Abstract
The prevalence of thrombosis in lymphoma patients is reportedly high and ranges from 3-10%. Vascular malfunction and inflammatory processes further contribute to the thrombotic activation process in these patients. Andexanet alfa (AA) is an antidote for factor Xa inhibitors and its usage has been reported with thrombotic complications. This study was designed to compare the effect of AA on the thrombin generation (TG) potential. Blood samples from 78 patients with confirmed diagnosis of non-Hodgkin lymphoma (NHL), Hodgkin lymphoma (HL) and Chronic lymphocytic leukemia (CLL) were collected from the University of Belgrade Clinic, Serbia. Normal human plasma (NHP) was used for referencing purposes. Individual samples were supplemented with AA at 100 ug/ml. TG studies were carried out using a commercially available fluorogenic substrate method. TG parameters such as peak thrombin (PT), lag time (LT) and area under the curve (AUC) were compiled. Cumulatively, lymphoma patients showed an increase in LT compared to NHP which decreases with AA. The PT and AUC levels were decreased compared to NHP and increases with AA. Upon sub-grouping of lymphoma patients, PT levels for all sub-groups were increased with AA. The AUC values increased for HL and NHL and decreased for CLL with AA. Variations in lag time were noted in all 3 sub-groups. Lymphoma represents a heterogenous group of patients where both the hypercoagulable state and inflammatory responses simultaneously occur. Increased thrombin generation in post AA supplemented samples suggest that the use of this agent may potentially be associated with thrombotic complications.
Keywords
Introduction
Lymphoma is a hematologic malignancy which approximates around 8.09% of all of the cancers in the US in the year 2020. Lymphoma originates from the lymphatic hematopoietic system, includes non-Hodgkin’s lymphoma (NHL, 4.274%) and Hodgkin’s lymphoma (HL, 0.469%.). 1 Chronic Lymphocytic leukemia/lymphoma (CLL, 3.349%) and originates from lymphoblastic cells in the bone marrow. 1 The thromboembolic event; venous thromboembolism (VTE) is one of the complications and second major cause of mortality in this malignancy. 2,3 The prevalence of thrombotic complications in lymphoma patients ranges from 1.5 to 59.5% in comparison to other cancers.
The pathogenesis of thromboembolism (TE) in lymphoma is complex and multifaceted depending on; a) inflammation, b) hemostatic dysregulation, c) fibrinolytic deficit, d) cellular and e) vascular dysfunctions. 2,4 Several other risk factors like chemotherapy. immobilization and anticoagulation contribute in developing VTE. It has been reported that immobility leads to the stasis of blood which increase the chance of VTE. 5 Use of chemotherapy is also reported to cause the higher rate of VTE in cancer patients compared to non-cancer patients. 6 –8 Oncology patients have a 4 to 7-fold increased risk of thrombosis compared with the general population and 15% of patients with cancer suffer a VTE episode. 8 –13 However, approximately 20% of patients presenting with VTE have an active cancer. 9,10,13 The survival rate of cancer patients with VTE is around 2.2-3.7% which is much lower in comparison to cancer patients without VTE. 14 –17
There are several studies, which have shown the risk of VTE in lymphoma patients. According to a study on cancer associated thrombosis (CAT), hematological malignancies showed comparable risk of VTE to solid malignancies, with an incidence of 35 per 1000 patient years. 18 A meta-analysis of 29 independent cohorts including 18,018 patients and 1,149 events reported the incidence rate (IR) of thrombosis observed in cases of NHL (15 cohorts; 997 events in 15,410 patients) was 6.5% (95% CI, 6.1%-6.9%), significantly greater (P < 0.001) than that observed for HL patients (7 cohorts; 2505 patients; 118 events; IR 4.7%; 95% CI, 3.9%-5.6%). 19 The apparent differences in the 2 meta-analysis may be possibly due to the methologic approach used and the population demographics. Another study in ambulatory patients showed the rate of VTE is 8.6% in HL and 1.5% in NHL. 20 Additionally, VTE treated cancer patients are at high risk of recurrence of TE as well as hemorrhagic complications. 21 Therefore, anticoagulant therapy is used for prophylactic or therapeutic purposes. Standard anticoagulant therapy for CAT includes unfractionated heparin (UFH), low molecular weight heparin (LMWH, tinzaparin), heparinoids (danaparoid), vitamin K antagonist (VKA’s), direct thrombin inhibitor (dabigatran) and factor-Xa inhibitors (apixaban, betrixaban, rivaroxaban and edoxaban). 22 –27 Use of anticoagulant also represents unique challenges and increases the risk of hemorrhage and recurrence of VTE. 21,28,29 Since these patients have a tendency to bleed, the use of anticoagulants should be considered with caution.
Andexanet alfa (AA) is an antidote for factor Xa inhibitors and its usage has been associated with thrombotic complications. 30 –33 While andexanet alfa is designed to act as a decoy to factor Xa inhibitors, it has multiple interactions with endogenous plasma proteins such as tissue factor pathway inhibitor (TFPI). 34,35 Moreover, because of this structural similarity to factor Xa, it may augment the effects of other serine proteases in the coagulation cascade presumably, by competing with the active sites of this enzyme. Lymphoma patients represent heterogenous group where the activation of hemostatic processes is multifactorial. Tissue factor and microparticles are generated at varying levels. Andexanet alfa may modulate these processes leading to an augmentation of thrombin generation. This study was designed to compare the effect of andexanet alfa on thrombin generation potential in healthy normals and patients diagnosed with various types of lymphoma.
Materials & Methods
Sample Collection
Citrated blood samples from 78 patients with histologically confirmed diagnosis of non-Hodgkin lymphoma (NHL), Hodgkin lymphoma (HL) and Chronic lymphocytic leukemia/Small lymphocytic lymphoma (CLL/SLL) were collected under the IRB approved protocol from the Clinic of Hematology Unit, University of Belgrade, Belgrade, Serbia. Normal human plasma (NHP) comprised of 25 male and 25 female samples were obtained from a commercial source George King Biomedical (Overland park, KS, US.) Both plasma cohorts from the control and patients’ groups were collected in identical anticoagulants tubes containing 3.8% sodium citrate in a 1:10 volume ratio and were processed in similar settings. Normal human pooled plasma was used for referencing purposes.
Thrombin Generation Studies
Thrombin generation studies were carried out by using a Fluoroskan Ascent Fluorimeter, Calibrated Automated Thrombogram from Diagnostica Stago (Parsippany, NJ, US). Reagents used in this assay included the fluo-substrate, fluo-buffer, tissue factor high reagent (mixture of tissue factor and phospholipids) and a thrombin calibrator. The thrombin generation assay was carried out in 96-well Immulon 2HB transparent round bottom plates. Individual plasma samples were either supplemented with saline or andexanet alfa at a final concentration of 100 µg/ml. The thrombin generation potential was measured in terms of the peak thrombin concentration, lag time and endogenous thrombin potential (ETP) / area under the curve (AUC). % peak thrombin and % endogenous thrombin potential were calculated for samples supplemented with andexanet alfa.
Statistical Analysis
Statistical analysis was carried out by using Prism, GraphPad. Paired t-test was used to determine the statistical differences between pre and post andexanet alfa in various groups of lymphomas. P value and correlation coefficient (r) was noted.
Results
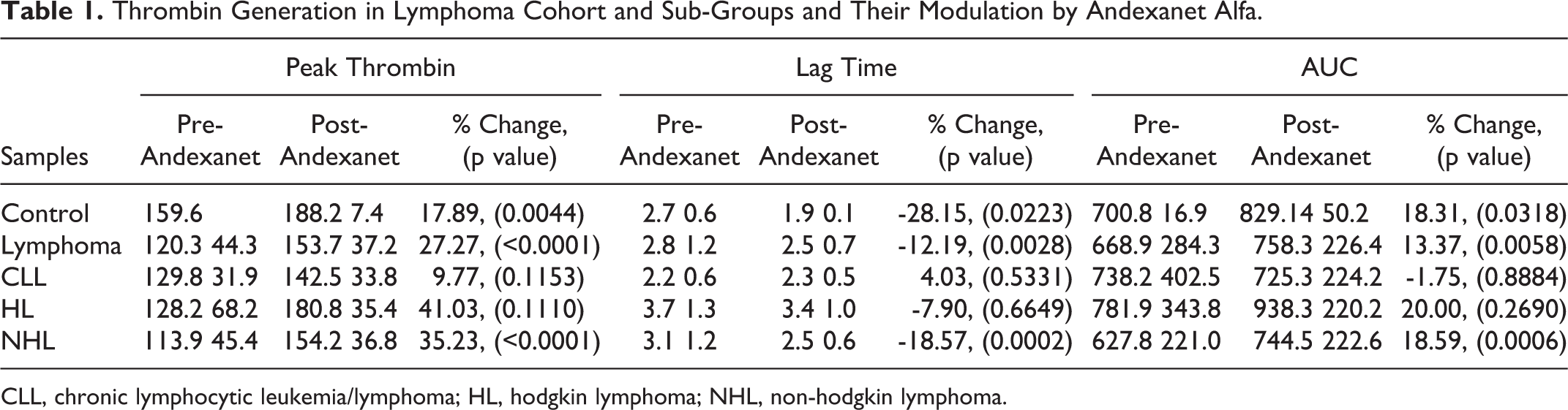
On a cumulative basis, lymphoma patients showed increase in lag time (2.87 ± 1.2) in comparison to the normal human plasma (2.06 ± 0.2). The peak thrombin levels were decreased (120.34 ± 44.3) in the lymphoma patients in comparison to normal human plasma (159.60 ± 31.5). The AUC was decreased (668.85 ± 284.3) in the lymphoma patients in comparison to normal human plasma (700.80 ± 16.9). When the lymphoma patients were sub-grouped, peak thrombin levels were similar in HL (128.16 nM), NHL (113.99 nM) and CLL (129.77 nM) and were decreased compared to normal human plasma (159.20 nM). However, the AUC was increased in the HL (781.91 nM*min), decreased in NHL (627.81 nM*min) and was comparable for CLL (738.18 nM*min) compared to normal human plasma (700.80 nM). Variations in lag time were noted in all 3 groups.
As shown on Table 1, when the lymphoma samples were supplemented with andexanet alfa, peak thrombin and AUC parameters increased, whereas the lag time decreased. This is also clear from the thrombokinetograms which are depicted in Figure 1. There was a wide scatter in the individual results obtained in the lymphoma cohort.
Thrombin Generation in Lymphoma Cohort and Sub-Groups and Their Modulation by Andexanet Alfa.
CLL, chronic lymphocytic leukemia/lymphoma; HL, hodgkin lymphoma; NHL, non-hodgkin lymphoma.

Thrombin generation in lymphoma cohort and sub-groups and their modulation by andexanet alfa. A. Thrombokinetogram for lymphoma samples B. Thrombokinetogram for chronic lymphocytic leukemia/lymphoma (CLL) samples, C. Thrombokinetogram for hodgkin lymphoma (HL) samples, D. Thrombokinetogram for non-hodgkin lymphoma (NHL) samples.
The peak thrombin levels were increased with andexanet alfa (153.70±37.2 nM) compared to controls (120.34 ± 44.3 nM). When the lymphoma patients were sub-grouped, peak thrombin levels were similar in HL (128.16 nM), NHL (113.99 nM) and CLL (129.77 nM) were decreased compared to normal human plasma (159.20 nM) which increases with andexanet alfa, in HL (180.75 nM), NHL (154.16 nM) and CLL (142.46 nM) compared to their respective controls. Figure 2 shows the results in terms of scattered plot for the cumulative and individual lymphoma groups. The average of all of the lymphoma groups and NHL group showed significant differences between pre and post andexanet alfa with p value < 0.0001 and the correlation coefficient (r) value is 0.4886 for average of all of the lymphomas and 0.5422 for NHL. Differences between pre and post andexanet alfa is not significant in HL and CLL groups.
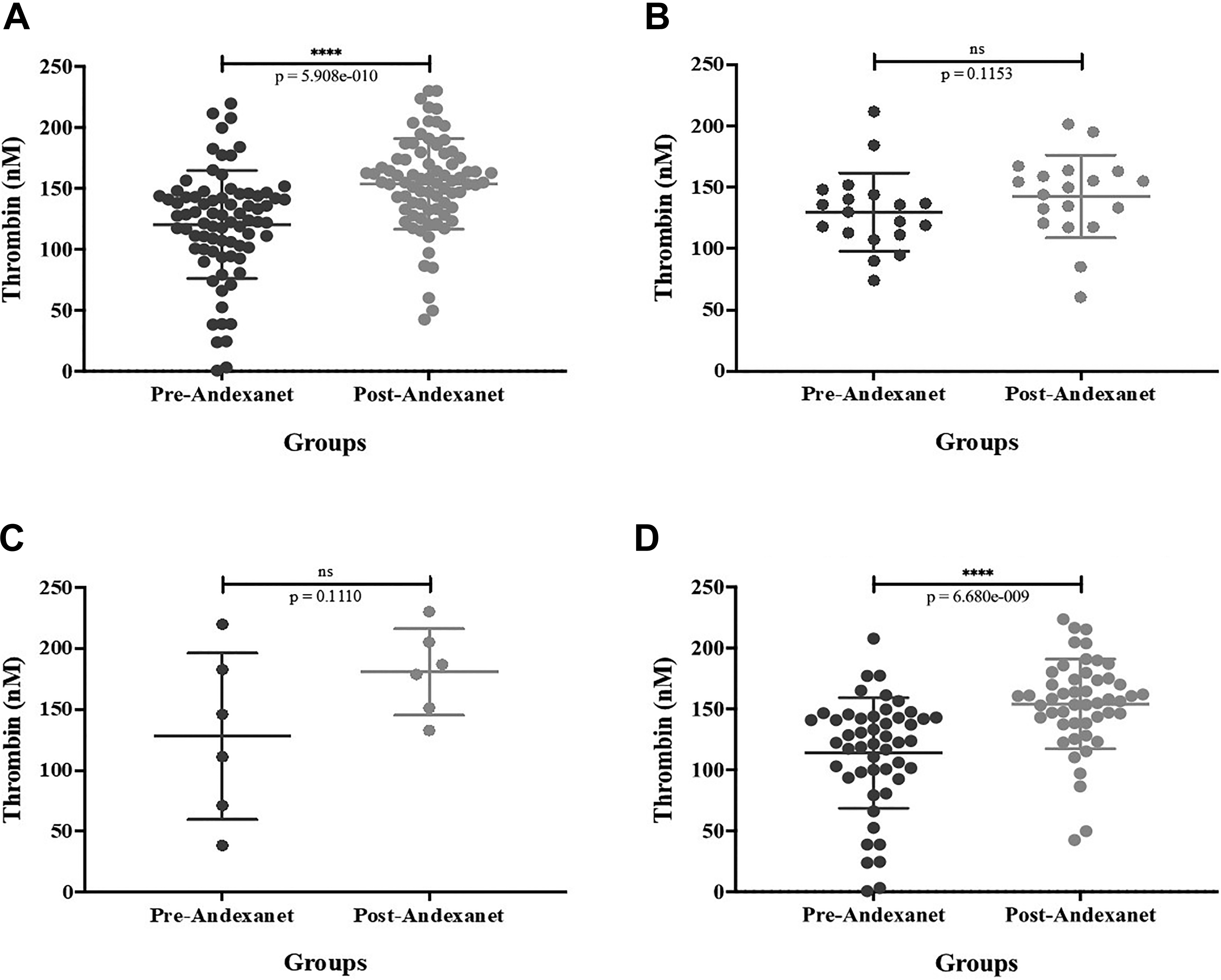
Peak thrombin levels in lymphoma cohort and subgroups and their modulation by andexanet alfa. A. Effect of andexanet alfa on lymphoma samples, B. Effect of andexanet alfa on chronic lymphocytic leukemia/lymphoma (CLL) samples, C. Effect of andexanet alfa on hodgkin lymphoma (HL) samples, D. Effect of andexanet alfa on non-hodgkin lymphoma (NHL) samples.
As depicted on Figure 3, on a cumulative basis, lymphoma patients showed decrease in lag time with andexanet alfa (1.94 ± 0.1 mins) in comparison to control samples (2.9 ± 1.15 mins). Upon sub-grouping, variations in lag time were noted in all of the 3 groups.

Onset of thrombin generation as measured by lag time in lymphoma cohort and subgroups and their modulation by andexanet alfa. A. Effect of andexanet alfa on lymphoma samples, B. Effect of andexanet alfa on chronic lymphocytic leukemia/lymphoma (CLL) samples, C. Effect of andexanet alfa on hodgkin lymphoma (HL) samples, D. Effect of andexanet alfa on non-hodgkin lymphoma (NHL) samples.
AUC (total thrombin generated) was also increased with andexanet alfa (758.32 ± 226.4 nM*min) compared to controls (668.85 ± 284.3 nM*min). When the lymphoma patients were sub-grouped, the AUC was increased in the HL (781.91 nM*min), decreased in NHL (627.81 nM*min) and was comparable for CLL (728.18 nM*min) compared to normal human plasma (756.46 nM*min). Variations were noted with andexanet alfa supplemented samples. However, with andexanet alfa the AUC values increased for HL (938.31 nM*min) and NHL (744.52 nM*min) and slightly decreased with CLL (725.26 nM*min) in comparison to their respective controls. Figure 4 shows the scattered plots for the cumulative and individual lymphoma groups. Similar to peak thrombin, average of all lymphoma groups and NHL group showed significant differences with p value < 0.0001 and the correlation coefficient (r) value is 0.4241 for cumulative lymphomas and 0.5027 for NHL group. HL and CLL groups did not show significant results.

Cumulative thrombin generation as measured by the area under the curve (AUC) in lymphoma cohort and subgroups and their modulation by andexanet alfa. A. Effect of andexanet alfa on lymphoma samples, B. Effect of andexanet alfa on chronic lymphocytic leukemia/lymphoma (CLL) samples, C. Effect of andexanet alfa on hodgkin lymphoma (HL) samples, D. Effect of andexanet alfa on non-hodgkin lymphoma (NHL) samples.
Discussion
The direct oral anticoagulant drugs such as the anti-Xa agents have added a new dimension for the management of thrombotic and cardiovascular disorders. The initial development of these drugs focused on the management of non-valvular atrial fibrillation and subsequently for the prophylaxis and treatment of venous thromboembolism. Bleeding complications associated with their use were difficult to manage due to the non-availability of an antidote. Andexanet alfa provided a novel molecular approach to neutralize the anti-Xa effects of rivaroxaban and apixaban. However, andexanet alfa can also neutralize other direct anti-Xa agents such as betrixaban and edoxaban. 36
Andexanet Alfa (AA, coagulation FXa recombinant, inactivated Zh-Zo; Portola Pharmaceuticals, South San Francisco, CA) is a recombinant modified version of human activated FXa, which is designed to reverse the inhibition of FXa bleeding associated with newer oral anti-Xa agent. 30, 31 In May 2018, the FDA approved andexanet alfa as an antidote for factor Xa inhibitors such as apixaban and rivaroxaban. 32 Andexanet alfa act as a decoy protein which lacks enzymatic activity, in this recombinant protein the serine active site is replaced by alanine and the gamma-carboxyglutamic acid (GLA) domain is removed to restrict its assemblage into prothrombinase complex. 34
Since the availability of andexanet alfa, its use has been reported to be associated with thrombotic complications. A full study report on andexanet alfa associated bleeding with FXa inhibitors, reported that 49 patients (14%) out of 352 patients died within 30 days. In this cohort of 352 patients, 34 patients (10%) had thrombotic events. Of these, 11 patients encountered thrombotic event within 5 days, 11 had an event within 6 to 14 days and 12 had an event between 13 and 30 days after andexanet alfa therapy. Ischemic stroke, myocardial infarction (MI), deep vein thrombosis (DVT) and pulmonary embolism (PE) were the main complications. 33
Although the mechanisms involved in the observed thrombotic complications are complex and multifactorial, andexanet alfa usage has been associated with the modulation of regulatory hemostatic mechanisms resulting in hypercoagulable state in both clinical and experimental studies. It has been reported that andexanet alfa reduces the TFPI activity, 35 which is a major inhibitor of FXa and tissue factor VIIa complex and its reduction may leads to increase in thrombogenic environment. 32,37 Another study reported that andexanet alfa also reverses the anticoagulant effects of low molecular weight heparin (LMWH) and fondaparinux by competing with FXa for binding with antithrombin. 38,39 Increased thrombin generation in antithrombin-deficient patients has also been reported. 40 Another possibility is that andexanet alfa treatment may also inhibit the endogenous heparan sulfate glycosaminoglycan (GAGs) complexes with endothelium bound antithrombin. 41 Taken together, andexanet alfa produces additional effects on endogenous modulators which regulates hemostasis including TFPI, antithrombin, GAGs and may also influence other heparin like mediators. Thus, beside neutralizing the anti Xa agents, andexanet alfa may promote a procoagulant environment by disrupting hemostasis at multiple sites. 42 The role of increased TG in promoting thrombotic complications in andexanet alfa administered to patients needs to be further investigated. Concerns regarding andexanet alfas potential prothrombotic effects were already stated in previous studies, including an increase of D-dimers induced by andexanet alfa. 30
In the previous study the augmentation of thrombin generation associated with andexanet has been reported in plasma-based systems. 36 Thrombotic complications have been reported in both solid tumors and hematologic malignancies. 18 Thrombin generation plays an important role in mediating its responses in cancer patients. Patients with different types of lymphoma exhibit 7-12% prevalence of VTE. The current studies were designed to investigate the thrombin generation potential in the cohort of 78 patients with various sub-types. The effect of andexanet alfa at therapeutic levels on the endogenous thrombin generation potential in these patients was investigated.
Thrombin generation studies in cancer patients have provided predictive outcomes in terms of its relationship with VTE. 43 Tumor cells can promote thrombin generation by producing tissue factor, expressing the coagulation factor X activator known as cancer procoagulant, and by displaying surface sialic acid residues that can support non-enzymatic factor X activation. 44,45 In the current studies, the lymphoma patient group as a whole showed a lower thrombin generation in comparison to the normal human pool plasma. This is suggestive of prothrombin and other coagulation factors due to ongoing activation of coagulation process. The relative amount of thrombin generation in various groups of lymphomas varied and followed the ranked order CLL >HL >NHL. This suggest that various lymphoma groups have differential potential of thrombin generation. All of these groups exhibited lower than normal human plasma values. Because of the wide variations in the data and small number of samples in these subgroups, the level of significance was not achieved, despite the differences in the cumulative results.
Supplementation of andexanet at therapeutic level of 100 ug/ml resulted in the augmentation of thrombin generation in comparison to the control group. In the NHP and lymphoma sub-groups, supplementation resulted in an increase in thrombin generation. The increased thrombin generation in the lymphoma sub-groups upon andexanet alfa supplementation was relatively higher in comparison to the saline control group. Thrombin generation values ranged in the order of HL >NHL > CLL. These results are consistent to the clinical observations where both HL and NHL exhibited higher prevalence of VTE. 19 This suggest that andexanet alfa is capable of producing differential activation of thrombin generation process in various lymphoma sub-types. This may be due to the compositional differences in the coagulation factors in these sub-groups. Although, upon the supplementation of andexanet alfa to the lymphoma patients’ sample, there was a cumulative increase of 27% in the whole cohort. Hodgkin and non-Hodgkin lymphoma showed relatively higher % change in peak thrombin. While there were trends toward increased peak thrombin both the pre and post andexanet samples showed wide variations with overlapping values, for this reason the p values were not meaningful.
Thrombotic complications in leukemias and lymphomas have been reported. 46,47 The relative risk of thrombosis in various types of leukemia depends on the types of leukemia. Similarly, the prevalence of thrombotic processes varies in lymphomas sub-groups.
The fact that thrombin generation values such as peak thrombin are lower in various types of lymphoma groups. However the reported incidence of VTE is higher which is suggestive of an ongoing activation of coagulation process due to the release of procoagulant mediators from blood cells, simultaneous measurement of thrombin generations markers including D-Dimer values has been reported in lymphoma group. 48 The procoagulant mediators from various cells release at different levels. Malignancy-associated inflammation can result in increased concentrations of acute phase proteins such as factor VIII, fibrinogen, and von-Willebrand factor that are associated with an increased risk of thrombosis. 49,50 Andexanet alfa most likely augments the effects of these mediators which may include tissue factor (TF) and factor Xa. Since these studies were carried out on cell free plasma, similar studies in whole blood may provide additional information on cellular contribution to the thrombogenic process. It is likely that in the whole blood environment, andexanet alfa may produce even stronger amplification of thrombin generation. In these studies, measurement of thrombin generation markers such at thrombin anti-thrombin complexes (TAT) and prothrombin fragment F1.2 may be useful.
Both the activation of inflammation and coagulation processes can simultaneously occur in lymphoma patients. 51 As discussed before, chemotherapy can also contribute to the thrombogenic process. 6 –8 The currents study was only carried out on 78 patients in 3 lymphoma sub-types. A larger study may provide more reliable data on the relevance of the sub-types of lymphomas and thrombin generation. These studies were only carried out at a fixed concentration of andexanet alfa. It will be of interest to determine the effect of different concertation of andexanet alfa on the relative generation of thrombin in various lymphoma sub-groups. Moreover, it would be of interest to study the effect of andexanet alfa on patients treated with factor Xa inhibitors in particular those with hematologic malignancies such as lymphoma and leukemia to determine the modulatory effects of andexanet alfa on thrombin generation. Despite these limitations, these studies clearly demonstrate that in lymphoma, there is an ongoing generation of thrombin which is related to the consumption of prothrombin, which is proportional to the lower peak thrombin values. Andexanet alfa augmentation of thrombin generation in lymphoma patients is clearly suggestive of the potential risk of thrombin mediated thrombotic complications in these patients. The extent to which the amplification of thrombin generation is observed in various lymphoma groups may be proportional to the risk of VE in these patients. Thus, patient’s predisposition and type of lymphoma along with chemotherapy may contribute to the overall thrombogenesis observed in these patients. These studies further validate the hypothesis that the endogenous thrombin generation potential in the hematologic malignancies such as lymphoma is amplified by andexanet alfa contributing to the reported thrombotic complications.
One of the limitations of the study is the size of the cohort with different lymphoma sub-groups with individual patient predisposition including therapeutic interventions. A larger study on a bigger cohort, may provide statistically valid data to further support our observations. The plasma samples were obtained from lymphoma patients, where the blood profile is variable. These patients may have varying plasma to cell ratios in comparison to the control group, where it is relatively constant, which may contribute to some differences in the thrombin generation profile. However, since each plasma sample tested was used to demonstrate augmentation of thrombin generation with identical composition, the observed increase in thrombin generation is valid. It may be useful to have comparative data on the complete blood profile and the effect of such variables as red blood cell (RBC) count and hematocrit on thrombin generation. The blood collection tubes for both control groups and lymphoma patients contained the same amount of sodium citrate and the processing of plasma was identical. In future studies, the thrombin generation values can be normalized by taken into account the plasma to cell ratios. Lastly, some of the chemotherapeutic approaches may also contribute to the alterations in the hemostatic profile of these patients, which may contribute to the observed variations. It may be useful to discuss the role of the chemotherapeutic approaches and their impact on hemostatic parameters such as thrombin generation. Despite these considerations, our studies have clearly demonstrated that due to an ongoing consumption of prothrombin and other coagulation factors, the thrombin generation is reduced in lymphoma patients upon activation with the reagent used in this assay. Moreover, in both the control and lymphoma patients, andexanet alfa produces augmentation of thrombin generation which may contribute to the procoagulant state observed in the lymphoma patients.
Footnotes
Acknowledgments
The authors gratefully acknowledge the skillful assistance of the staff of the Hemostasis and Thrombosis Research Unit, Loyola University Health Science Division. A special thanks to the nursing and laboratory staff of the lymphoma center, University of Belgrade, Belgrade, Serbia. We gratefully acknowledge Dr. Paul Riley of Diagnostica Stago (Paris, France) for providing us the instrument and reagents for the thrombin generation studies. We are also thankful to Ms. Erin Healy-Erickson for her expert assistant in preparing this manuscript. We also acknowledge the support of Dr. Eva Wojcik, Chairperson, Department of Pathology and Laboratory Medicine and Dr. Seth Robia, Co-director of the Cardiovascular Institute for facilitating these studies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
