Abstract
Background
Since several studies have examined the use of direct oral anticoagulants (DOACs) in treating patients with splanchnic vein thrombosis (SVT), we conducted a meta-analyses to assess the safety and efficacy of DOACs compared to vitamin K antagonists (VKAs) in this population.
Methods
We conducted a comprehensive search using the PubMed, Embase, and Cochrane Library databases until June 2024. We used odds ratios (ORs) and 95% confidence intervals (CIs) as the effect measures to compare DOACs with VKAs.
Results
A total of 9 observational studies were included. The pooled analysis revealed that a trend towards higher complete recanalization rates with DOACs (71.4%) compared to VKAs (55.3%), though not statistically significant (OR 1.95; 95%CI 0.70 to 5.44). For SVT extension, a significant effect was observed favoring DOACs (OR 0.12; 95%CI 0.03 to 0.54). No significant differences were found in other efficacy outcomes or safety outcomes, except for major bleeding, which was significantly lower with DOACs (OR 0.27; 95%CI 0.13 to 0.56).
Conclusion
DOACs are superior to VKAs in SVT extension and major bleeding, suggesting that DOACs may be a favorable treatment option in the treatment of SVT.
Introduction
Splanchnic vein thrombosis (SVT) is a type of blood clotting disorder that occurs in the splanchnic venous system, which includes the portal vein, mesenteric vein, and splenic vein.1,2 In addition, SVT within the hepatic veins, a condition referred to as Budd-Chiari syndrome, is recognized as the rarest variant of SVT. In the past, splanchnic thrombosis was considered rare, but recent studies have shown an increase in its occurrence, likely due to advancements in diagnostic methods. Several studies have found that SVT is commonly associated with various underlying conditions such as liver cirrhosis, abdominal infections, malignancies, abdominal surgery, thrombophilia, and myeloproliferative disorders. However, there is still limited evidence available to guide treatment recommendations for SVT patients.
Currently, the treatment of SVT involves the use of different anticoagulant medications depending on the specific clinical context.3,4 These may include vitamin K antagonists (VKAs), low-molecular-weight heparins (LMWHs), or direct oral anticoagulants (DOACs). However, traditional VKAs such as warfarin, have several drawbacks (eg, complex dose adjustments, drug-drug interactions, and drug-food interactions) when compared to DOACs. Furthermore, studies have shown that DOACs provide greater net clinical benefits compared to VKAs, such as reduced risk of stroke, lower rates of major bleeding, and improved patient adherence. These advantages have contributed to the widespread recommendation and adoption of DOACs as the preferred anticoagulant treatment option in preventing thromboembolic events among patients with atrial fibrillation or treating venous thromboembolism (VTE).5–7 However, the optimal anticoagulant modality for SVT is still not well established, primarily due to the different underlying causes of the condition. Due to the emergence of numerous studies examining the use of DOACs in treating patients with SVT, we conducted a systematic reviews and meta-analysis to evaluate the safety and efficacy of these DOACs compared to VKAs in the treatment of SVT patients.
Methods
The findings of our meta-analysis were presented according to the the PRISMA checklist (Supplemental Table 1).
Study Search
We conducted a comprehensive search using the PubMed, Embase, and Cochrane Library (CENTRAL) databases. Our search was limited to articles published until June 2024. We specifically looked for studies that provided data on the effectiveness and safety of DOACs compared to VKAs in patients with SVT. To ensure the search included relevant studies, we used specific MeSH terms and keywords (Supplemental Table 2). Furthermore, we manually reviewed the reference lists of previous review articles to identify any additional studies that might have been missed through the database search. The search was not limited to any specific language.
Eligibility Criteria
In accordance with the PICOS (Population, Intervention, Comparison, Outcomes, Study design) framework, our eligibility criteria for the meta-analysis were as follows: 1)Population: we focused on patients diagnosed with SVT, encompassing those with various underlying conditions that contribute to this thrombotic disorder. 2)Intervention: Our intervention of interest was the use of DOACs, specifically dabigatran, rivaroxaban, apixaban, and edoxaban. 3)Comparison: The comparison in this study was between the use of DOACs and VKAs. 4)Outcomes: The efficacy outcomes included complete or partial recanalization of SVT, SVT extension, recurrent thrombotic events, and all-cause mortality, while the safety outcomes included major bleeding, 8 any bleeding, intracranial bleeding, and gastrointestinal bleeding. 5)Study design: randomized controlled trial or observational studies.
Studies that met at least one of the following exclusion criteria were excluded: 1) Guidelines, expert position, or consensus; brief reports, case reports, letters, comments, or protocol studies; review and meta-analysis articles; 2) Studies that did not provide any data on the efficacy or safety outcomes of interest; 3) Duplicate data; 4) Single-arm studies that solely focused on DOACs, as these studies would not provide a comparative analysis between DOACs and VKAs; and 5) Studies not published in a peer-reviewed format.
Study Selection
Once the search was completed, two independent reviewers went through the titles and abstracts of the retrieved studies. In line with our predefined criteria for exclusion, we removed studies that were animal experiments, case reports, reviews, meta-analyses, comments, conference abstracts, or any other irrelevant articles. After this initial screening, we proceeded with the full-text screening process to select studies that met our eligibility criteria. Any disagreements between the two reviewers were resolved through discussion or consultation with the senior author.
For the studies included in this meta-analysis, we extracted several baseline characteristics. This information consisted of the authors’ names and publication years, details about the study population (including sample size, age, and sex), study design, data source, enrollment period, type or etiology of SVT, methods used for SVT assessment, the type of DOAC and VKA used, cirrhosis rate, the outcomes assessed, and the duration of follow-up.
Study Quality Assessment
To assess the quality of observational studies included in our analysis, we utilized the Newcastle-Ottawa Scale (NOS) tool. 9 This tool encompasses three domains: the selection of cohorts, comparability of cohorts, and the assessment of the outcome. Each domain can score a maximum of 4, 2, and 3 points, respectively. In this study, we considered scores of 6 or higher as indicative of moderate to high-quality studies, while scores below 6 indicated studies of lower quality.10–13
Statistical Analysis
We assessed the consistency of the included studies using the Cochrane Q test and I² statistic. A P value of <0.1 and an I² value of >50% indicated substantial heterogeneity. We collected data on the number of events and sample size from each included study. The choice of the model was based on the assessment of heterogeneity using the I² statistic, and a random-effects model was used when I² > 50% in the pooled analysis. We used odds ratios (OR) and 95% confidence intervals (CIs) as the effect measures to compare DOACs with VKAs. We employed a leave-one-out approach for conducting the sensitivity analysis. To evaluate potential publication bias, the study employed funnel plots for visual inspection of asymmetry. For the statistical analyses, the study followed the guidelines outlined in the Cochrane Handbook for Systematic Reviews of Interventions version 6.2. The data were analyzed using the Review Manager version 5.4 software (the Cochrane Collaboration 2014, Nordic Cochrane Centre Copenhagen, Denmark).
Results
Study Selection
Figure 1 shows a visual representation of the step-by-step process undertaken to retrieve relevant literature for this meta-analysis. To begin with, the primary sources for the search were the electronic databases of PubMed, Embase, and Cochrane Library. After conducting a comprehensive search, a total of 346 records were initially identified as potentially relevant to the meta-analysis. We excluded 52 duplicated studies. In the first phase, title and abstract reviews were conducted for 270 records. This meticulous examination resulted in the exclusion of several studies that did not align with the research objectives. In the second phase, the full texts of the 24 remaining studies were carefully examined to determine their suitability for inclusion according to the predetermined criteria. As a result, 15 studies14–28 was excluded from the meta-analysis because they did not meet the established criteria. Finally, our current meta-analysis included a total of 9 observational studies.29–37
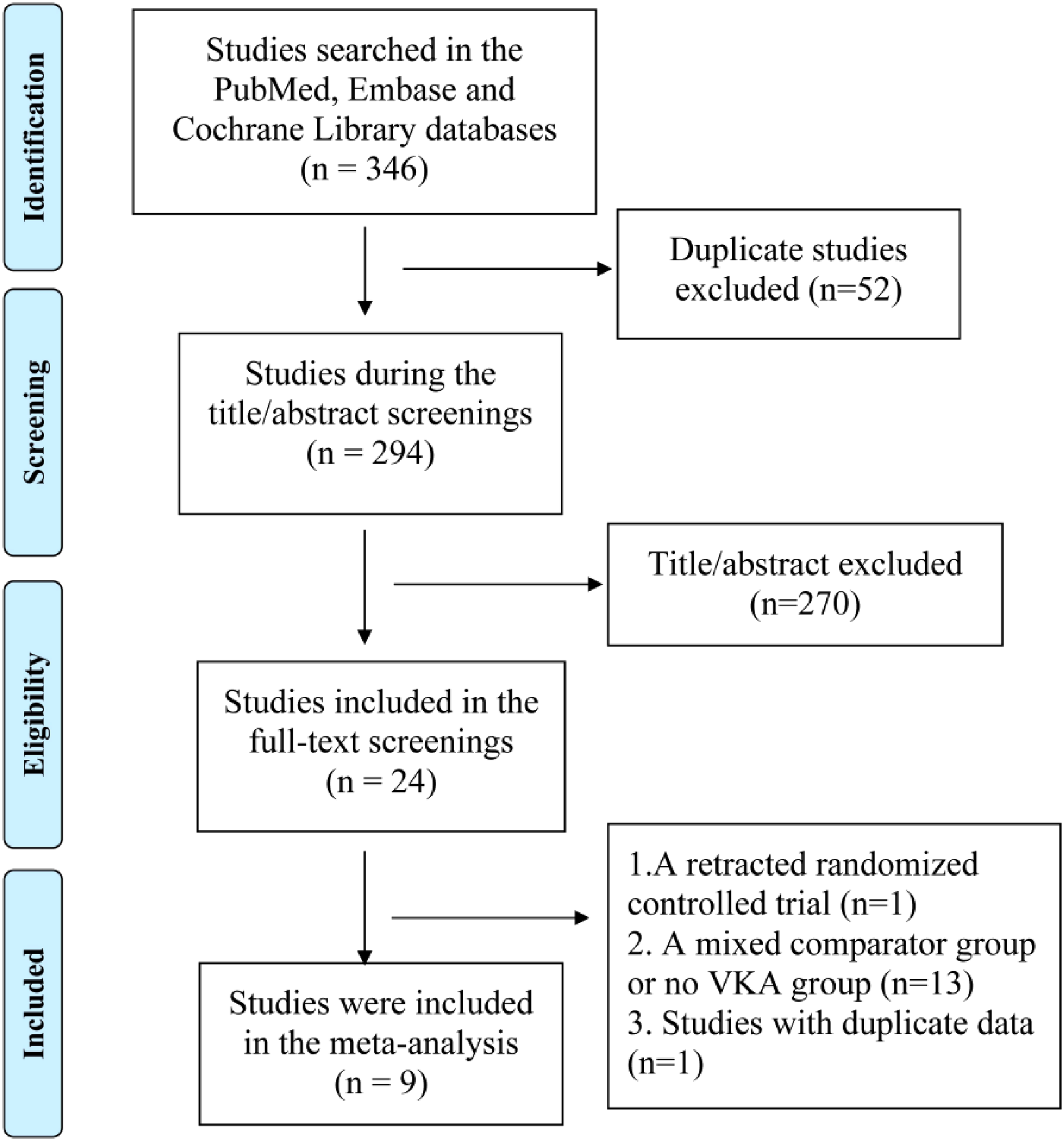
A visual representation of the step-by-step process undertaken to retrieve relevant literature for this meta-analysis.
In Table 1, we presented the baseline characteristics of these studies that were included in our analysis. We found that SVT predominantly occurred in the portal vein. We included both cirrhosis and non-cirrhosis patients with SVT in our review. Among the nine studies included, four focused on patients with liver cirrhosis, one on non-cirrhotic patients, and four on a mixed population comprising both liver cirrhosis and non-cirrhotic patients. The quality of observational studies was assessed using the NOS tool, 9 and all of the studies a NOS score of six or above (Supplemental Table 3). The definitions of efficacy and safety outcomes from the included studies are presented in Supplemental Tables 4 and 5.
Baseline Characteristics of the Included Studies in This Review.
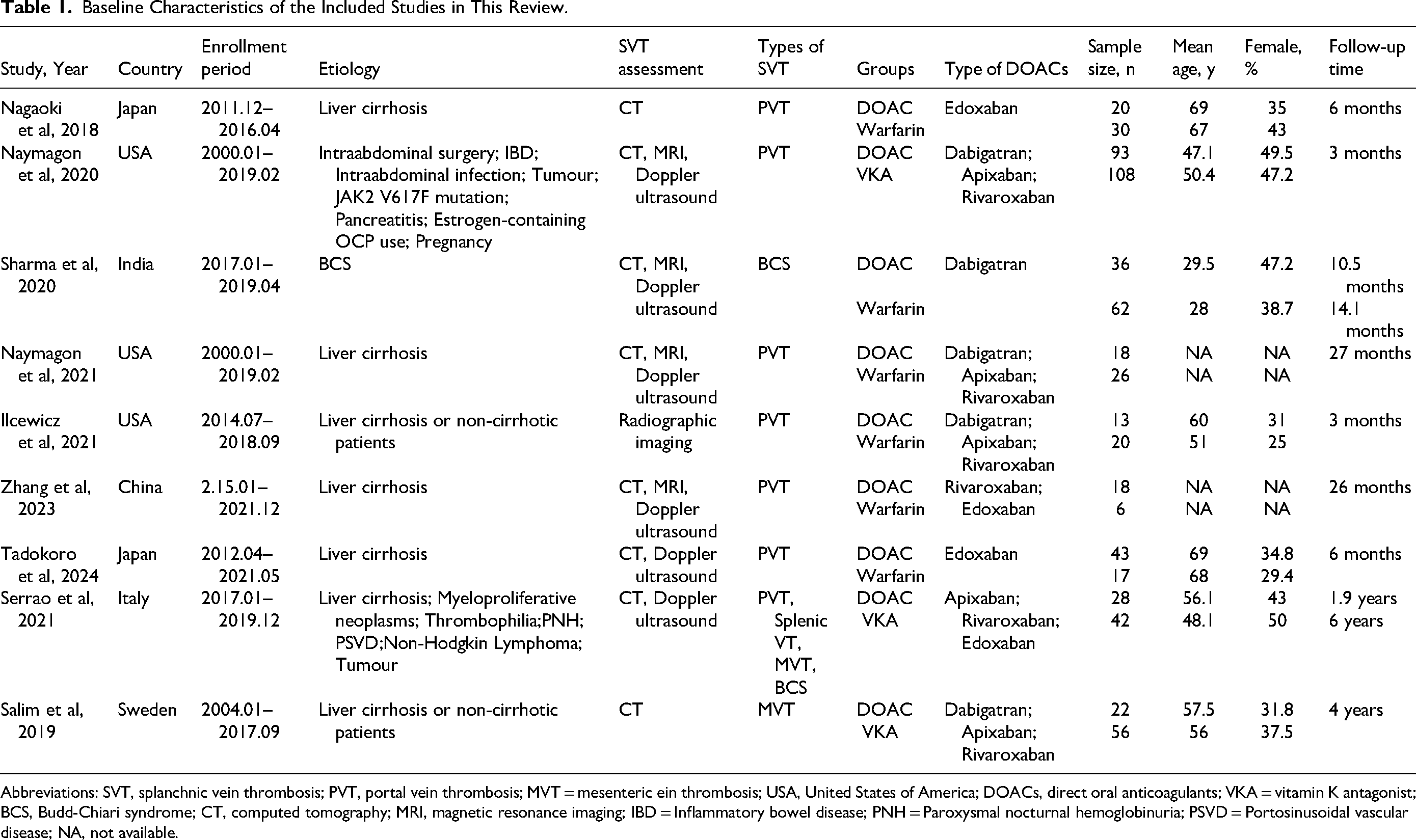
Abbreviations: SVT, splanchnic vein thrombosis; PVT, portal vein thrombosis; MVT = mesenteric ein thrombosis; USA, United States of America; DOACs, direct oral anticoagulants; VKA = vitamin K antagonist; BCS, Budd-Chiari syndrome; CT, computed tomography; MRI, magnetic resonance imaging; IBD = Inflammatory bowel disease; PNH = Paroxysmal nocturnal hemoglobinuria; PSVD = Portosinusoidal vascular disease; NA, not available.
Efficacy Outcomes Between DOACs Versus VKAs in Patients with SVT
The pooled analysis showed that 71.4% of patients treated with DOACs achieved complete recanalization of affected blood vessels, whereas 55.3% of those treated with VKAs achieved the same outcome, although not statistically significant between the two treatment groups (OR 1.95; 95%CI 0.70 to 5.44; P = 0.20). There were no significant difference in partial recanalization of SVT (OR 2.72; 95%CI 0.65 to 11.37; P = 0.17), recurrent thrombotic events (OR 0.89; 95%CI 0.33 to 2.42; P = 0.83), and all-cause mortality (OR 0.78; 95%CI 0.24 to 2.58; P = 0.68) between the DOACs and VKAs groups. However, for SVT extension, there is a significant effect favoring DOACs with an OR of 0.12 (OR 0.12; 95%CI 0.03 to 0.54; P = 0.006) (Figures 2 and 3).
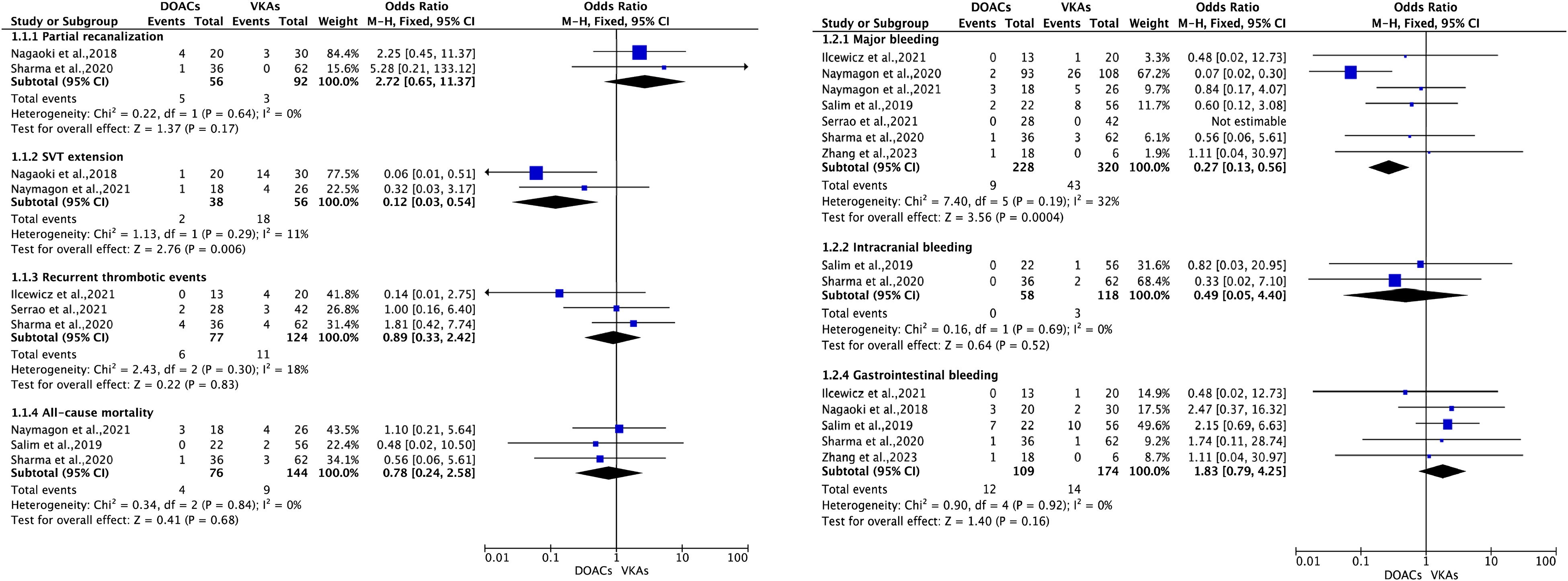
The outcomes between DOACs versus VKAs in patients with SVT (fixed-effects model). DOACs = direct oral anticoagulants; VKAs = vitamin K antagonists; SVT = splanchnic vein thrombosis; RD = risk difference; CI = confidence interval.
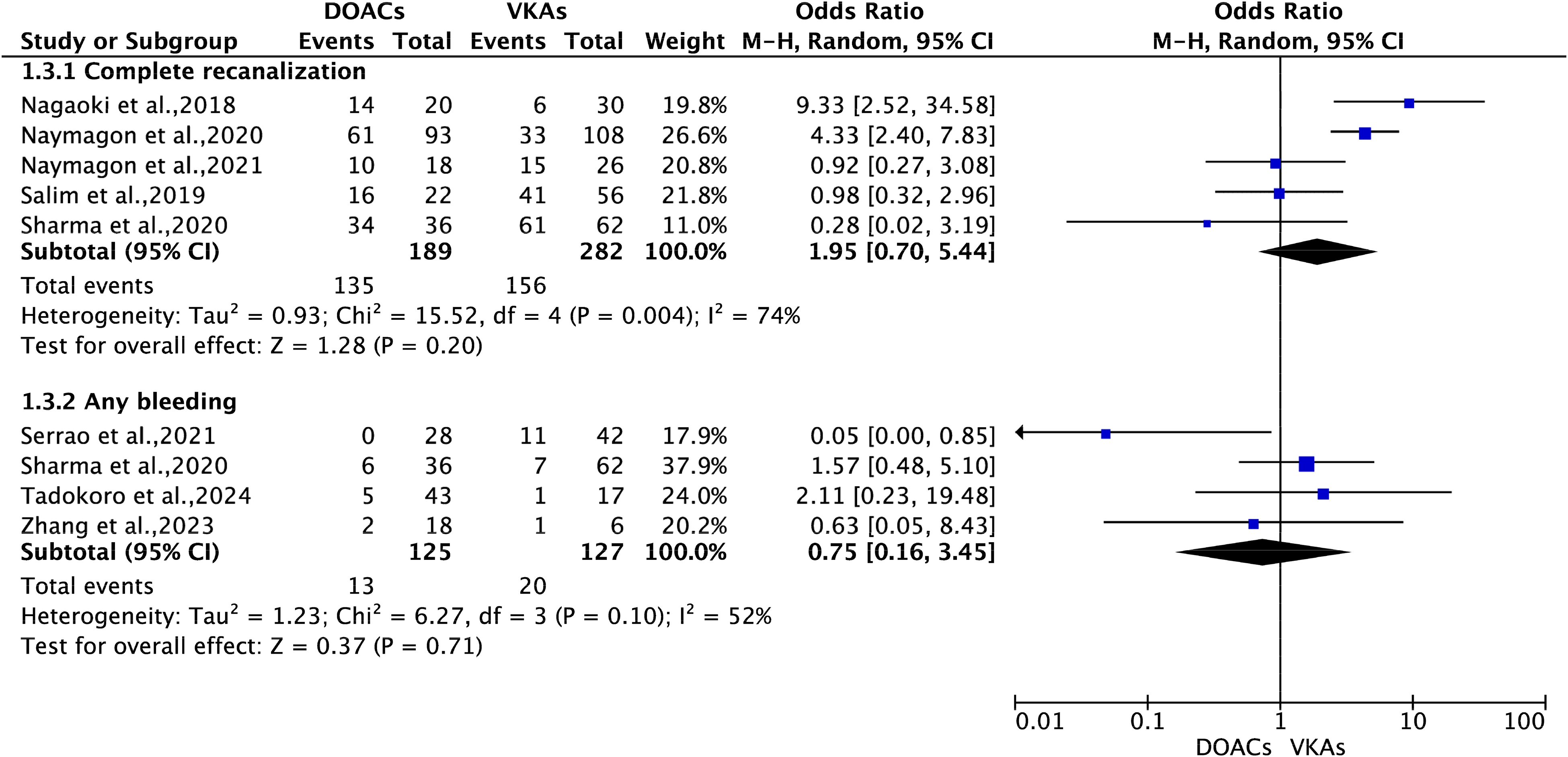
The outcomes between DOACs versus VKAs in patients with SVT (random-effects model). DOACs = direct oral anticoagulants; VKAs = vitamin K antagonists; SVT = splanchnic vein thrombosis; RD = risk difference; CI = confidence interval.
Safety Outcomes Between DOACs Versus VKAs in Patients with SVT
For the safety outcomes, the analysis found that major bleeding showed a significant overall effect with an OR of 0.27 (OR 0.27; 95%CI 0.13 to 0.56; P = 0.0004), suggesting a lower risk of major bleeding in patients treated with DOACs. There were no significant differences between DOACs and VKAs in the risks of any bleeding (OR 0.75; 95%CI 0.16 to 3.45; P = 0.71), intracranial bleeding (OR 0.49; 95%CI 0.05 to 4.40; P = 0.52), and gastrointestinal bleeding (OR 1.83; 95%CI 0.79 to 4.25; P = 0.16) among patients with SVT (Figures 2 and 3).
The analysis of the four studies (Nagaoki et al,2018; Zhang et al,2023; Tadokoro et al,2024; Naymagon et al,2021) that exclusively included cirrhotic patients has been conducted, yielding the following results for the specified outcomes: complete recanalization (n = 2; OR 2.89, 95%CI 0.30 to 28.4), partial recanalization (n = 1), SVT extension (n = 2; OR 0.12, 95%CI 0.03 to 0.54), recurrent thrombotic events (n = 0), all-cause mortality (n = 1), major bleeding (n = 2; OR 0.89, 95%CI 0.21 to 3.65), any bleeding (n = 2; OR 1.35 95%CI 0.26 to 6.97), intracranial bleeding (n = 0), and gastrointestinal bleeding (n = 2; RD 2.02, 95%CI 0.39 to 10.61) (Supplemental Figure 1). Given the limited data included in this analysis, it was noted that the results be interpreted with caution.
Sensitivity Analysis and Publication Bias
In the sensitivity analysis, we sequentially removed the included studies and found that the results were stable for the outcomes of complete recanalization of SVT and major bleeding. Supplemental Figure 2 provides the funnel plots for the efficacy and safety outcomes showing no obvious publication bias.
Discussion
The present meta-analysis provides valuable insights into the comparative effectiveness and safety of DOACs versus VKAs in the clinical management of SVT. While our study did not identify a statistically significant overall advantage of DOACs in terms of efficacy, the notable reduction in major bleeding events with DOACs is clinically significant. This finding is particularly important given the narrow therapeutic window in SVT patients, where the balance between preventing thrombotic complications and avoiding bleeding is critical. The trend observed in favor of higher complete recanalization rates with DOACs, despite not reaching statistical significance, aligns with the known benefits of these agents in other thrombotic conditions and warrants further investigation.
For patients with liver cirrhosis, the coagulopathy associated with the disease makes it challenging to monitor anticoagulation effectively using VKAs. Additionally, the reduced hepatic function in these patients may impact the metabolism of DOACs and increase the risk of bleeding. Furthermore, the presence of varices, which are enlarged and fragile blood vessels, in anticoagulated SVT patients can also contribute to bleeding complications. In non-cirrhotic patients with malignancies or myeloproliferative disorders, the management of SVT is further complicated by the multifaceted nature of these underlying conditions. The choice of anticoagulation therapy must take into consideration the unique risks and benefits associated with each individual's specific condition. Therefore, the treatment of SVT requires a thorough understanding of the underlying etiology and careful consideration of the potential risks and benefits associated with different anticoagulant modalities.
Due to the emergence of numerous studies examining the use of DOACs in treating patients with SVT,3,38 it has become necessary to conduct systematic reviews and meta-analyses to evaluate the safety and efficacy of these DOACs compared to VKAs. Previous reviews39,40 attempted to fulfill this purpose; however, their findings were compromised by the inclusion of the retracted randomized clinical trial conducted by Hanafy et al 15 Consequently, in order to provide a more accurate evaluation, we have excluded the study by Hanafy et al 15 and incorporated recently published research into our updated systematic review and meta-analysis of the current body of evidence about DOACs versus VKAs in the treatment of SVT patients. Our findings of this meta-analysis suggests that DOACs could serve as a feasible alternative to VKAs in the management of SVT, presenting a comparable safety profile while holding the promise of enhanced efficacy. The absence of significant differences in bleeding complications between the two groups further supports the notion that DOACs can be just as safe and effective in treating SVT as VKAs.
When compared with DOACs, traditional VKAs like warfarin present several disadvantages. These include the necessity for regular monitoring of anticoagulant activity, the complexity of dose adjustments, and susceptibility to drug-drug and drug-food interactions. LMWHs, despite their efficacy in preventing thrombosis, often necessitate injections, which can be inconvenient and distressing for patients, particularly those with a needle phobia or aversion to injections. Moreover, the prolonged use of LMWHs may precipitate complications such as heparin-induced thrombocytopenia and a reduction in bone mineral density. Given these considerations, even in the absence of significant differences in efficacy and safety outcomes between DOACs and VKAs, the use for DOACs in the treatment of SVT could be preferred. Their oral administration, fewer dietary restrictions, and reduced need for routine coagulation monitoring offer a more patient-friendly and convenient therapeutic option.
The present meta-analysis was not registered. In addition, there were several limitations that should be noted in this study. First, one of the main limitations of the study was that it only considered retrospective studies, which means that the data used in the study was derived from past events and may not accurately represent the current situation. Second, another limitation was the absence of subgroup analyses based on the different types of DOACs, the various types or etiologies of SVT, and the severity of cirrhosis. This lack of subgroup analysis was due to the scarcity of available data on these specific factors. Third, our study was unable to perform a subgroup analysis based on the Child-Pugh classification because the necessary data for such an analysis were not reported in the included studies. To address these limitations and provide more conclusive findings, further randomized clinical trials are essential, which would involve a controlled experimental framework and enable researchers to establish more definitive treatment recommendations for patients with SVT.
Conclusions
In summary, our meta-analysis indicates that DOACs are superior to VKAs in SVT extension and major bleeding in treating SVT. These findings support the consideration of DOACs as an alternative to VKAs for SVT treatment. Further randomized clinical trials should confirm these findings.
Supplemental Material
sj-pdf-1-cat-10.1177_10760296241274750 - Supplemental material for Effect of Direct Oral Anticoagulants in Patients with Splanchnic Vein Thrombosis: A Systematic Reviews and Meta-Analysis
Supplemental material, sj-pdf-1-cat-10.1177_10760296241274750 for Effect of Direct Oral Anticoagulants in Patients with Splanchnic Vein Thrombosis: A Systematic Reviews and Meta-Analysis by Yun Wan, MS, Linjuan Guo, MD, and Meimei Xiong, MS in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgment
None.
Availability of Data and Materials
All relevant data and materials are presented in the paper.
Author Contributions
Initials (Meimei Xiong), conceptualization; Initials (Yun Wan and Linjuan Guo), methodology; Initials (Meimei Xiong), investigation; Initials (Yun Wan) wrote the manuscript; Initials (Linjuan Guo), validation, revised the manuscript. All authors contributed to editorial changes in the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not required.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
