prospective studies are needed to confirm these results.
Conclusion
In our cohort of patients with chronic HBV infection and lower extremity fractures, the incidence of DVT was 30.4%. Multivariate analysis identified multiple fractures, elevated HBsAg, a history of cirrhosis, and a history of cerebral infarction were independently associated with a higher risk of DVT, while higher HGB levels were associated with a lower risk. Based on these results, individualized thromboprophylaxis strategies may be needed in HBV-infected orthopedic trauma patients, such as closer monitoring for DVT in those with HBsAg > 826.35 IU/mL, cirrhosis, multiple fractures, or a history of cerebral infarction, and correcting anemia to maintain adequate hemoglobin levels.
Footnotes
Abbreviations
Acknowledgements
We thank the staff of the Third Hospital of Hebei Medical University for their assistance with data retrieval and patient care.
Ethics Approval and Consent to Participate
This study was approved by the Ethics Committee of the Third Hospital of Hebei Medical University (approval number: S2023-002-1). Informed consent was waived due to the retrospective nature of the study.
Author Contributions
ZYH and LJ designed the study. HPL, QYL and EDZ collected the data. WXL and HFW performed the analysis. EDZ and JQZ drafted the manuscript. All authors revised the manuscript critically for important intellectual content and approved the final version.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key R&D Program of the China Ministry of Science and Technology, Department of Finance of Hebei, Natural Science Foundation of Hebei Province, (grant number 2024YFC2510600, ZF2024086, H2023206159).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
