Abstract
This study investigates the prevalence and risk factors associated with venous thrombotic events in patients receiving (ECMO) support. Systematic review and meta-analysis of case-control and cohort studies. PubMed, Cochrane Library, Embase, CINAHL, Web of Science, Scopus, and ProQuest databases from inception through November 25, 2023.Case-control and cohort studies focusing on the prevalence and risk factors for venous thrombotic events in patients supported by ECMO. Identification of risk factors and calculation of incidence rates. Nineteen studies encompassing 10,767 participants were identified and included in the analysis. The pooled prevalence of venous thrombotic events among patients receiving ECMO support was 48% [95% confidence interval (CI) 0.37–0.60, I2 = 97.18%]. Factors associated with increased incidence rates included longer duration of ECMO support (odds ratio [OR] 1.08, 95% CI 1.07-1.09, I2 = 49%), abnormal anti-coagulation monitoring indicators (OR 1.02, 95% CI 1.00-1.04, I2 = 84%), and type of ECMO cannulation (OR 1.77, 95% CI 1.14-3.34, I2 = 64%). The pooled prevalence of venous thrombotic events in patients with ECMO support is high. Increased risk is associated with extended duration of ECMO support, abnormal anti-coagulation monitoring, and specific types of ECMO cannulation.
Introduction
Extracorporeal membrane oxygenation (ECMO) is a critical extracorporeal life support technology predominantly utilized for managing cardiac and/or respiratory insufficiency and failure. 1 The ECMO system extracts venous blood from the body, oxygenates it, removes carbon dioxide via an extracorporeal membrane oxygenator, and then returns the oxygenated blood to the body through a centrifugal pump. This process facilitates gas exchange and promotes circulation. ECMO is categorized based on the method of blood flow into two types: VA-ECMO, where blood is withdrawn from and returned to the arterial system, providing both circulatory and respiratory support, and veno-venous ECMO (VV-ECMO), where blood is extracted and reinfused into the venous system, offering primarily respiratory assistance. 2
Venous thrombosis frequently occurs during and after using ECMO 3 and can manifest as catheter-associated thrombosis or systemic thromboembolism. 4 Venous thromboembolism (VTE) includes deep venous thrombosis (DVT) and pulmonary thromboembolism (PTE). 5 DVT refers to abnormal clot formation in deep veins, whereas PTE involves a thrombus obstructing the pulmonary artery. One study has indicated a notably high incidence of VTE, reaching 43.8%, following lung transplantation, according to data from the Cleveland Respiratory Center in the United States. 6 This study involved 701 lung transplant recipients and revealed that the elevated VTE incidence in these patients was primarily attributed to the high use rate of ECMO during the waiting period for lung transplantation.
ECMO carries the potential for severe and, in some cases, fatal complications. 7 An analysis of data from 100 000 adult patients receiving extracorporeal life support, as registered by Extracorporeal Life Support Organization (ECLS), identified bleeding, thrombosis, infection, and hemolysis as the most common complications leading to ECMO failure. 8 These complications can occur throughout adjuvant support therapy, with thrombus-related problems being particularly prevalent. 9 Studies have confirmed that both catheter-related and systemic thrombosis in patients following ECMO support and extubation constitute significant complications that are closely related to patient prognosis. 10
The relationship between ECMO support and venous thrombosis and its potential contributing factors remains unclear. To address this ambiguity, this study conducted a meta-analysis to elucidate the risk factors for VTE in patients supported by ECMO. The aim is to furnish evidence for the early detection of VTE and to inform the implementation of preventive measures and nursing interventions.
Material and Methods
Protocol and Guidelines
This study adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 11 The review protocol for this review was registered with PROSPERO (CRD42023472498).
Inclusion Criteria
Case-control and cohort studies published in English were deemed eligible if they involved adult patients (age ≥18 years) receiving ECMO support with confirmed VTE/, DVT/PTE/, or Catheter-related venous thrombosis (CRVT).DVT is diagnosed by ultrasound, but PE is diagnosed by computer tomographic pulmonary angiography (CTPA) or lung scintigraphy. Additionally, these studies are needed to investigate thrombosis incidence and risk factors during ECMO support.
Exclusion Criteria
Studies were excluded if they were case reports or case series, duplicates, or scored less than seven points on the Newcastle-Ottawa Scale (NOS). Additionally, studies were omitted if they provided incomplete information, could not support data extraction, or if the original data could not be converted and utilized effectively.
Outcomes
The primary outcome was the incidence rate, and the secondary outcome was risk factors.
Search Strategy
One of the authors (YZ) conducted literature searches across multiple databases, including PubMed, Cochrane Library, Embase, CINAHL, Web of Science, Scopus, and ProQuest, from their inception to November 25, 2023 (Supplementary Table S1-S7). ClinicalTrials.gov and the World Health Organization International Clinical Trials Registry Platform were also searched to identify ongoing and unpublished studies that met our inclusion criteria. In addition, the reference lists of identified studies and systematic reviews were reviewed to ensure comprehensiveness. While there was no language restriction during the database search, only English studies are potentially eligible for this study.
Study Selection
After eliminating duplicate records, two independent researchers (YZ and JL) screened the titles and abstracts of all selected studies. Full texts were retrieved for those studies that met the eligibility criteria. Any discrepancies between the two researchers were discussed with a third researcher and resolved through consensus based on the inclusion criteria.
Data Collection Process
Two researchers above (YZ and LY) used a standardized data extraction spreadsheet to extract details such as author name, publication year, study location, study design, sample size, and risk factors from the included studies. A third researcher settled any discrepancies during this process.
Assessment of Risk of Bias and Quality of Evidence
The quality of all included trials was independently assessed and determined by the two researchers mentioned above (YZ and JL) using the Newcastle-Ottawa Scale, which consists of three parts: selection of study population, comparability between groups, and exposure factors. 12 A perfect score on this scale is 9 points, with literature scoring 6 or higher considered high quality.
Data Synthesis and Statistical Analysis
Data were analyzed by STATA (version 17.0) and Rev Man (version 5.4). We also assessed outcomes using the Odds ratio and their 95% confidence intervals (CIs). P < .05 was considered as statistically significant. Heterogeneity was assessed by the I2 test (I2 < 50%). 13 Fixed effects models were utilized to combine the outcomes. However, random effects models were used when the I2 value was more than 50%. The presence of minor study effects was evaluated qualitatively, through funnel plot inspection, and quantitatively, using Egger's test. 14
Sensitivity Analyses
We evaluated the sensitivity of datasets and the resultant impact on the combined effect size (ES) in each study. Also, the methodology was performed to investigate the impact of each study on all analyses.
Results
Eligible Studies and Study Characteristics
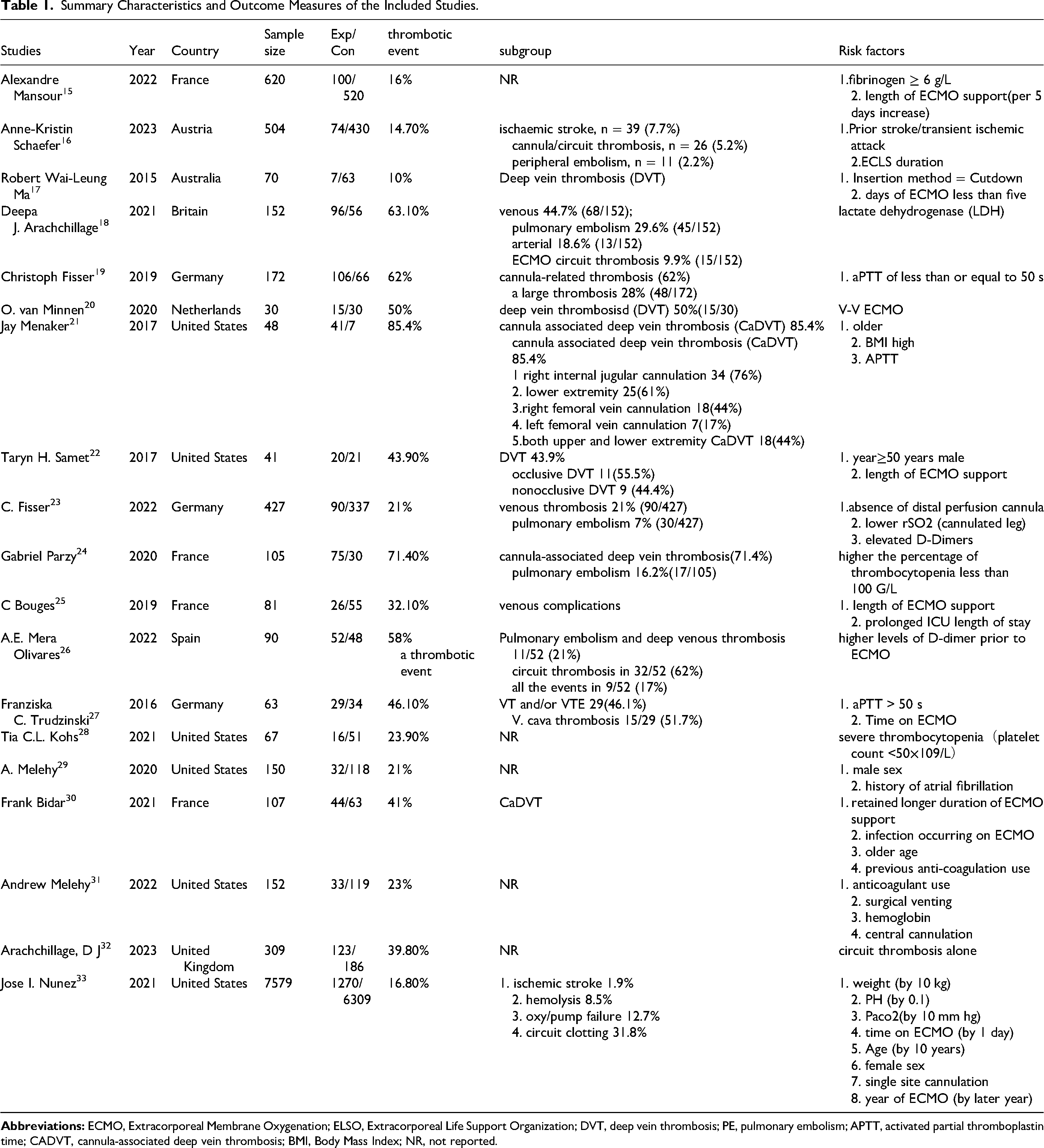
Initially, 10 767 studies were identified, from which nineteen eligible studies were selected for the final meta-analysis (Figure 1). Nineteen of these included studies scored 6 points at least in the methodological quality assessment, indicating a high quality of the included studies for meta-analysis. Table 1 summarizes the included studies.
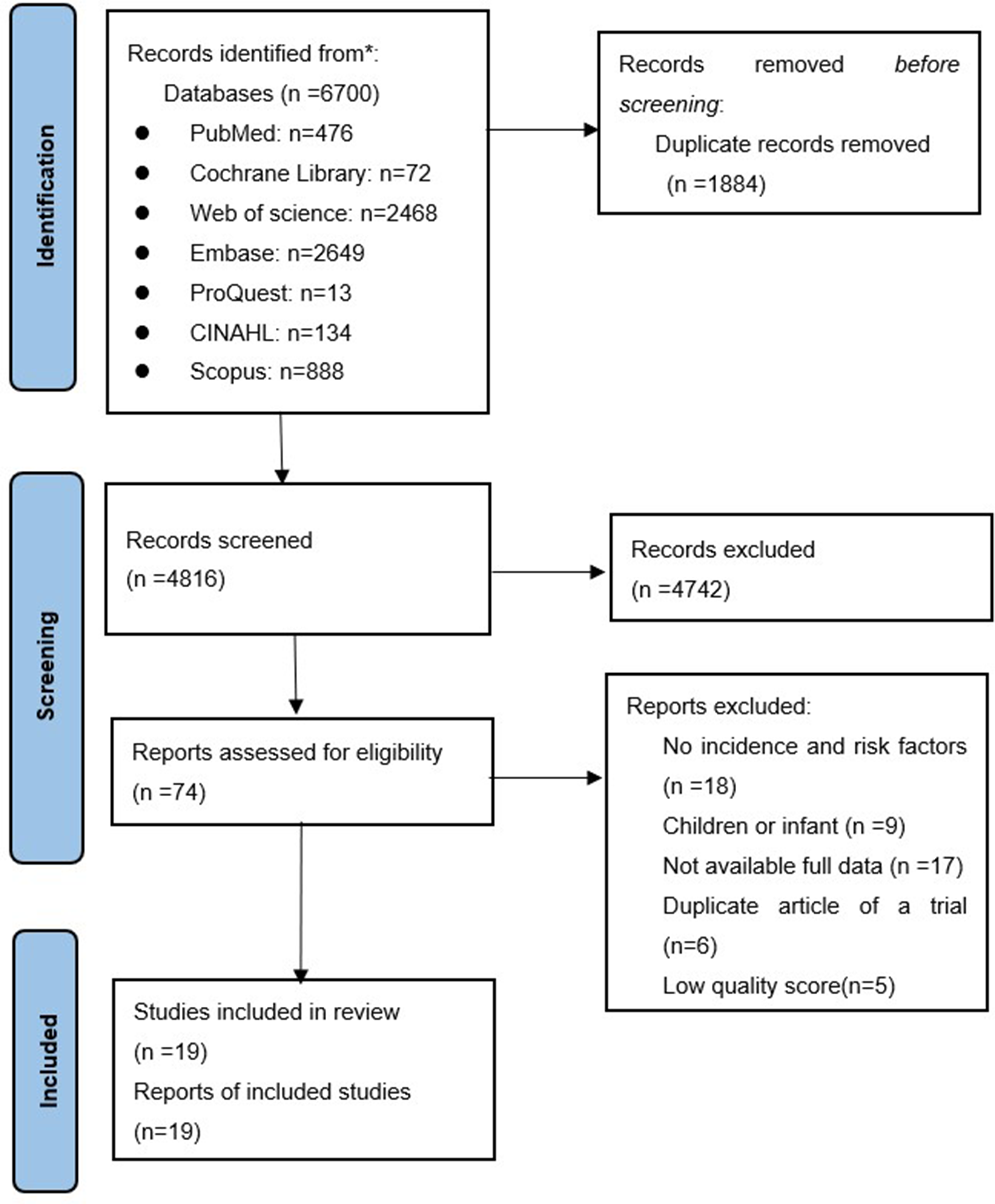
PRISMA (preferred reporting items for systematic reviews and meta-analysis) flowchart showing the study selection process.
Summary Characteristics and Outcome Measures of the Included Studies.
Primary Outcome: Incidence Rate
Nineteen studies reported the incidence rate of ECMO support in patients, showing heterogeneity among them. Therefore, fixed effect analysis was performed. The pooled prevalence of venous thrombotic events in patients with ECMO support was 48% (95%CI 0.37-0.60, I2 = 97.18%, Figure 2). The high pooled prevalence underscored the importance of further research into risk factors associated with venous thrombotic events in ECMO-supported patients. The funnel plot analysis suggested symmetry (P > .05), and Egger's tests revealed no significant minor study effects (Supplementary Figure S1).
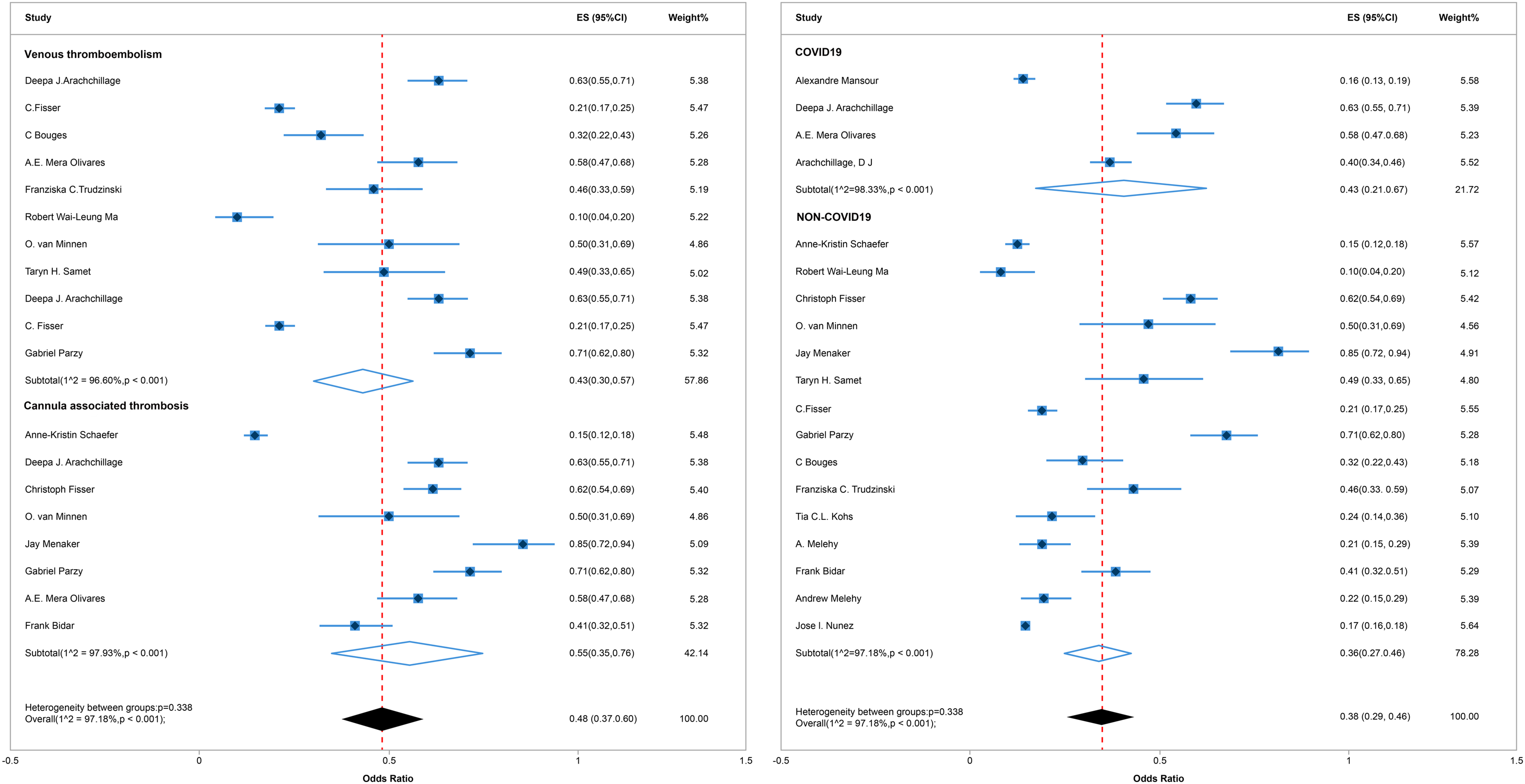
Forest plot of incidence of venous thromboembolism, cannula-related thrombosis and COVID 19-associated thrombosis on or after ECMO.
Subgroup analysis demonstrated that the incidence of VTE was 43%, whereas the occurrence specific to CRVT was notably higher at 55%. The subgroup analysis delineating ECMO-associated thrombosis prevalence in the contexts of COVID-19 and non-COVID-19 cases holds considerable significance. Within the results segment, we have meticulously conducted subgroup analyses, which have revealed no statistically significant disparity in the occurrence of clotting events between subjects afflicted with COVID-19 and those unaffected (P = .338, Figure 2). Specifically, the incidence of thrombus formation in COVID-19 patients undergoing ECMO support was observed to be 43%, whereas in non-COVID-19 patients receiving ECMO assistance, the rate stood at 36%.
Secondary Outcome: Risk Factors
All nineteen studies investigated various risk factors. Longer ECMO support duration (risk ratio 1.08, 95% CI 1.07-1.09, I2 = 49%, Figure 3), abnormal anti-coagulation monitoring indicators (risk ratio 1.02, 95% confidence interval 1.00-1.04, I2 = 84%, Figure 4) and cannulation type of ECMO (risk ratio 1.77, 95% confidence interval 1.14-3.34, I2 = 64%, Figure 5) were associated with venous thrombotic events. Sensitivity analysis indicated that excluding any single study had minimal impact on the combined effect size. Funnel plot analysis showed symmetry (P > .05), and both Egger's and Harbord's tests did not detect any significant minor study effects (Supplemental Figures S2–S4).
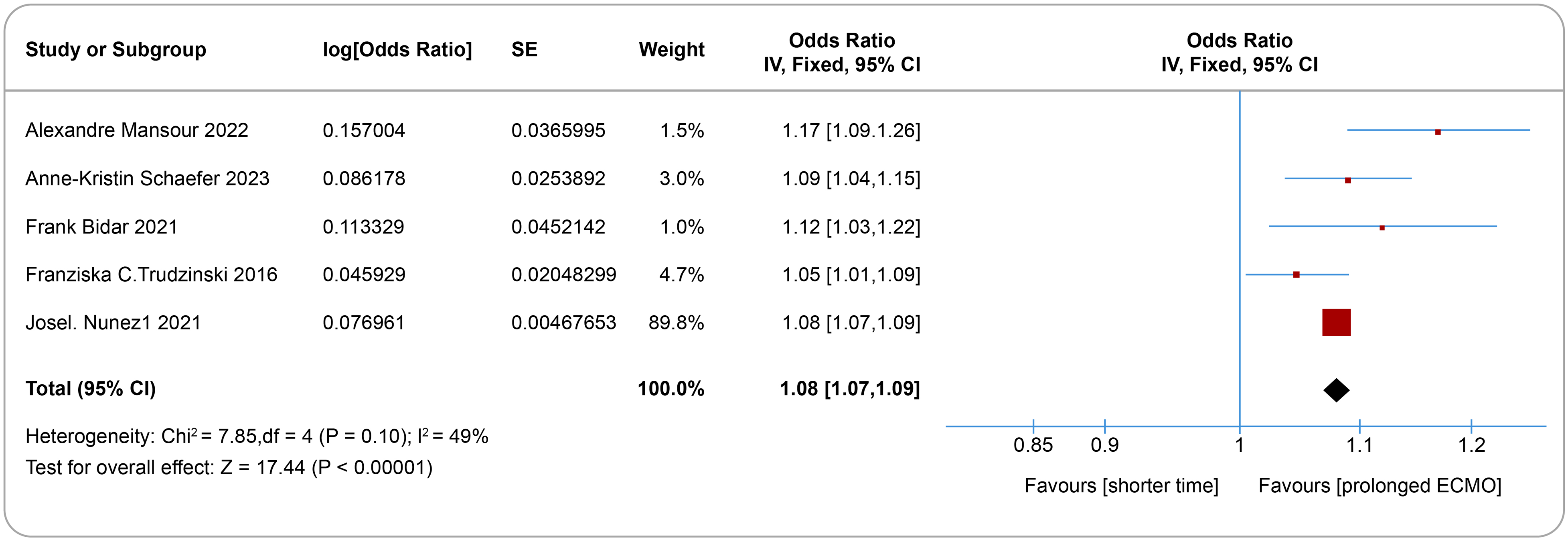
Forest plot of the effects of prolonged ECMO support on the development of venous thromboembolism and cannula-related thrombosis.
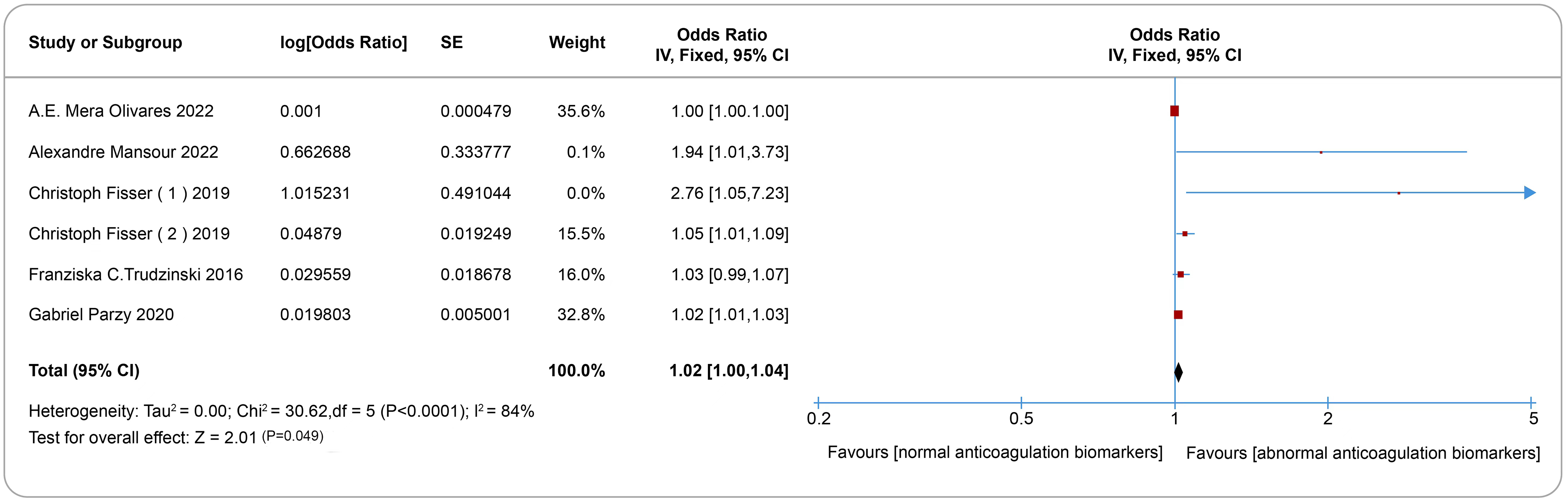
Forest plot of the effects of abnormal anti-coagulation on the development of venous thromboembolism and cannula-related thrombosis.
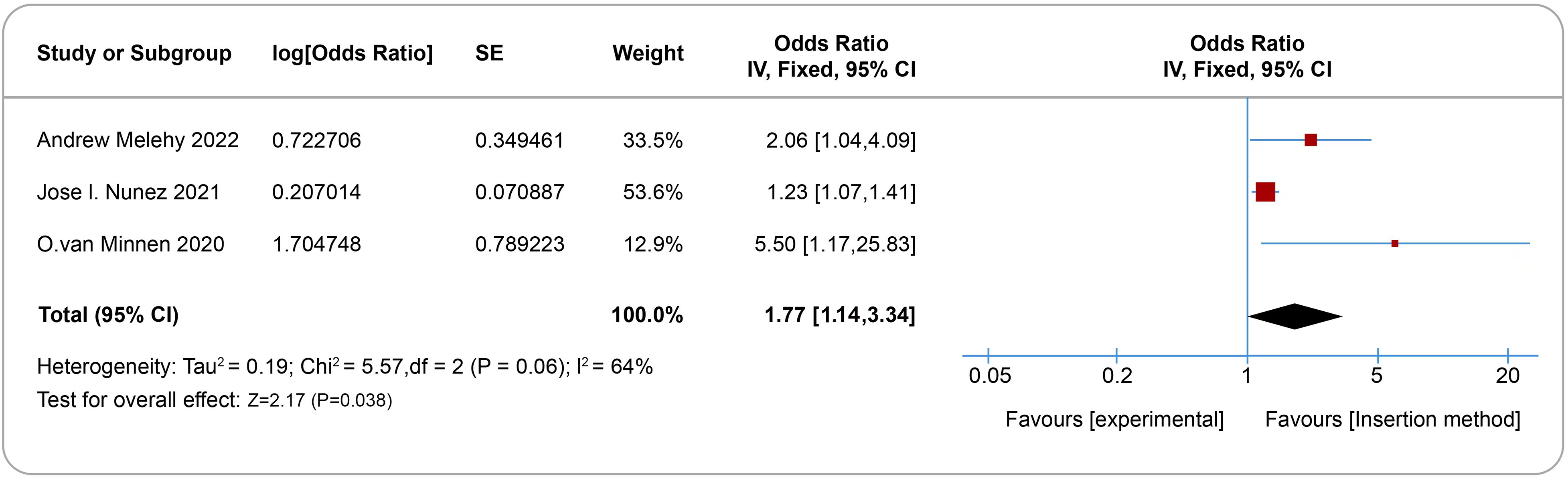
Forest plot of the effects of insertion method on the development of venous thromboembolism and cannula-related thrombosis.
Discussion
Principal Findings and Comparison with Other Studies
This meta-analysis of 19 studies involving 10 767 participants identified a significant association between ECMO support and venous thrombotic events. Identified risk factors included prolonged ECMO duration, abnormal anti-coagulation monitoring, and cannulation type. Despite the heterogeneity of the study, sensitivity analyses had minimal impact on the results. The symmetry of the funnel plot and non-significant results from Egger's and Harbord's tests indicated no substantial small-study effects. However, this study's findings on total venous thrombotic event rates diverge from previous meta-analyses.
A 2022 systematic review with meta-analysis, analyzing 18 studies with 1095 participants, reported an overall DVT incidence of 52.8% (95% confidence interval, 49.8-55.8%). Similarly, a systematic review by Abruzzo and Gorantla included 30 studies with 11 984 participants and highlighted VTE in patients receiving ECMO, noting the association between ECMO and VTE. 34 These reviews did not delve into the risk factors for VTE in ECMO patients. In contrast, our current analysis excluded over 10 studies featured in these prior meta-analyses but included additional 15 studies published post-2019. The studies included in our analysis accounted for 98% (10 593/10 767) of the total participants, allowing for a more comprehensive investigation of DVT incidence in ECMO patients. Research has validated the correlation between thrombosis and inflammation within the pathology of COVID-19. 35 However, our findings reveal no significant disparity in the incidence of VTE between COVID-19 and non-COVID-19 patients supported by ECMO.
Long-term ECMO support stands as a risk factor for the development of venous thrombosis development. This study found that the risk of venous thrombosis increased exponentially with the duration of ECMO use. Due to the compositional difference between ECMO biomaterial and normal vascular endothelium, when the artificial biomaterial on the surface of the ECMO tubing system comes into contact with the bloodstream, its surface structure will increasingly bind to free fibrin and platelets from the blood, both of which will form a complex to release a variety of procoagulant substances, activate endogenous coagulation pathways, and further enhance coagulation. 36 Plus, prolonged ECMO support induces venous stasis in the oxygenator because of overwork, which promotes thrombosis. Therefore, the oxygenator should be replaced promptly to ensure that ECMO achieves the ideal oxygenation effect and prevents VTE.
Abnormal anti-coagulation is another risk factor for venous thrombosis in patients undergoing ECMO support. Our study examined several coagulation monitoring parameters, including D-dimer, activated partial thromboplastin time (APTT), platelets, and fibrinogen. D-dimer, an indicator of fibrinolysis during thrombosis in ECMO patients, is critical in transforming stable fibrin into soluble byproducts. This transformation hinges on the conversion of plasminogen to plasminase, which actively breaks down fibrin into smaller fragments, releasing a variety of degradation products. First, the APTT test indicates a patient's intrinsic coagulation pathway and reflects the function integrity of the coagulation system. Studies have highlighted that a reduced APTT value may indicate an increased risk of thrombosis. 37 Second, elevated fibrinogen levels have been identified as contributing to a hypercoagulable state of the blood, amplifying the thrombotic risk. 38 Fibrinogen also interacts with various inflammatory factors, thereby intensifying the inflammatory response and contributing to the formation of inflammatory thrombosis. Third, platelets, derived from the cytoplasm of mature megakaryocytes in bone marrow, are an integral part of the process of thrombosis. During ECMO support, there is often a decline in platelet count and function. 39 This decrease can be attributed to factors like platelet consumption, mechanical damage due to ECMO components, and dilution of blood.
Besides, multiple catheterizations, peripheral cannulation, and V-VECMO mode can directly injure the venous wall and lead to the formation of DVT. Vascular wall injury, blood stasis, and blood hypercoagulability are known as Virchow's Triad, which can activate platelets to release a variety of bioactive substances, initiate the endogenous coagulation system, and cause platelet aggregation and adhesion, thus forming thrombus, underlying the pathogenesis of DVT. 40 The present study suggests multi-catheterization, peripheral cannulation, and VV-ECMO mode as the risk factors for developing venous thrombosis in patients on or after ECMO.
Strengths and Limitations
This systematic review with meta-analysis boasts several methodological strengths. We adhered to the recommendations of the Cochrane Collaboration and the PRISMA statement and established a priori protocol to ensure transparency and consistency. Furthermore, this study utilized the GRADE approach for a rigorous assessment of the quality of evidence, ensuring high quality for the primary outcome. It also reached the minimum information size required in trial sequential analysis, confirming that the study met the optimum size.
However, our study has several limitations that merit attention. Variations in assessing and quantifying risk factors across different studies may impact our findings. Our meta-analysis exclusively incorporated published literature, thus omitting gray literature, which could introduce potential publication bias. Additionally, while certain factors influencing DVT formation were identified, they could not be analyzed due to insufficient data or extraction limitations from the included studies. Moreover, language and search strategies limitations might have restricted our search's comprehensiveness.
Implications
ECMO is an invasive adjunctive therapy with an exceptionally high incidence of venous thrombosis complications, which warrants clinical vigilance. Despite this, there is still a lack of standardized guidelines for managing certain clinical complications. Some issues related to mechanical conduit complications stem from inadequate operational procedures, device management, and adherence to standards. These areas require further research and discussion for improvement. To prevent venous thrombosis complications, clinicians should increase their expertise, effectively anticipate problems, use evidence-based practices, and intervene promptly to improve the safety and efficacy of ECMO.
Patients on ECMO are at increased risk for VTE, emphasizing the need for increased VTE monitoring. Periodic laboratory testing should be performed, including coagulation function, D-dimer, platelet count, and APTT. In the presence of symptoms suggestive of new VTE, such as chest pain, dyspnea, or decreased oxygen saturation, clinicians should promptly perform CTPA or radionuclide lung V/Q imaging. Similarly, patients with signs of new DVT, such as asymmetric swelling, pain, or superficial venous dilation, warrant immediate compressive ultrasound (CUS) evaluation. 41
In addition to an increased risk of thrombosis, patients on ECMO also face a significant risk of bleeding, occurring in 12% to 52% of cases and contributing to a poor prognosis. Bleeding may originate from surgical incisions or ECMO insertion sites. However, it can also occur in areas such as the gastrointestinal tract, urinary tract, intracranial system, lungs, pericardium, or abdominal cavity. Intracranial bleeding is severe and life-threatening. Several factors contribute to bleeding in ECMO patients, including stress ulcers, excessive anti-coagulation, coagulation factor depletion, ECMO-induced thrombocytopenia, platelet dysfunction, hyperfibrinolysis, DIC, acquired von Willebrand syndrome, and heparin-induced thrombocytopenia. 42 Balancing anti-coagulation to mitigate both thrombotic and bleeding risks presents a significant clinical challenge for these complex, critically ill patients on ECMO.
Conclusions
This meta-analysis revealed a pooled prevalence of venous thrombotic events in ECMO-supported patients at 48%. Contributing factors included a longer duration of ECMO support, abnormal anti-coagulation monitoring, and the method of ECMO insertion. Deeming the systematic evaluation for DVT in every patient subsequent to the cessation of extracorporeal membrane oxygenation (ECMO) therapy as a commendable tactical measure worthy of contemplation. Healthcare providers ought to promptly recognize these predisposing factors and institute efficacious interventional measures, thereby mitigating the prevalence of VTE incidents associated with ECMO utilization.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241279293 - Supplemental material for Assessing Venous Thrombotic Risks in Extracorporeal Membrane Oxygenation-Supported Patients: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-cat-10.1177_10760296241279293 for Assessing Venous Thrombotic Risks in Extracorporeal Membrane Oxygenation-Supported Patients: A Systematic Review and Meta-Analysis by Yan Zhu, Mei-Juan Lan, Jiang-Shu-Yuan Liang, Ling-Yun Cai, Lu-Yao Guo, Pei-Pei Gu and Fei Zeng in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Author Contributions
FZ and YZ conceptualized the study and developed the protocol. YZ and JL conducted the literature search. YZ and JL selected the studies and extracted the relevant information. LY G, PP G, and LY C synthesized the data. YZ drafted the initial version of the paper, and all authors reviewed and made critical revisions to subsequent drafts of the paper. The final version was approved by all authors. FZ and MJ L are the study guarantors. The corresponding author confirms that all listed authors meet the criteria for authorship and that no eligible authors have been excluded.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the study are available from the corresponding author on reasonable request.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the Health Commission of Zhejiang Province, China, (grant number 2021KY713).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
