Abstract
Objective
This study aimed to investigate the associations between total serum bilirubin levels and the incidence of venous thromboembolism (VTE) among patients with influenza infection.
Methods
A retrospective cohort study was conducted among outpatients with laboratory-confirmed influenza using data from the Veterans Affairs Informatics and Computing Infrastructure (VINCI). Propensity score weighting was applied to balance study groups across baseline covariates. Cox proportional hazards models assessed VTE risk by total bilirubin levels, adjusting for important covariates including age, sex, race, comorbidity index, BMI, and smoking status.
Results
A total of 487 patients with total bilirubin levels <0.3 mg/dL, 8608 patients with levels between 0.3–1 mg/dL, and 1148 patients with levels >1 mg/dL were included. Patients with bilirubin <0.3 mg/dL exhibited a 6-fold higher risk of VTE compared to those with levels 0.3–1 mg/dL within 30 days of infection (HR = 6.2, 95% CI = 1.46–26.42). Elevated risks were noted through 90 days post infection (HR = 4.71, 95% CI = (1.42–15.67))
Conclusions
Serum bilirubin levels, particularly below 0.3 mg/dL, were significantly associated with an increased risk of VTE among individuals with influenza. These findings suggest that lower bilirubin levels may contribute to heightened inflammatory responses and subsequent thromboembolic events in patients with influenza. The underlying mechanisms and potential therapeutic implications for VTE prevention among patients with acute respiratory infection warrants further consideration.
Introduction
Bilirubin is a byproduct of heme catabolism and found to be inversely correlated with the incidence of thromboembolic and cardiovascular events.1–3 This relationship suggests a potential protective role for bilirubin, attributed to its antioxidant properties. Conversely, influenza virus infection is associated with an increased risk of venous thromboembolism (VTE), encompassing both deep vein thrombosis (DVT) and pulmonary embolism (PE).4–8
The mechanisms underlying influenza-associated thrombosis are multifaceted, with pro-oxidant and inflammatory responses playing pivotal roles. Influenza infection induces excessive production of reactive oxygen species (ROS),9–12 creating a pro-oxidant and inflammatory environment favorable to coagulation.13–15 Influenza triggers a cascade of proinflammatory cytokines, including IL-1β, TNF-α, and IL-6.16,17 These inflammatory mediators contribute to endothelial dysfunction and activate the coagulation pathway through tissue factor (TF) induction, creating a prothrombotic state.
The interaction between bilirubin levels and VTE after influenza infection is currently unexplored. We hypothesize that lower serum bilirubin levels during influenza infection may exacerbate the pro-oxidant state, potentially amplifying the risk of subsequent VTE. Our study aims to investigate the association between total serum bilirubin levels and the incidence of thromboembolic events in a national cohort of patients with influenza.
Materials and Methods
Data Source
This retrospective study examined VTE among outpatients with laboratory confirmed influenza. Data on patient demographics, medical claims history, hospitalization, and outpatient medication dispensation was obtained using the Veterans Affairs Informatics and Computing Infrastructure (VINCI). The study received Dorn Research Institute Institutional Review Board (IRB No.1139248) and VA Research and Development approval and was conducted in compliance with the Department of Veterans Affairs requirements. This paper used STROBE guidelines for the reporting of observational studies.
Cohort Creation
Patients were included in the study if they had a positive influenza polymerase chain reaction (PCR) or antigen test along with an outpatient diagnosis code indicating fever, cough, acute respiratory infection, or influenza. Encounters for influenza with a prior positive influenza lab result within 180 days were excluded. Patients were required to have a bilirubin laboratory taken on the influenza diagnosis date and were excluded if they had no bilirubin lab result, were hospitalized on the day of diagnosis or had a hospital admission or discharge date within 30 days prior to diagnosis. Patients with any outpatient dispense of an anticoagulant medication including warfarin, apixaban, rivaroxaban, edoxaban, dabigatran, or enoxaparin were excluded. Patients with a missing body mass index (BMI) were also excluded.
Study Outcome
Venous thromboembolism was categorized similar to prior work, 18 utilizing a combination of diagnosis codes and anticoagulant medication dispenses. VTE was indicated if patients had an inpatient diagnosis code indicating VTE, codes listed in Supplementary Table S1, or at least 1 outpatient VTE diagnosis code and an initial anticoagulant medication within 10 days after the outpatient diagnosis. Anticoagulant medications include an outpatient dispense record for warfarin, apixaban, rivaroxaban, edoxaban, dabigatran, or enoxaparin, a common procedure terminology (CPT) code indicating administration for an anticoagulant (J1642, J1643, J1644, J1650, J1652) or an IV infusion of heparin or fondaparinux. VTE incidence was evaluated until a maximum of 180 days post influenza diagnosis. Patients with records indicating a VTE within 182 days prior to influenza were excluded.
Statistical Analysis
Patients were categorized into mutually exclusive cohorts based on their total bilirubin levels on the diagnosis date. Cohorts were broken down into three mutually exclusive groups based on their bilirubin levels including less than 0.3 mg/dL, between 0.3 and 1 mg/dL and greater than 1 mg/dL. Baseline characteristics were summarized using means, proportions, and standard deviations. Statistical tests, including T-tests and Chi-square tests, assessed cohort differences. Standardized differences (Std.Diff) were calculated to quantify the degree of similarity between the groups, with values less than 0.2 or ideally, 0.1 indicating good balance between cohorts. To analyze the risk of VTE among the cohort groups the cumulative hazard of VTE was assessed through 180 days stratified by the bilirubin level. Cumulative event counts and the number of patients at risk are also presented.
To minimize the possibility of confounding we used propensity score weighting. The propensity score model was specified using a multinomial logistic regression model that includes all baseline covariates. Weights we created as 1 divided by the estimated propensity score for respective treatment received. Weights were truncated at the 99th percentile. Means and percentages were compared with standardized differences across weighted samples. Subsequently, weighted Cox proportional hazards models evaluated hazards of VTE among influenza patients with varying bilirubin levels, adjusting for covariates including age, sex, race, comorbidity index, BMI, smoking status, and year of influenza diagnosis. Models were fit at different follow-times including withing 30, 60,90, and 180 days. A weighted Cox model was fit to examine differences in mortality within 90 days of diagnosis.
2020 and Later Subgroup Analysis
Variation among strains of influenza over time along with other respiratory viruses such as COVID-19 could play a role in the development of VTE. We conducted a sub-analysis among patients with influenza diagnosed in 2020 and later. Among this subgroup, propensity score weighting was conducted by first estimating a multinomial logistic regression to create the propensity score specific for the subgroup. Inverse propensity score weights were then calculated by one divided by the probability of treatment received. Weights were then truncated at the 99th percentile. Weighted Cox models were fit to estimate hazards of VTE.
Results
Sample Characteristics
Table 1 displays the the baseline characterstics for the influenza patients included in the sample. Among these patients a total of 487 patients had bilirubin levels <0.3 mg/dL, 8608 patients had bilirubin levels between 0.3–1 mg/dL, and 1148 patients had bilirubin levels >1 mg/dL were included. Mean bilirubin levels for each cohort were as follows: 0.2 in the <0.3 mg/dL cohort, 0.58 in the 0.3–1 mg/dL cohort, and 1.47 in the >1 mg/dL cohort. Antiviral treatment usage did not exhibit a significant difference across bilirubin groups (p = 0.45, Std.Diff =0.038). Age was positively correlated across the groups (p < 0.001, Std.Diff = 0.348), with older age being associated with higher bilirubin levels. Race distribution did not significantly differ among the groups (p = 0.1, Std.Diff = 0.124), nor was there significant variation among BMI (p = 0.263, Std.Diff = 0.119), drug or alcohol abuse (p = 0.07, Std.Diff = 0.113). Higher proportions of smokers were reported in the lower bilirubin cohorts (p = 0.047, Std.Diff = 0.11). The Charlson comorbidity index exhibited significant differences (p = 0.011, Std.Diff = 0.122), reflecting higher average comorbidity burdens as bilirubin increased. Table 2 illustrates propensity-weighted characteristics of the cohort groups. Propensity weighting achieved a balanced distribution for covariates across the study cohorts, as reflected by low (all less than 0.1) standardized differences.
Sample Characteristics.
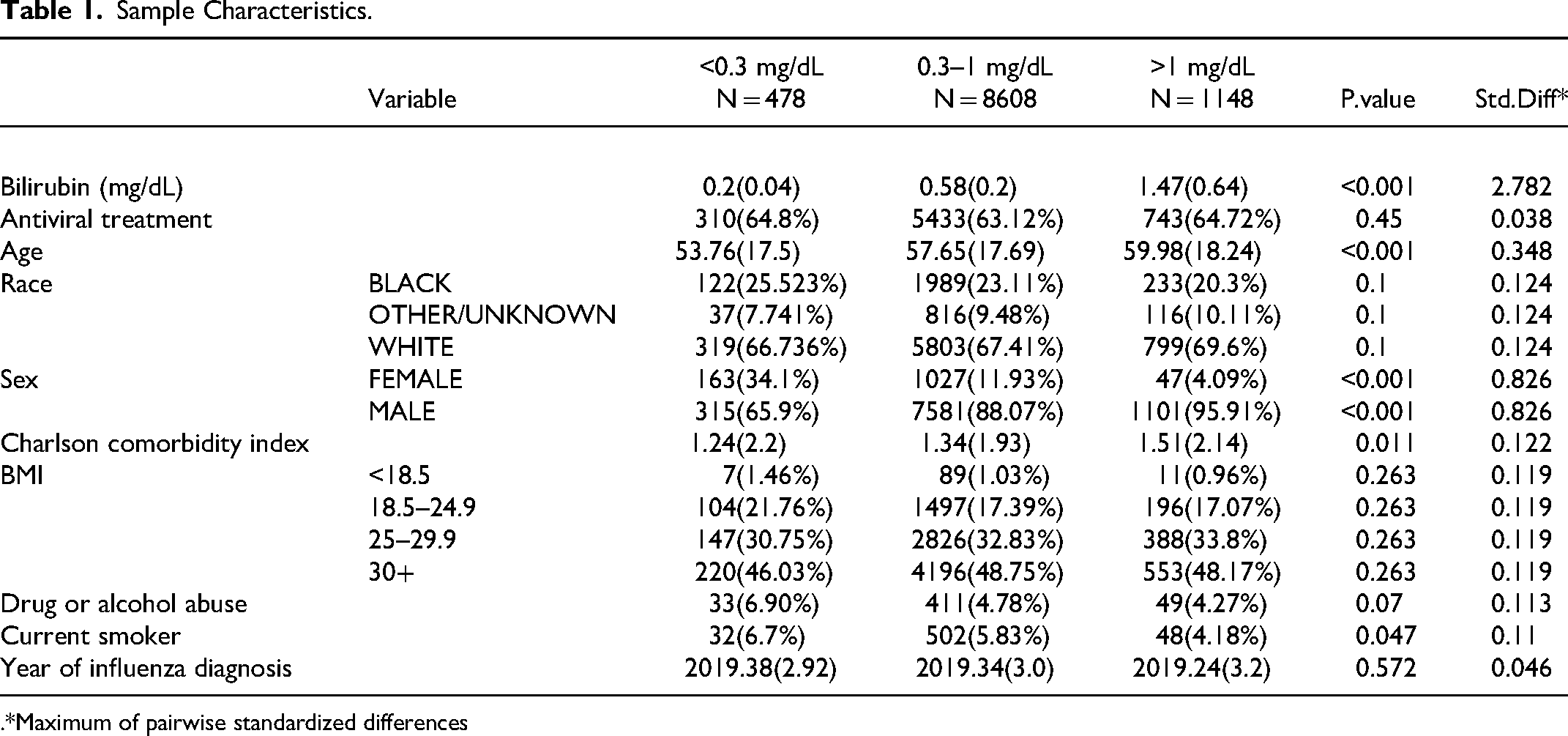
.*Maximum of pairwise standardized differences
Propensity Weighted Characteristics.
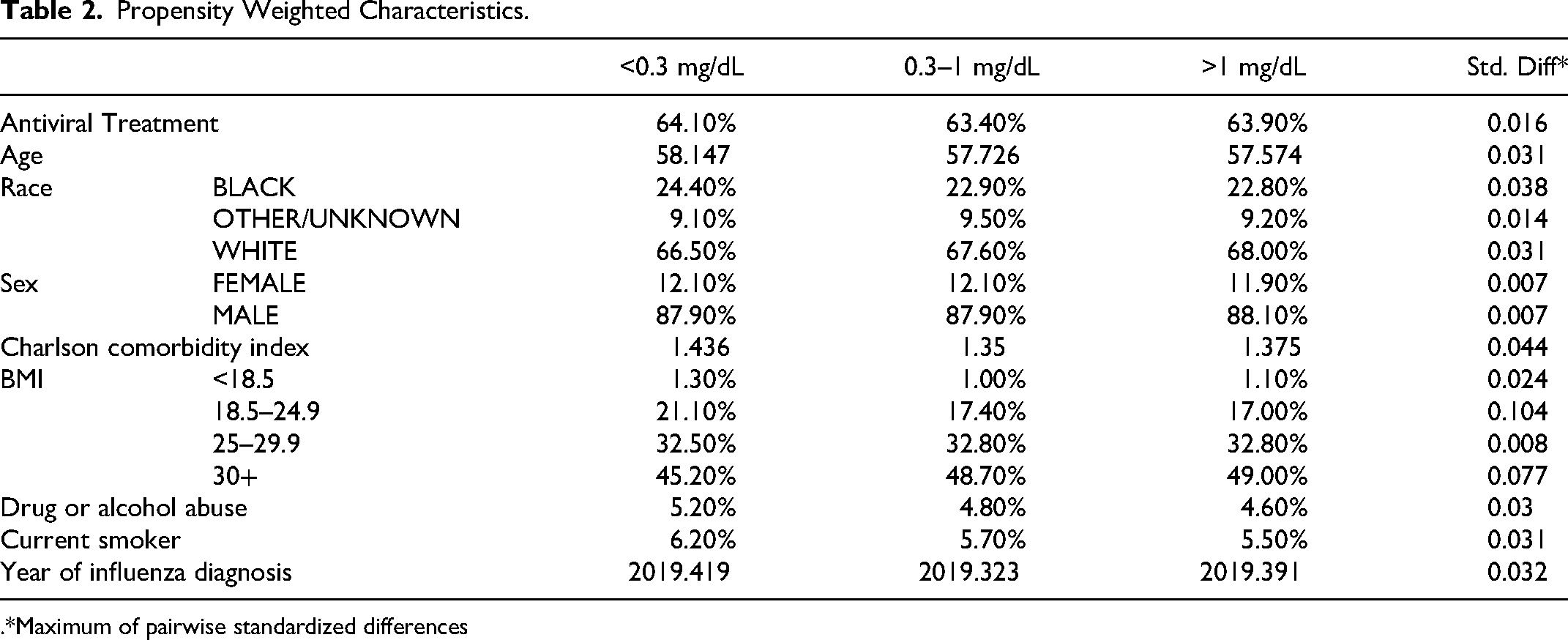
.*Maximum of pairwise standardized differences
Bilirubin and the Risk of VTE
Figure 1 presents cumulative incidence of VTE across the study period among the original study data. Across the study period, patients with low bilirubin levels (<0.3 mg/dL) and those with bilirubin greater than 1 mg/dL have higher cumulative VTE hazards compared to the 0.3–1 mg/dL cohort. The cohort with the lowest bilirubin levels (<0.3 mg/dL) had the highest hazard of VTE over the study period.
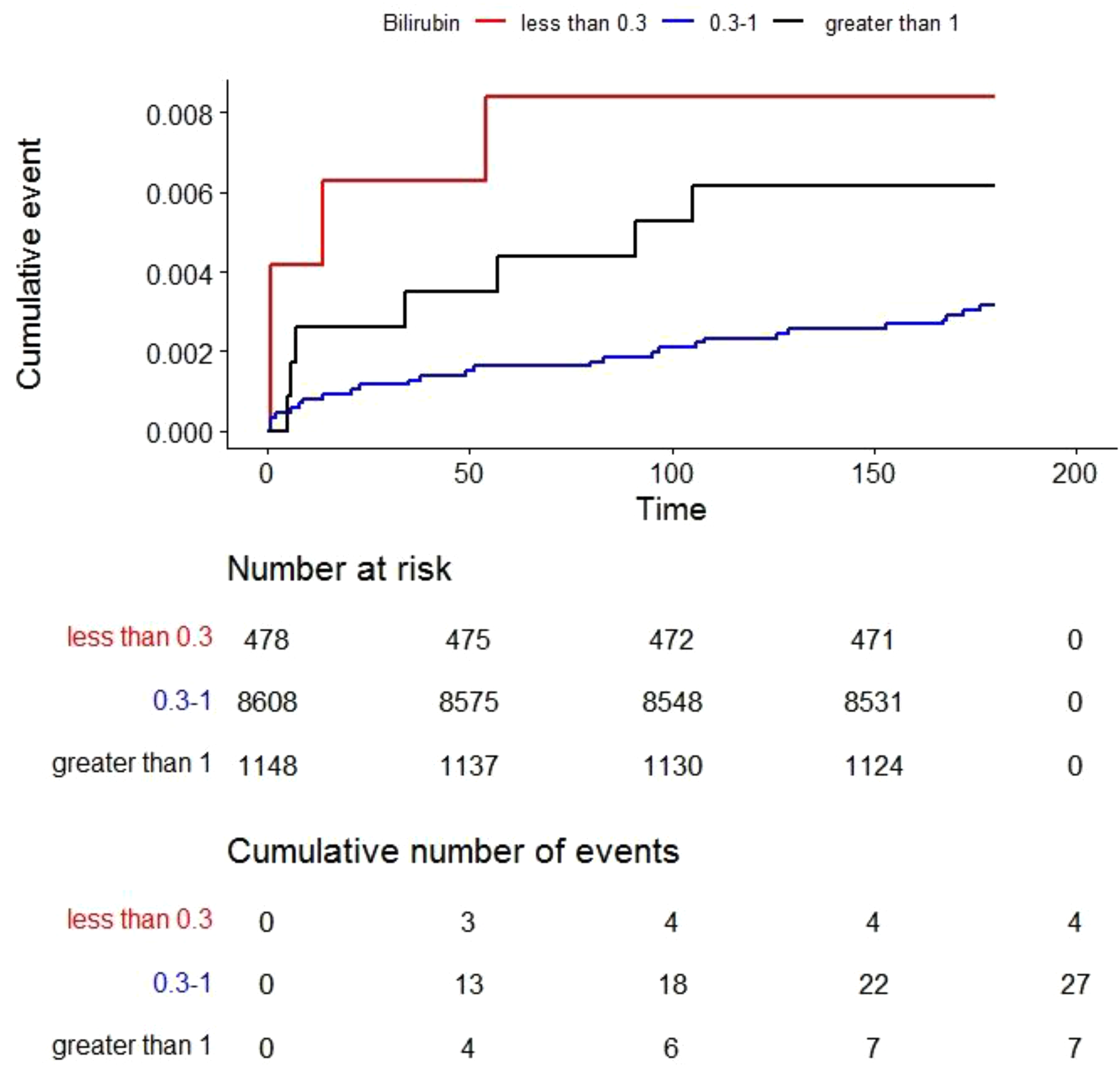
Kaplan-Meier cumulative incidence curves.
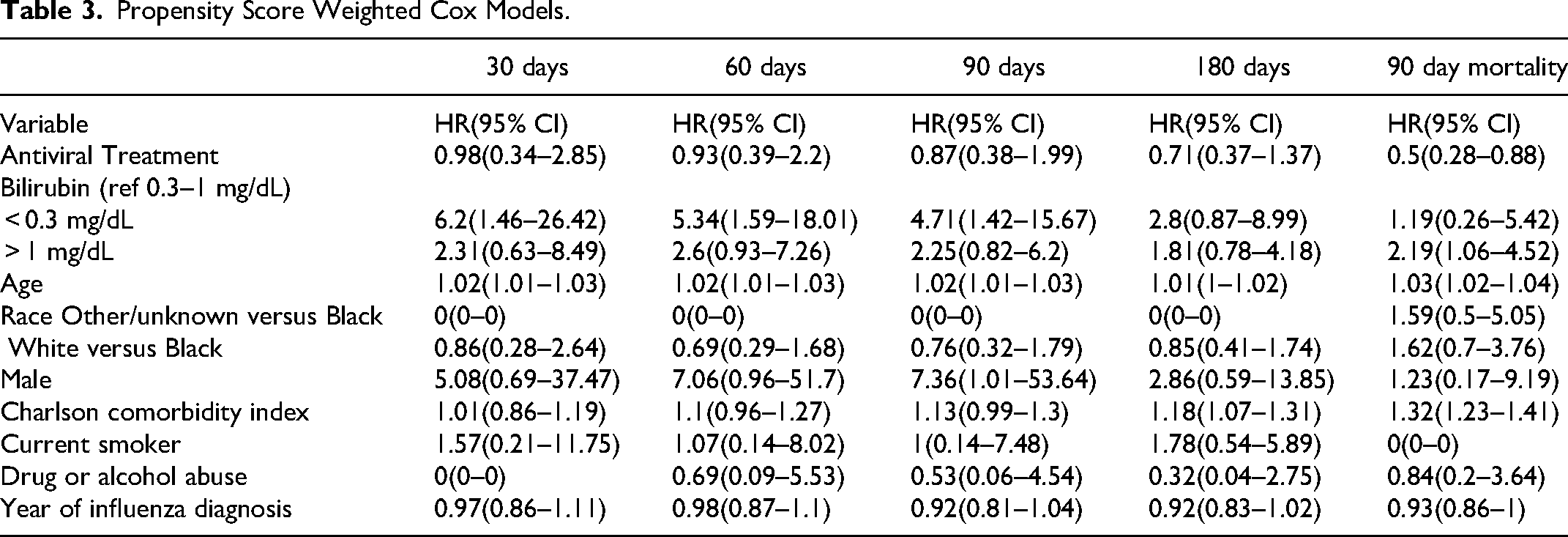
Table 3 presents the results of propensity weighted Cox proportional hazards models evaluating the hazard of VTE associated with bilirubin levels across time points. Hazard ratios (HR) with 95% confidence intervals are reported. Patients with bilirubin levels lower than 0.3 mg/dL exhibited a six-fold increased hazard of VTE at 30 days (HR = 6.2, 95% CI = 1.46–26.42). At subsequent follow-up time points the magnitude of the HR reduced: 60 days (HR = 5.34, 95% CI = 1.59–18.01) and 90 days (HR = 4.71, 95% CI = 1.42 −15.67), 180 days (HR = 2.8, 95% CI = 0.87–8.99) compared to those with levels between 0.3 and 1.0 mg/dL. Patients with bilirubin levels greater than 1 mg/dL did not have a statistically significantly greater risk of VTE in any time period compared to patients with bilirubin levels between 0.3–1 mg/dL. However, while not statistically significant, point estimates for the patients with >1 mg/dL all indicated higher risk compared to patients with bilirubin levels between 0.3–1 mg/dL. Moreover, the HRs were more consistent over the time frame ranging from a low of 1.81 (180 days) to 2.6 (60 days). This is a noticeable difference compared to the hazard ratio estimated for the patients with <0.3 mg/dL which peaked at the earliest time point (30 days) and descended with all subsequent time points.
Propensity Score Weighted Cox Models.
Bilirubin and the Risk of VTE: Years 2020 Onward
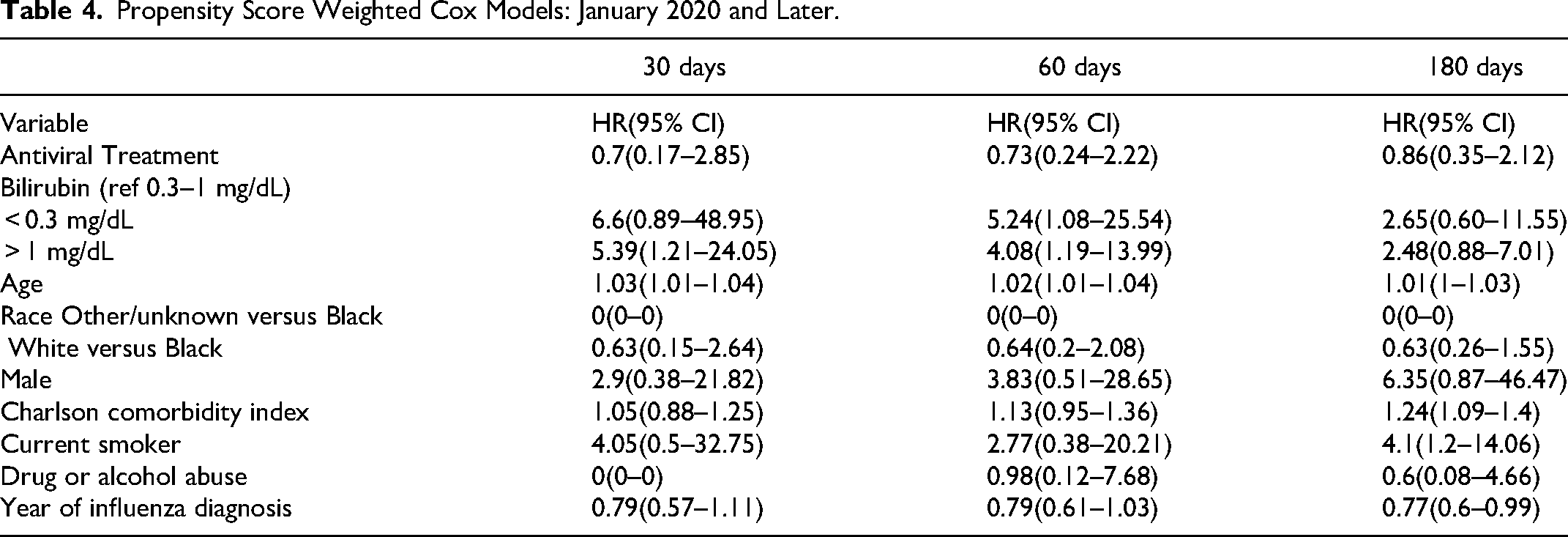
Table 4 presents the results of Propensity Score Weighted Cox models for the period January 2020 and later, evaluating the impact of bilirubin on risk of VTE across the study period (30 days, 60 days, and 180 days). Bilirubin levels lower than 0.3 mg/dL demonstrate a significantly increased risk of VTE compared to bilirubin levels of 0.3 to 1.0 mg/dl. Moreover, similar to the previous result, hazards ratios were highest at the 30-day time point and decline at subsequent time points. Interestingly, patients with bilirubin greater than 1 mg/dL also have a higher risk compared to those with 0.3–1 mg/dL at 30-days and hazard ratio magnitudes taper by 180 days.
Propensity Score Weighted Cox Models: January 2020 and Later.
Discussion
Influenza virus infection imposes a considerable public health burden, affecting healthcare systems and causing individual morbidity and mortality. Recent evidence suggests a potential connection between influenza and an elevated risk of venous thromboembolism (VTE), including DVT and pulmonary embolism (PE). Influenza virus infection is associated with a significant overproduction of ROS,9–12 contributing to viral pathogenesis. This increase in ROS contributes to influenza pathogenicity through multiple mechanisms, including activation of innate immunity and promotion of inflammation.10,11 Recent studies have demonstrated that the influenza viral matrix 1 protein exacerbates viral pathogenicity by inducing TLR4-mediated ROS production and apoptotic cell death. 10 Furthermore, mitochondrial ROS have been implicated in the pathological inflammation observed during influenza A virus infection in mice. 12
The elevated levels of ROS during influenza infection can promote a procoagulant state, which is conducive to thrombus formation. ROS has been shown to increase the expression of tissue factor (TF), a key initiator of the coagulation cascade.13,14 Additionally, ROS-mediated NADPH oxidase activation in vascular smooth muscle cells can lead to tissue factor-dependent procoagulant activity. 15
Beyond promoting a procoagulant state, ROS can also impair anticoagulant mechanisms. Oxidative stress has been demonstrated to inactivate activated protein C, an important anticoagulant protein. 19 Moreover, oxidation of thrombomodulin, another crucial anticoagulant factor, can inhibit its cofactor activity, potentially providing a rapid mechanism for modulating coagulation. 20
While the link between excessive ROS and procoagulant states during influenza infection is well-established, recent studies have also investigated endogenous factors, such as bilirubin, that could also have a role in VTE formation. Previous studies reported an inverse relationship between bilirubin levels and thromboembolic and cardiovascular events.1–3
Bilirubin is a metabolite of heme that scavenges ROS preventing oxidation. Bilirubin is oxidized to biliverdin by taking on oxygen free radicals and then subsequent conversion back to bilirubin through NADPH/biliverdin reductase.21–25 Bilirubin's ability to prevent oxidative stress is thought to be linked to its structure, with the ability of its exposed hydrogen molecule to combine with oxygen radicals.21,26,27 Moreover, bilirubin could act as a regulator of inflammation through its action on vascular cell adhesion molecule-1(VCAM-1). 28 Therefore, it is possible that, in the context of viral infections, patients with low bilirubin levels may be more at risk for complications such as VTE formation that are in part due to oxidative stress and inflammation.
Using a national cohort of patients with influenza and bilirubin lab results, we found that patients with total serum bilirubin levels below 0.3 mg/dL exhibited a significantly higher hazard of VTE following influenza infection compared to those with higher bilirubin levels. Moreover, we found that lower serum bilirubin levels were more significant predictors of VTE during follow-up periods closer to the influenza infection point suggesting that low bilirubin levels are particularly important in the context of acute viral or inflammatory illness.
Given the possibility of acute inflammation exacerbating VTE risk, we investigated if two medications, montelukast or metformin could alter the subsequent risk of VTE among our sample of influenza patients. Montelukast, a commonly used leukotriene receptor antagonist, has been previously identified as being protective against VTE 29 and preclinical evidence purports its benefit in preventing thrombus formation among broad cardiovascular benefits. 30 Prior work found metformin associated with lower mortality among obese patients with influenza. 31 Metformin has a growing body of research examining its anti-inflammatory effects,32,33 which could be beneficial in VTE prevention. For each medication patients were classified as either exposed or unexposed on their influenza infection date and Cox models were fit for each medication separately, estimating the hazard ratio (HR) at each follow up period. Neither montelukast nor metformin demonstrated any association with subsequent VTE among influenza patients (Supplementary Table S2).
Subgroup analysis of more recent influenza infections (year 2020+) revealed a consistent association between lower bilirubin levels and VTE risk. Different strains of influenza could result in different risks for thromboembolic events. For example, H1N1 influenza during the 2009 influenza season was particularly associated with thrombotic events. 6 The results of the current study, while not parsing out specific influenza virus variants, indicate that lower bilirubin levels remained an important predictor of VTE development among influenza patients in the post-2020 era.
Point estimates for bilirubin levels >1 mg/dL consistently pointed to an increased risk compared to 0.3–1 mg/dL levels, however in the main analysis the finding was not statistically significant. Interestingly in the post-2020 era, bilirubin levels greater than 1 mg/dL were found to increase VTE risk. This result, along with the consistent point estimates in the main analysis, suggests a possible U-shape risk profile with patients having low or high bilirubin levels conferring higher VTE risk. Our results suggesting a U-shape risk profile between bilirubin values and VTE is consistent with other results examining bilirubin and cardiovascular disease risk.34–38 While low bilirubin levels may allow an environment favorable to coagulation, excessively high bilirubin levels might be an indicator of liver damage, which can increase risk of VTE.39–43 Moreover, preclinical data suggest that increased levels of bilirubin could result in a switch from an antioxidant to a pro-oxidant effect. 44
Limitations of this study warrant consideration. Being a non-randomized study the potential for confounding exists. To minimize the confounding bias, we used propensity score weighting to make the cohorts as similar as possible. However, despite inclusion of important covariates, there may be other factors such as genetics, lifestyle factors and differences in the influenza strains not captured in the medical records that could impact the risk of VTE. Moreover, the incidence of other respiratory illnesses such as COVID-19 could impact the risk of VTE and this was not accounted for in our analysis.
The results of this study demonstrated a significant association between low total serum bilirubin levels and increased VTE risk in patients with influenza infection. Importantly, sub-analysis also suggests that increased total bilirubin levels may be associated with increased VTE risk. These results are consistent with previously observed U-shape risk profile found for bilirubin levels in cardiovascular disease. Further investigations to examine bilirubin and VTE risk among broader populations are warranted.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241275138 - Supplemental material for Serum Bilirubin Levels and Risk of Venous Thromboembolism among Influenza Patients: A Cohort Study
Supplemental material, sj-docx-1-cat-10.1177_10760296241275138 for Serum Bilirubin Levels and Risk of Venous Thromboembolism among Influenza Patients: A Cohort Study by S. Scott Sutton, PharmD, Joseph Magagnoli, PhD, Tammy Cummings, PhD, and James W. Hardin, PhD in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Additional Contributions
We thank Charles L. Bennett, MD, PhD, MPP, Professor, College of Pharmacy, University of South Carolina, for discussions and editing of the manuscript. Dr Bennett did not receive financial compensation for this contribution.
Author Disclosures
Sutton has received research grants from Boehringer Ingelheim, Gilead Sciences, Alexion Pharmaceuticals, and EMD Serono, all for projects unrelated to study. The other authors declare no competing interests. The content of this article is solely the responsibility of the authors and does not necessarily represent the official views of the US Department of Veterans Affairs, nor does mention of trade names, commercial products or organizations imply endorsement by the US government. This paper represents, in part, original research conducted using data from the Department of Veterans Affairs and is the result of work supported with resources and the use of facilities at the Dorn Research Institute, Columbia VA Health Care System, Columbia, South Carolina.
Data Availability
Analyses of the Veterans Health Administration Database were performed using data within the US Department of Veterans Affairs secure research environment, the VA Informatics and Computing Infrastructure (VINCI). The completeness, utility, accuracy, validity, and access methods are described on the VA website, ![]() .
.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Sutton, Magagnoli, and Cummings are supported by NIH grant R01DA054992 and the South Carolina Center for Rural and Primary Healthcare unrelated to this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
